Abstract
Background
Gastric intestinal metaplasia is a pre-cancerous condition associated with multiple factors.
Objective
We evaluated whether cumulative proton pump inhibitor dose is associated with the diagnosis of gastric intestinal metaplasia while controlling for multiple variables.
Methods
We retrospectively identified patients who underwent upper endoscopy with gastric biopsy between 2005 and 2014. Covariate data retrieved included age, sex, ethnicity, smoking status,
Results
Of the 14,147 included patients (median age 63.4 years; women 54.4%;
Conclusions
Among
Introduction
Interest in the pathogenesis, diagnosis and management of gastric intestinal metaplasia (GIM), a precursor lesion to gastric dysplasia and cancer, is increasing.1–8 Emerging evidence from several observational studies suggests that the long-term use of proton pump inhibitors (PPIs) is associated with a higher risk of gastric cancer development, especially in patients who have undergone
On the other hand, data regarding the effect of long-term PPI use on the development of GIM are conflicting. Previously, two randomised controlled trials have shown that long-term PPI use was not associated with gastric atrophy or GIM.14,15 More recently, a Cochrane systematic review of four randomised controlled trials reported a non-significant increase in the risk of atrophic gastritis and GIM in patients with long-term PPI use.
16
A systematic review that pooled 16 studies (1920 patients) concluded that
However, understanding the association of PPI use with the development of GIM is complex, as histological examination is required and other multiple risk factors are associated with the diagnosis of GIM, such as
Hence, to clarify further the association between PPI use and the development of GIM, we conducted a retrospective endoscopic pathology-based study to assess whether PPI use is associated with the diagnosis of GIM while controlling for quantitative PPI use,
Methods
Patient selection
This retrospective study was conducted at Rabin Medical Center, an academic referral centre of the Clalit Health Services (CHS), the largest health maintenance organisation (HMO) in Israel. 22 Patients were eligible for inclusion if they were members of the CHS for at least 10 consecutive years before undergoing endoscopy, aged over 18 years, underwent upper endoscopy with a stomach biopsy between January 2005 and December 2014 at Rabin Medical Center, and did not have evidence of previous GIM. They were identified from the electronic database of Rabin Medical Center. Patients were excluded from the study if they had undergone an upper gastrointestinal surgery or had been diagnosed as having gastric cancer, had a previous diagnosis of GIM or cancer predisposition syndromes (Lynch syndrome or familial polyposis).
Endoscopic and pathological data
Endoscopy results were extracted from the patients’ electronic medical records (EMRs). EMRs were linked to the pathology database for the diagnosis of GIM (International Classification of Disease, version 10 (ICD-10) code K318). All pathology reports with GIM were manually reviewed, and GIM was classified for dysplasia according to the Vienna system (absent, low-grade dysplasia (LGD), or high-grade dysplasia) and the extent of intestinal metaplasia (extensive (the involvement of both the antrum and body), focal (the involvement of the antrum or stomach body), or unspecified (indeterminate)).23,24 All cases with GIM were manually reviewed for evidence of prior GIM in the pathology archive of the CHS (updated from January 2000).
Cumulative proton pump inhibitor use
PPI use was defined in accordance with PPI prescriptions issued within 10 years before endoscopy. The cumulative PPI dose (number of daily drug doses (DDD)) within 10 years (prior to endoscopy) was calculated in accordance with the anatomical therapeutic chemical (ATC) classification system 21 (omeprazole, 20 mg; pantoprazole, 40 mg; lansoprazole, 20 mg; and esomeprazole, 30 mg). The total DDD was divided into quartiles (Q1: 0–19, Q2: 20–89, Q3: 90–503, Q4: ≥504).
H. pylori infection status
Other variables
Other variables included age at endoscopy, sex, ethnicity (Jewish or Arab Israeli), smoking status (current, past, or never smoker), 25 presence of anti-parietal cell antibodies (APCAs) at a threshold of 1/80 or greater, BMI divided into quantiles (<22.86 kg/m2, 22.86–<25.35 kg/m2, 25.35–<27.68 kg/m2, 27.68–<30.83 kg/m2, ≥30.83 kg/m2) and by the World Health Organization (WHO) classification (underweight <18.5 kg/m2, normal weight 18.5–24.9 kg/m2, overweight 25–29.9 kg/m2 and obese ≥30 kg/m2, the age-adjusted Charlson co-morbidity index (none, mild, moderate and severe) 26 and a family history of gastrointestinal tract cancer.
Statistical analyses
Binary logistic regression analysis of GIM diagnoses was performed. The variate data included age group, sex, ethnicity,
Study oversight and conduct
The Helsinki institutional review board of Rabin Medical Center approved the study and waived the requirement for written informed consent (RMC 544-17).
Results
Patients
During the study period, 40,178 patients underwent upper gastrointestinal endoscopy. Of the patients, 5888 were excluded (3796 without consecutive CHS membership, 1201 with previous gastric cancer, 705 with current gastric malignancy, 101 with previous GIM and 85 with familial polyposis or Lynch syndrome). Of the remaining 34,391 patients (85.4%), 14,147 (41.4%) had a biopsy taken during endoscopy and were included in the final data set (Figure 1). The patients’ median age was 63.4 years. Of the patients, 54.4% were women, 96.8% were Jewish Israeli, 35.5% were current or past smokers and 29% received a clarithromycin-based triple therapy treatment for
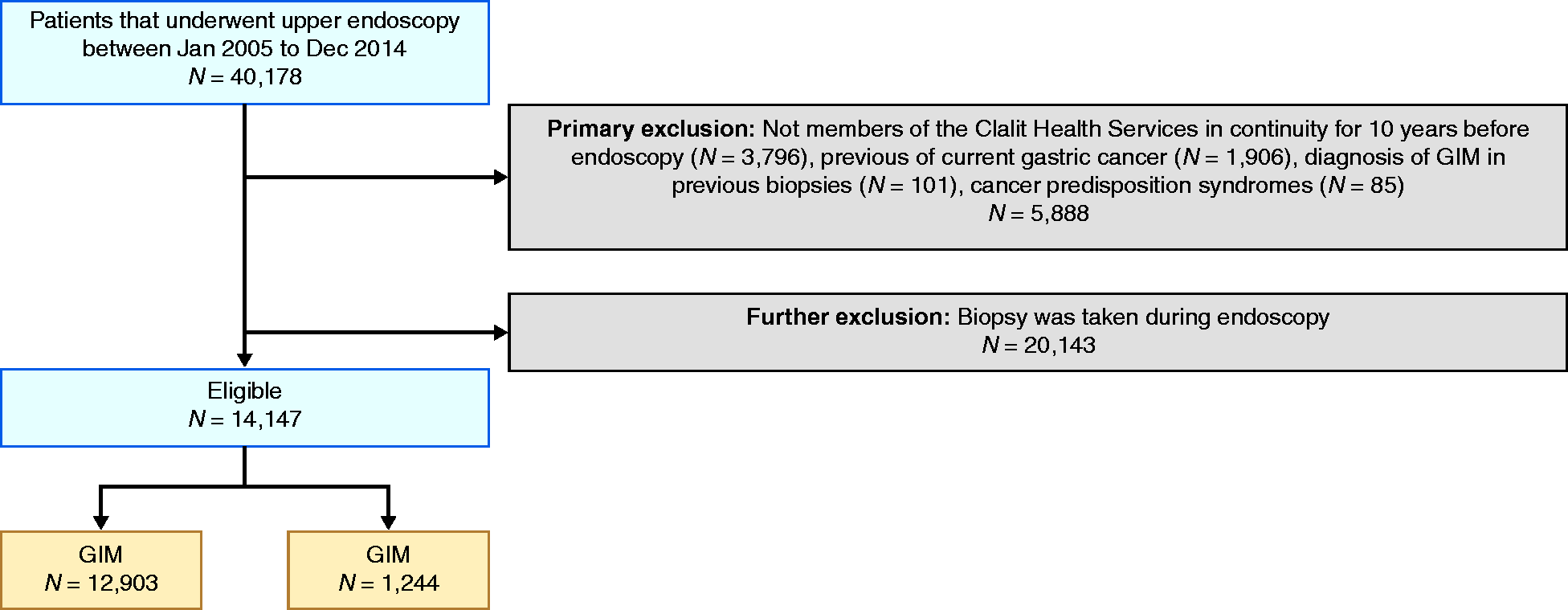
Study flowchart.
Patient characteristics and cumulative PPI use.
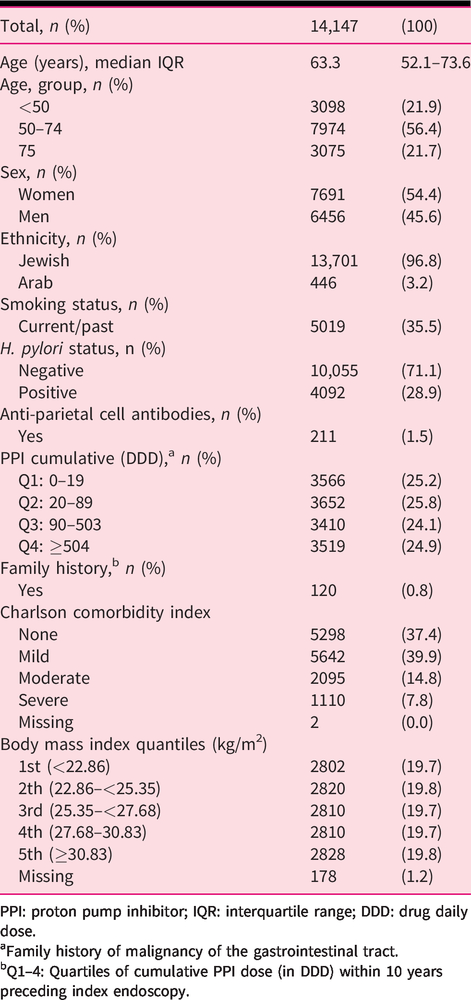
PPI: proton pump inhibitor; IQR: interquartile range; DDD: drug daily dose.
aFamily history of malignancy of the gastrointestinal tract.
bQ1–4: Quartiles of cumulative PPI dose (in DDD) within 10 years preceding index endoscopy.
Factors associated with the diagnosis of GIM
GIM was evident in 13.8% of those aged 75 years old and older, 9.0% of those aged 50–74 years old and 3.3% of those aged under 50 years old (
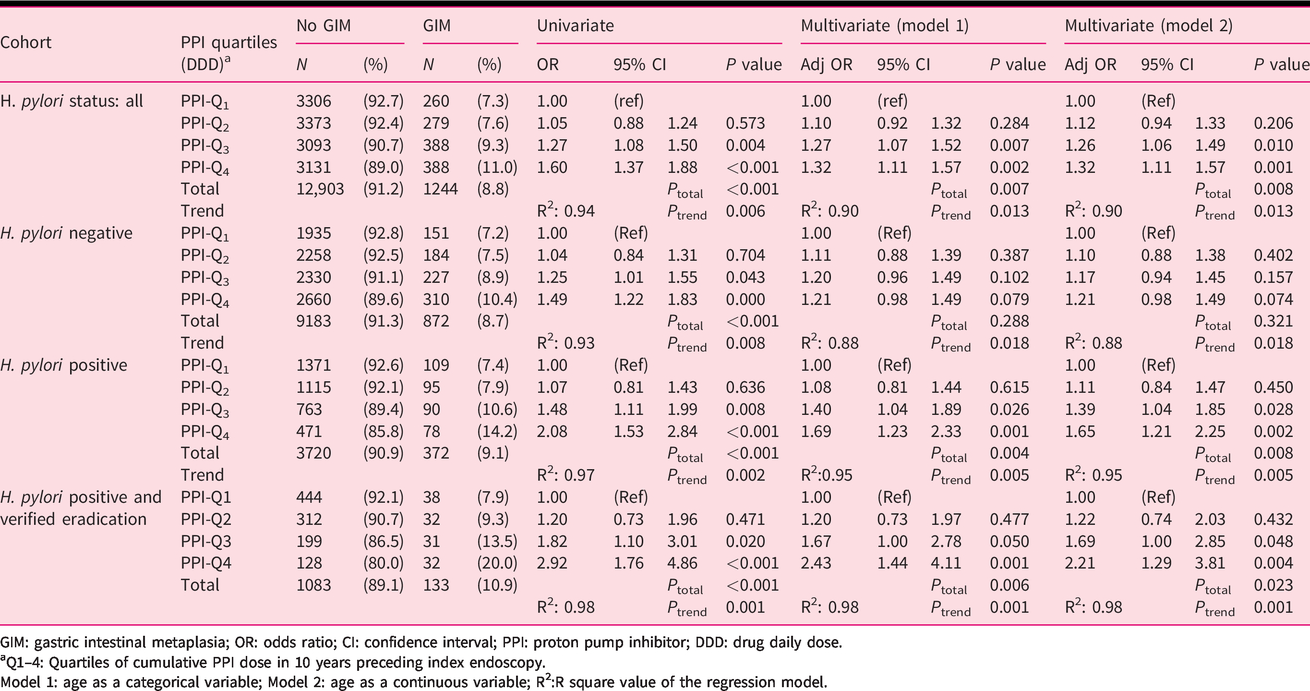
Univariate and multivariate analysis for diagnosis of GIM.
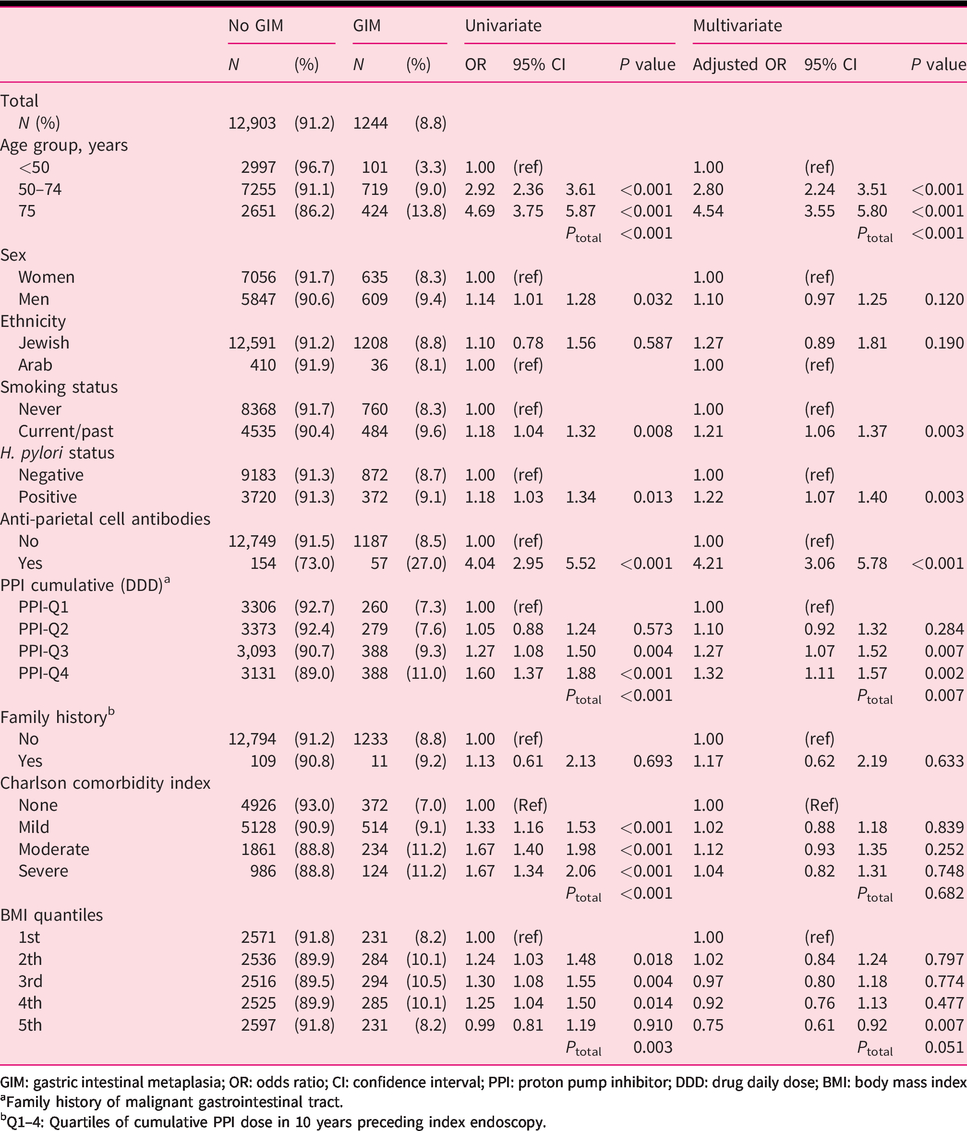
GIM: gastric intestinal metaplasia; OR: odds ratio; CI: confidence interval; PPI: proton pump inhibitor; DDD: drug daily dose; BMI: body mass index
Family history of malignant gastrointestinal tract.
Q1–4: Quartiles of cumulative PPI dose in 10 years preceding index endoscopy.
Age,
As compared to the lower quartile of cumulative PPI doses (PPI-Q1), PPI-Q4 and PPI-Q3 were significantly associated with a diagnosis of GIM: adjusted OR 1.32 (95% CI 1.11–1.57) and 1.27 (95% CI 1.07–1.52), respectively, for the whole cohort; 1.69 (95% CI 1.23–2.33) and 1.40 (95% CI 1.04–1.89), respectively, for
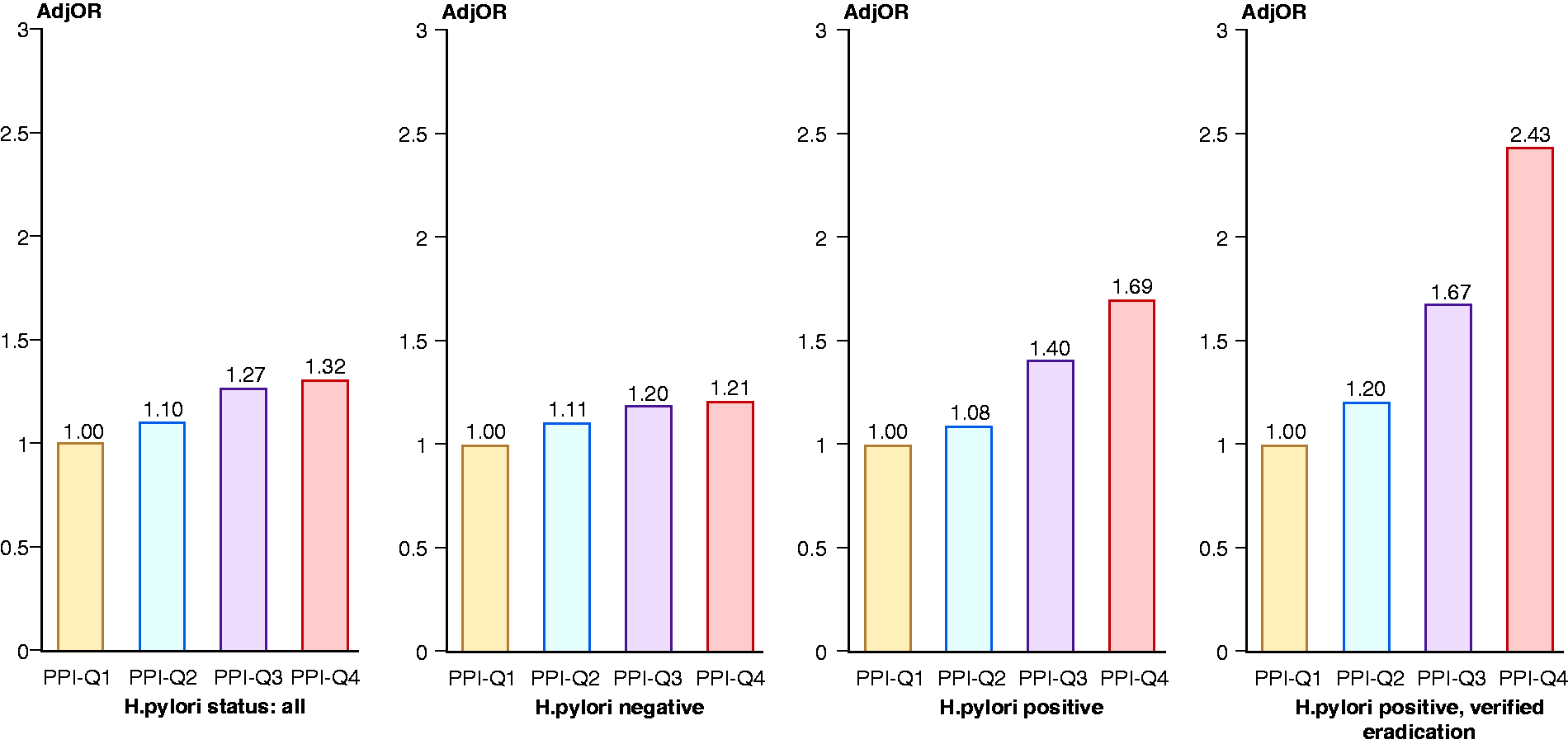
Adjusted odds ratios (ORs) for the association of proton pump inhibitor (PPI) use and the diagnosis of pathologically confirmed gastric intestinal metaplasia (GIM) among the whole cohort,
Univariate and multivariate analysis for diagnosis of GIM with cumulative PPI exposure, stratified by
GIM: gastric intestinal metaplasia; OR: odds ratio; CI: confidence interval; PPI: proton pump inhibitor; DDD: drug daily dose.
aQ1–4: Quartiles of cumulative PPI dose in 10 years preceding index endoscopy.
Model 1: age as a categorical variable; Model 2: age as a continuous variable; R2:R square value of the regression model.
As can be seen in Table 3, these associations remained stable when age was used as either a continuous variable or a categorical variable. As can be seen in Table 3, the trend analysis yielded a positive dose–response with a strong fit (R2 ≥0.95) for the
After adjusting for multiple variates, gender, ethnicity and the comorbidity index were not significantly associated with the diagnosis of GIM. Analysis of the association between BMI and diagnosis of GIM showed that after adjusting for all variables, as compared to the 1st quintile, the 5th quintile was associated with reduced odds for a diagnosis of GIM (adjusted OR, 0.75, 95% 0.61–0.92). Repeated analysis with BMI classified according to WHO classification yielded consistent results (obese: adjusted OR 0.76, 95% CI 0.64–0.89; normal weight: referent) (see Supplementary Table 1).
GIM with LGD was diagnosed in 43 patients (0.3%). In multivariable analysis, independent variables associated with the diagnosis of GIM with LGD were increasing age, the presence of APCAs (adjusted OR 6.76, 95% CI 2.02–22.6), and upper quartiles (PPI-Q4 and PPI-Q3) of PPI use (adjusted OR 10.1, 95% CI 2.32–44.1 and adjusted OR 5.66, 95% CI 1.24–25.83, respectively; PPI-Q1: reference) (Table 4).
Univariate and multivariate analysis for diagnosis of GIM with LGD.
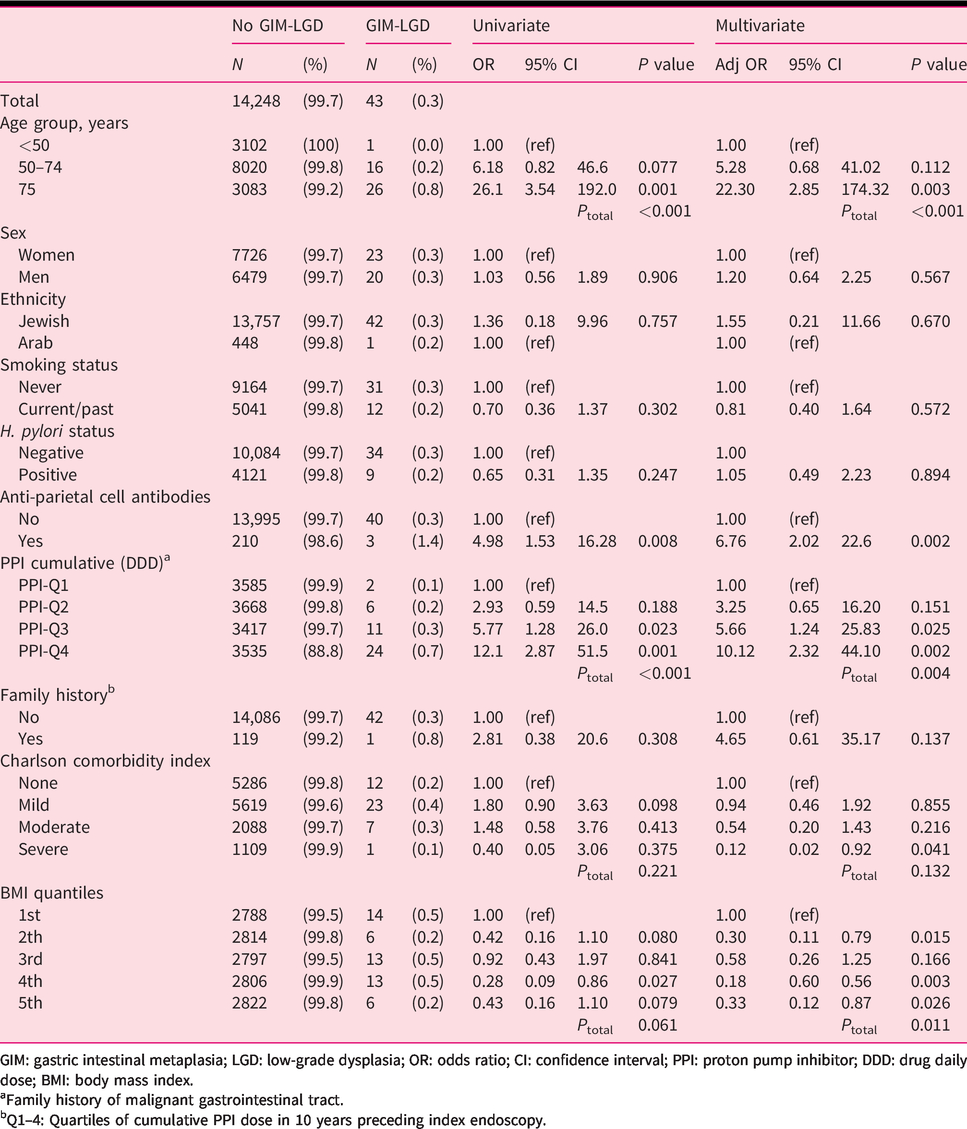
GIM: gastric intestinal metaplasia; LGD: low-grade dysplasia; OR: odds ratio; CI: confidence interval; PPI: proton pump inhibitor; DDD: drug daily dose; BMI: body mass index.
aFamily history of malignant gastrointestinal tract.
bQ1–4: Quartiles of cumulative PPI dose in 10 years preceding index endoscopy.
Extensive GIM was evident in 232 of the 804 subjects with GIM and separate biopsies that were obtained from both the antrum and stomach body (28.9%). After adjusting for age, only the presence of APCAs was significantly associated with extensive GIM (adjusted OR 2.05, 95% CI 1.14–3.70).
Discussion
Our findings show a dose-dependent association of cumulative PPI dose with the diagnosis of GIM. We have also shown that the association of cumulative PPI use with the diagnosis of GIM is dependent on
As stated earlier, data regarding the effects of long-term PPI use on the development of pre-cancerous gastric lesions such as gastric atrophy or GIM are conflicting.12,27 While two randomised controlled trials reported no association between PPI use and gastric atrophy or GIM,14,15 a recent Cochrane review of four randomised controlled trials reported non-significant increases in the risks of atrophic gastritis and GIM in patients with long-term PPI use.
16
Finally, a systematic review that pooled 16 studies concluded that
As guidelines are conflicting regarding routine H.
A recent large Korean study reported that obesity was independently associated with an increased incidence of GIM. 21 In our study, the univariate analysis did show an association between increasing BMI and a diagnosis of GIM. However, after adjusting for multiple variables, this association was no longer observed, and a negative association was observed for the obese (as defined by either the WHO classification and by quintiles). Our findings are consistent with a recent study performed among US veterans, which found that smoking is associated with a diagnosis of GIM. 20
The precise mechanism by which PPIs may induce gastric cancer is unclear, although several mechanisms have been proposed. PPIs induce hypergastrinemia and hypochlorhydria, which may contribute to enterochromaffin-like cell hyperplasia and proliferation of gastric mucosa. PPI-induced hypergastrinemia occurs due to inhibition of the somatostatin-mediated negative feedback of gastrin release on antral G cells.
17
Similarly, gastrin may exert a direct trophic effect on the oxyntic mucosa. It has also been hypothesised that PPI-induced alterations of the gastric microbiome may play a role in carcinogenesis. However, a recent study showed that PPI-treated patients showed similar microbial diversity compared with normal subjects, while patients with
The main strength of our study is that it is a large-scale endoscopic pathology-based study conducted in an HMO. We verified continuous membership, quantified PPI use for up to 10 years before endoscopy, defined
As a retrospective association study, the main limitation of this study is that it did not clarify whether the association between PPI use and the diagnosis of GIM is causative. Also, our sample size was not large enough to allow stratification according to the timing of
In summary, we report a dose-dependent association of PPI doses with the diagnosis of GIM among patients treated for
Study highlights
The established knowledge on this subject:
Data regarding the effects of long-term PPI use on the development of pre-cancerous gastric lesions such as gastric atrophy and GIM, especially in patients treated for
What are the significant findings of this study?
Among Upper quartiles of PPI use are also associated with 5–10-fold increased odds for the diagnosis of GIM with dysplasia.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620951403 - Supplemental material for Dose-dependent association of proton pump inhibitors use with gastric intestinal metaplasia among Helicobacter pylori -positive patients
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620951403 for Dose-dependent association of proton pump inhibitors use with gastric intestinal metaplasia among
Footnotes
Author contribution
Specific author contributions: conceptualisation and design: YS, ZL and ADC; data extraction: IF and HL; analysis and interpretation of data: IF, AV and TS; drafting of the manuscript: YS and YLW; critical revision of the manuscript for valuable intellectual content: DB, YN and ID; final approval of the paper: ZL and DB; guarantor of the article: ZL. All authors approved the final version of the paper.
Declaration of conflicting interests
Arnon Cohen received research grants from Janssen, Novartis, AbbVie, Janssen, and Sanofi. Arnon Cohen served as a consultant, advisor, or speaker to AbbVie, Amgen, Boehringer Ingelheim, Dexcel Pharma, Janssen, Kamedis, Lilly, Neopharm, Novartis, Perrigo, Pfizer, Rafa, Samsung Bioepis, Sanofi, Sirbal and Taro. Iris Dotan served as a consultant to Abbvie, Janssen, Takeda, Pfizer, Genentech/Roche, Gilead, Rafa, Celltrion, Ferring and Medtronic; and a speaker to AbbVie, Janssen, Takeda, Pfizer, Genentech/Roche, Ferring and Falk Pharma. The remaining authors report no other potential personal or financial conflicts of interest.
Ethics approval
The Helsinki institutional review board of Rabin Medical Center approved the study.
Funding
This study received financial support from the Rabin Medical Center Research Fund.
Informed consent
The Helsinki institutional review board of Rabin Medical Center approved the study and waived the requirement for written informed consent (RMC 544-17).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
