Abstract
Background and aims
Histopathology is the most accurate test to detect H. pylori when performed correctly with unknown validity in daily practice clinic settings. We aimed to determine the rate of potentially false-negative H. pylori results that might be due to continued use of proton pump inhibitors (PPIs) in routine endoscopy practice. We also aimed to establish whether gastroenterologists recommend routine cessation of PPIs before esophagogastroduodenoscopy (EGD) and whether they regularly document that biopsies for H. pylori testing have been taken while the patients are on PPI treatment.
Methods
Detailed information about three known factors (PPIs, antibiotics and prior H. pylori eradication treatment), which may cause histology or rapid urease test (RUT) to be unreliable, had been prospectively collected through interviews using a questionnaire before each test. Gastric biopsies were stained with H&E for histological analysis.
Results
A total of 409 individuals at three academic gastroenterology institutions were tested 200 times with histology. Fifty-six per cent (68 of 122) of all negative tests fell in the category of continuing PPI use, which had the potential to make the histology and RUT results unreliable.
Conclusions
These data demonstrate a clear and important gap between current guidelines and real-world practice with regards to the diagnosis of H. pylori during EGD. A negative histology or RUT should be considered false negative until potential protocol violations are excluded. Documentation of PPI use during the EGD should be an integral part of the EGD report. The current practice of taking biopsies for H. pylori testing in patients under PPIs should be reevaluated.
Introduction
Proton pump inhibitors (PPIs) constitute one of the most significant medical breakthroughs in the treatment of acid-related disorders. They are highly effective in the symptomatic relief and treatment of peptic ulcer, gastro-oesophageal reflux disease (GERD), and as part of different combination therapies for Helicobacter pylori (H. pylori) eradication.1,2 Use of PPIs may cause false-negative results of the urea breath test (UBT), the stool antigen test (SAT), rapid urease test (RUT) and gastric histopathology or culture. This may be explained by a combination of marked gastric acid suppression3–6 and antimicrobial activity of these compounds against H. pylori. 7 Direct inhibition of the bacterial urease activity8,9or formation of sulphides of benzimidazoles as demonstrated in culture media may be the reasons for this selective antibacterial effect. 10 However, growth inhibition has also been reported in urease-negative H. pylori derivatives. 11 In addition, in H pylori-positive patients, PPIs alter the distribution and shape of the bacteria,12,13 making the bacteria more difficult to detect.
Previously published reports have described false-negative UBT rates of 10%–40% in patients using PPIs.14,15 PPIs can similarly influence SAT16,17 results, but some monoclonal antibody-based SATs that are currently available are not affected by PPIs.18,19 This may also be the case with biopsy-based tests (including culture, RUT and histology) although previous studies of this type have involved relatively few patients.7,20,21
According to the latest Maastricht IV Consensus Conference published by the European Helicobacter Study Group, Statement 4 declares that ‘In patients treated with PPIs: 1) if possible, PPI should be stopped for 2 weeks before testing by culture, histopathology, RUT, UBT or stool test’ (Evidence level: 1b; Grade of recommendation: A). 22 The American Gastroenterological Association (AGA) and American College of Gastroenterology (ACG) also recommend discontinuing PPIs two weeks before endoscopy, and taking biopsies both from the body and antrum.23,24 The categorisation of H. pylori gastritis according to gastritis subsites together with the assessment of gastritis severity is also recommended by the Kyoto Global Consensus Report on H. pylori gastritis. 25 These are critically important for identifying those patients who remain at high risk after H. pylori eradication.
Based on these studies, most centres currently recommend cessation of PPIs 7–14 days before H. pylori UBT testing. This allows the bacteria to repopulate in the stomach and the tests previously negative can once again become positive.
In the present study we prospectively evaluated the diagnosis of H. pylori in routine endoscopy practice and determined the rate of potentially false-negative results that might be due to continued use of PPIs before esophagogastroduodenoscopy (EGD). We aimed to establish whether gastroenterologists recommend routine cessation of PPIs before EGD and whether they regularly document that biopsies for H. pylori testing have been taken while the patients are on PPI treatment. Additionally we aimed to reveal independent causes that lead to high probability of EGD histology being false negative.
Materials and methods
Study population and sample size
Patients undergoing upper gastrointestinal endoscopy for symptom evaluation at three academic (affiliated to Tel-Aviv University) medical centres in Israel (Sheba (A), Assaf Harofeh (B) and Sourasky (C)) were evaluated prospectively. This study was approved separately by each of the ethics committees of the institutions. Data were collected from March to September 2014. The inclusion criteria were patients older than 18 years who had an EGD with evaluation for the presence of H. pylori by histology.
Available data at the time of planning the study indicated that the sero-prevalence of H. pylori infection was 45.2% in the Jewish population, increasing with age, reaching 60% at age ≥ 50 years. Seropositivity ranged from 24.3% in individuals originating from North America/Western Europe/Australia, to 63.2% in those from Asia/Africa/South America. 26 Among Arabs individuals, aged 0–19 years, H. pylori sero-prevalence was 42.1% and reached 65% in adolescents. A sample size of 322 participants was calculated as sufficient to estimate a prevalence of 60% with a confidence level of 95% and a precision of 5%. Our aim was to recruit about 400 patients.
Data collection
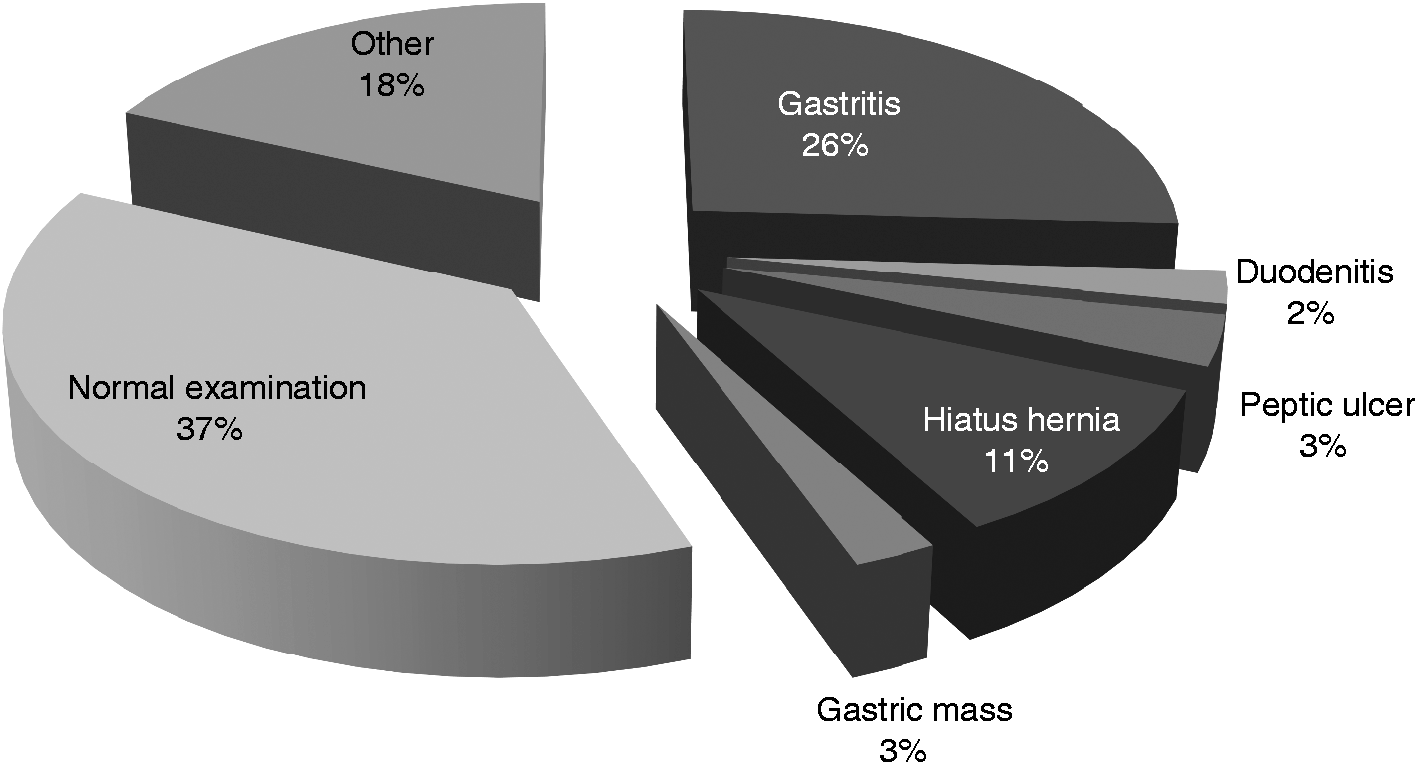
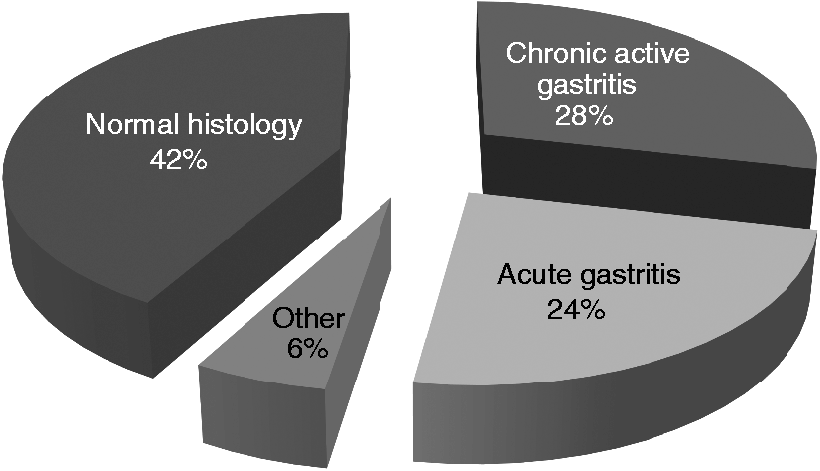
Data were collected through interviews using a questionnaire that looked for the causal factors of false-negative H. pylori results on histology or RUT (recent use of PPIs, antibiotics or medicines containing bismuth, prior H. pylori eradication treatment) as well as the clinical indication for the EGD (Figure 1), endoscopic diagnosis (Figure 2) and histopathologic diagnosis (Figure 3). The structured questionnaire was completed by the local medical staff just before the EGD.
Indications for gastroscopy and H. pylori testing. Endoscopic diagnosis. Histopathology results.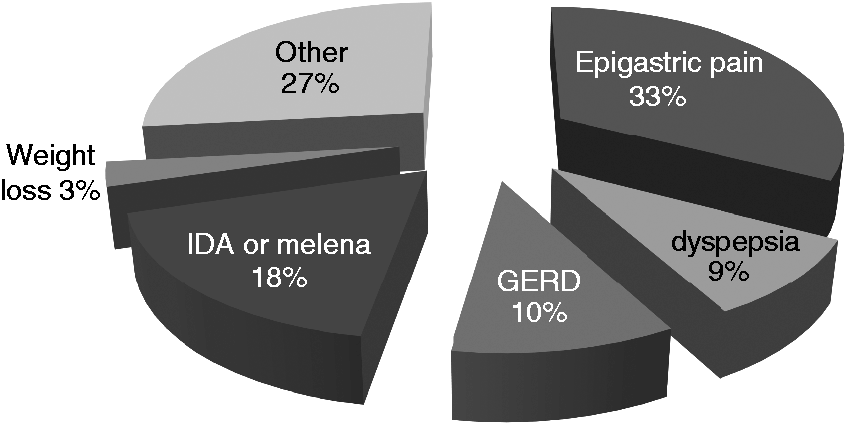
The site and the number of the gastric biopsies that were obtained from each patient during EGD were documented. In cases suggestive of malignancy, biopsies were obtained from the suspected lesion and the adjacent area for histological analysis. Gastric biopsies were stained with hematoxylin-eosin (H&E) for histological analysis. The gastric histopathological parameters were scored from 0 to 3 according to the Sydney classification system for the analysis of polymorphonuclear and mononuclear cell infiltration. Statistical analysis was performed using SPSS for Windows (16.0; SPSS Inc, Chicago, IL, USA) with the Chi-square test and Fisher’s exact probability test (where appropriate) for dichotomous variables and with p values < 0.05 being reported as significant. All participating gastroenterologists were practicing physicians actively engaged in endoscopy practice. Ten of 86 (11.6%) had been in specialist practice between one and five years, 41/86 (47.7%) between six and 10 years, and 35/86 (40.7%) more than 10 years.
To examine independent predictors of high-probability false-negative tests (using an outcome of negative histology under PPI treatment), a multivariate logistic regression model was conducted and variables with p values < 0.05 were considered statistically significant. The variables examined were patient age, the hospital and physician years in practice.
Results
Baseline patients' characteristics
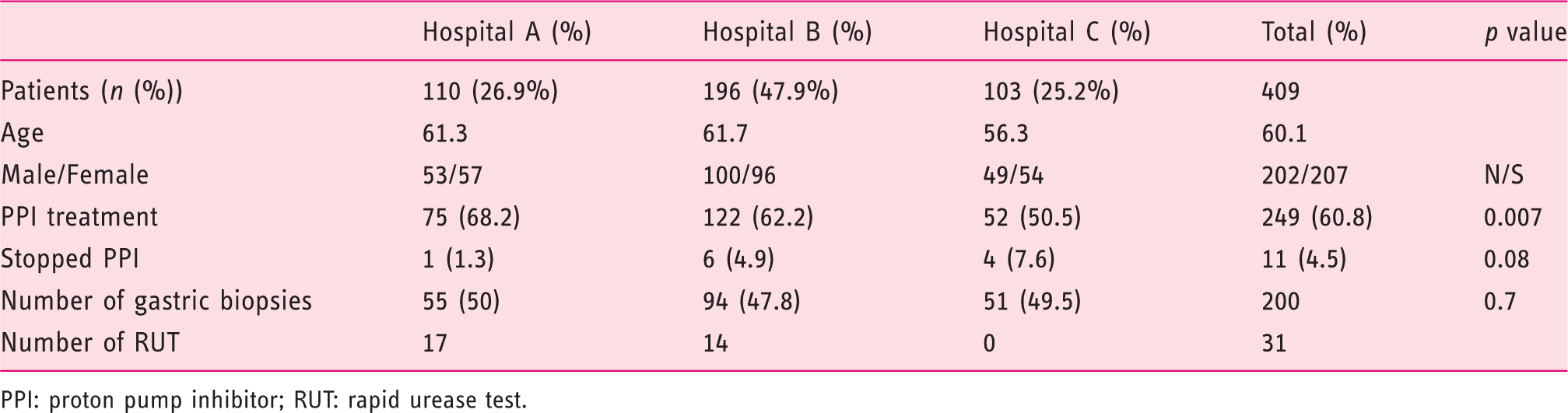
PPI: proton pump inhibitor; RUT: rapid urease test.
Sixty-one per cent of the patients (249/409) were taking PPIs before the EGD and only 11 (4.5%) of them stopped PPIs at least two weeks before the procedure. Among patients continuing PPIs, 81 biopsies have been performed. Of those 68 (84%) were negative and 13 (16%) were positive. Of 81 biopsies taken from patients under PPI treatment, only 17 (20.9%) were taken from both the body and the antrum, as indicated by the Sydney protocol. Fifty-six per cent (68/122) of all negative tests by histology fell in the category of continuing PPI use, which had the potential to make the histology results unreliable (Figure 4).
HP results in patients with or without PPIs.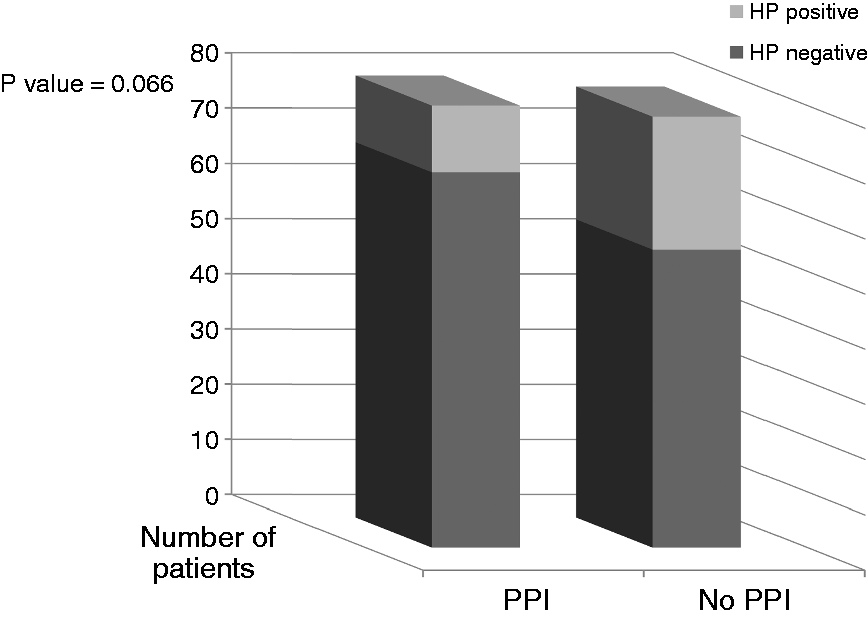
H. pylori status, evaluated by histology or RUT or both (reported in 159 patients)
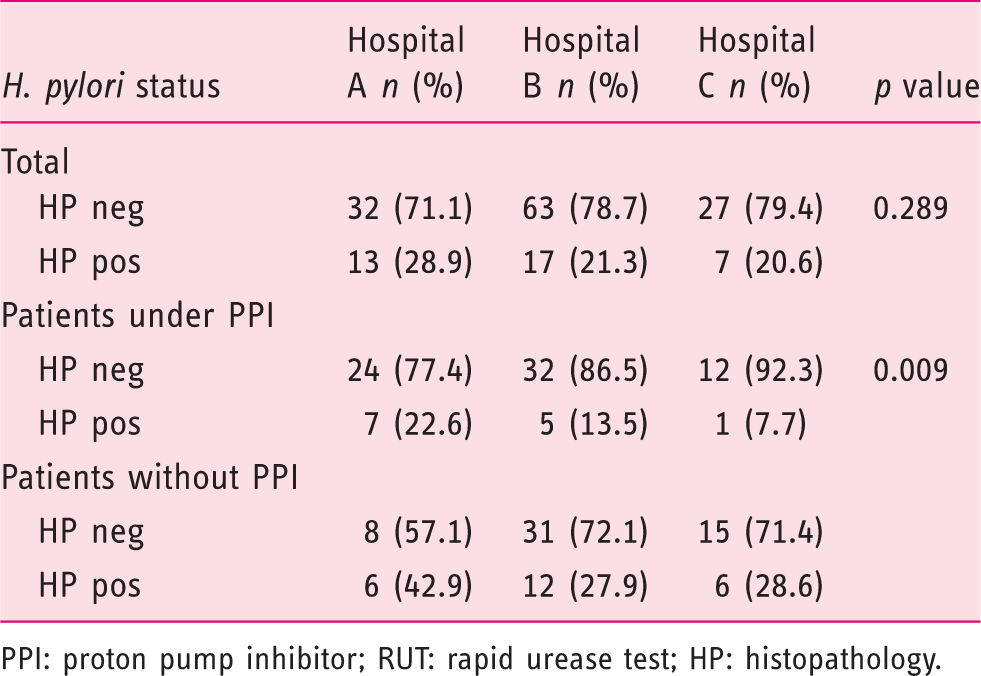
PPI: proton pump inhibitor; RUT: rapid urease test; HP: histopathology.
Forty-four patients were on antibiotic treatment within the past four weeks of the EGD. H. pylori was evaluated in 18 of these patients (17 negative and one positive). Thirteen patients were also on PPIs. Among those referred for EGD, 119 had been diagnosed with H. pylori in the past. Eighty-one of those received different eradication regimens and 59 confirmed the eradication of the bacteria.
No significant difference in the number of biopsies taken from patients on a PPI was observed between the three specialist groups. Multivariate logistic regression analysis revealed a significant difference (p = 0.004) between patient age regarding the probability to have false-negative results (adjusted odds ratio (AOR) = 1.028, 95% confidence interval (CI): 1.009–1.048). In an additional variable examined, no significant difference was demonstrated regarding physician’s experience in years (0.669).
Comparison between hospitals
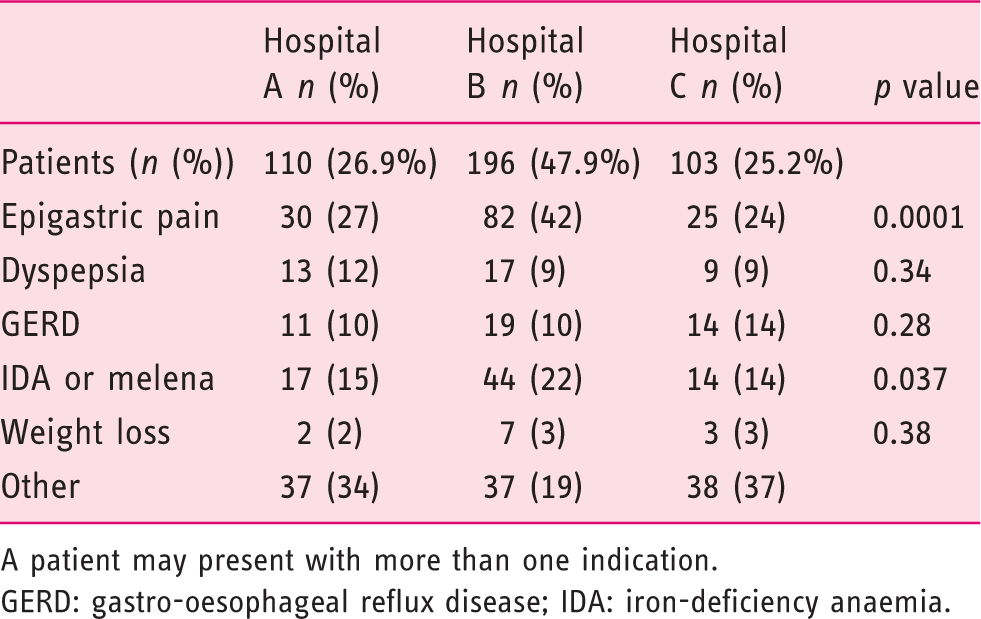
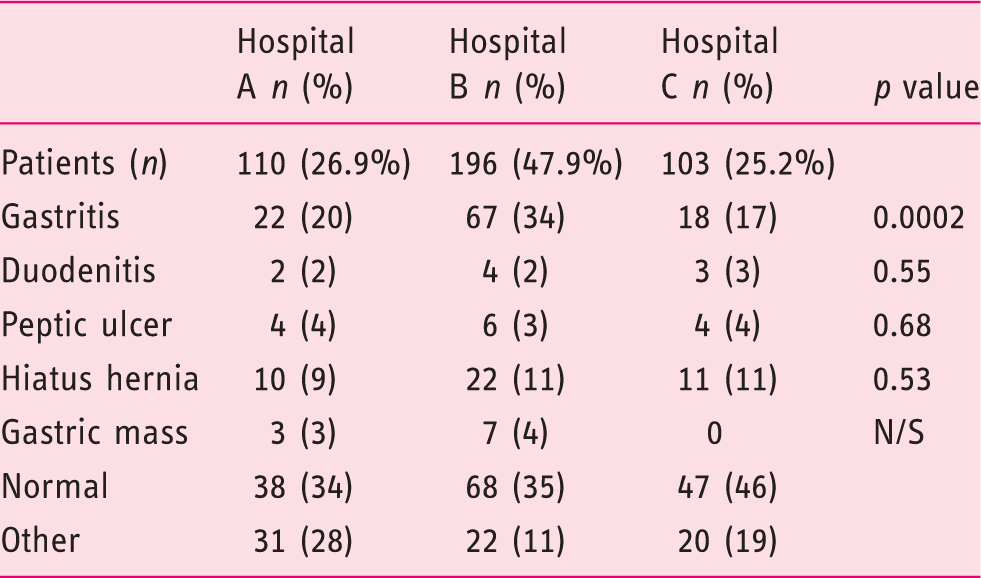
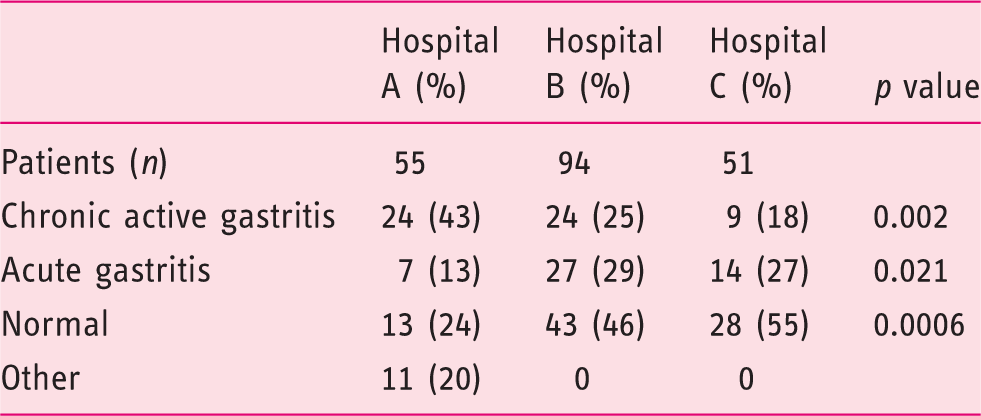
Gastroenterology units did not differ regarding the percentage of patients using PPIs during the EGD, number of biopsies taken (Table 1) and the prevalence of H. pylori among their patients (Table 2 and Figure 5). No significant difference was obtained in the number and sites of the gastric biopsies in patients on PPI treatment (p = 0.107), indicating a similar nonadherence rate to the Sydney recommendations. Nevertheless, the three medical centres did differ significantly with regard to patients’ indications for EGD. Epigastric pain and iron-deficiency anaemia or melena were more common in Hospital B compared to Hospital A or C (Table 3). The only significant difference in the endoscopic findings was a higher prevalence of gastritis in Hospital B (Table 4). However, chronic active gastritis was significantly more common in Hospital A (Table 5).
Biopsies taken under PPI – comparison between three medical centres. Indications for gastroscopy – comparison between three medical centres A patient may present with more than one indication. GERD: gastro-oesophageal reflux disease; IDA: iron-deficiency anaemia. Endoscopic diagnosis – comparison between three medical centres Histologic diagnosis – comparison between three medical centres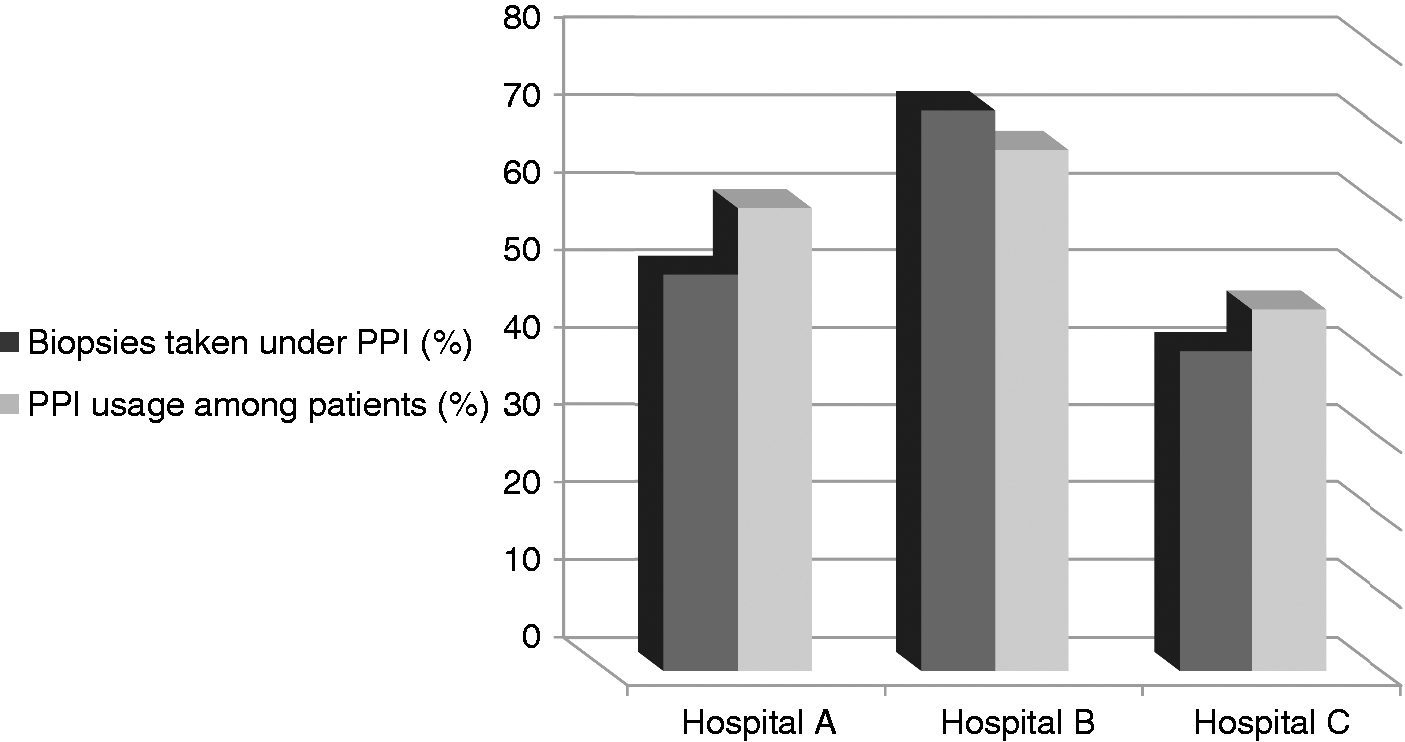
Logistic regression analysis revealed a significant difference (p = 1.2 E−5) between the hospitals regarding the probability to have false-negative results. Hospital A demonstrated higher risk of false-negative results compared to Hospital B (AOR = 4.29, 95% CI: 2.18–8.4, p = 2.1 E−5), and Hospital C (AOR = 5.95, 95% CI: 2.44–14.7, p = 8.3 E−5). No significant differences were demonstrated between Hospital B and C (p = 0.429).
Discussion
The idea for the current study came from a recent publication by Olafsson et al. which showed that 23% (120 of 526) of all negative UBTs had the potential to be unreliable. 27 Based on our daily clinical practice, we hypothesised that the negative predictive value of H. pylori diagnosed by histopathology or RUT will probably be lower. Indeed, our results indicate that a negative histology test or RUT for H. pylori could be a false negative in up to 56%, making these results meaningless. We also found that there was no routine documentation of PPI usage among patients tested for the bacteria, giving the patients and the physicians a false assurance that H. pylori was not present.
The need to withhold PPI therapy in symptomatic patients while awaiting EGD is a common clinical problem with medico-legal aspects. Evaluation of the preparation instructions of the three medical centres participating in the study revealed that none of them recommend cessation of PPIs before EGD. Moreover, only a minority of the patients (6.8%, 17 of 249) were asked to stop PPIs before the procedure.
Treatment with PPIs before endoscopy negatively affects the presence of H. pylori by histopathology and RUT in 50% to 82% of cases.7,21,22,28,29 Recently, Saniee et al. demonstrated that, in vitro, PPIs exert transient negative effects on H. pylori viability, morphology, and urease test. Complete recovery was observed 12 days after removal of PPIs, suggesting that cessation of PPIs before endoscopy, at least for this time period, may help avoid false-negative results of H. pylori. 30
As expected, comparison of the presence of H. pylori infection in our study showed a higher prevalence of the bacterium in patients without PPIs when compared to patients on PPIs, with a strong trend although not statistically significant (p = 0.066). In previous studies, Yakoob et al. demonstrated that histology is comparatively less affected by PPIs than is the RUT.21,29 However, after four weeks of treatment, PPIs may also improve H. pylori-associated gastritis in half of patients, confusing those who believe that in the absence of chronic inflammation, H. pylori infection can be reliably excluded. 31 The Rodger C. Haggitt Gastrointestinal Pathology Society recommends the use of ancillary stains when biopsies show chronic or chronic active gastritis without detectable H. pylori in H&E-stained sections. 32 However, because of higher sensitivity many authors advocate the use of immunostains rather than histochemical stains if ancillary testing is indicated.33,34
Given the design of the present study, we can’t estimate the true false-negative rate due to the use of PPIs. Our study did not take into account two other certain specific conditions that may also be considered to cause false negative by histopathology: a) atrophic gastritis and intestinal metaplasia, which are associated both with decreased or absence of the bacteria in the gastric mucosa, especially in elderly patients;35–37 and b) very low adherence of endoscopists to the Sydney System guidelines (20.9% in our study) which recommend, in PPI receiving patients, taking biopsies both from the antrum and corpus. 38 A recent Canadian study that evaluated the sites of gastric sampling and PPI use at endoscopy also concluded that these guidelines are infrequently adhered to, and sampling only one region increases the likelihood of missing active infection by at least 15%. 39 Considering these two additional pitfalls, older patients may be even more liable to have false-negative results.
Our study provides important information about the accuracy of the commonly used RUT and histopathology for the diagnosis of H. pylori infection in clinical practice. Gastroenterologists should be aware whether the patient is taking PPIs, not only because of the potential false-negative results but also because the negative H. pylori result becomes the conclusive result of the patient. Most experts would agree that if a negative histology or RUT test for H. pylori could be false negative in up to 56% of cases, the common practice of taking biopsies for H. pylori in patients on PPIs should be reevaluated.
In conclusion, we suggest that patients, referring physicians and gastroenterologists should be informed by a detailed instruction sheet which patients may stop PPIs two weeks before EGD. In patients on PPIs, biopsies should be taken both from the antrum and corpus. In the absence of H. pylori with chronic or chronic active gastritis with H&E staining, immunohistochemical staining for H. pylori is indicated. In all patients documentation of PPI use during the EGD should be an integral part of the EGD report.
Survey questionnaire
Potential parameters which may cause false-negative H. pylori results
What is the indication for performing the endoscopic procedure? (epigastric pain, heartburn, discomfort/postprandial fullness/bloating, anaemia/dark stools, weight loss, halitosis, history of malignancy in the family, other). Have you ever been informed of H. pylori colonisation in the past? (Y/N) Have you received treatment for eradication of the bacteria? (Y/N) Has the eradication been verified? (Y/N) Are you treated with drugs of the PPI group? (lansoprazole, omeprazole, esomeprazole, pantoprazole, H2 blockers)? (Y/N) Had the treatment been temporarily stopped prior to the examination today? (Y/N) If so, for how long? (one week, two weeks, three weeks, one month, more than one month). Has your physician advised you to stop the PPI treatment prior to the examination? (Y/N) Have you been informed by the form given prior to the EGD that you should stop PPI treatment? (Y/N) Have you received antimicrobial drugs or bismuth in the last four weeks? (Y/N) Have you undergone gastric surgery in the past? (Y/N)
Data regarding the endoscopic procedure
How long has the physician who performed the procedure been practicing gastroenterology? (<5 years, 5 to 10 years, > 10 years) What is the endoscopic diagnosis? (normal, hiatal hernia, gastritis, peptic ulcer, duodenitis, mass, other). Has a test for H. pylori been performed during the procedure? (Y/N) If so, which test? (histology, RUT) Pathologic diagnosis? (normal, chronic active gastritis, other type of gastritis (atrophic, acute, gastritis has not been evaluated)
Footnotes
Acknowledgement
The authors would like to thank Professor Steven F Moss, Brown University, Providence, RI, USA, for helpful comments in preparing the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
