Abstract
Background
Hepatic encephalopathy is associated with altered gut microbiota. Proton pump inhibitors increase the risk of small bowel bacterial overgrowth.
Objectives
This was a case-control study aimed at exploring the relationship of proton pump inhibitor use with the risk of hepatic encephalopathy during hospitalization in liver cirrhosis.
Methods
Case and control groups were defined as cirrhotic patients who developed hepatic encephalopathy during hospitalization and those without hepatic encephalopathy at admission or during hospitalization, respectively. Age, gender, and Child-Pugh score were matched between the groups. Odds ratios with 95% confidence intervals were calculated to express the association of proton pump inhibitors with the risk of hepatic encephalopathy. Four subgroup analyses were performed after excluding patients with acute upper gastrointestinal bleeding, infections, and in-hospital death, and after matching model for end-stage liver disease score.
Results
In the overall analysis, 128 patients were included in each group of cases and controls. The proportion of proton pump inhibitor use was significantly higher in the case group than the control group (79.7% vs 43%, p < 0.001). Proton pump inhibitor use (odds ratio = 3.481, 95% confidence interval: 1.651–7.340, p = 0.001) was independently associated with the development of hepatic encephalopathy in the multivariate analysis. In the four subgroup analyses, proton pump inhibitor use remained independently associated with the risk of hepatic encephalopathy.
Conclusion
Proton pump inhibitor use might increase the risk of hepatic encephalopathy during hospitalization.
Key summary
1. Summarise the established knowledge on this subject.
HE is associated with small bowel bacterial overgrowth. PPIs may alter the gut microbiota in cirrhotic patients. Association of PPIs with the risk of HE in cirrhotic patients has become a hot debate. 2. What are the significant and/or new findings of this study?
An overall analysis demonstrated that use of PPIs might increase the development of HE during hospitalization. Four subgroup analyses were performed after excluding acute upper gastrointestinal bleeding, infection, and in-hospital death and after matching MELD score, which further confirmed the association of PPIs with risk of HE.
Introduction
Hepatic encephalopathy (HE) is a serious complication of liver cirrhosis1,2 associated with poor prognosis. 3 Overt HE, which refers to grade II–IV HE according to the West Haven criteria, is characterized by paradoxical sleep, altered personality and consciousness, impaired memory, disordered orientation and coordination, even coma.1,2 The incidence of overt HE is 30–40% in liver cirrhosis. Minimal HE can be detected only by neuropsychiatric and psychomotor tests.1,2 The incidence of minimal HE is reportedly 20–80% in liver cirrhosis. 4 Hypotheses for explaining the development of HE include the hyperammonemia hypothesis, the gamma-aminobutyric acid ergic neurotransmission theory, and inflammatory mediators, etc. 5 Predisposing factors of HE include electrolyte imbalance, severe portosystemic shunting, high protein intake, gastrointestinal bleeding, infection, previous HE, constipation, and medication, etc. 6
Proton pump inhibitors (PPIs) are commonly prescribed drugs that can effectively manage various acid-related disorders, such as gastroesophageal reflux disease, peptic ulcer, non-variceal upper gastrointestinal bleeding, as well as being used for bleeding prophylaxis in selected users of nonsteroidal anti-inflammatory drugs. 7 PPIs are frequently used in cirrhotic patients. 8 It has been reported that PPIs may effectively decrease the size of ulcers after endoscopic variceal banding in cirrhotic patients. 9 However, numerous recent studies have suggested that there are potential risks in increasing the incidence of clostridium difficile infection and spontaneous bacterial peritonitis.10,11
Recent evidence suggests that gut microbiome alteration is more frequent in cirrhotic patients with HE and that small bowel bacterial overgrowth increases the risk of minimal HE in cirrhosis.12,13 Several studies have suggested that PPI use might be a risk factor for the occurrence of HE in patients with cirrhosis, cirrhosis with ascites, and hepatitis B virus-related acute-on-chronic liver failure.14–16 However, it is uncertain whether cirrhotic users of PPIs are prone to the development of HE during hospitalization.
Herein, we report on the conduct of a case-control study to explore the association between the use of PPIs and the development of HE in liver cirrhosis during hospitalization.
Methods
Study design
The study protocol was approved by the Medical Ethical Committee of the General Hospital of Shenyang Military Area on 29 March 2017 (approval number: k(2017)15). The requirement of informed written consent was waived, as this was a retrospective study. Similarly, trial registration was not necessary, as this was not a prospective randomized controlled trial. We reviewed the medical records from all patients with a diagnosis of liver cirrhosis consecutively admitted to our hospital between January 2011–June 2014. Some patients had been involved in our previous studies.17–19 A diagnosis of liver cirrhosis was based on the history of liver disease, laboratory tests, clinical manifestations, abdominal imaging, and liver biopsy, if available. Exclusion criteria were as follows: (a) patients with malignancy; (b) patients with HE at admission; (c) unavailability of medical records; and (d) inability to calculate the Child-Pugh score to assess the severity of cirrhosis.
Case and control groups
Patients who developed a new episode of HE during hospitalization were included in the case group; and patients who did not present with HE at admission nor develop any new episode of HE during hospitalization were included in the control group. Case and control groups were matched on a ratio of 1:1 according to three major variables: age (±2 years), gender, and Child-Pugh score (±1).
Data collection
The data we collected included age, gender, Child-Pugh scores, etiology of liver cirrhosis, ascites, acute upper gastrointestinal bleeding (AUGIB), infection, regular laboratory data, PPI use, dose of PPIs, in-hospital death, and causes of death.
Definitions
AUGIB was defined as a new episode of haematemesis or melena within five days before admission. 17 Ascites was evaluated according to the current consensus and guidelines. 20 Minimal HE was not considered an endpoint of our study.1,2 Overt HE was evaluated according to the West Haven criteria.1,2 PPI users were defined as the patients who used PPIs during hospitalization. Defined daily dose (DDD) was defined as the average dose per day for a drug used due to its primary indication in adults. 21 Cumulative DDD (cDDD) of PPIs was defined as the sum of dispensed DDD regardless of type of PPIs before the development of HE and during hospitalization in the case group or during the whole hospitalization in the control group. Infection was defined as bacterial peritonitis, pneumonia, urinary tract infection, and/or bacteremia before the development of HE and during hospitalization in the case group or during the whole hospitalization in the control group. The causes of death were assessed according to the medical records. They were mainly divided into liver-related and other causes.
Statistical analysis
Statistical analyses were performed by using SPSS Statistics version 21.0.0 (International Business Machines Corp., USA). Categorical variables (i.e. gender, etiology of liver diseases, complications of liver cirrhosis, PPI use, and in-hospital death) were presented as frequencies (percentage), and were compared between case and control groups by using the Pearson’s chi-square test or Fisher exact test. Continuous variables (i.e. age, Child-Pugh score, hemoglobin, white blood cell, platelet, total bilirubin, direct bilirubin, albumin, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, gamma-glutamyl transpeptidase (GGT), creatinine, blood urea nitrogen (BUN), potassium, international normalized ratio (INR), sodium, ammonia, and cDDD of PPIs) were expressed as mean ± standard deviation or median (range), and were compared between case and control groups by using the independent sample t-test or Wilcoxon signed-rank test. A two-sided p value of < 0.05 was considered to be statistically significant. Statistically significant variables were further enrolled into the multivariate logistic regression analyses. Notably, considering the potential co-linearity between PPI use and cDDD of PPIs, only PPI use was included in the multivariate analysis. Then, the independent risk factors that were significantly associated with HE in the multivariate analyses were obtained. Meanwhile, odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for each variable. Since AUGIB and infections are known precipitants of HE, 6 they might influence the statistical results. Additionally, the patients who died during hospitalization might use larger doses of PPIs, so in-hospital death was also considered as a potential confounding factor for our analysis. Therefore, in both case and control groups, the patients with AUGIB, infections, or in-hospital death were excluded from our specific subgroup analyses. We further performed a subgroup analysis after matching model for end-stage liver disease (MELD) score (±1) between the two groups.
Results
Overall analysis
A total of 2445 cirrhotic patients without malignancy were admitted to our hospital between January 2011–June 2014. Of these, 128 patients who developed HE after admission were selected as cases and 128 patients without HE at or after admission were selected as controls (Figure 1). Among them, 102 and 55 patients used PPIs in case and control groups, respectively. Indications of PPIs were clear in 60 (60/102, 58.8%) and 27 (27/55, 49.1%) patients in case and control groups, respectively (p = 0.313); on the contrary, indications of PPIs were unclear in 42 (42/102, 41.2%) and 28 (28/55, 50.9%) patients in case and control groups, respectively (Supplementary Material Table 1). In addition, indications of hospital admission in case and control groups are summarized in Supplementary Material Table 2.
Flow chart of patient selection. HE: hepatic encephalopathy.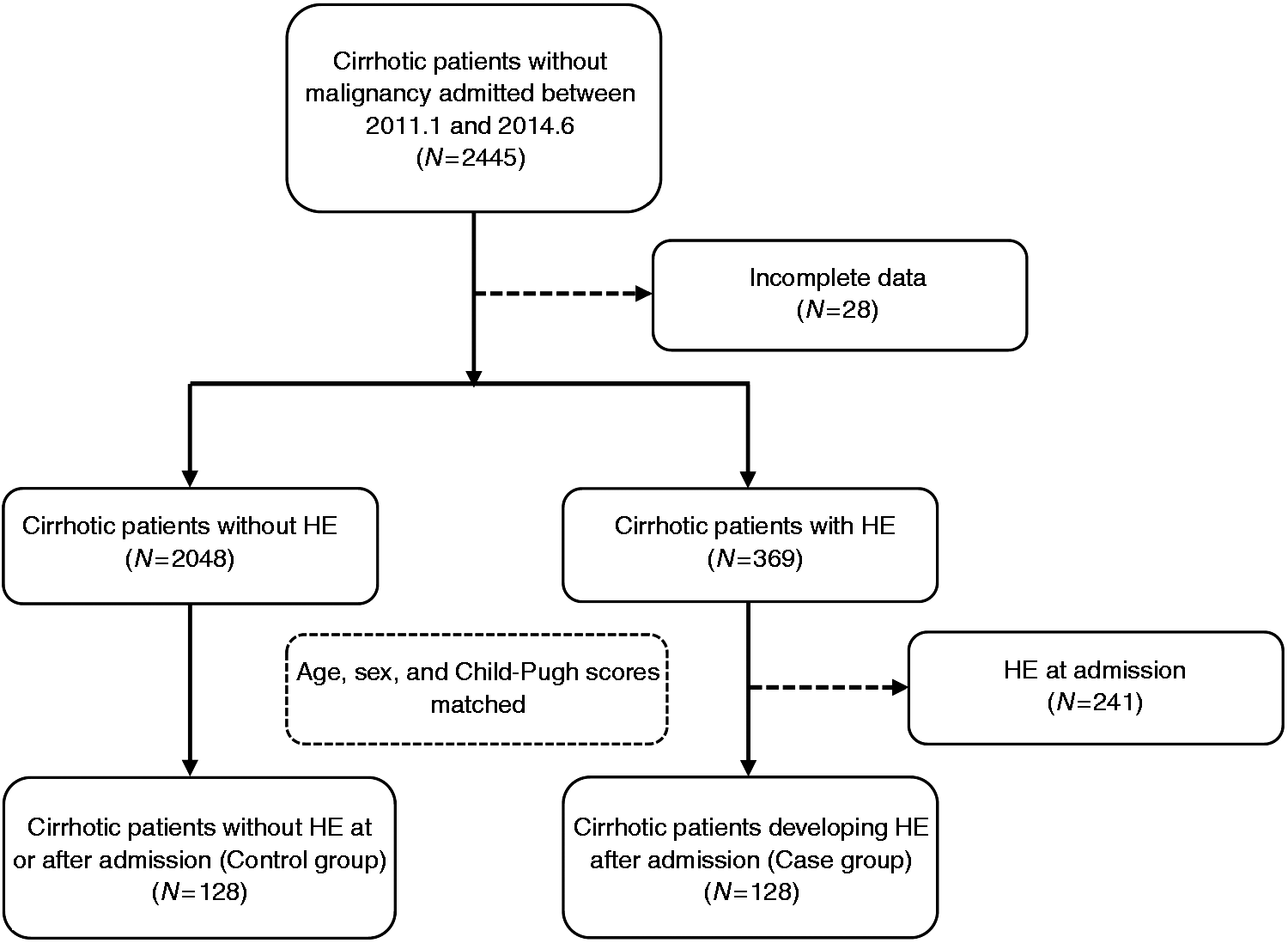
Characteristics between case and control groups.
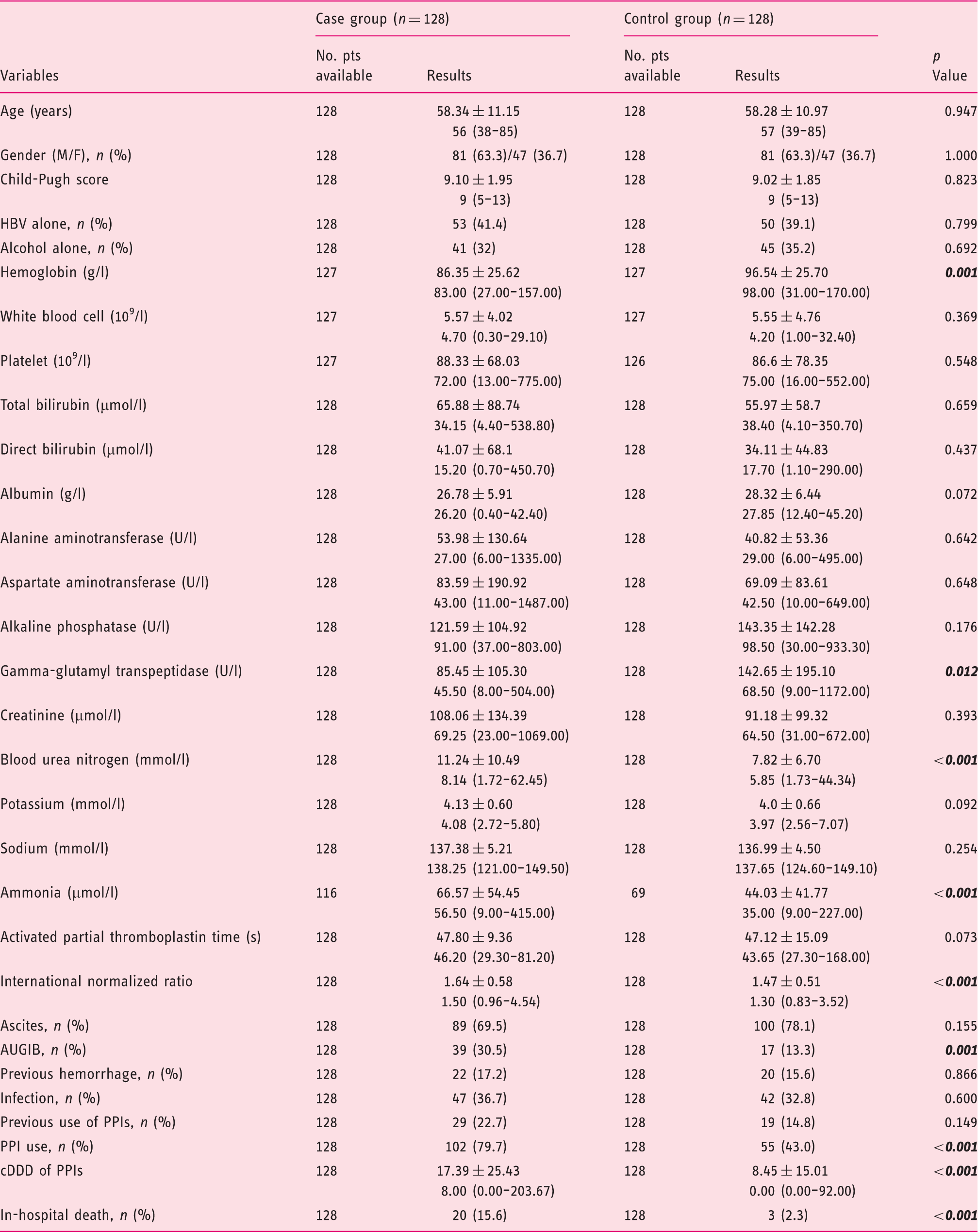
AUGIB: acute upper gastrointestinal bleeding; cDDD: cumulative defined daily dose; HBV: hepatitis B virus; PPI: proton pump inhibitor; Pts: patients; SD: standard deviation. Bold italic p values indicate statistically significant.
Results are presented as mean ± SD, median (range) or n (%).
Multivariate analysis of predictive factors of hepatic encephalopathy.
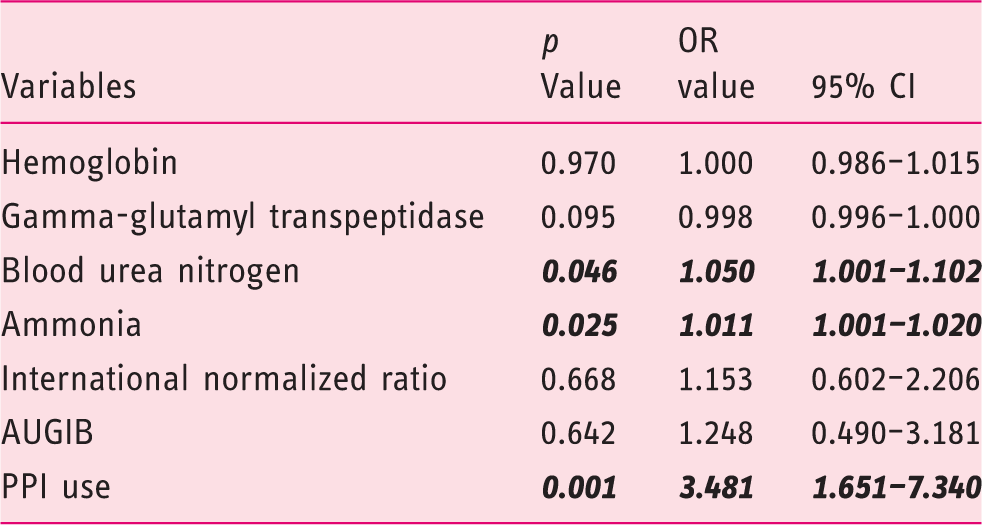
AUGIB: acute upper gastrointestinal bleeding; CI: confidence interval; OR: odds ratio; PPI: proton pump inhibitor. Bold italic values indicate statistically significant.
The case group had a significantly higher in-hospital mortality than control group (15.6% vs 2.3%, p < 0.001). Causes of death are summarized in Supplementary Material Table 3.
Subgroup analysis after excluding patients with AUGIB
We performed a subgroup analysis specifically excluding patients with AUGIB, in which 78 patients with HE and 78 patients without HE were selected as case and control groups, respectively.
Compared with the control group, the case group had significantly higher INR, ammonia level, and cDDD of PPIs and higher proportion of PPI use (Supplementary Material Table 4).
Multivariate analysis showed that ammonia level (OR = 1.014, 95% CI = 1.002–1.026, p = 0.026) and PPI use (OR = 4.573, 95% CI = 1.991–10.504, p < 0.001) were independently associated with the development of HE during hospitalization (Supplementary Material Table 5).
Subgroup analysis after excluding patients who died during hospitalization
We performed a subgroup analysis specifically excluding patients who died during hospitalization, in which 106 patients with HE and 106 patients without HE were selected as case and control groups, respectively.
Compared with the control group, the case group had significantly lower hemoglobin concentration and serum GGT, higher INR, ammonia level, BUN, and cDDD of PPIs, and higher proportions of AUGIB and PPI use (Supplementary Material Table 6).
Multivariate analysis revealed that serum GGT (OR = 0.997, 95% CI = 0.994–1.000, p = 0.048), ammonia level (OR = 1.013, 95% CI = 1.002–1.025, p = 0.016), and PPI use (OR = 4.983, 95% CI = 2.089–11.884, p < 0.001) were independently associated with the development of HE during hospitalization (Supplementary Material Table 7).
Subgroup analysis after excluding patients with infection
We performed a subgroup analysis specifically excluding patients with infection, in which 57 patients with HE and 57 patients without HE were selected as case and control groups, respectively.
Compared with the control group, the case group had significantly lower hemoglobin concentration and serum GGT, higher INR, ammonia level, BUN, and cDDD of PPIs, and higher proportions of AUGIB and PPI use (Supplementary Material Table 8).
Multivariate analysis revealed that ammonia level (OR = 1.024, 95% CI = 1.006–1.043, p = 0.007) and PPI use (OR = 4.530, 95% CI = 1.270–16.162, p = 0.020) were independently associated with the development of HE during hospitalization (Supplementary Material Table 9).
Subgroup analysis after matching MELD score (±1)
We performed a subgroup analysis specifically after matching MELD score (±1), in which 83 patients with HE and 83 patients without HE were selected as case and control groups, respectively.
Compared with the control group, the case group had significantly higher INR, ammonia level, cDDD of PPIs, and higher proportion of AUGIB and PPI use (Supplementary Material Table 10).
Multivariate analysis showed that PPI use (OR =4.327, 95% CI = 1.681–11.134, p = 0.002) was independently associated with the development of HE during hospitalization (Supplementary Material Table 11).
Discussion
By systematically reviewing the previously published papers regarding association between PPIs and HE (Appendix 1), we identified three retrospective studies (Supplementary Material Figure 1).14–16 We reviewed the characteristics and quality of included studies and patients (Supplementary Material Table 12). A meta-analysis further supported a positive association between PPI use and development of HE (Supplementary Material Figure 2). In spite of its striking advantages, 22 a number of potential weaknesses of the study by Tsai et al. 14 should be recognized as follows: (a) laboratory data were largely lacking; (b) case and control groups were matched by advanced/decompensated cirrhosis status, but not Child-Pugh or MELD scores, 23 which are the most important methods to evaluate the severity of cirrhosis; (c) only a low OR of <3 for assessing the association between PPIs and development of HE was found, which suggested the existence of residual confounding factors; 24 (d) no subgroup analyses were performed after excluding patients with or without pre-existing risk factors of HE, such as infection and gastrointestinal bleeding, which might be associated with the use of PPIs. Apart from these above-mentioned weaknesses, the study by Dam et al. 16 enrolled only patients with ascites treated with satavaptan or other diuretic drugs with or without paracentesis, which are related to the risk of HE and could increase the risk of bias; and the study by Lin et al. 15 enrolled a relatively small number of patients. Neither of the latter two studies by Dam et al. 16 and Lin et al. 15 calculated the cDDD of PPIs on the occurrence of HE.
By comparison, our study had some unique features. First, the endpoint of our study was the development of HE during hospitalization. Second, the laboratory data, including ammonia level, were comprehensively collected. Third, the cDDD of PPIs was more accurately calculated according to the medical records and hospitalization costs. Fourth, the subgroup analyses were performed to validate the reliability of our findings by excluding the patients with AUGIB, infection, and in-hospital death.
In our study, a high proportion of cirrhotic patients included (61.33%) were treated with PPIs during hospitalization. Besides, the proportion of PPI use was significantly higher in the case group than the control group (79.7% vs 43%, p < 0.001). The cDDD of PPIs was also significantly higher in the case group than the control group (p < 0.001). The overall analysis supported the association of PPI use with the development of HE during hospitalization (OR = 3.481, 95% CI = 1.651–7.340, p = 0.001).
Gastrointestinal bleeding is a known precipitant of HE, 6 and PPI use can be a non-independent variable reflecting the burden of gastrointestinal bleeding in the case group. Infection is another known precipitant of HE, 6 and PPI use may increase the risk of bacterial infection in liver cirrhosis. 10 The patients who die during hospitalization are more severe cases and may use larger doses of PPIs in the absence of any clear indication. Additionally, our study demonstrated a higher proportion of in-hospital death in the HE group. Thus, the inclusion of patients who die during hospitalization may bring the risk of bias into our analysis, especially a false positive association between HE and use of PPIs. In order to determine if PPI use in and of itself, is associated with HE, we performed four subgroup analyses after excluding patients with AUGIB, infection, and in-hospital death and after matching MELD score (±1). All of them suggested that PPI use is an independent variable in terms of risk of HE during hospitalization. We speculate that hospitalization may increase the likelihood of small bowel bacterial colonization (i.e. a more bacterially contaminated environment than a home environment) and adversely change the bacterial flora, all of which could contribute to the development of HE. The addition of PPIs would then accentuate these adverse changes to the microbiome.
The overall and subgroup analyses did not show any significant difference in the proportion of infection between the two groups. This unexpected finding might be explained by the matched Child-Pugh score between case and control groups, which was associated with the risk of infections. 25
The impact of PPIs on the development of HE could be explained by the following mechanisms. First, PPI use is associated with small intestinal bacterial overgrowth in patients with cirrhosis. 26 Meanwhile, the overgrowth of small intestinal bacteria and altered intestinal permeability have been confirmed in patients with cirrhosis and found to be associated with the development of minimal HE.12,27 The modulation of gut flora by prebiotics, probiotics, and synbiotics decreases the risk of minimal HE. 28 By contrast, PPI use increases small intestinal bacterial overgrowth and translocation, thereby increasing the risk of HE. Second, higher ammonia level induces the development of HE by primarily influencing the brain energy metabolism and central nervous system.29,30 Intestinal bacteria in the colon produces ammonia from the glutamine and the catabolism of nitrogenous sources. 31 PPI use alters the gastrointestinal motility and mucosal barrier.32,33 Thus, PPIs may increase the absorption of nitrogenous substances, thereby increasing the risk of HE.
There were some major limitations in our study. First, the patient selection bias and misclassification were unavoidable due to the retrospective nature of our study. Second, minimal HE was not considered in our study due to its sub-clinical features.1,2 Third, PPI use before admissions could not be assessed. Fourth, not all potential precipitating factors for developing HE, such as previous HE or constipation etc., were clearly identified, which might increase the risk of bias.
In conclusion, the prescription of PPIs might increase the likelihood of developing a new episode of HE in cirrhotic patients during hospitalization. Although PPI use may be clinically indicated, depending on circumstance in these patients, our findings also suggest that physicians should be cautious about the use of PPIs in cirrhotic patients. Specifically, they should not be overused or prescribed without a clear indication and it may be that HE associated with PPIs constitutes a form of iatrogenesis. Considering the potential study limitations, our findings are hypothesis generating, supportive of other studies reporting an association of HE and PPIs, but may not be definitive. More prospective studies are needed to explore the association of HE with PPI treatment in cirrhotic patients, especially given the almost ubiquitous use of these drugs.
Supplemental Material
Supplementary Figure 1 -Supplemental material for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis
Supplemental material, Supplementary Figure 1 for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis by Jia Zhu, Xingshun Qi, Haonan Yu, Eric M Yoshida, Nahum Mendez-Sanchez, Xintong Zhang, Ran Wang, Han Deng, Jing Li, Dan Han and Xiaozhong Guo in United European Gastroenterology Journal
Supplemental Material
Supplementary Figure 2 -Supplemental material for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis
Supplemental material, Supplementary Figure 2 for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis by Jia Zhu, Xingshun Qi, Haonan Yu, Eric M Yoshida, Nahum Mendez-Sanchez, Xintong Zhang, Ran Wang, Han Deng, Jing Li, Dan Han and Xiaozhong Guo in United European Gastroenterology Journal
Supplemental Material
Supplementary Tables -Supplemental material for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis
Supplemental material, Supplementary Tables for Association of proton pump inhibitors with the risk of hepatic encephalopathy during hospitalization for liver cirrhosis by Jia Zhu, Xingshun Qi, Haonan Yu, Eric M Yoshida, Nahum Mendez-Sanchez, Xintong Zhang, Ran Wang, Han Deng, Jing Li, Dan Han and Xiaozhong Guo in United European Gastroenterology Journal
Footnotes
Acknowledgements
The following author contributions were made: Jia Zhu: reviewed the literature, wrote the protocol, collected the data, performed the statistical analysis, interpreted the data, and drafted the manuscript. Xingshun Qi: conceived the work, wrote the protocol, performed the statistical analysis, interpreted the data, and drafted the manuscript. Haonan Yu, Xintong Zhang, Ran Wang, Jing Li, Han Deng, and Dan Han: collected and checked the data. Eric M Yoshida, Nahum Mendez-Sanchez, and Xiaozhong Guo: interpreted the data, gave critical comments, and revised the manuscript. All authors have made an intellectual contribution to the manuscript and approved the submission.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
The study protocol was approved by the Medical Ethical Committee of the General Hospital of Shenyang Military Area on 29 March 2017 (approval number: k(2017)15).
Informed consent
The requirement of informed written consent was waived because of a retrospective study.
Appendix 1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
