Abstract
The 48 national member societies and 17 specialist member societies which operate under the umbrella of United European Gastroenterology (UEG) increasingly develop clinical practice guidelines for both national and international implementation. The methodologies and strategies used in these guidelines vary considerably. The UEG Quality of Care Taskforce aimed to provide a framework for quality guidelines in order to assist member societies in the process of developing guidelines, and to provide a tool for readers of guidelines to critically appraise their quality. We outline the steps necessary to begin the guideline development process, how to build working groups, how to search for evidence, how to grade the quality of the evidence, how to reach consensus on statements and how to write the guideline document. We believe that using this framework will increase the potential to produce a high-quality guideline which is transparent, independent, reproducible and implementable.
Introduction
Clinical guidelines are fundamental to assist clinicians and other healthcare professionals optimize the care of people and patients, based on the latest and best available scientific knowledge and, where evidence is scarce, consensus opinion of the experts from the respective field. Sometimes, guidelines are expressed in other formats such as consensus statements, expert recommendations or position papers. Hence, guidelines are not uniform and the process by which guidelines are developed can be very heterogeneous. Factors that influence the quality of the guidelines are the composition of the guideline panel, the methodology of the evidence review, the evidence and recommendation rating, the decision-making and consensus process and, finally, the implementation of the guideline. There are guidelines with a very stringent process, including different guideline committees, systematic literature reviews, PICO (population, intervention, comparator, outcome) questions, Delphi technique and/or consensus conferences, which are all important for objective and unbiased evidence-based recommendations. However, feasibility and also costs are potential obstacles to universal application. Other guidelines are written by only a few experts with a very lean systematic process. A lean guideline process might allow for a faster response and timely publication of practice recommendations when there are new developments on a topic. However, the potential bias of a small expert steering group needs to be considered. A very sensitive topic to deal with is the conflict of interest (COI) of the expert panel. The experts for a particular topic might be involved in clinical trials or research activities that are supported by industry. This COI has the potential to influence the expert’s position with respect to the subject matter being considered or, at the very least, to raise concerns regarding bias. However, excluding these experts from the guideline process may significantly reduce the panel’s expertise and experience. Thus, all aspects of the composition of the guideline panel and the methodology have advantages and disadvantages.
National and international health organizations and medical societies have increasingly issued their own guidelines and, as a result, the quality of the guidelines can vary considerably. In addition, there are often various guidelines on a topic, some of which contain differing, sometimes even contradictory, recommendations. Different recommendations may be due to national circumstances but also due to a bias in the guideline process. Thus, it is important to develop standards for guideline development which can help organizations optimize their guideline process and ensure that recommendations being developed are transparent, unbiased and evidence-based. In 2003, the Guidelines International Network was founded with the aim to develop and implement international consensus for minimum standards for high-quality guidelines (Appendix 1). These standards can help users to better classify the quality of guidelines.
Several national and international organizations have proposed standards for guideline development. These can assist guideline developers to use appropriate methodologies, and similarly assist guideline readers to assess the quality of a guideline. One of the most widely applied tools for this purpose is the Appraisal of Guidelines for REsearch & Evaluation (AGREE II) Instrument (Appendix 1). In addition, several medical societies have proposed standards or guidance for their guideline process (Appendix 2). Accordingly, United European Gastroenterology (UEG) has developed a “guideline for guidelines” document that will discuss the key steps in the guideline process and thereby offer a framework for future UEG-supported guideline projects (Figure 1).
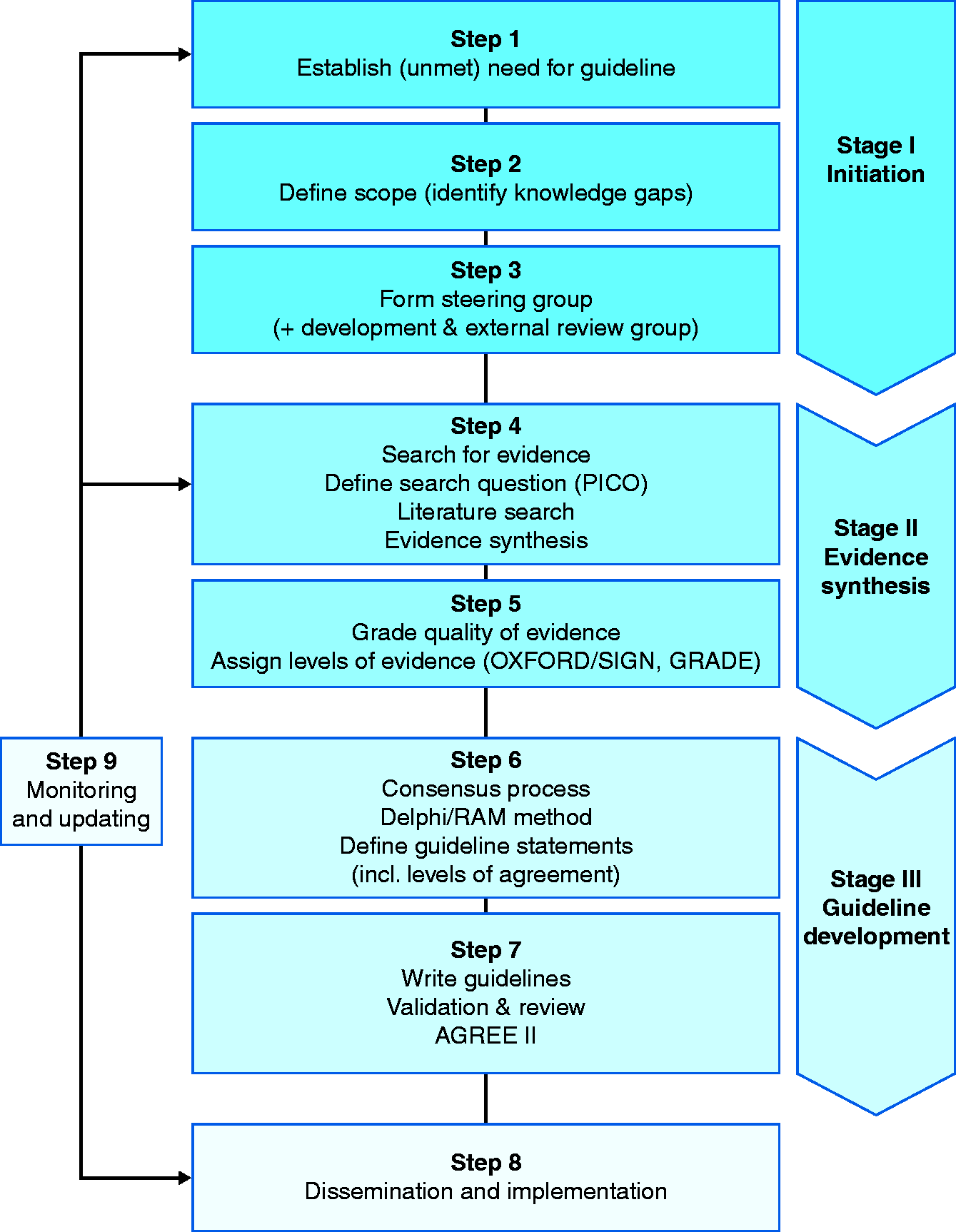
Schematic overview of guideline development process.
This document was developed by the UEG Quality of Care Taskforce (QoC TF), which includes representatives from specialist member societies and the National Societies Committee. A six-member working group was formed, consisting of five QoC TF members and a methodologist (SB). We identified published guideline appraisal tools and standards of practice from different medical subspecialties. These documents were retrieved and critically reviewed by working group members. An appropriate methodology for the UEG framework was developed through consensus discussion at face-to-face and virtual meetings. Following approval of the draft by the working group, the final draft was approved by all QoC TF members.
How to start a guideline process
Identifying the unmet need
Before developing new guidelines, the need for creating a guideline should be carefully assessed. New guidelines should not duplicate an existing document, but rather focus on relevant questions which have not been addressed sufficiently.
1
Similarly, in non-English speaking countries, it may be preferable to translate and adapt an existing guideline rather than develop a non-English guideline de novo. Accordingly, the unmet need for a guideline should arise after a thorough investigation of existing guidelines and the current evidence available. Firstly, the need for new guidelines may arise due to the emergence of new diagnostic or therapeutic modalities.
Defining the scope
After the unmet need for a guideline is established, the scope of the guideline should be defined. This process involves deciding both the breadth and the depth of the document to be developed. It should consider what should and should not be covered in the guideline, with respect to the target audience, patient population, healthcare setting and treatment or clinical outcomes of interest. The scope of the guideline should be based on the gaps identified above, rather than aiming to produce an all-inclusive document.
4
Building working groups and formulating PICO questions
Once the scope of the guidelines is defined, a steering group should be formed to continue the process. 5 Members of the steering group should be chosen based on their pre-eminence in the field, their ability to contribute to the work process and their personal experience as effective team members (Figure 2). In addition to choosing recognized experts in the field, the steering group may wish to consult experts in methodology and research synthesis, health economists, representatives of stakeholders, representatives of potential users and, in some cases, experts in health systems or policymakers. These experts should be consulted at this early stage; however, they do not all need to be included in the steering group. For practical reasons, a steering group should not include more than a handful of participants who are able to effectively deliver a joint decision on key issues and literally steer the work process when problems arise. The steering group should also liaise with a secretarial team that can provide support throughout the work process.
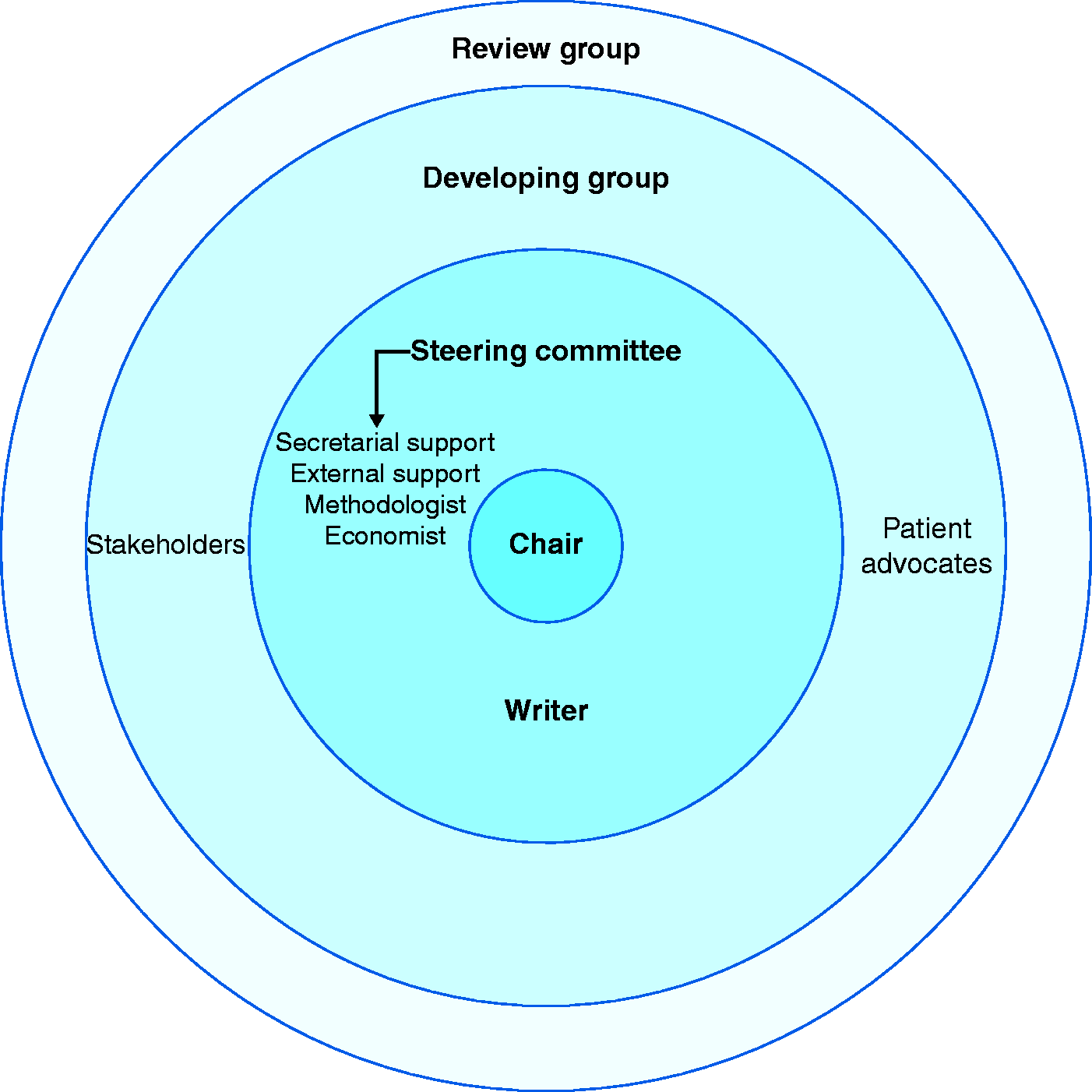
Structure of the guideline working groups.
Once a steering group has been formed, the group should identify and isolate relevant topics within the desired scope. The topics that need to be addressed should be listed and prioritized based on joint decision-making of the steering group. The chosen topics are then elaborated upon, and a list should be made of key issues to be addressed, including knowledge gaps and controversies. It is advisable to select key issues that are adequately focused, of manageable size and are amenable to practical change. The topics should be restricted as much as possible at this stage, as the topics often expand and multiply later in the development process.
How to involve patients in the guideline process

After the steering group has been formed, the scope has been defined and priority topics selected, it is useful to formulate potential recommendations in the framework of PICO questions.
6
,
7
PICO refers to four elements that should be in a question governing a systematic search of the evidence. The PICO formulation defines the
Once PICO questions have been drafted, a broader developing group should be formed, involving a greater number of participants (ideally not exceeding 20). It is useful to draw a list of key external organizations and stakeholders who should be invited to participate in the development group based on their good standing and their achievements in the field.
Finally, an external review group should be formed, made up of experts and stakeholders who were not involved in the work process. The review group’s aim is to provide a peer-based high-quality critique of the drafted guidelines. Ideally, the review group should have a wide scientific, geographical and philosophical reach, being able to illuminate the gaps that remain and solidify the guideline’s statements. From a practical perspective, the review process should be balanced and should not impede the work process. It is worthwhile to consider that while an extensive validation of the guidelines is desirable, a review group that is too large has the potential to encumber the process.
Managing COIs
COIs should be addressed early in the process of establishing the working group. Since numerous COIs can arise during the work process and potentially introduce substantial bias in the final document, we recommend that all individuals provide a declaration of interest (DOI) prior to being allowed to participate in the work process. 11 These forms should include financial, academic and public disclaimers. COIs of a scientific nature are more difficult to identify and address. For example, a working group member who is involved in research which is directly related to a guideline statement may have a viewpoint which could introduce bias. The steering group should review participants’ declarations and decide if any conflicts exist that might preclude a specific participant from commencing the work process. During the work process, COIs may change. 12 Therefore, we recommend reassessing COIs at selected intervals of time during the work process (e.g. every 3–6 months, or at every major meeting). A practical approach could be re-signing a draft of the previously signed declaration. Any changes in a DOI should be re-approved by the steering group. In certain cases, it may be prudent to consult an unbiased, certified external consultant. All declared interests must be reported in the final guideline document. 13
It is a matter of judgement as to what actually constitutes a potentially significant COI with regards to industry funding. Being employed by, consulting for, acting as a paid speaker for or owning shares in a company with a product related to the guideline should be considered a potential COI. On the other hand, it is our opinion that having received research funds from a company, either directly or indirectly, might not automatically constitute a COI.
Budget and timeline
Budget and sources of funding should be transparent to members of the working groups. Sources of funding should also appear in the final guideline document. Direct funding from industry should be avoided, whether or not they have an explicit commercial interest in the outcome of the guideline. Industry funding fundamentally undermines the quality and objectivity of any guideline and introduces concerns regarding commercial bias, which will be a barrier to publication and dissemination. Funding from institutional or government bodies is acceptable provided that they are not involved with the guideline development or review process.
A timeline and a timetable should be addressed at the beginning and end of every meeting. Establishing a timeline early in the process and maintaining strict adherence to the timetable is of utmost importance. The timetable should be available to all participants. Realistically, a good quality guideline will take at least 12–36 months to complete (Box 2). 18
How to start the guideline process

How to search for evidence
Framing the question
Although preliminary questions in the form of PICOs may have already been drafted when deciding the guideline’s scope, these questions must be clearly defined, agreed upon and finalized prior to commencing the literature search (Figure 3). Several frameworks, besides PICO, have been described; however, PICO remains the most common and easy to apply.
18
For example, the ECLIPSE framework addresses questions related to health policy and management, rather than clinical questions.19–21 ECLIPSE is structured according to the mnemonic:
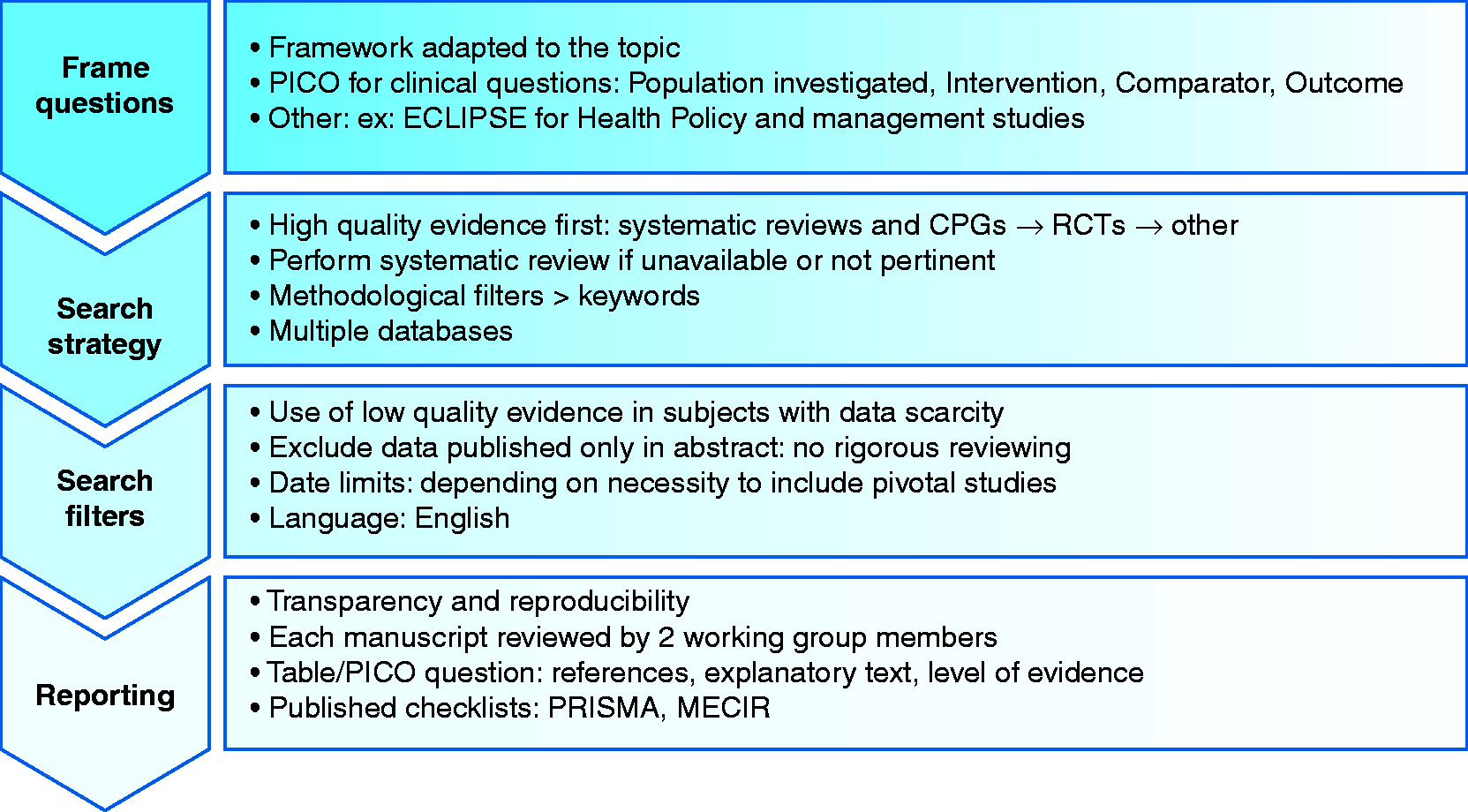
Searching for evidence.
Search protocol and strategy
The quality of a literature search directly impacts the quality of the guideline. Evidence must be unbiased and comprehensive. 22 Before a search is performed, a search protocol must be agreed upon. The aim of this protocol is to outline what strategy will be used in order to answer each question in the framework; which inclusion and exclusion criteria will be applied; which databases will be utilized; and how the results will be assessed and documented.
The most commonly used search protocol (favoured by National Institutes of Health (NIH), National Institute for Health and Clinical Excellence, World Health Organization (WHO) and UEG member societies; Appendices 1 and 2) is a step-by-step iterative approach that allows the working group to first consider high-quality evidence from systematic reviews and meta-analyses, then randomized controlled trials (RCTs) and then all other levels of evidence. 4 , 23 , 24
It may even be necessary to perform a new systematic review and evidence synthesis if there are no high-quality reviews recently published. In order to decide whether to update an existing systematic review or commission a new document, we recommend the stepwise approach developed by the WHO. This involves developing key questions, identifying a relevant systematic review, assessing its quality and determining whether it is sufficiently up to date. 23
At this stage, a search for primary sources, which may not appear in the high-quality review, should be performed. It is important to utilize methodological filters rather than simply searching keywords or MeSH (medical subheading) terms as free text. Both EMBASE and TRIP allow searching according to the PICO format. Other databases such as PubMed and Medline have advanced search options and are also acceptable. It is preferable to use more than one database, and it may be useful to seek the assistance of a medical information specialist.
Search filters
At this stage it is not advisable to limit the search by excluding studies with a low quality of evidence. This could lead to the exclusion of topics where a paucity of evidence exists. Similarly, limiting the dates in the search may lead to the exclusion of pivotal studies. In some situations, however, such as when a guideline is being updated or when addressing a new development, limiting publications by date may be useful. In many cases, for practical reasons, the search language is restricted to English, because a reliable translation is not readily available. This could introduce publication bias. If a foreign language guideline is being produced and non-English local data are available, then a language restriction should not be applied. Including data published in abstract form only should be avoided, as these data are often preliminary and not subject to rigorous peer review.
Documenting and reporting
All stages of the search should be documented to ensure transparency and reproducibility, and to enable future guideline updates. We recommend that each manuscript is reviewed by at least two working group members who both agree to inclusion of the publication. The final search results should be summarized in an output document and include the key references together with a short explanatory text and level of evidence (see
How to search for evidence

How to grade the quality of evidence
There are several systems available to grade the quality of evidence; however, we recommend adopting one of the three most commonly encountered: the Oxford system, Scottish Intercollegiate Guidelines Network (SIGN) and the Grading of Recommendations Assessment, Development and Evaluation (GRADE) 25 ,28–30 (Appendix 1). Both the Oxford and SIGN classification systems focus on the quality of the individual studies, while the GRADE approach views the available evidence from the outcome perspective (Table 1).
Systems to grade the quality of evidence.
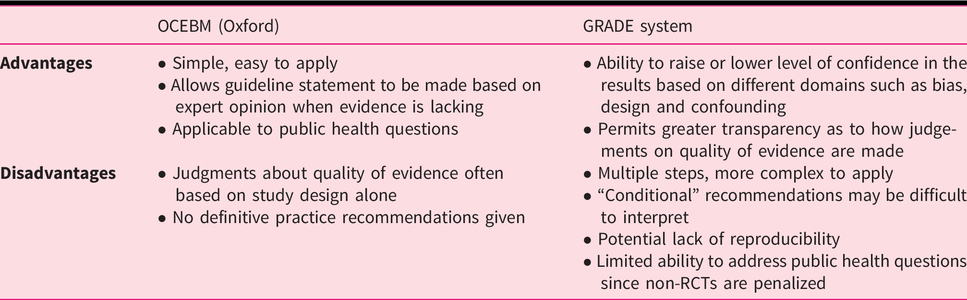
The Oxford and SIGN grading systems
The Oxford Centre for Evidence-Based Medicine (OCEBM) appraises data according to five levels of evidence (Levels 1–5). This system may be applied to different types of studies, with seven different outcome measures, including disease prevalence, accuracy of diagnostic tests, disease prognosis, therapeutic effects, harms of screening and usefulness of screening. The level of evidence assigned depends upon which outcome is measured in the particular study.
The GRADE system
GRADE is unique in that it allows the level of evidence to be downgraded, such as in the case of limitations in study design (risk of bias), inconsistencies between studies, indirectness, imprecision or publication bias.
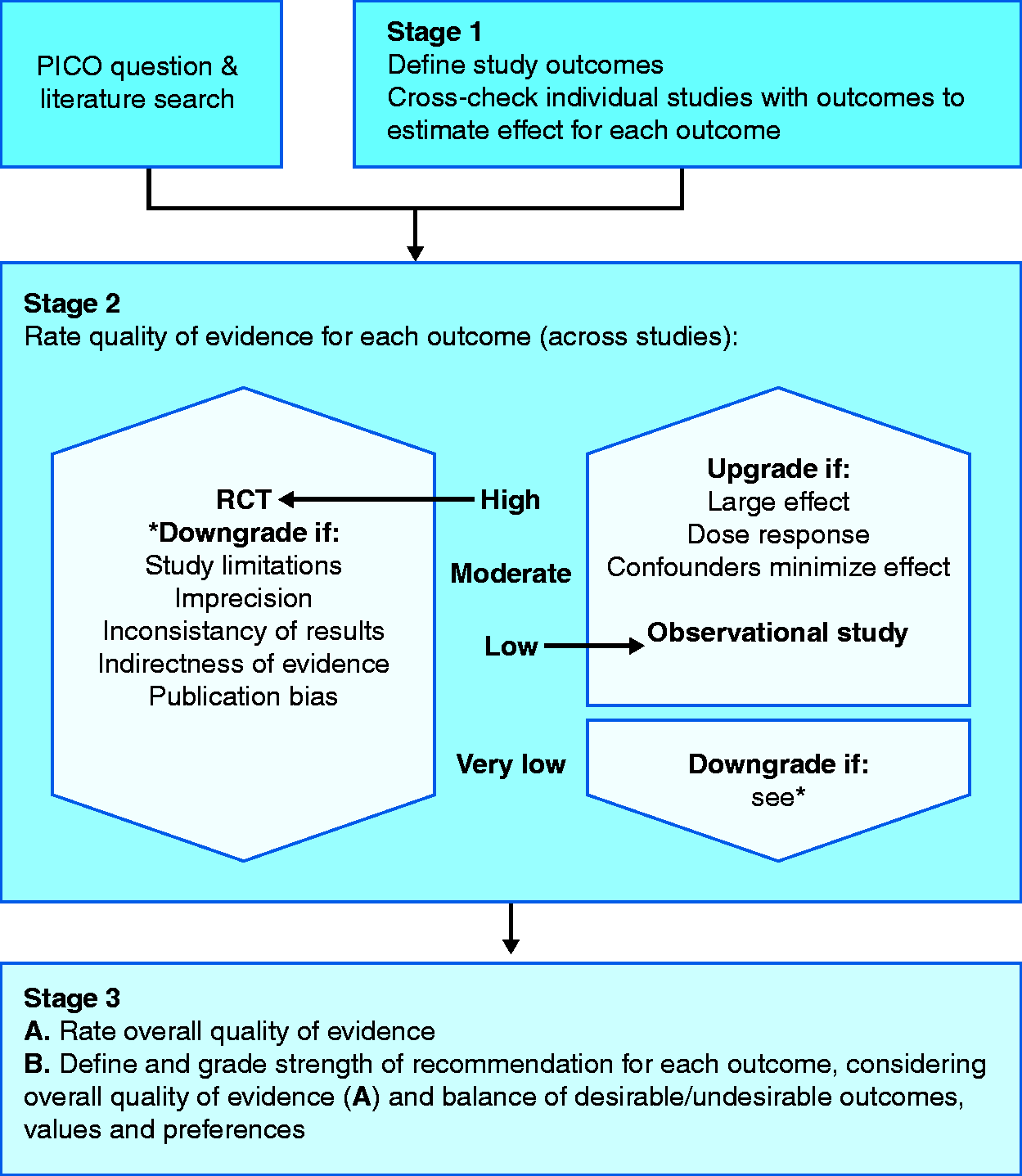
GRADE approach to assessing level of evidence.
How to evaluate the evidence for guidelines using consensus methods
After an evidence table has been constructed for a particular PICO question, a draft recommendation can be written. Once all draft recommendations have been completed, they should be discussed by the panel of participants as a whole. The participants should be presented with the evidence tables (indicating the number, type, quality and grade of evidence of studies for each review question; see
The most common formal consensus method in use is the Delphi method (Figure 5). The Delphi method is a stepwise process in which an anonymous questionnaire is circulated among the developing group. The results are reported back to the group, after which the questionnaire is again circulated. This process is repeated until there is a convergence of opinion (to a predetermined level of agreement) or until no further changes in the replies are elicited. An advantage of this method is that it allows a large number of participants. 33 The RAND/UCLA appropriateness method (RAM) is a modified two-round Delphi process consisting of two independent groups: a core panel and an expert panel.34 The core panel oversees the consensus process and provides synthesized literature evidence to ensure that the expert panel has all pertinent information available to guide evidence-based decision-making. Regardless of which method is used, it is important that the process is defined in advance and should contain at least two rounds of voting to establish whether agreement can be reached. 34 A second voting round allows for revision of statements and recommendations with a view to achieving consensus. Ideally, the final round of voting should occur at a face-to-face meeting of all participants to allow debate and discussion where a consensus on draft statements has been difficult to achieve.
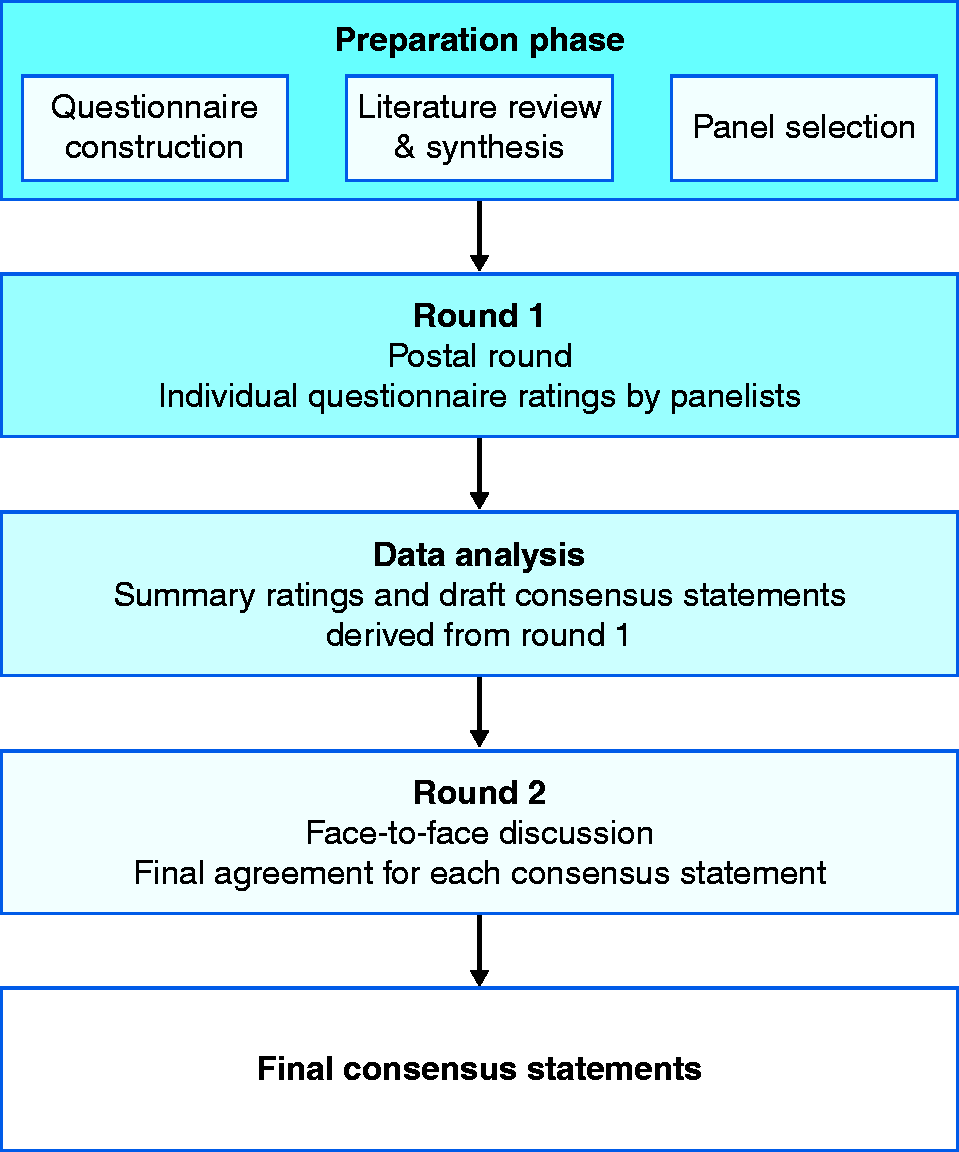
Methodology for reaching consensus.
The level of agreement required for a statement or recommendation (generally expressed as a mean rating or percentage of agreement) should be decided in advance, and this threshold should not be modified during the process.
How to evaluate evidence and reach a consensus

How to write the guidelines
Writing draft recommendations
Recommendations should be drafted that relate directly to the PICO questions formulated and the outcome of the literature search that was performed. The language used should ideally follow a uniform format and refer to the population, the intervention and the context. Recommendations should also each contain an indication of the strength of the recommendation and the quality of evidence that underpin them, followed by a summary of evidence. Domain 4 of the AGREE II instrument provides an outline for how to clearly present summary recommendations (Appendix 1).
Constructing a guideline statement
Guideline statements are generally written in English, but can also be drafted in the primary language for the country or geographical area to which they pertain. Whatever the language used, the statement should be drafted in the simplest language possible, while ensuring clarity of meaning. Statements should generally adopt a broad health-system perspective, based on the needs of the patient population on a national and/or European level. This perspective is key when determining benefit, harm and costs of any intervention. Recommendations should clearly state the assumed values and preferences of the population for which it is intended. Recommendations should be concise, unambiguous and actionable, with consistent language across the guideline. It is better to keep sentences short and avoid long statements with multiple elements as they are more likely to give rise to lack of agreement, in which case it may be difficult to define which element is generating the difference in opinion. Guidelines should use language such as “
The participant who drafts the guideline should provide justification for the recommendation and include relevant considerations about implementation strategies, practice implications, monitoring and future research priorities. This can then be included in the supporting text. It should be emphasized in the final guideline manuscript that the recommendations/statements should not read in isolation but considered in the context of the supporting text and evidence. Guidelines should support shared decision-making and allow for flexibility to reflect the need for individualized treatments – that is, it should be recognized that not all strong recommendations based on high-quality evidence will be suitable for every patient.
Final manuscript and publication
There should be a clear outline of who will write the final guideline document. This should contain all of the statements or recommendations that achieved the threshold for agreement in the final voting round. The evidence supporting each statement should also be included in the final manuscript (either in the body of the manuscript or as supplementary material that is readily accessible for anyone who has access to the final published manuscript). The supporting text is important as it allows a context to be offered for each recommendation and allows potential caveats to any recommendation to be outlined.
The structure of the guideline document should ideally also be decided in advance and may comprise an executive summary, a main body and one or more appendices. The executive summary is a concise summary of the recommendations, their strength and evidence quality. The background to the recommendations should include the overall objective of the guidelines, the target population and target users and the main clinical questions to be addressed. The main text of the guideline should include a table of contents, background, objectives, development process of the guideline, evidence summary, recommendations, resource and service implications and recommendations for future research. All participants, roles and affiliations should be listed, with their COIs and how these were managed. The systematic review(s), outcome ratings, summaries of findings, GRADE evidence profiles, evidence-to-decision frameworks and tables, and any other relevant documentation can be supplied in appendices and/or may be published electronically as supplementary material, as long as these remain readily accessible.
Where appropriate, guideline panels should actively consider including a specific section in the guideline manuscript addressing how the recommendations might be implemented in countries or populations with resource limitations, which will serve as a barrier to comprehensive guideline implementation. Ideally, implementation strategies should be proposed, and specific tools and/or strategies should be suggested to assess implementation.
Prior to submission for publication, the AGREE-II appraisal instrument should be applied to ensure the guideline meets international quality standards (Appendix 1). COI declarations should also be shown clearly. A timeframe for updating the guideline should be agreed and outlined in the final published manuscript. It is useful to consider adding a disclaimer in any published format of a guideline reminding readers that a guideline does not replace the need for clinical decision-making for each individual patient and cannot always take account of variations in patient population, facilities and resource availability, thus avoiding the inappropriate use of the guideline in medico-legal proceedings.
Validation and review
A robust validation process is an integral part of guideline development and may involve consultation with relevant stakeholders, public consultation and external peer review. Completed draft guidelines should be published on an appropriate website with a designated feedback form to facilitate structured feedback from relevant stakeholders. External peer review may also take place during the consultation process and peer review by a panel of expert peer reviewers not involved in the guideline development or voting may serve as a further useful validation. Following submission of comments on the draft guideline, the participants or a panel of coordinators should respond to all feedback and meet to discuss if any changes are required to the guideline. Guidelines should be refined and prioritized prior to final publication, and comments and responses made during consultation should ideally be made available with the final publication of the guideline.
Guideline implementation
Prior to publication, strategies to increase the implementation of a guideline should be considered. Guidelines should be published on an open-access platform or made available for download free of charge to maximize the dissemination of the guideline and ensure it is accessible.
Guideline updates
In the medium- to long-term period following publication, the steering group should convene at predetermined intervals in order to assess the need for a guideline update (e.g. on the side-lines of scientific meetings). The steering group may wish to form a designated working group for this purpose. Literature searches, as described above, should be performed periodically in order to identify new evidence. The importance and relevance of this evidence should be assessed at guideline update meetings. If new evidence substantially impacts or alters the recommendations of the published guideline such that the guideline is no longer valid, then an update is required.
Conclusion
Clinical practice guidelines have the potential to improve the quality of patient care and population health in the regions where they are applied. However, in order to produce a high-quality guideline, it is crucial to adhere to a transparent and rigorous methodology. UEG supports the development of high-quality guidelines, which are independent, reproducible and implementable. Future UEG-funded guidelines will adhere to this framework.
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
