Abstract
Background
Optimizing colonoscopy resources is challenging, and information regarding performing diagnostic quantitative faecal immunochemical test (FIT) in daily clinical practice in primary health care is still limited. This study aimed to assess the sensitivity, specificity, positive predictive value and negative predictive value of varying FIT positivity thresholds on colorectal cancer (CRC) detection in primary health care.
Methods
A retrospective cohort study of 38,675 asymptomatic and symptomatic patients with a FIT (OC-Sensor™) performed between 2012 and 2016 in a primary health-care setting, using a clinical laboratory database of two Spanish areas linked with the National Health System’s Hospital Discharge Records Database. The primary outcome was 2-year CRC incidence.
Results
The mean age of the participants was 63.2 years; 17,792 (46.0%) were male. CRC prevalence was 1.7% (650/38,675). The percentage of patients with a FIT result above the threshold was 20.7% and 14.6% for 10 µg Hb/g faeces and 20 µg Hb/g faeces thresholds, respectively. Sensitivity was 90.5% (95% confidence interval 88.0–92.5%) at a 10 µg Hb/g faeces threshold, and this decreased by 3.1% when a 20 µg Hb/g faeces threshold was used. The negative predictive value for CRC was at least 99.2% in any subgroup analysed. At a 20 µg Hb/g faeces threshold, less than one additional CRC would be missed per 1000 patients investigated, while approximately 1.3 times more colonoscopy examinations were needed to identify an incidence of CRC using the lowest threshold for any situation analysed.
Conclusions
In primary health care, a quantitative FIT threshold should be tailored to colonoscopy capacity and CRC prevalence in specific populations.
Keywords
Introduction
Although colorectal cancer (CRC) screening programmes have been extended widely in Western Europe, most cases of CRC are still diagnosed in symptomatic patients. 1 Unfortunately, the majority of lower gastrointestinal symptoms have a low positive predictive value (PPV) for CRC (3–4%), 2 and several studies included in previous meta-analyses have revealed that a quantitative faecal immunochemical test for haemoglobin detection (FIT) can be used to identify those symptomatic patients with a higher CRC risk. Therefore, this assists primary health care in determining who should be referred for colonoscopy. 3 , 4
The threshold to determine which patients would benefit from further investigation plays a crucial role in any diagnostic strategy for CRC detection, as it may be locally selected on the basis of regional variables such as CRC incidence, colonoscopy capacity and population demographics (sex, age) which could affect FIT diagnostic performance among others. 5 , 6
Furthermore, the National Institute for Health and Care Excellence (NICE) recommends (NICE DG30) FIT to guide referral in primary care for suspected CRC. This would be in people without rectal bleeding who have unexplained symptoms but who do not meet the criteria for a suspected cancer pathway referral using a 10 µg Hb/g faeces threshold regardless of sex or age. 7 However, the main concern for implementation of this strategy is that information regarding FIT diagnostic performance in primary health care is still limited, and large studies are required to validate its use. 8
Since then, some studies have been conducted in this scenario using different FIT platforms.9–12 A recent study revealed that faecal haemoglobin (f-Hb) <10 µg Hb/g faeces, in the absence of iron-deficient anaemia, rectal bleeding, a palpable mass or persistent diarrhoea, identifies patients with an extremely low risk of developing CRC. 9 However, use of f-Hb, regardless of variables which define the clinical spectrum of an individual patient, could lead to missing CRC in some subgroups of patients while at the same time overusing colonoscopy resources in others.
We designed a retrospective study that aimed to assess the diagnostic accuracy of FIT in daily clinical practice in primary health care for CRC diagnosis in two areas of northern Spain between 2012 and 2016.
Methods
We followed the Standards for Reporting of Diagnostic Accuracy Group initiative checklist for diagnostic tests and the Strengthening the Reporting of Observational studies in Epidemiology statement to conduct and report our study. 13 , 14
Participants and setting
This population-based retrospective cohort study included asymptomatic and symptomatic patients aged ≥18 years from two northern Spanish health areas (Ourense and San Sebastián) who consulted their general practitioners between 1 January 2012 and 31 December 2016, who requested a FIT as part of their medical treatment.
Data on their consultations and laboratory results were linked to the Spanish Health System’s Hospital Discharge Records Database (CMBD). This database receives information about hospital discharges from approximately 98% of public hospitals in Spain and, since 2005, has gradually covered private hospitals. This information includes diagnoses made during hospital admission, which are mainly coded based on the Spanish version of the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) and 10th revision. Patients with a history of CRC in the 2 years prior to FIT determination were excluded.
Index test
The FIT used in both health areas is OC-Sensor™ (Eiken Chemical Co., Tokyo, Japan). Use of this FIT in our community has been reported before. 15 In short, patients were told to collect a faecal sample from one bowel movement without specific diet or medication restrictions, and each sample was processed as previously reported at each regional reference hospital’s laboratory. 16 Estimates of f-Hb were quantitated as µg Hb/g of faeces so that results could be compared across analytical systems. 17
FIT performance was assessed using the thresholds of 10 and 20 µg Hb/g faeces. When a patient had more than one FIT determination in the database, only the former was used. Reasons for FIT request were classified into three groups: (a) opportunistic screening (outside the scope of regional CRC screening programmes), (b) study of symptoms and (c) follow-up of gastrointestinal pathology. An overview of FIT data collection is shown in Supplemental Figure S1.
Outcome variables
The main outcome was 2-year CRC incidence. Patients were included as having CRC if a diagnosis was recorded in the CMBD, and latency was defined as the time elapsed between FIT determination and the earliest record of CRC assigned in the CMBD. CRC was recorded as ‘right-sided’ when located proximal to the splenic flexure.
Data analysis
Differences between both cohorts were evaluated using chi-square and Mann–Whitney U-tests for qualitative and quantitative variables, respectively. Discriminatory ability for detecting CRC was assessed using the receiver operating characteristics curve and its area under the curve (AUC). Sensitivity, specificity, PPV, negative predictive value (NPV), positive and negative likelihood ratio, diagnostic odds ratio and their 95% confidence interval (CI) were calculated using 10 and 20 µg Hb/g faeces as thresholds. Subgroup analysis was conducted to assess differences between centre, age category (<50, 50–69 and >69 years), sex, presence of symptoms and CRC location due to their potential association with FIT accuracy. The number of colonoscopy examinations needed to detect a subject with CRC (number necessary to scope) and the number of missed CRC per 1000 patients evaluated were calculated for each subgroup. A p-value of <0.05 was deemed statistically significant. Statistical analysis was performed using SPSS v15.0 (SPSS, Inc., Chicago, IL).
Results
Participants
In the study period, 54,327 FIT samples were submitted from primary health care to San Sebastián’s and Ourense’s referral laboratories. Reasons for exclusion are detailed in the study population flow chart (Figure 1). We analysed data from 38,675 participants. Their median age was 65.2 years (interquartile range 25.1), and 54.0% (20,883/38,675) were female. Patient cohort characteristics are provided in Table 1.
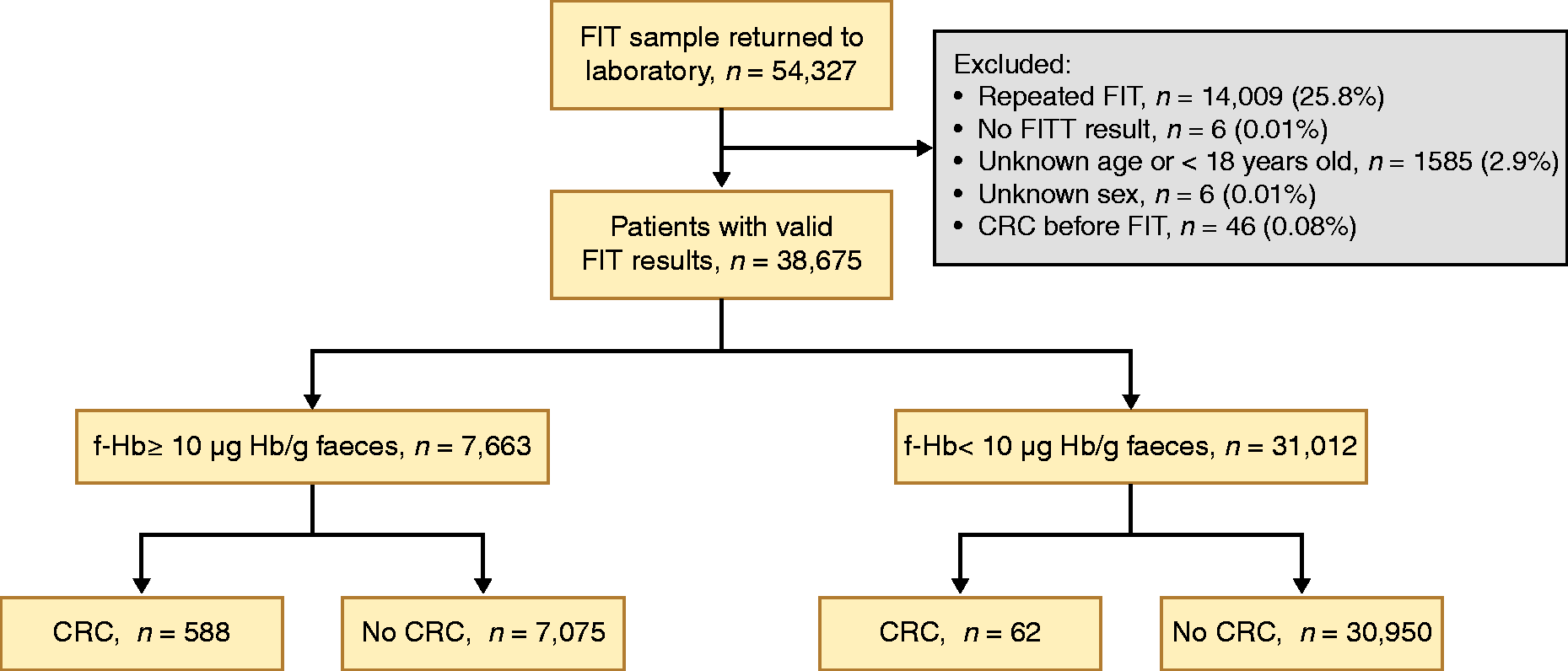
Study population flow chart. CRC: colorectal cancer; f-Hb: faecal haemoglobin concentration; FIT: faecal immunochemical test (threshold 10 µg Hb/g faeces).
Characteristics of the individuals included in the analysis.
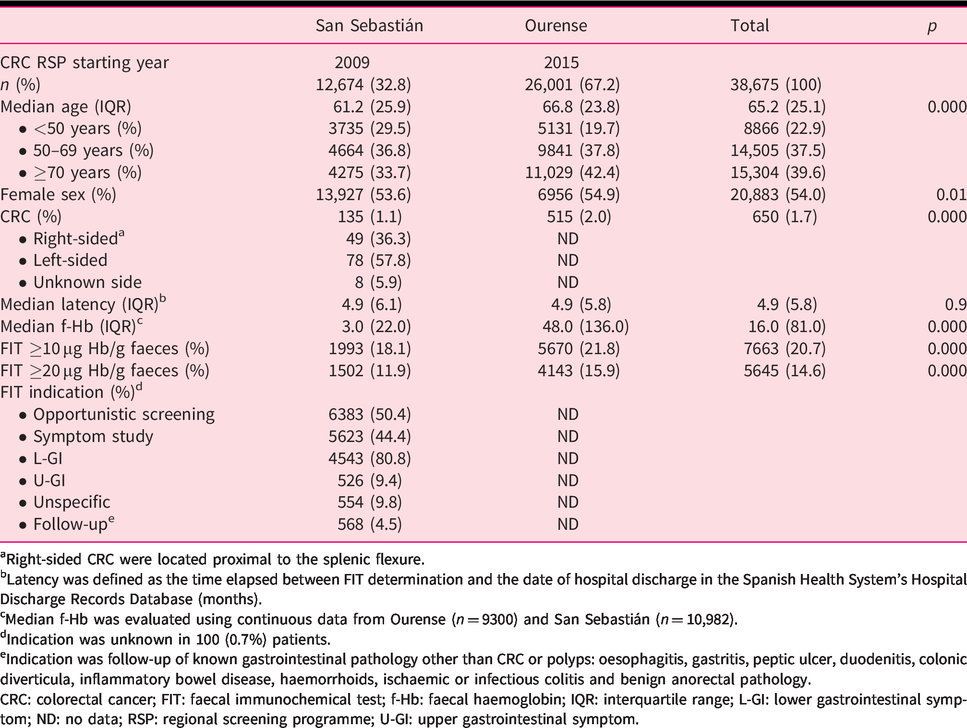
aRight-sided CRC were located proximal to the splenic flexure.
bLatency was defined as the time elapsed between FIT determination and the date of hospital discharge in the Spanish Health System’s Hospital Discharge Records Database (months).
cMedian f-Hb was evaluated using continuous data from Ourense (n = 9300) and San Sebastián (n = 10,982).
dIndication was unknown in 100 (0.7%) patients.
eIndication was follow-up of known gastrointestinal pathology other than CRC or polyps: oesophagitis, gastritis, peptic ulcer, duodenitis, colonic diverticula, inflammatory bowel disease, haemorrhoids, ischaemic or infectious colitis and benign anorectal pathology.
CRC: colorectal cancer; FIT: faecal immunochemical test; f-Hb: faecal haemoglobin; IQR: interquartile range; L-GI: lower gastrointestinal symptom; ND: no data; RSP: regional screening programme; U-GI: upper gastrointestinal symptom.
CRC
CRC was detected in 650 (1.7%) patients, with differences in prevalence regarding health area (San Sebastián = 1.1%, Ourense = 2.0%; p<0.001), age group (<50 years old = 0.3%, 50–69 years old = 1.4%, >69 years old = 2.8%; p<0.001) and sex (female =1.1%, male = 2.3%; p<0.001). Information regarding FIT indication and CRC location was only available for San Sebastián. The prevalence of CRC according to FIT indication was 1.4% in patients with gastrointestinal symptoms, 0.8% in opportunistic CRC screening and 0.4% in patients with gastrointestinal tract disorders different from CRC (p = 0.02). The rate of right-sided CRC was more common in females (females 49.1%, males 31.1%; p = 0.04). The delay between FIT determination date and the first recorded date in CMBD was significantly higher in FIT negative patients (<10 µg Hb/g faeces = 10.6 months, ≥10 µg Hb/g faeces = 5.9 months; p<0.001) and inversely correlated with the amount of f-Hb detected (r = –0.2; p<0.001).
Percentage of FIT above threshold
For the whole cohort, the percentage of patients with FIT result above the threshold was 20.7% and 14.6% at 10 and 20 µg Hb/g faeces thresholds, respectively, with statistically significant differences (p<0.01) according to area, sex and CRC location (Tables 2 and 3).
Diagnostic accuracy of a FIT for colorectal cancer by category and threshold.
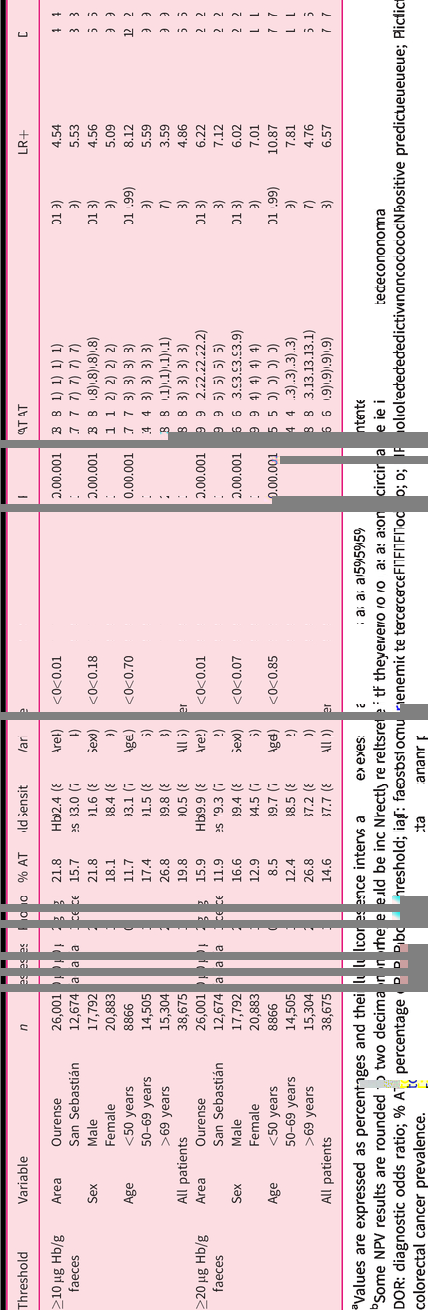
aValues are expressed as percentages and their 95% confidence interval.
bSome NPV results are rounded to two decimals, as they could be incorrectly interpreted if they were rounded to one decimal (100.0).
DOR: diagnostic odds ratio; % AT: percentage of FIT above threshold; FIT: faecal immunochemical test; LR: likelihood ratio; NPV: negative predictive value; PPV: positive predictive value; P: colorectal cancer prevalence.
Diagnostic accuracy of a FIT for colorectal cancer by indication, location and threshold in San Sebastián.
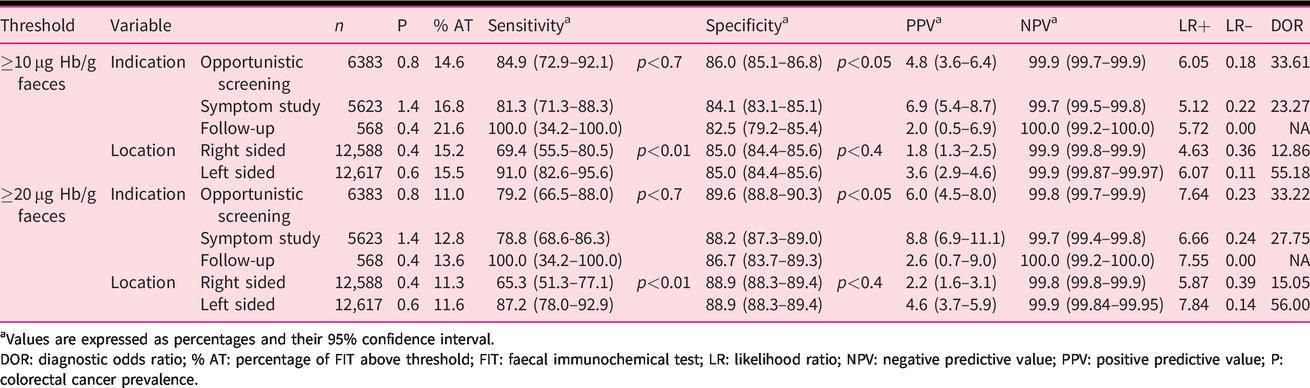
aValues are expressed as percentages and their 95% confidence interval.
DOR: diagnostic odds ratio; % AT: percentage of FIT above threshold; FIT: faecal immunochemical test; LR: likelihood ratio; NPV: negative predictive value; PPV: positive predictive value; P: colorectal cancer prevalence.
Diagnostic accuracy for CRC detection
The AUC of FIT for CRC detection was 0.89 (95% CI 0.88–0.91), as shown in Supplemental Figure S2. At the 10 µg Hb/g faeces threshold, the sensitivity and specificity for detecting CRC were 90.5% (95% CI 88.0–92.5%) and 81.4% (95% CI 81.0–81.8%), respectively (Table 2). In contrast, at the 20 µg Hb/g faeces threshold, sensitivity decreased by 3.1% and specificity increased by 6.5%. The PPV for CRC increased from 7.7% to 10.1% without changes in the NPV.
Effect of demographic variables on FIT performance characteristics
FIT sensitivity was not significantly different between sex or age category (p > 0.1). However, there were differences in specificity between health area, sex and age category (p < 0.001). The PPV for CRC detection in the different subgroups ranged from 2.6% to 9.9%. However, the NPV for CRC was at least 99.6% for all subgroups analysed (Table 2).
Influence of symptoms and CRC location
Table 3 shows FIT characteristics based on CRC location and reason for FIT request in the San Sebastián cohort. These are detailed in Table 4. Sensitivity did not change significantly between symptomatic and asymptomatic patients (p = 0.7) despite differences in FIT positivity in those groups. However, specificity was significantly higher in opportunistic screening setting at both 10 and 20 µg Hb/g faeces thresholds (p<0.05). Conversely, FIT specificity was similar regardless of CRC location (p = 0.4), while sensitivity decreased significantly in right-sided CRC at 10 and 20 µg Hb/g faeces thresholds (p < 0.05).
Diagnostic accuracy of a FIT for colorectal cancer by indication and threshold in San Sebastián.
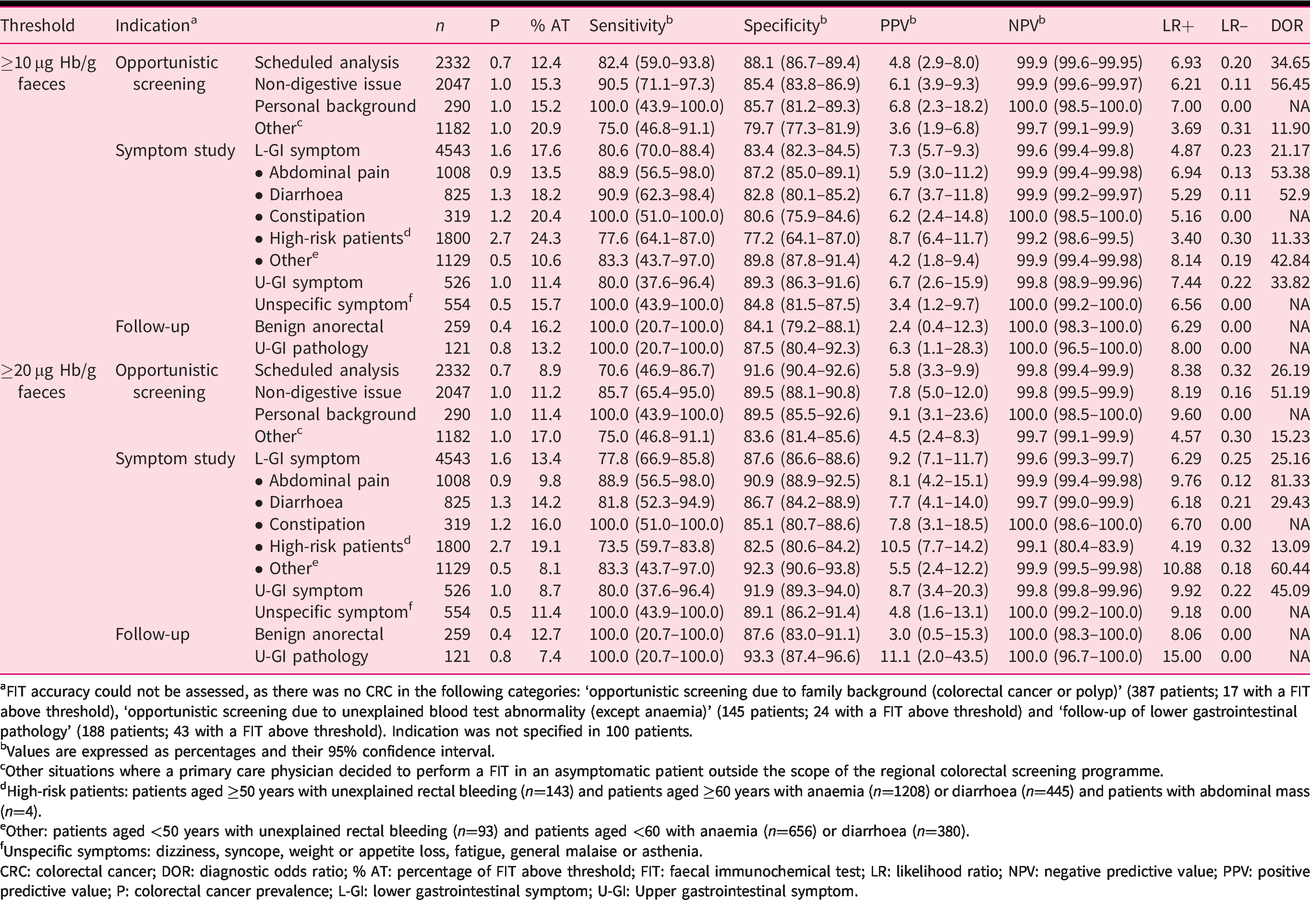
aFIT accuracy could not be assessed, as there was no CRC in the following categories: ‘opportunistic screening due to family background (colorectal cancer or polyp)’ (387 patients; 17 with a FIT above threshold), ‘opportunistic screening due to unexplained blood test abnormality (except anaemia)’ (145 patients; 24 with a FIT above threshold) and ‘follow-up of lower gastrointestinal pathology’ (188 patients; 43 with a FIT above threshold). Indication was not specified in 100 patients.
bValues are expressed as percentages and their 95% confidence interval.
cOther situations where a primary care physician decided to perform a FIT in an asymptomatic patient outside the scope of the regional colorectal screening programme.
dHigh-risk patients: patients aged ≥50 years with unexplained rectal bleeding (n=143) and patients aged ≥60 years with anaemia (n=1208) or diarrhoea (n=445) and patients with abdominal mass (n=4).
eOther: patients aged <50 years with unexplained rectal bleeding (n=93) and patients aged <60 with anaemia (n=656) or diarrhoea (n=380).
fUnspecific symptoms: dizziness, syncope, weight or appetite loss, fatigue, general malaise or asthenia.
CRC: colorectal cancer; DOR: diagnostic odds ratio; % AT: percentage of FIT above threshold; FIT: faecal immunochemical test; LR: likelihood ratio; NPV: negative predictive value; PPV: positive predictive value; P: colorectal cancer prevalence; L-GI: lower gastrointestinal symptom; U-GI: Upper gastrointestinal symptom.
Effect of threshold on diagnostic yield for CRC
The difference in missed CRC using both thresholds was less than one in 1000 patients evaluated for any subgroup analysed. However, FIT positivity was higher using the lowest threshold (Tables 5 and 6).
Performance of FIT when threshold is increased from 10 µg Hb/g faeces to 20 µg Hb/g faeces by sex and age.
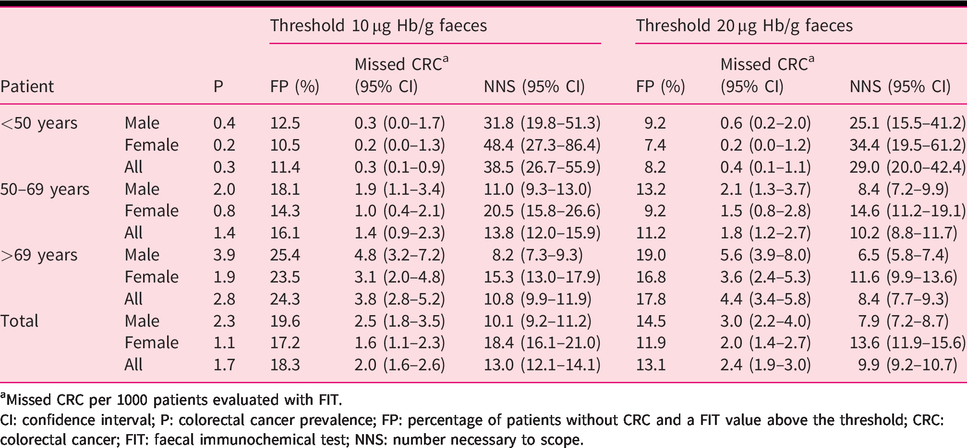
aMissed CRC per 1000 patients evaluated with FIT.
CI: confidence interval; P: colorectal cancer prevalence; FP: percentage of patients without CRC and a FIT value above the threshold; CRC: colorectal cancer; FIT: faecal immunochemical test; NNS: number necessary to scope.
Performance of FIT when threshold is increased from 10 µg Hb/g faeces to 20 µg Hb/g faeces by indication and location (San Sebastián).
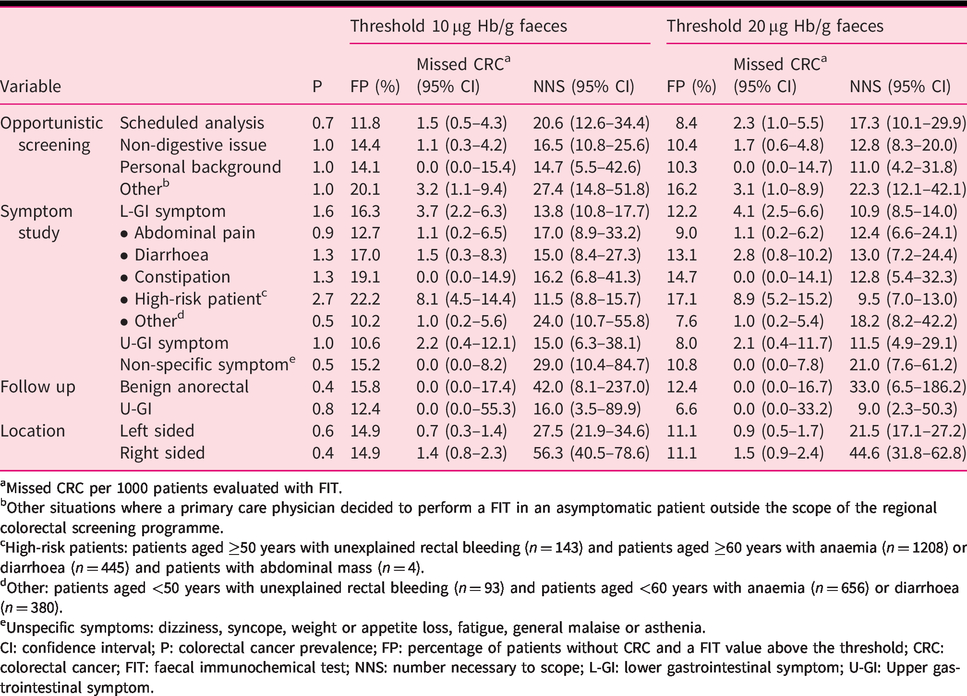
aMissed CRC per 1000 patients evaluated with FIT.
bOther situations where a primary care physician decided to perform a FIT in an asymptomatic patient outside the scope of the regional colorectal screening programme.
cHigh-risk patients: patients aged ≥50 years with unexplained rectal bleeding (n = 143) and patients aged ≥60 years with anaemia (n = 1208) or diarrhoea (n = 445) and patients with abdominal mass (n = 4).
dOther: patients aged <50 years with unexplained rectal bleeding (n = 93) and patients aged <60 years with anaemia (n = 656) or diarrhoea (n = 380).
eUnspecific symptoms: dizziness, syncope, weight or appetite loss, fatigue, general malaise or asthenia.
CI: confidence interval; P: colorectal cancer prevalence; FP: percentage of patients without CRC and a FIT value above the threshold; CRC: colorectal cancer; FIT: faecal immunochemical test; NNS: number necessary to scope; L-GI: lower gastrointestinal symptom; U-GI: Upper gastrointestinal symptom.
Discussion
Statement of principal findings
In this study, we evaluated performing FIT (OC-Sensor™) at different thresholds in daily clinical practice in primary health care, outside the scope of regional CRC screening programmes. We confirm FIT has high sensitivity to detect CRC in this setting using both 10 and 20 µg Hb/g faeces thresholds. Furthermore, unlike specificity, FIT sensitivity was not significantly influenced by characteristics related to the patient clinical spectrum such as demographics (sex and age group) or symptoms. Conversely, sensitivity was significantly impaired in right-sided lesions. Most importantly, NPV was >99.2% in any situation evaluated, covering a wide range of CRC prevalence. Thus, at the 20 µg Hb/g faeces threshold, fewer than one additional CRC would be missed per 1000 patients evaluated, while approximately 1.3 more colonoscopy examinations were needed to identify a CRC using the lowest threshold for any situation analysed.
Strengths and weaknesses
The main strength of this study is the large sample size. In addition, our data were collected from a daily clinical practice setting where initial suspicion of CRC arises and comprised any requested FIT in the aforementioned scenario. The main limitation of this study was the absence of colonoscopy as a reference standard. Previous studies reported overestimation of sensitivity in registry-based studies evaluating diagnostic performance of FIT, but that bias mainly affected studies with 1 year of follow-up. 18 Furthermore, one meta-analysis detected similar sensitivity and specificity between studies using colonoscopy to follow-up all participants and those using 2-year registry follow-up. 19 Moreover, our study included cases of CRC requiring hospitalization, which is not equivalent to the true CRC incidence in the population, as in situ CRC would not be detected by the CBMD. The effect of this information bias could be to overinflate sensitivity, as a significant number of supposed ‘true negatives’ might actually be fully endoscopically resectable CRC which therefore would not require hospital admission. However, the effect of this bias could also be the opposite. Our study could underestimate FIT sensitivity, as detectable f-Hb has been revealed to correlate with the severity of an underlying lesion, and many false positives could be related not only to advanced adenomas or other significant colonic lesions but also to in situ colorectal carcinomas. 20 Another weakness of the study is the lack of detailed information on the clinical spectrum of patients. Non-specific gastrointestinal symptoms commonly associated with CRC are common and sometimes unreported among apparently healthy people, 21 and categorizing a patient as ‘asymptomatic’ based on a recorded reason for FIT request has a high risk of bias.
Strengths and weaknesses in relation to other studies
Our findings are consistent with previous meta-analyses summarizing studies performed in different settings. 3 , 4 Information on sex differences in population-based FIT screening has been conflicting, and sex-tailored thresholds were proposed by some authors to increase the optimal use of colonoscopy resources. 22 A recent meta-analysis did not detect any statistically significant differences in FIT accuracy by sex or age, 19 and our results were in line with this meta-analysis conclusion.
An interesting finding in the subgroup analysis is the different FIT sensitivity between areas. Despite cohorts from San Sebastián and Ourense being statistically different in terms of demographic characteristics, stratified subgroup analysis by sex and age groups in both cohorts, as well as the previously mentioned studies discussing the effect of age and sex on FIT sensitivity, suggest that those differences do not account for the different performance of FIT to rule out CRC between them.
For proper interpretation of results, it is important note that the regional CRC screening programme began in San Sebastián and Ourense in 2009 and 2015, respectively. The impact of those preventive programmes on pathology detected in subsequent colonoscopy explorations has been reported before. 23 , 24 Our study revealed both lower CRC prevalence and FIT positivity for any demographic subgroup evaluated in San Sebastián, which is in line with those studies and could explain the decrease in FIT sensitivity in that population with regard to the population of Ourense.
Moreover, FIT-based screening programmes have an impact both on proximal and distal CRC surgery rates. 25 Since right-sided lesions are known to be more difficult to detect by FIT, 26 subsequent screening rounds could select not only CRC with lower rates of bleeding (i.e. early-stage CRC) but also right-sided CRC.
It was also noteworthy that our data revealed a downward trend in FIT sensitivity for the group of females older than 69 years from San Sebastián compared to data for males of the same age. Although data are only available for the area of San Sebastián, females older than 69 years have been shown to present a right-sided CRC prevalence, which is significantly higher than males in the same age group, 27 thus providing a possible explanation for this finding. However, we would need to know the percentage of right-sided CRC in Ourense to confirm this assumption. Another hypothesis suggested to account for differences in FIT screening between females and males (i.e. different amount of globin or colonic transit time between the sexes) could not account for the differences in FIT sensitivity between females of the same age group from both areas. 19 , 28
Implications for clinical practice and research
Our data confirm that FIT can be used as an aid to daily clinical practice in primary health care, as reported in recent studies,9–12 but also suggest that an increase in the NICE recommended threshold does not lead to a rise in the number of missed CRC in any demographic subgroup and avoids unnecessary colonoscopy examinations. This may be of particular relevance to special situations such as the current coronavirus disease 2019 pandemic in which colonoscopy availability is severely curtailed, and also in the many European countries that have limited colonoscopy capacity.
Despite this, it can be argued that some of these examinations could lead to an advanced adenoma diagnosis, thus contributing to CRC prevention. However, CRC develops from a premalignant lesion (adenomatous polyp) in >70% of cases throughout a process that can last approximately 10 years. 29 Therefore, it is likely that this kind of lesion could be subsequently diagnosed when it is still in an endoscopically fully resectable stage. A recent study in a screening setting used three categories of FIT below 20 µg Hb/g faeces – 0–3.8 µg Hb/g faeces, 3.9–9.9 µg Hb/g faeces and 10.0–19.9 µg Hb/g faeces – and demonstrated that the probability of testing positive and being diagnosed in subsequent screening rounds of advanced neoplasia or CRC interval rose with increasing values of FIT. 30 Thus, repeating FIT determination in a scheduled interval could also be an alternative strategy in the assessment of patients in primary health care to ‘rescue’ those early-stage lesions without increasing colonoscopy resource demand. Another recent proposal is to refer for colonoscopy those patients with cumulative f-Hb concentration ≥20 µg Hb/g faeces over two ‘negative’ tests. 31 We believe that these data provide the basis to justify a clinical trial in which the risks and benefits of both thresholds could be prospectively compared.
Meanwhile, prioritizing individuals for colonoscopy examinations by f-Hb concentration could diminish latency time to diagnosis. 5 Ideally, this should be in a dynamic waitlist manner. The Model for End-Stage Liver Disease is also used to prioritize a liver transplant waitlist. Furthermore, close monitoring of FIT characteristics locally could enable rapid adjustment of FIT thresholds to optimize each area’s resources. 32
Moreover, managing colonoscopy resources efficiently goes beyond the costs. 33 A recent study reported that latency higher than 12 months after the initial positive FIT was associated with more advanced disease and higher mortality due to CRC. 34 In our study, the mean time from FIT determination to initial hospital discharge with a CRC diagnosis exceeded 10 months for FIT negative patients, and was almost twice with respect to patients with a positive FIT result. Another study revealed that a direct referral to colonoscopy from primary health care reduces the risk of mortality. 35 It is therefore important that FIT can be introduced into daily clinical practice at this care level at an optimal threshold.
Conclusions
Our study confirms that FIT is highly sensitive for CRC detection in daily primary health care using a threshold of either 10 or 20 µg Hb/g faeces. The use of a threshold higher than that recommended by NICE (20 µg Hb/g instead of 10 µg Hb/g faeces) could reduce the number of colonoscopy examinations and therefore the latency time of FIT positive patients to be evaluated without missing more than one CRC per 1000 patients evaluated belonging to the low-risk group defined by the NICE recommendation. Right-sided CRC are more likely to be missed by FIT and may justify a relevant percentage of false-negative results in elderly, particularly female, patients. Any strategy using FIT to aid clinical assessment of this particular demographic subgroup must be especially monitored.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620949714 - Supplemental material for Optimal diagnostic accuracy of quantitative faecal immunochemical test positivity thresholds for colorectal cancer detection in primary health care: A community-based cohort study
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620949714 for Optimal diagnostic accuracy of quantitative faecal immunochemical test positivity thresholds for colorectal cancer detection in primary health care: A community-based cohort study by Noel Pin-Vieito, Laura García Nimo, Luis Bujanda, Begoña Román Alonso, María Ángeles Gutiérrez-Stampa, Vanessa Aguilar-Gama, Isabel Portillo and Joaquín Cubiella in United European Gastroenterology Journal
Footnotes
Acknowledgements
This manuscript has been professionally edited by Jason Willis-Lee (professional biomedical translator and author’s editor). Part of our data were previously presented as an oral presentation at a Spanish scientific meeting (Spanish Gastroenterology Association; March 2018) and was accepted for poster presentation at the United European Gastroenterology week in 2018 (Vienna).
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics approval
The study protocol is compliant with the ethics guidelines of the 1975 Declaration of Helsinki and was approved by the Clinical Research Ethics Committee of Galicia (Code 2017/277) under the resolution dated 20 June 2017. This committee confirmed that no formal written consent for ethics approval was required in this study. Data were anonymised after linkage between databases.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Spain’s Carlos III Healthcare Institute by means of project PI17/00837 (co-funded by the European Regional Development Fund/European Social Fund ‘A way to make Europe’/‘Investing in your future’).
Informed consent
No formal written consent was required in this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
