Abstract
Background
Aspirin (ASA) is a drug that can cause gastrointestinal lesions and symptoms. Colorectal cancer (CRC) is the most prevalent type of cancer in Western countries. We assessed the effect of aspirin on the diagnostic accuracy of the faecal immunochemical test (FIT) for CRC and/or advanced neoplasia (AN) in patients undergoing colonoscopy for gastrointestinal symptoms.
Methods
We conducted a prospective multicentre observational study of diagnostic tests that included patients with gastrointestinal symptoms undergoing colonoscopy between March 2012 and 2014 (the COLONPREDICT study). Symptoms were assessed and a FIT and blood tests assessing haemoglobin and carcinoembryonic antigen (CEA) levels were performed.
Results
The study included 3052 patients: A total of 2567 did not take aspirin (non-user group) and 485 (16%) took aspirin (user group). Continuous treatment with ASA did not change the AUC (0.88, 0.82; p = 0.06), sensitivity (92%, 88%; p = 0.5) or specificity (71%, 67%; p = 0.2) of the FIT for CRC detection. Similarly, we found no differences in the AUC (0.81, 0.79; p = 0.6), sensitivity (74%, 75.5%; p = 0.3) or specificity (76%, 73.6%; p = 0.3) for AN detection. Patients with an aspirin use of ≥ 300 mg/day had a lower prevalence of AN and the sensitivity, specificity and AUC for AN for these patients were 54%, 68% and 0.66, significantly lower than for the non-user group (p = 0.03).
Conclusions
Aspirin does not modify the diagnostic accuracy of FIT for CRC and/or AN in patients with gastrointestinal symptoms. Aspirin use of ≥ 300 mg/day decreases the accuracy of the test.
Introduction
Colorectal cancer (CRC) is the most prevalent type of cancer in Western countries, and screening for this type of cancer has led to a decrease in its incidence and associated mortality.1,2 The two most widely accepted techniques for screening in most countries are colonoscopy and the faecal immunochemical test (FIT).1,3 Most CRC diagnoses are made in people who seek medical attention for symptoms that include rectal bleeding, change in intestinal rhythm, abdominal pain and anaemia. These symptoms are very common in the general population, hence, various risk factors have been used to identify individuals who are more likely to have CRC. Recently, published studies have suggested that FIT is the approach with greatest diagnostic accuracy for CRC detection in symptomatic patients4,5 as a sole biomarker or within prediction models.
Studies conducted by our research group have found that the combination of certain signs and symptoms, older age, male sex and a positive FIT result are associated with a 10× higher risk of CRC.4,5 In patients who meet these criteria, colonoscopy must be performed. It has been speculated that FIT results may be modified by certain drugs such as aspirin. Neither antiplatelet drugs nor oral anticoagulants affect the predictive value of the test in asymptomatic populations taking part in screening programmes; however, we do not know the potential effect of aspirin use on FIT in people with gastrointestinal symptoms.6,7 Aspirin has been shown to, on the one hand, decrease the risk of colon polyps 8 and on the other, increase the likelihood of gastrointestinal complaints and anaemia. 9
Our objective was to assess whether aspirin use changes the diagnostic accuracy of FIT in patients with gastrointestinal symptoms.
Patients and methods
Design
We carried out a post-hoc analysis to assess whether aspirin use affects the diagnostic accuracy of FIT in the derivation and validation cohorts from the COLONPREDICT study.4,5 COLONPREDICT was a blind, multicentre cross-sectional study of diagnostic tests. The study sought to create and validate a CRC-predictive index based on available biomarkers and clinical and demographic data.
Population
The study population consisted of consecutive patients with gastrointestinal symptoms referred for colonoscopy from primary and secondary care. Patients were excluded if they were younger than 18 years, pregnant, or asymptomatic and undergoing colonoscopy for CRC screening, had a history of colonic disease and were undergoing surveillance colonoscopy, required hospital admission, or had symptoms that had ceased within three months before evaluation. The trial protocol was approved by the Clinical Research Ethics Committee of Galicia (Code 2011/038). Patients provided written informed consent before inclusion.
Laboratory testing
Patients were administered by nurses specifically trained in the assessment of gastrointestinal symptoms or doctors a questionnaire that included questions related to medication use. In addition, all patients underwent a blood test that included measurement of haemoglobin and carcinoembryonic antigen (CEA) levels, a FIT and colonoscopy.
All individuals collected a stool sample the week before colonoscopy from one bowel movement without specific diet or medication restrictions. They were specifically instructed to sample a stool with no visible blood. FIT was performed using the automated OCsensor™ (Eiken Chemical Co, Tokyo, Japan). Samples were processed as previously described 4 at each regional reference hospital. Patients with ≥ 20 µg of haemoglobin per gram of faeces were classified as positive. Moreover, we measured serum haemoglobin levels and mean corpuscular volume with a Beckman Coulter Autoanalyzer (Beckman Coulter Inc, CA, USA) and serum CEA levels using a chemiluminescent microparticle immunoassay (UniCel DXI 800; Beckman Coulter, CA, USA).
Definition of cohorts
Patients were divided into two groups: users and non-users. We defined the ‘users’ as those reporting chronic use of aspirin. Chronic use was defined as patients taking aspirin for more than one year at least five days a week. ‘Non-users’ were individuals who did not report chronic use of aspirin. Patients who reported a sporadic intake of aspirin were excluded. The dose and duration of aspirin use were verified during the patient’s interview with the physician or nurse when the medical history was reviewed. At some participating centres, dosage and duration of aspirin use were also verified by inspecting their electronic prescription records and the number of pack of pills withdrawn from the pharmacy. It was recommended that patients not suspend aspirin use before FIT or prior to colonoscopy.
Colonoscopy
All colonoscopies were performed by experienced endoscopists (each performing >200 colonoscopies per year). Colonoscopy was performed blind to the questionnaire and other test results. Before colonoscopy, endoscopists performed a digital rectal examination and an anuscopy. Colonoscopy quality was ensured by following the Spanish Gastroenterological Association and Spanish Society for Digestive Endoscopy guidelines. 10 We considered colonoscopy complete if cecal intubation is achieved or a complete neoplastic stenosis is found.
Outcomes
The main outcome was the diagnosis of CRC and advanced neoplasia (AN). Adenomas ≥10 mm in size, with villous architecture, high-grade dysplasia or intramucosal carcinoma were classified as advanced adenomas. The location of the CRC was described as rectum, or distal or proximal to the splenic flexure. Tumour staging was performed according to the seventh edition of the American Joint Committee on Cancer (AJCC) classification. 11 AN was defined as advanced adenoma or invasive cancer. Data from each individual were recorded in an online database.
Statistical analysis
A descriptive analysis of the sample was performed. Comparisons between the groups (non-users and users) were conducted using chi-square and/or Fisher’s exact tests for categorical variables, and the non-parametric Mann-Whitney test for continuous variables.
Patients were classified into three groups by level of aspirin use: none, less than 300 mg/day and 300 mg/day or more. The effect of aspirin use on the positivity of the FIT was analysed with logistic regression, calculating odds ratios (both crude and adjusted for covariates reflecting other factors potentially involved). To assess the accuracy of the FIT in the diagnosis of CRC and AN, sensitivity, specificity and areas under the receiver operating characteristic curve (AUCs) were calculated, together with their confidence intervals. These estimators were compared between groups of aspirin use with a chi-square test to compare proportions and homogeneity of the areas.
All analyses were performed using IBM SPSS Statistics for Windows, version 22.0 and MedCalc, version 12.7.0.0.
Results
Description of the sample
We included 3116 patients, of whom 64 were subsequently excluded, yielding a final sample of 3052 (Figure 1). Of the 3052 participants, 2567 (84%) reported not taking aspirin (non-user group), and 485 (16%) taking aspirin (user group). In the user group, 427 (88%) took less than 300 mg/day of aspirin while 58 individuals took more ≥ 300 mg/day. As expected, more men were taking aspirin than women (p < 0.05) and users were older than non-users (p < 0.001) (Table 1).
Recruitment and participant flow through the study. Characteristics of individuals included in the study. p value Mann-Whitney U. FIT: faecal immunochemical test.
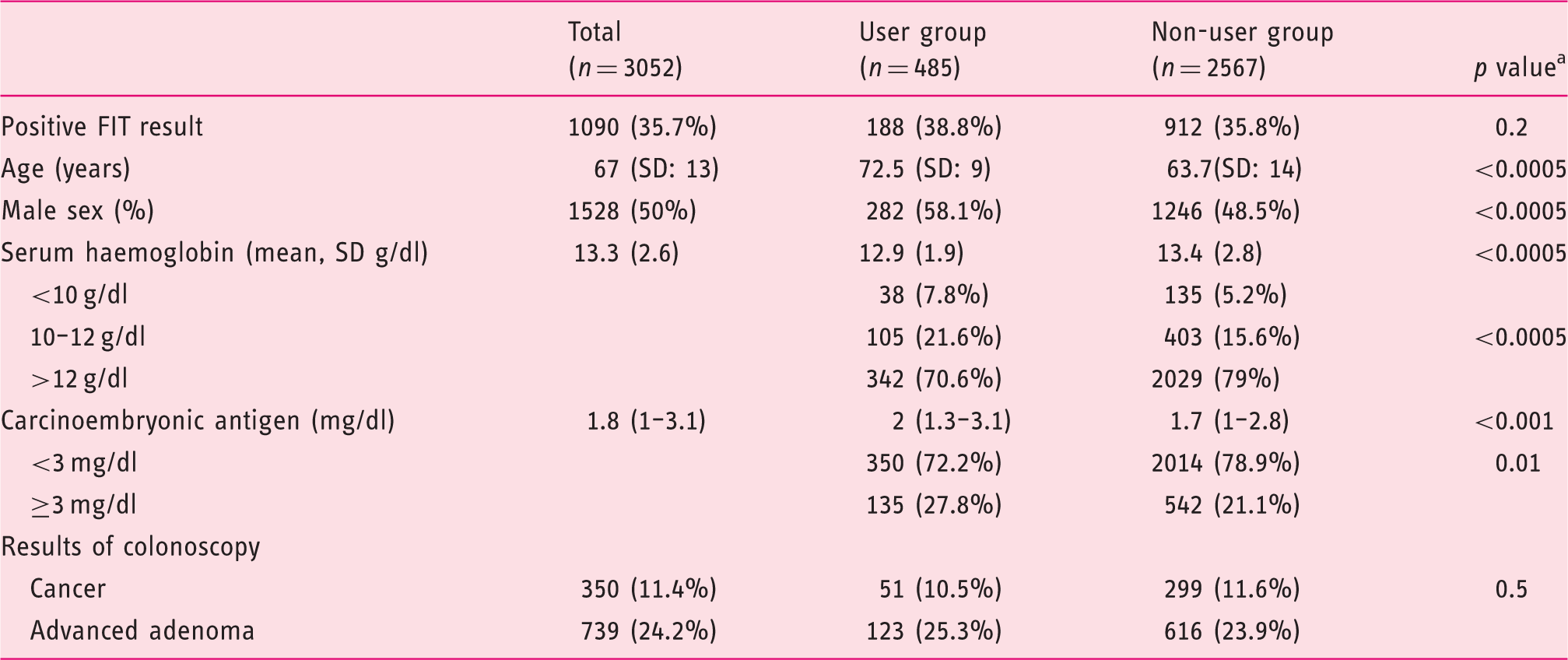
We detected CRC in 350 (11.4%) patients, located in the rectum, in the distal and proximal colon respectively in 28.4%, 50.7% and 20.9% of cases. The tumour staging was: 0 in 2.8% of cases, I in 18.6%, II in 25.1%, III in 37.7% and IV in 15.8%. Additionally, we found advanced adenomas in 739 (24.2%) patients. Overall, we detected AN in 1089 (35.6%) patients.
Effect of aspirin use on FIT results
Factors associated with a diagnosis of advanced neoplasia in patients with a positive faecal immunochemical test result.
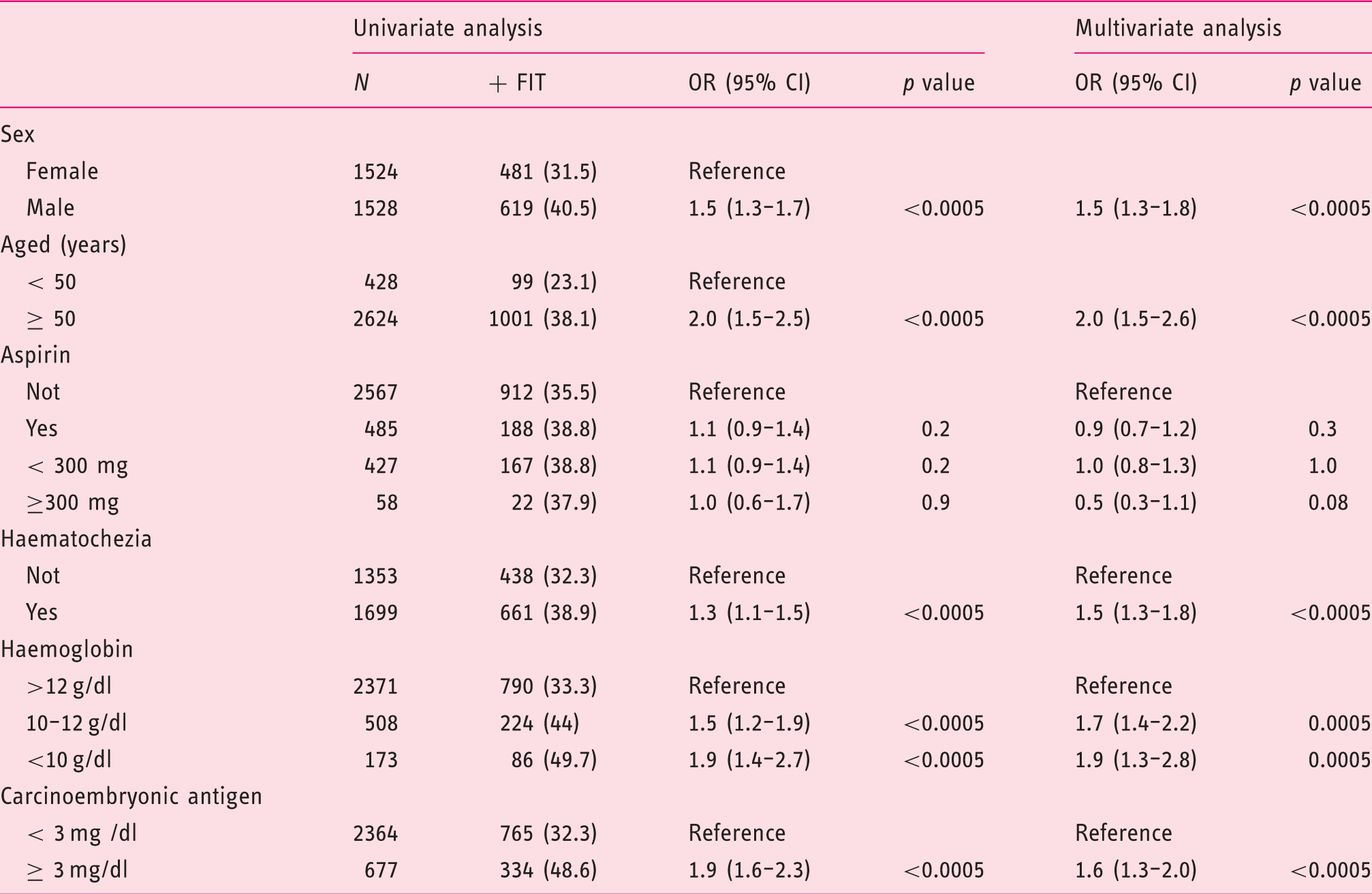
FIT: faecal immunochemical test; OR: odds ratio; CI: confidence interval.
Diagnostic accuracy for CRC in users and non-users
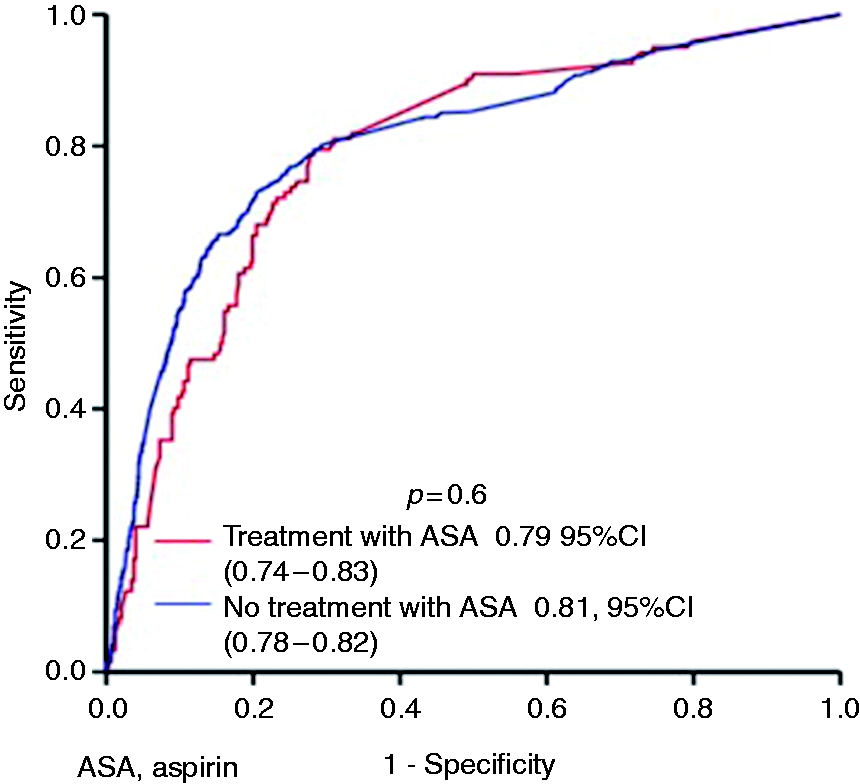
In the user group, there were 51 cases (10.5%) of CRC, of whom 44 (88%) had a positive FIT result. The sensitivity and specificity of the FIT for CRC in the user group were 88% and 67%, respectively, while the AUC was 0.82 (Figure 2). In this group, 123 (25.3%) individuals were found to have advanced adenoma, 82 (67%) of the cases being detected with the FIT. The sensitivity and specificity of the FIT for AN, in the user group were 75% and 73%, respectively (Table 3), while the AUC was 0.79 (Figure 3).
Receiver operating characteristic curve analysis for colorectal cancer diagnosis with faecal immunochemical test as a function of aspirin use. Sensitivity, specificity and area under the receiver operating characteristic curve (AUC) for colorectal cancer and advanced neoplasia as a function of aspirin use. Differences with p < 0.05 are considered statistically significant. Values are expressed as percentage and 95% confidence interval. Receiver operating characteristic curve analysis for advanced neoplasia diagnosis with faecal immunochemical test as a function of aspirin use.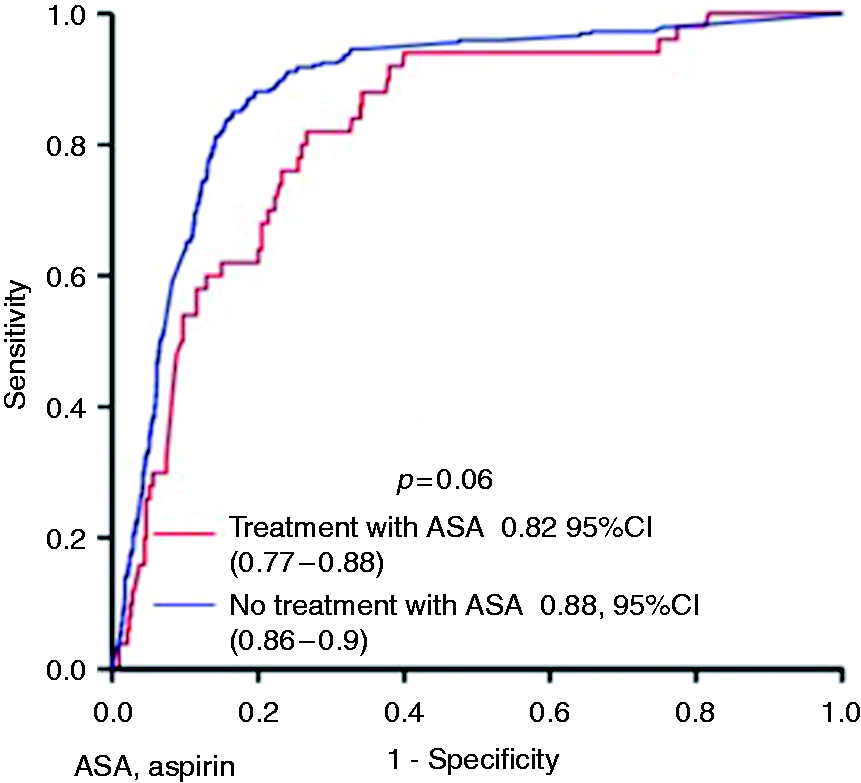
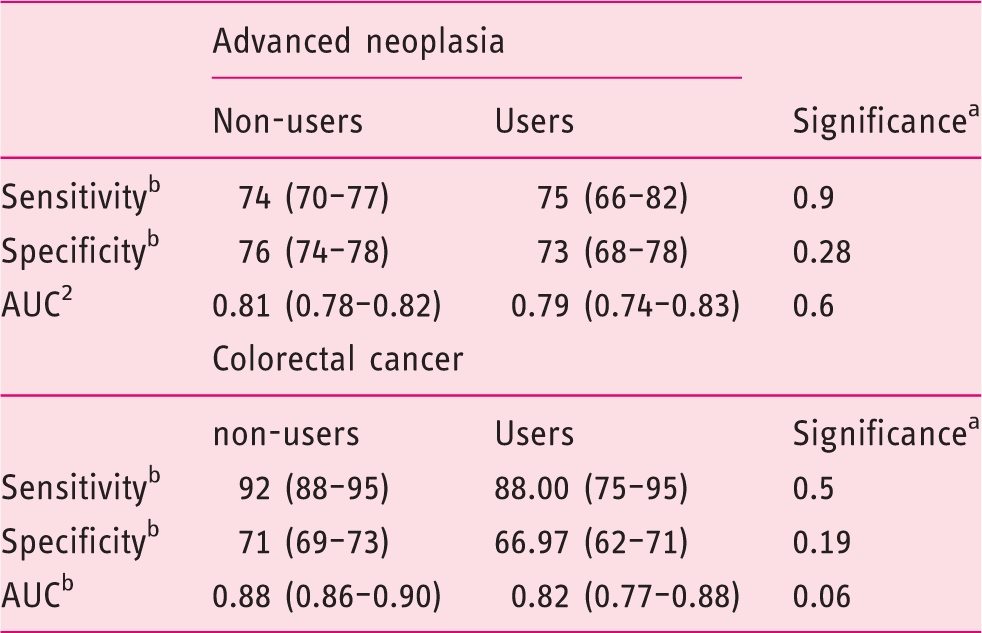
In the non-user group, there were 299 (11.6%) cases of CRC, of whom 275 (92%) had a positive FIT result. The sensitivity and specificity of the FIT for CRC in the non-user group were 92% and 71%, respectively, and the AUC was 0.88 (Table 3). In this group, we detected 616 (23.9%) cases of advanced adenoma, of which 351 (56%) cases were detected with the FIT. The sensitivity and specificity of the FIT for AN in the user group were 74% and 76%, respectively, while the AUC was 0.81. We did not observe significant differences between the user and non-user groups in terms of sensitivity, specificity and AUCs for AN.
Effect of aspirin dose on diagnostic accuracy
Prevalence of advanced adenoma and cancer as a function of aspirin dose.

AA: advanced adenoma; CRC: colorectal cancer
Sensitivity, specificity and area under the receiver-operating characteristic curve (AUC) for advanced neoplasia as a function of aspirin dose.
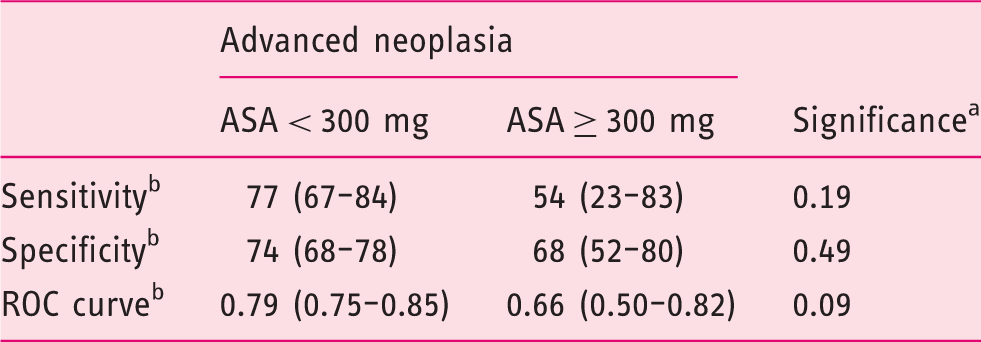
Differences with p < 0.05 are considered statistically significant.
Values are expressed as percentage and 95% confidence interval.
ASA: acetylsalicylic acid; ROC: receiver operating characteristic.
Discussion
In this study, we have observed no significant difference in the rate of positive FIT results in patients seeking medical attention for digestive symptoms as a function of whether they reported aspirin use. The sensitivity of the FIT was similar in the two groups (75% users vs 74% non-users).
Levi et al. 12 in a retrospective study with fewer aspirin users (980 non-users and 170 users) also found no differences in sensitivity or specificity of the FIT in symptomatic patients. In that study, the sensitivity, specificity and positive predictive value (PPV) of the FIT for AN in aspirin users were 66%, 92% and 52%, respectively, while these values were 46%, 93% and 38%, respectively, in the control group. Our findings differ from these results in that we found sensitivity, specificity and PPV in the non-user group of 74, 76% and 50%, respectively. Levi et al. do not provide information about the positivity of the test. The differences observed in the test sensitivity may be due to the differences in prevalence of the lesions detected and the design of the study (retrospective vs prospective). In contrast to our study, the prevalence of AN in the study by Levi et al. 12 was very low (9.3%), comparable to values of 1.3% to 10.7% found in asymptomatic individuals in prospective studies.13,14 Research based on asymptomatic individuals, such as the COLONPREV study, 14 has also failed to find differences in the predictive value of the test between aspirin users and non-users. Analysing the predictive value as a function of aspirin use, we found that the predictive value of the FIT was lower for those taking the highest doses of the drug. Further, the prevalence of AN was lower in patients on higher doses of aspirin (>300 mg/day). It is known that aspirin reduces the occurrence of adenomas8,15 and that a lower prevalence of lesions decreases the predictive value of diagnostic tests. With regards to the most suitable dose of aspirin to reduce the occurrence of adenomas, there is no consensus between studies15,16 and this may be due to the number of patients included, the type of patients (moderate vs high risk) or the duration of aspirin use.
The prevalence of AN in our study was high (35.6%). Blumenstein et al. 17 observed in a prospective study that the prevalence of CRC in individuals of at least 55 years of age was equivalent in symptomatic individuals (1.21%) and the control group (1.02%); advanced adenomas were observed in significantly fewer symptomatic patients (5.67%) than matched asymptomatic controls (8.03%); and finally, polyps were found significantly less often in symptomatic patients (25.0%) than in matched controls (33.6%). However, in another study, 18 the prevalence was somewhat higher, with colonic neoplasia and CRC being identified in 17.3% and 11.3% cases, respectively of a total 1013 symptomatic individuals (mean age 59.9 years, 52.3% females) from a multi-ethnic Asian background. The high prevalence we found could be attributable to the inclusion of patients referred for in-hospital high-resolution consultation for colon cancer.
The prevalence of aspirin use in the general population is very high. Low-dose aspirin use is indicated for primary prevention of cardiovascular events. Aspirin is taken by about 17% of asymptomatic individuals aged over 65 years 19 and 25% of patients admitted to hospital for gastrointestinal bleeding. This drug is known to cause lesions in the stomach and duodenum, and can lead to an increase in the bleeding of colon lesions. 20 In our study, 16% of symptomatic patients took aspirin. Aspirin use among asymptomatic people who attended screening tests ranged between 8.9% and 12% with a mean age between 59 and 62 years of age, respectively.6,13 As in previous studies, we found that aspirin users were older and more likely to be male. 13
Further, aspirin may increase the onset of digestive symptoms that can lead patients to seek medical attention. A meta-analysis of 59 studies found that 5.2% of individuals receiving aspirin reported a minor gastrointestinal complaint (abdominal pain, dyspepsia, or nausea/vomiting), vs 3.7% of placebo participants. The corresponding summary odds ratio was 1.46, while the odds ratios for dyspepsia and abdominal pain were 3.17 and 1.92, respectively. 9 Further, aspirin enhances the onset of anaemia and gastrointestinal bleeding. 21 In our study, we found that aspirin users had lower levels of haemoglobin, and this may be related to the harmful effect of this drug on the gastrointestinal mucosa, since the prevalence of AN was similar in both groups. Despite the potential adverse effects of aspirin, treatment cessation may increase the likelihood of cardiovascular complications (primarily, cerebral stroke and ischaemic heart disease).
The present study is strengthened by a number of observations. First, it is a post-hoc analysis within a prospective clinical trial in symptomatic patients without selection bias, which should minimise confounders for the detection of advanced adenomas and AN. Second, the number of cases analysed is higher than that reported to date with a high prevalence of CRC. Third, this is the largest study that assesses the effect of a treatment with aspirin in terms of diagnostic accuracy for CRC and AN.
The study also has some limitations. First, we do not know to what extent patients who were taking aspirin may show a positive FIT result due to lesions in the stomach and small intestine. Additionally, we were not able to determine whether the clinical signs that prompted patients to seek medical attention were related to aspirin intake.
In summary, aspirin use did not change the diagnostic accuracy of FIT in patients with gastrointestinal symptoms. For this reason, we do not recommend discontinuation of aspirin treatment in patients who seek medical addition for symptoms suggestive of CRC.
Footnotes
Acknowledgements
The authors’ contributions were as follows: LB, JC, PV and MS participated in the study design; JC, EQ, MS, VAS, FFB, MA, JB, RC, AG, AF, LT, DR, MD, VH, and LB in the collection, analysis, and interpretation of data; JC, CS and LB in the writing of the report; and all the authors decided to submit the article for publication. All authors had full access to all the data (including statistical reports and tables) in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
None declared.
Ethics approval
The study was approved by the Clinical Research Ethics Committee of Galicia (Code 2011/038).
Funding
This work was supported by a grant from the Carlos III Health Institute (PI11/00094). This institute had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication. JC and VH have received an intensification grant through the European Commission-supported ‘BIOCAPS’ project (FP-7-REGPOT 2012-2013-1, grant agreement no. FP7- 316265). CIBERehd is also funded by the institute.
Informed consent
Patients provided written informed consent.
Supplementary note
Researchers of the COLONPREDICT study: Complexo Hospitalario Universitario de Ourense: Joaquín Cubiella, Pablo Vega, María Salve, Marta Díaz-Ondina, Irene Blanco, Pedro Macía, Eloy Sánchez, Javier Fernández-Seara University of Vigo: María Teresa Alves Hospital Universitario de Canarias: Enrique Quintero, Natalia González-López Complejo Hospitalario de Pontevedra: Victoria Álvarez Sánchez, José Mera, Juan Turnes Hospital Universitari Mútua de Terrassa: Fernando Fernández-Bañares, Victoria Gonzalo, Mar Pujals Registre del Càncer de Catalunya Pla Director d'Oncologia de Catalunya, Hospital Duran i Reynals, L’Hospitalet de Llobregat: Josepa Ribes, Ramón Cleries, Xavier Sanz Consorci Sanitari de Terrassa: Jaume Boadas, Sara Galter Corporació Sanitària i Universitària Parc Taulí: Rafel Campo, Marta Pujol, Eva Martínez-Bauer Departamento de Bioquímica, CATLAB, Viladecavalls, Barcelona: Antonio Alsius. Donostia Hospital: Luis Bujanda, Cristina Sarasqueta, Mauro D’Amato, Jesús Bañales, María J Perugorria Hospital de Sagunto: Joan Clofent, Ana Garayoa Hospital Clínico Universitario de Zaragoza: Ángel Ferrández, Marina Solano Sánchez Hospital Dr. Josep Trueta: Leyanira Torrealba, Virginia Piñol Hospital Universitario de Móstoles: Daniel Rodriguez-Alcalde, Jorge López-Vicente Complexo Hospitalario Universitario de Vigo: Vicent Hernández, Felipe Iglesias
