Abstract
Background
The diagnostic yield of the faecal immunochemical test and sigmoidoscopy in detecting proximal serrated polyps in a colorectal cancer screening programme has not been fully assessed.
Aim
We determined the detection rate of proximal serrated polyps by simulated sigmoidoscopy and faecal immunochemical test compared with total colonoscopy in a population-based, multicentre, nationwide, randomised controlled trial (ColonPrev study).
Methods
Sigmoidoscopy yield was simulated based on the UK-Flexible Sigmoidoscopy Trial for total colonoscopy referral. Definitions were: proximal serrated polyp (proximal serrated polyp): sessile serrated polyp or hyperplastic polyp of any size and proximal at-risk serrated polyp (at-risk proximal serrated polyp): sessile serrated polyp of any size or hyperplastic polyp ≥ 10 mm, both located proximally to the splenic flexure.
Results
A total of 10,611 individuals underwent faecal immunochemical test and 5059 underwent total colonoscopy and were evaluated by simulated sigmoidoscopy. Sigmoidoscopy and faecal immunochemical test were less accurate in detecting proximal serrated polyps (odds ratio: 0.13; 95% confidence interval: 0.10–0.18 and 0.13; 0.09–0.18, p < 0.0001, respectively). Both tests were inferior to colonoscopy in detecting at-risk proximal serrated polyps, and sigmoidoscopy was inferior to faecal immunochemical test in detecting these lesions (odds ratio: 0.17; 95% confidence interval: 0.10–0.30 and 0.25; 0.17–0.37, p < 0.0001, respectively).
Conclusion
Sigmoidoscopy and faecal immunochemical test are less accurate in detecting proximal serrated polyps than colonoscopy, particularly in women.
Key summary
Despite evidence of the efficacy of sigmoidoscopy and faecal occult blood testing (FOBT) in colorectal cancer (CRC) screening, both strategies have potential limitations in detecting proximal lesions. Recent evidence suggests that up to 30% of CRC cases develop from serrated polyps (SPs), so the detection of proximal and large SP may be one of the future goals of CRC screening strategies. We were able to clarify that sigmoidoscopy and the faecal immunochemical test (FIT) have major limitations in detecting proximal SPs compared with total colonoscopy (TC). These limitations are more marked in women, independently of age. More studies are needed to determine the effectiveness of repeat FIT testing over time in proximal SP detection.
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and the second leading cause of cancer-related death and studies has shown that screening with both faecal occult blood testing (FOBT) and flexible sigmoidoscopy is effective and reduces CRC-specific incidence and mortality in randomised controlled trials. Accordingly, these two strategies, along with colonoscopy, have been universally accepted and recommended for CRC screening.1,2
Until recently, CRC was thought to develop from adenomas through the adenoma-carcinoma-pathway. However, recent evidence suggests that up to 30% of CRC cases develop from serrated polyps (SPs) through an alternative pathway, known as the serrated pathway.3,4 In this regard, recently published studies have shown an increase risk of CRC in individuals with large SPs (≥10 mm) and proximal sessile serrated polyps (SSPs),5–7 suggesting that these lesions may be one of the future goals of CRC screening strategies.
Although colonoscopy may be imperfect in the detection of SPs, it is probably superior to alternative methods of CRC screening. Inconsistent results have been reported regarding the influence of sigmoidoscopy on the incidence of proximal CRC.8,9 Moreover, conflicting results have also been reported on the ability of FOBT to detect advanced proximal neoplasms (APNs). Similar to the findings in the baseline screening examination of the ColonPrev study, 10 some studies have reported a lower sensitivity in APN detection, 11 while other studies have reported comparable sensitivities in both locations. 12 In fact, regarding CRC detection, a recent meta-analysis in colonoscopy verified studies reported better diagnostic performance for the detection of CRC in the distal than in the proximal colon. 13
The ColonPrev study, a randomised controlled trial designed to assess the efficacy of one-time colonoscopy and biennial faecal immunochemical test (FIT) in reducing CRC mortality at 10 years 10 constitutes a unique opportunity to estimate the risk of proximal SPs by comparing different screening strategies, FIT and simulated sigmoidoscopy, in a large cohort of average-risk individuals screened by colonoscopy. Therefore, this analysis aimed to compare FIT and sigmoidoscopy strategies in terms of accuracy and the resources needed to detect one proximal SP, both overall and in age- and sex-specific subgroups.
Methods
The current analysis is a post-hoc analysis of the ColonPrev study (ClinicalTrials.gov number: NCT00906997), details are described elsewhere.
10
The study protocol (Figure 1), in compliance with the ethical guidelines of the 1975 Declaration of Helsinki, was approved by the institutional review board of Parc de Salut Mar (protocol number: 2008/3050/, 15 April 2008), and all participants provided written informed consent.
Enrolment and outcomes of individuals included in the study.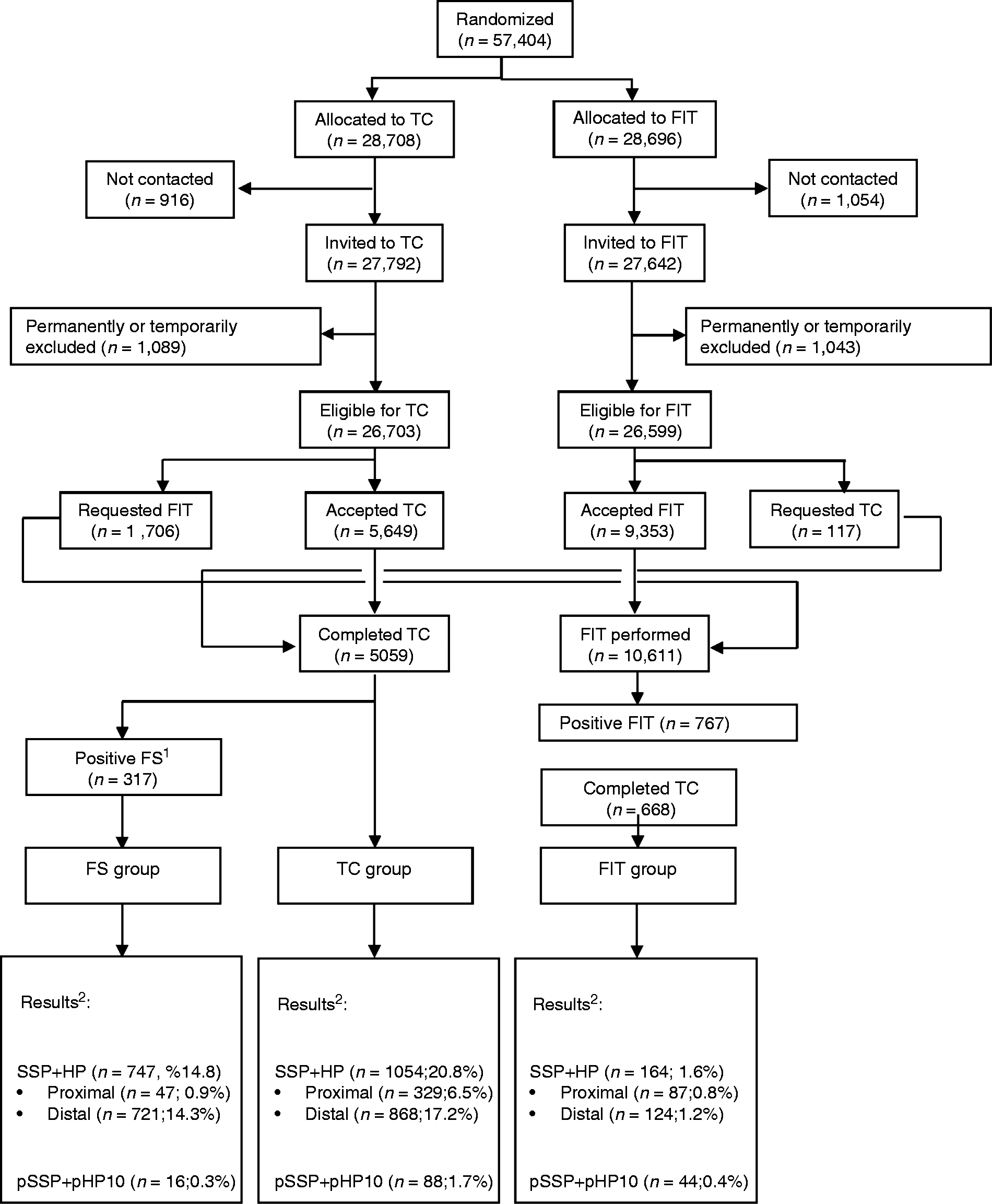
FIT strategy
The FIT strategy consisted of a single stool sample analysis with the use of the automated semiquantitative OC-Sensor (Eiken Chemical). 10 Participants who were found to have a haemoglobin level of 15 µg Hb/g faeces or more were invited to undergo colonoscopy.
Sigmoidoscopy simulation
Simulation of sigmoidoscopy yield was estimated from the results obtained in the colonoscopy arm by considering lesions detected in the rectum and sigmoid colon and according to the criteria proposed in the UK Flexible Sigmoidoscopy Trial for colonoscopy referral (one distal polyp ≥10 mm, tubulovillous or villous histology, high-grade dysplasia, ≥3 adenomas, CRC, or ≥20 hyperplastic polyps above the distal rectum). 9 Patients were classified according to the most advanced lesion present in the proximal and distal segments. To minimise any potential bias caused by the segments included in the sigmoidoscopy simulation, a sensitivity analysis was performed by including the descending colon in the distal colon.14,15
Data analysis
The main outcome of the analysis was the detection rate of proximal SPs, which was defined as any SSP, traditional serrated adenoma (TSA) or hyperplastic polyp (HP), regardless of size (proximal SP group) or at-risk proximal SPs, defined as any SSP or TSA regardless of size plus any HP larger than 10 mm (at-risk proximal group), both located proximally to the splenic flexure. The performance characteristics of each strategy in detecting proximal serrated lesions were estimated both overall and upon stratification of the whole series in arbitrarily defined subsets of individuals by age (50– 59 years and 60–69 years) and sex. Detection rates for each strategy, were compared with the corresponding detection rate in the colonoscopy arm by logistic regression analysis and are reported as odds ratios (ORs) with 95% confidence intervals (CIs), adjusted by participating centre. The analysis of resources was done by calculating the number of individuals needed to screen and the number of individuals needed to scope to detect one proximal SP based on the strategy: sigmoidoscopy or FIT.
Finally, a forward stepwise logistic regression analysis was performed to identify distal findings independently associated with the presence of proximal SP. Variables included were the presence in the rectum and sigmoid colon of any HP > 10 mm, any SSP < 10 mm, any non-advanced adenoma, any SSP ≥ 10 mm, or an advanced neoplasm, defined as advanced adenoma or CRC. Patients were classified by the most advanced lesion.
Analyses were done by using the SPSS statistical software, version 15.0 (SPSS Inc., Chicago, Illinois, USA). All statistical tests were two-sided and p values <0.05 were considered statistically significant.
Results
A total of 10,611 individuals underwent FIT, and 5059 underwent colonoscopy at the baseline screening examination of the ColonPrev study, either as the initial randomised allocation or after crossover from the alternative arm (Figure 1). Among those screened by FIT, 767 individuals (7.2%) tested positive, and 663 of them (86.4%) completed colonoscopy, thus resulting in 10,507 individuals evaluable for final outcomes in the FIT arm. Simulation of sigmoidoscopy yield based on the results obtained in the colonoscopy arm and according to the criteria proposed in the UK Flexible Sigmoidoscopy trial9 resulted in 317 individuals (6.2%) being referred to colonoscopy when lesions detected in the rectum and sigmoid colon were considered (primary analysis) (Figure 1) and in 365 individuals (7.2%) when lesions in the descending colon were also considered (sensitivity analysis).
Overall SP detection rate
Detection rate of serrated polyps according to faecal immunochemical test (FIT) and sigmoidoscopy-based screening strategies with respect to total colonoscopy (TC).
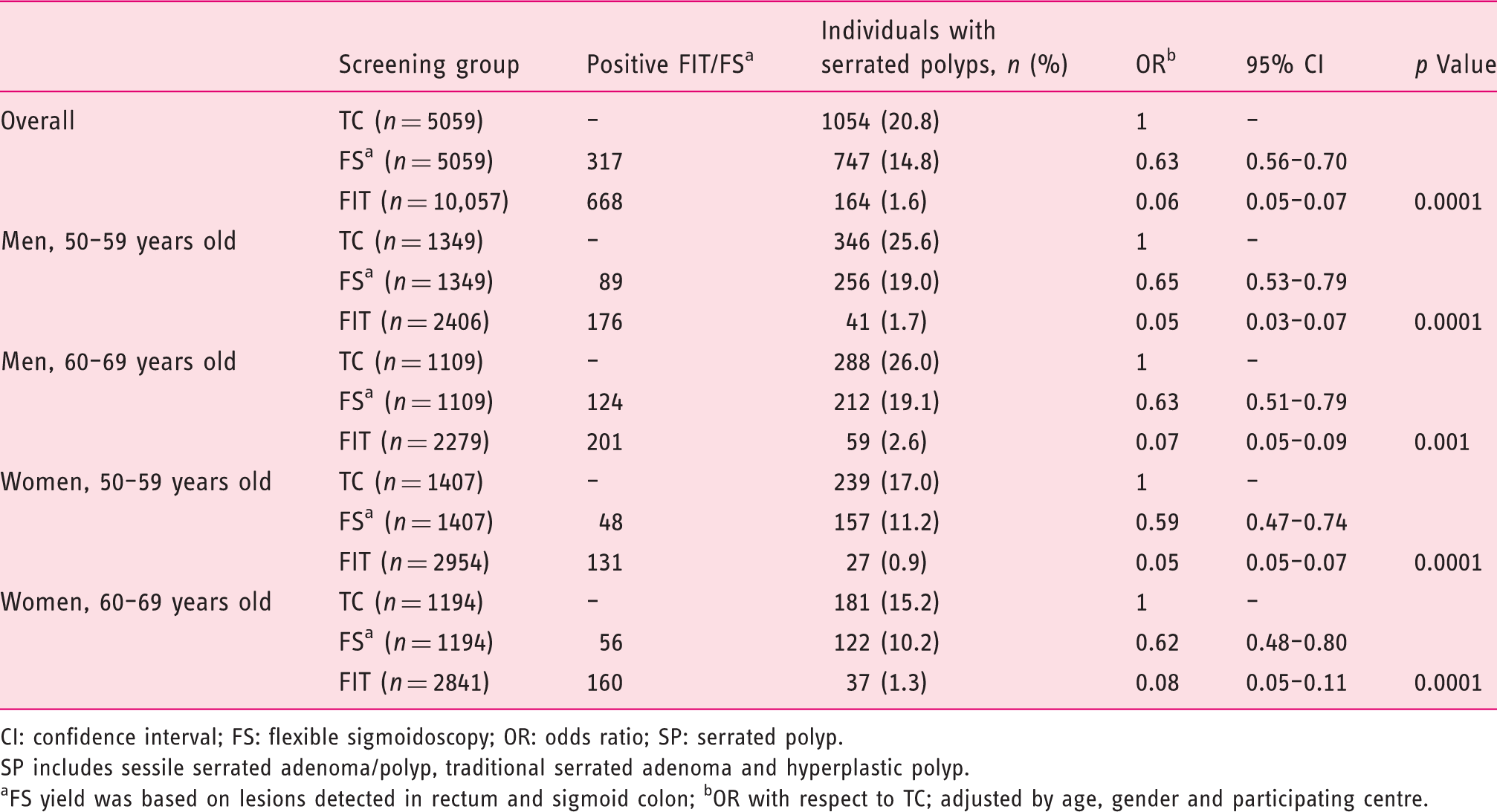
CI: confidence interval; FS: flexible sigmoidoscopy; OR: odds ratio; SP: serrated polyp.
SP includes sessile serrated adenoma/polyp, traditional serrated adenoma and hyperplastic polyp.
FS yield was based on lesions detected in rectum and sigmoid colon; bOR with respect to TC; adjusted by age, gender and participating centre.
Colon side-specific SP detection
Detection rate of proximal serrated polyps according to faecal immunochemical test (FIT) and sigmoidoscopy-based screening strategies with respect to total colonoscopy (TC).
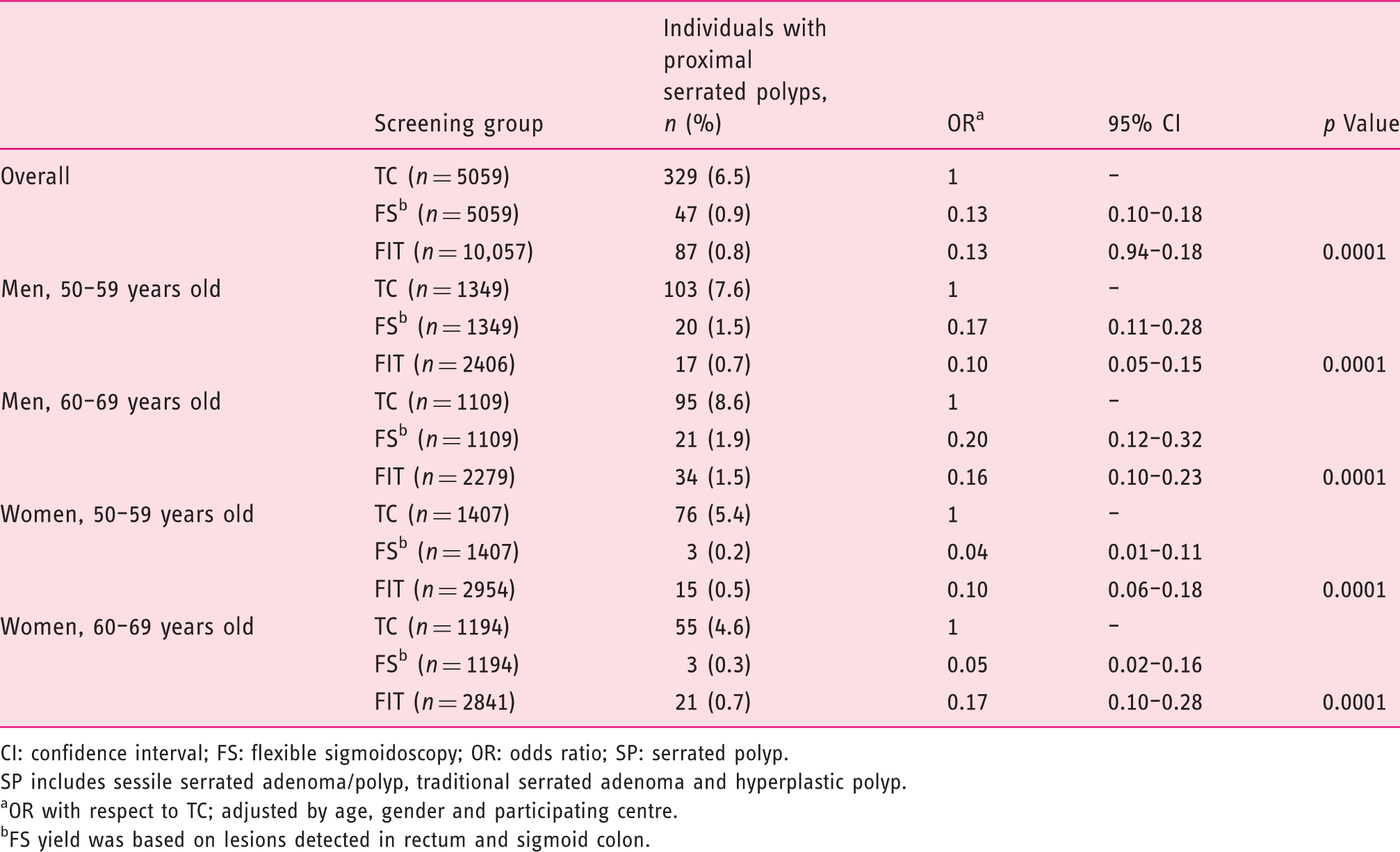
CI: confidence interval; FS: flexible sigmoidoscopy; OR: odds ratio; SP: serrated polyp.
SP includes sessile serrated adenoma/polyp, traditional serrated adenoma and hyperplastic polyp.
OR with respect to TC; adjusted by age, gender and participating centre.
FS yield was based on lesions detected in rectum and sigmoid colon.
Detection rate of at-risk proximal serrated polyps according to faecal immunochemical test (FIT) and sigmoidoscopy-based screening strategies with respect to total colonoscopy (TC).
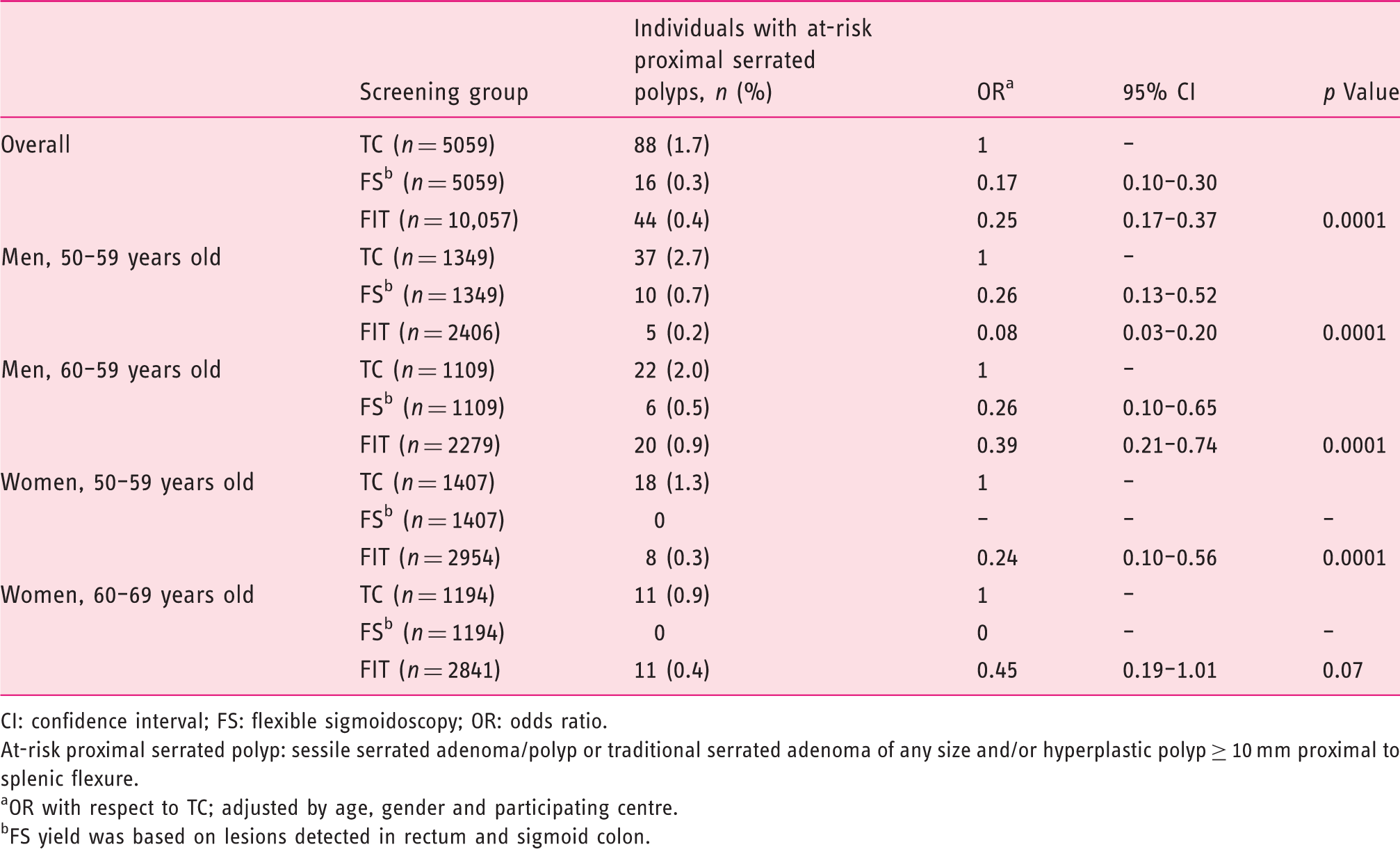
CI: confidence interval; FS: flexible sigmoidoscopy; OR: odds ratio.
At-risk proximal serrated polyp: sessile serrated adenoma/polyp or traditional serrated adenoma of any size and/or hyperplastic polyp ≥ 10 mm proximal to splenic flexure.
OR with respect to TC; adjusted by age, gender and participating centre.
FS yield was based on lesions detected in rectum and sigmoid colon.
In the sensitivity analysis, the performance characteristics of FIT- and sigmoidoscopy-based strategies were similar to those observed in the primary analysis regarding proximal (Supplementary Material Table 2) and at-risk proximal SPs (Supplementary Material Table 3).
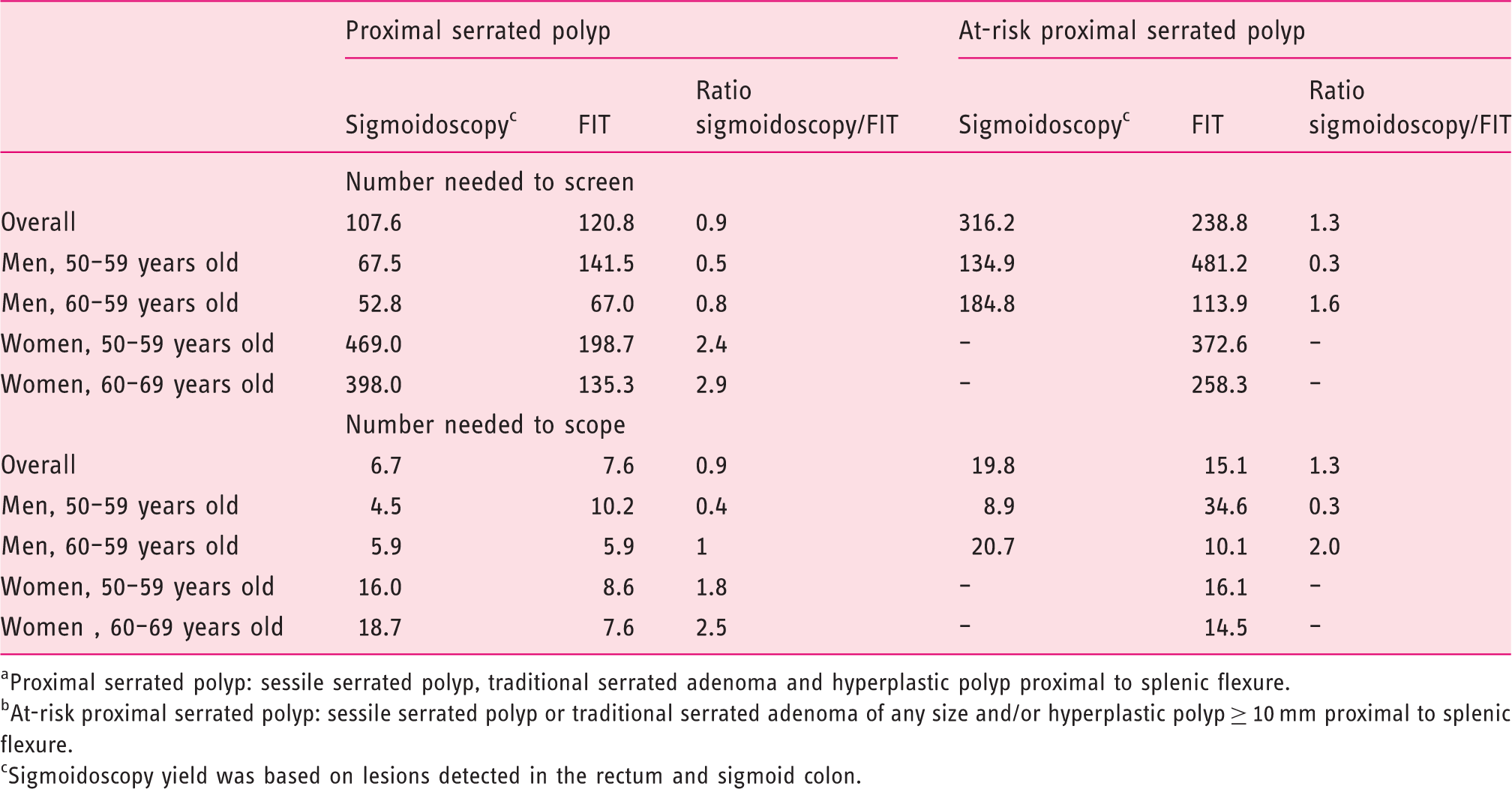
Analysis of resources
Proximal serrated polyp: sessile serrated polyp, traditional serrated adenoma and hyperplastic polyp proximal to splenic flexure.
At-risk proximal serrated polyp: sessile serrated polyp or traditional serrated adenoma of any size and/or hyperplastic polyp ≥ 10 mm proximal to splenic flexure.
Sigmoidoscopy yield was based on lesions detected in the rectum and sigmoid colon.
In both cases, these results were reproduced on evaluation of the number of individuals needed to refer for colonoscopy (Table 4). The results obtained in the primary analysis were similar to those obtained in the sensitivity analysis, in which the descending colon was also included in the sigmoidoscopy simulation (Supplementary Material Table 4).
Distal findings associated with proximal SPs
Characteristics independently associated with the presence of any proximal serrated polyp. a
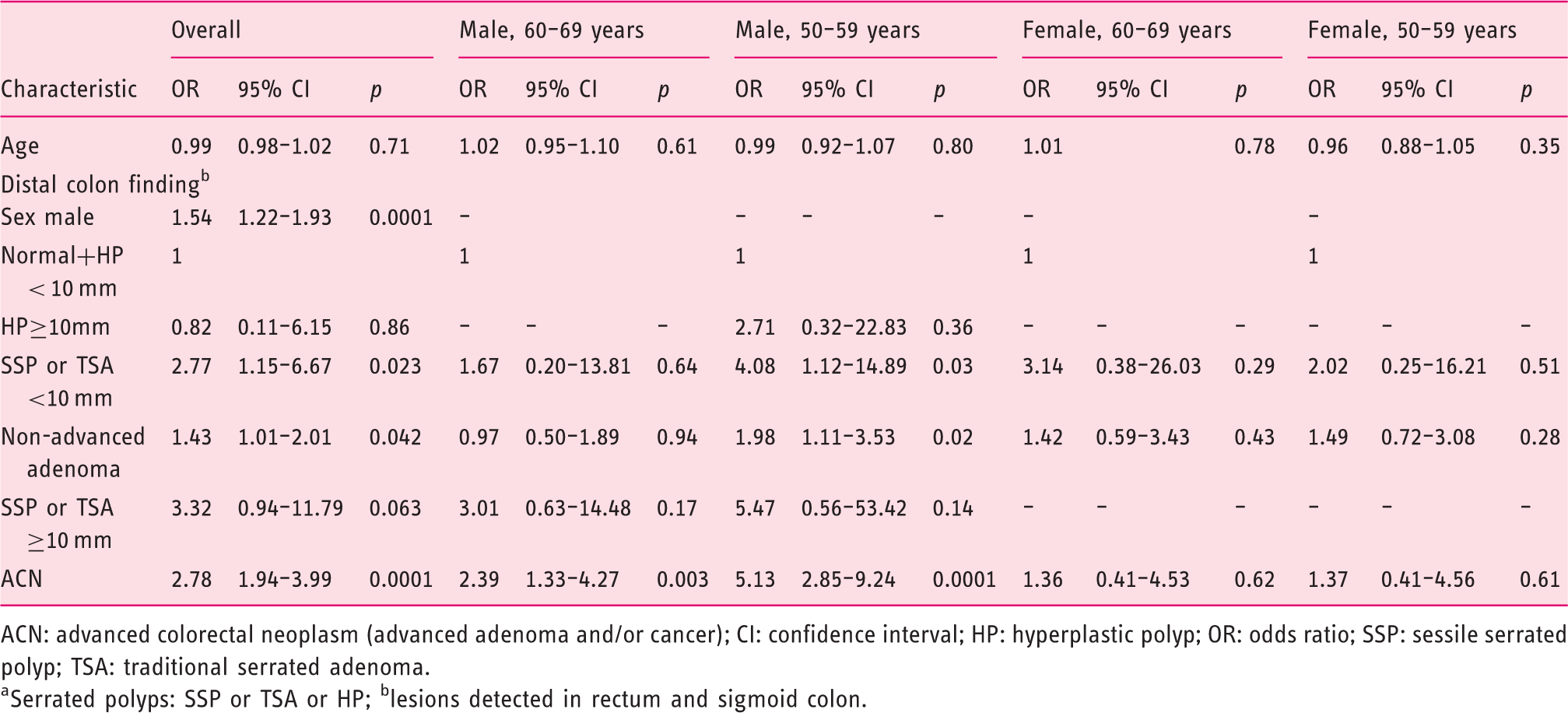
ACN: advanced colorectal neoplasm (advanced adenoma and/or cancer); CI: confidence interval; HP: hyperplastic polyp; OR: odds ratio; SSP: sessile serrated polyp; TSA: traditional serrated adenoma.
Serrated polyps: SSP or TSA or HP; blesions detected in rectum and sigmoid colon.
Characteristics independently associated with the presence of at-risk proximal serrated polyp. a
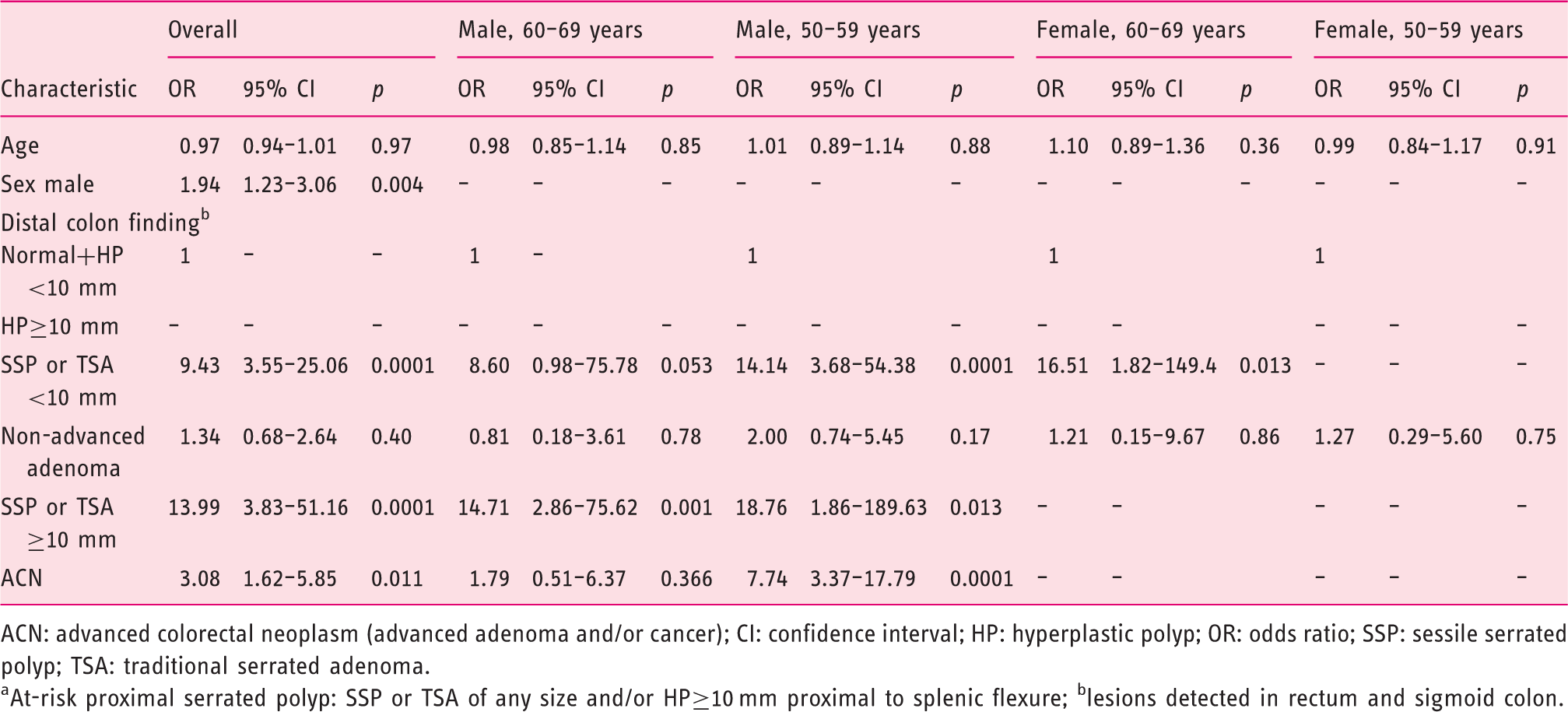
ACN: advanced colorectal neoplasm (advanced adenoma and/or cancer); CI: confidence interval; HP: hyperplastic polyp; OR: odds ratio; SSP: sessile serrated polyp; TSA: traditional serrated adenoma.
At-risk proximal serrated polyp: SSP or TSA of any size and/or HP≥10 mm proximal to splenic flexure; blesions detected in rectum and sigmoid colon.
Discussion
The results of our study based on the baseline screening examination of a multicentre, nationwide, randomised controlled trial clearly indicates the superiority of TC over FIT and sigmoidoscopy in the detection of proximal and at-risk proximal SPs. Moreover, when individuals were stratified by age and sex, significant differences were observed in the efficacy of screening strategies. In fact, although sigmoidoscopy detected more proximal SPs than FIT in men, the results were the opposite in women. In this sense, FIT detected more proximal SPs, especially in older women, than sigmoidoscopy. Reported prevalence 16 of proximal SPs in both arms of the study (TC and FIT) was within that of previously published papers.17–20
Previous studies have shown that FIT essentially failed to detect SSPs. Reported sensitivities ranging from 0–18% to 5.1–10.5% for large SSPs by FIT-20 and FIT-10 µg Hb/g faeces buffer positive threshold, respectively.21,22 A recent study, based on three rounds of a FIT-based screening program using a 10 µg Hb/g faeces level cut-off, showed that no differences in Hb levels were found for those with SPs (independently of size or subtypes) or non-advanced adenomas compared with individuals without polyps at colonoscopy. 23 In fact, except for the TSA usually located in the distal colon, SPs have a median absolute FIT result ranging between 15–22 µg Hb/g faeces. Finally, a study comparing FIT versus a noninvasive multitarget stool DNA test showed a sensitivity of FIT-20 µg Hb/g faeces of 5.1% in SSPs measuring 1 cm or more. 24 Although our results are based on the first screening round only, and the putative advantages of this strategy could be accentuated by its periodic nature with a potential cumulative effect in the long term, published results are in agreement with our finding of low sensitivity in proximal SP detection. In fact, in the setting of an organised CRC screening programme with FIT a low prevalence of SSPs and a lack of association with the FIT round was observed, 19 suggesting that SSPs are not a suitable target for FIT-based organised/population-based programmes.
An explanation for this low sensitivity may lie in the biology of these precursor lesions. SSPs are typically sessile or flat, non-ulcerated, and without haemorrhagic features, 25 thus suggesting that serrated lesions are coincidental, nonbleeding findings in FIT positive individuals. 25 Nevertheless, previous studies have observed wide variability in the presence of concurrent adenoma in patients with a large SP, ranging from 20–63%.6,20,26
In fact, the SP is considered the key precursor lesion of some colonoscopy interval colorectal cancers (iCRCs), and the rate of occurrence is strongly related to the sensitivity of the test. A recent meta-analysis observed an overall pooled incidence rate of iCRC after a negative FIT test of 20 (95% CI 14–29) per 100,000 patients-year. In the studies where tumour location was assessed, iCRCs were located distal from the splenic flexure in 67% (95% CI 64–70%) of the cases). 27
Regarding sigmoidoscopy, the results of our study clearly show that detection of proximal and at-risk proximal SPs with sigmoidoscopy was inferior to its ability to detect APNs. Contrasting evidence of the protective effect of sigmoidoscopy in proximal CRC incidence in different sigmoidoscopy trials has been observed. Analysis of three randomised sigmoidoscopy trials (PLCO, SCORE and NORCCAP trials) 28 has shown a significant reduction on CRC incidence, both distal and proximal by 26% and 14%, respectively. However, although the effectiveness of sigmoidoscopy in the incidence of distal CRC was similar in men and woman, irrespective of age (29% and 24% reduction, respectively), the protective effect was different regarding proximal CRCs. In fact, incidence of proximal CRCs was significantly reduced in men but not in women (27% and 9%, respectively). Moreover, screening reduced the incidence of proximal CRCs by 35% in younger women while women aged 60 years and older did not benefit from screening. Moreover, the extended follow-up of the UK flexible Sigmoidoscopy Trial (UKFSST) 9 through a median of 17 years of follow-up has shown a reduction in incidence of distal CRCs by 56%, but a 5% non-significant effect on proximal CRC incidence. This small effect with respect to the other trials was expected since referral for a colonoscopy was restricted to individuals with high-risk polyps, and as a result, only 5% of the screened group had a colonoscopy. Epidemiological studies have shown a rightward shift in the distribution of CRCs that may be related to population aging, with patients having fewer distal and more proximal neoplasms over time.29,30 Some studies have reported that proximal advanced neoplasia without distal lesions is more common in women than in men.31,32 The screening arm of the NORCCAP study, based on sigmoidoscopy with or without FOBT, identified 103 individuals (0.8%) with at least one large SP. Of these, 22 had concomitant advanced adenomas and 81 individuals had isolated SPs, of which four (4.9%) fulfilled the World Health Organization criteria for serrated polyposis. 6 Only seven out of 81 had an isolated proximal large SP, whereas 39 out of 81 had both a distal and proximal large SP. Like our study, a retrospective cross-sectional study in a small sample showed that average-risk patients aged 50 years old or older with distal colorectal polyps were no more likely to harbour advanced serrated lesions (ASLs) (defined as TSA and SSP with dysplasia or size ≥10 mm) in the proximal colon than patients without distal colorectal polyps. 17 The study also evaluated predictive factors of proximal SPs and only age was associated with proximal ASLs and, by contrast, only the presence of SSP<10 mm with or without HP was independently associated with the presence of a proximal SP. With respect to the previous study, to estimate the risk of proximal SPs, we differentiated those individuals with distal HP>10 mm and a SSP. Proximal and at-risk proximal SPs were associated with male sex, the presence of advanced colorectal neoplasm (advanced adenoma or colorectal cancer) and the presence of a distal SSP, irrespective of size. However, only 12.5% and 13.6% of patients with proximal and at-risk proximal SPs had neoplasms in the distal colon.
This study has several strengths. First, it is based on the results of a large, prospective, multicentre, nationwide, randomised controlled trial that followed a strict quality assurance program, 10 thus ensuring the reliability of the data. Secondly, the analysis of the results, both overall and after stratification by age and sex, permits more accurate ascertainment of the usefulness of screening strategies.
However, we are aware of some limitations. First, the definition of at-risk proximal SPs, was arbitrary. Initial studies suggest that proximal and large serrated lesions have an increased risk of neoplasia and advanced neoplasia at follow-up.16,33 The Norwegian CRC prevention trial observed that patients with large (≥10 mm) SPs, compared with patients not invited to screening and patients without polyps at sigmoidoscopy, have a hazard ratio for CRC of 2.5 (95% CI, 0.8–7.7) and 4.2 (95% CI, 1.13–13.3), respectively, equivalent to having an advanced adenoma. 6 Moreover, a nationwide population-based study reported that a history of SSPs was associated with a substantially increased CRC risk compared with patients without these polyps. The CRC risk was particularly high for SSPs with dysplasia (OR 4.76; 95% CI 2.59–8.73), for women with SSPs (OR 5.05; 95% CI, 3.05–8.73), and for patients with proximal SSPs, the highest risk being for CRC (OR 12.42; CI 95% 4.88–31.58). 7 In these studies, with a centralised pathological review, although no information about polyp size was available, 28.8% and 24.9% of the lesions originally thought to be a HP were reclassified as a SSP and TSA. In fact, agreement on the SP subtypes was only moderate to low, both for expert and non-expert pathologists.34–36 In our retrospective study, the vast majority of proximal SPs were classified as HP. A key difficulty is that the minimum criteria for the diagnosis of SPs have varied between guidelines and between groups of pathologists, and in our study, the pathologic criteria for the diagnosis of SPs were not centrally reviewed and, although location and size of the HPs were independently described, the database analysis prevented also differentiation of SSA, with or without dysplasia, from TSA.
Secondly, as previously described,14,15 data on the sigmoidoscopy yield were extrapolated from the colonoscopy results. This potential bias, however, was minimised by the performance of a sensitivity analysis in which the descending colon was included in the distal colon. In addition, because of the nature of this post hoc analysis, the long-term diagnostic yield of both sigmoidoscopy and FIT cannot be established because participation in each screening strategy could not be evaluated and data on the detection rate were limited to the first screening round.
Although a retrospective sigmoidoscopy simulation is not real-life screening and the fact that a single round underestimates the role of a FIT-based screening programme, the results of this study indicate that sigmoidoscopy and FIT have major limitations in detecting proximal SPs compared with TC. These limitations are more marked in women, independently of age. Further prospective studies are needed to determine the effectiveness of repeat FIT testing over time in proximal SP detection. Moreover, our study suggests that not only conventional adenomatous lesions, but also distal SSPs, independently of size, are associated with proximal SPs.
Supplemental Material
Supplementary Material - Supplemental material for Detection of serrated lesions in proximal colon by simulated sigmoidoscopy vs faecal immunochemical testing in a multicentre, pragmatic, randomised controlled trial
Supplemental material, Supplementary Material for Detection of serrated lesions in proximal colon by simulated sigmoidoscopy vs faecal immunochemical testing in a multicentre, pragmatic, randomised controlled trial by Laura Carot, Antoni Castells, Cristina Hernández, Cristina Alvarez-Urturi, Francesc Balaguer, Angel Lanas, Joaquín Cubiella, Jose D Tasende, Rodrigo Jover, Vicent Hernandez, Fernando Carballo, Luis Bujanda, Enrique Quintero, Montserrat Andreu and Xavier Bessa in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics approval
The study protocol, in compliance with the ethical guidelines of the 1975 Declaration of Helsinki, was approved by the institutional review board of Parc de Salut Mar (protocol number: 008/3050/, 15 April 2008)
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
All participants provided written informed consent.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.