Abstract
Alcoholic hepatitis should be suspected in every patient with excessive chronic alcohol consumption and recent onset of jaundice. Diagnosis of alcoholic hepatitis is based on clinical and laboratory findings, and confirmed by a liver biopsy when available. Several scores are available to assess severity and prognosis of alcoholic hepatitis. The 1-month mortality of patients with severe alcoholic hepatitis, as defined by Maddrey’s discriminant function, is 20–30%. Therefore, severe alcoholic hepatitis should be treated with a 28-day course of oral prednisolone after systematic screening for infection. In this review, we discuss diagnosis of alcoholic hepatitis, the different scores to assess severity of the disease, indications for corticosteroid therapy and alternative therapeutic options for non-responders to medical therapy.
Introduction
Excessive alcohol consumption is one of the leading causes of liver disease worldwide. Alcohol abuse can cause a spectrum of liver injuries, collectively termed alcohol-related liver disease. 1 Alcoholic hepatitis (AH) is an acute and severe presentation of alcohol-related liver disease and is characterised by the recent onset of jaundice. The 1-month mortality for patients with severe AH can be as high as 17–50%. 2 , 3 Patients with non-severe AH have a less than 10% risk of 1-month mortality. 4 However, it has recently been shown that 1- and 5-year survival rates for patients with non-severe AH who were admitted for recent liver decompensation were only 80% and 50%, respectively. 5 Although the incidence of AH remains largely unknown, one study suggested an increase in incidence between 1999 and 2008, both in men and women. 6 Despite decades without novel effective therapeutics, the management of patients with AH has evolved considerably during the last few years. The aim of this review is to provide an update on diagnosis, severity assessment and available therapeutic options in AH, and to suggest a personalised approach through a clinical management algorithm. We also provide a clinical case to illustrate management of AH.
Case report
A 56-year-old man presented with malaise and progressive jaundice that had persisted for 10 days. The patient had a history of excessive alcohol consumption (80 g/day) for more than 15 years. Physical examination revealed jaundice, spider naevi on the chest wall, moderate ascites, and no encephalopathy. Blood test results included: total bilirubin 18 mg/dl, aspartate aminotransferase (AST) 85 IU/l, alanine aminotransferase (ALT) 36 IU/l, gamma-glutamyltranspeptidase 220 IU/l, prothrombin time 21.9 s (30%), international normalised ratio (INR) 2.14, albumin 25 g/l and creatinine 0.85 mg/dl. Abdominal ultrasound demonstrated dysmorphic liver with steatosis and ascites but there was no sign of biliary obstruction. A diagnosis of AH was suspected.
Diagnosis of AH
Symptoms and clinical signs
In clinical practice, AH should be suspected in every patient with excessive chronic alcohol consumption and recent onset of jaundice. It is not uncommon for patients to have ceased drinking alcohol days or even weeks before the onset of jaundice. A period of less than 60 days of abstinence before the onset of jaundice is generally observed. 7
Progressive jaundice is often associated with fever, malaise and malnutrition. Other signs of liver decompensation, such as ascites and/or encephalopathy, can be observed in severe forms of the disease. Blood analysis typically reveals hyperbilirubinaemia (>5 mg/dl, mainly conjugated), neutrophilia and serum AST levels greater than the upper limit of normal range, although rarely above 300 IU/l, with an AST/ALT ratio typically greater than 1.5–2.0. In severe forms, prolonged prothrombin time and hypoalbuminaemia are frequently observed. 1 AH should be distinguished from other liver diseases, such as ischaemic hepatitis and drug-induced liver injury, two conditions in which ALT and AST are more elevated, typically above 300–400 IU/l. Liver ultrasound should exclude biliary obstruction. Viral hepatitis and severe autoimmune liver disease testing should be performed.
Liver biopsy
When AH is suspected based on clinical and laboratory findings, a liver biopsy, performed by the transjugular route in the presence of ascites or coagulation disorders, is useful to confirm the diagnosis and to rule out other diagnoses (10–20% of cases). Histologically, typical findings include signs of steatohepatitis, defined by steatosis, hepatocyte ballooning, an inflammatory infiltrate with neutrophils, bilirubinostasis and advanced fibrosis with a typical chicken-wire pattern. 8 However, many centres do not routinely perform liver biopsy to confirm a diagnosis of AH. The main restrictions in routine clinical practice are access to transjugular liver biopsy, potential risks, the costs of the procedure and the absence of well-validated grading systems.
Proposed criteria for diagnosis
Recently, National Institute on Alcohol Abuse and Alcoholism (NIAAA) Alcoholic Hepatitis Consortia recommendations have defined AH patients for inclusion in clinical studies as follows: ‘definite AH’ defined as clinically diagnosed and biopsy proven AH, ‘probable AH’ defined as clinically diagnosed but not biopsy proven AH, without confounding factors, and ‘possible AH’ defined as clinically diagnosed but not biopsy proven AH, with potential confounding factors: i.e. possible ischaemic hepatitis, possible drug-induced liver injury, uncertain alcohol use assessment, atypical laboratory tests or presence of antinuclear antibodies (Table 1). 7
Degree of diagnostic certainty of alcoholic hepatitis (AH) based on National Institute on Alcohol Abuse and Alcoholism (NIAAA) consortium criteria.

ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Based on these criteria, which still need to be validated, a transjugular liver biopsy is mandatory in possible AH, while liver biopsy is not absolutely mandatory in cases of probable AH.
Biomarkers
Cytokeratins have emerged as serum biomarkers of hepatocyte damage. Circulating fragments of cytokeratin-18 (CK-18), M65 and M30 are of particular interest in AH. A recent study reported higher levels of total and microvesicle-bound M65 and M30 in the circulation of patients with biopsy-proven AH. A cut-off of 2000 IU/l for M65 has a positive predictive value of 91% and a cut-off of 642 IU/l has a negative predictive value of 88%. 9 Although promising, these biomarkers need further investigation before being used in routine clinical practice.
Case report (cont.)
Screening for infection (urine, blood, ascites) was negative. Maddrey discriminant function was calculated at 69.5, Model for End-stage Liver Disease (MELD) score at 25.9, and Glasgow Alcoholic Hepatitis Score (GAHS) score at 12. Severity, prognosis, and therapeutic options were discussed.
Prognostic assessment in AH
Several scores are available to assess severity and prognosis in AH. The aims of these scores are, first, to estimate the likelihood of short-term mortality and, second, to determine whether the patient should be treated with corticosteroids (CSs).
Maddrey’s discriminant function (MDF) is frequently used for predicting the severity of AH and assessing the need for CS therapy. The limitations of this score include its use of prothrombin time expressed in seconds, which is not standardised between clinical laboratories, and the absence of renal function assessment, as acute kidney injury is a strong predictor of mortality in AH. 10
The MELD score, the GAHS and the Age-Bilirubin-International normalised ratio-Creatinine (ABIC) score are also useful for predicting 30-day and 90-day mortality in patients with AH and take into account renal function assessment.11–14 Although the MDF remains the most commonly used score in clinical studies and in many centres, the MELD, GAHS and ABIC scores appear to have similar efficacy for predicting short-term mortality in AH patients 15 and are used according to the local practice of each centre. The variables included in the scoring systems for AH and the cut-offs used to determine the severity of the disease are shown in Table 2.
Variables included in the most commonly used prognostic scores for alcoholic hepatitis patients

ABIC: Age-Bilirubin-International normalised ratio-Creatinine; FR: full responder; GAHS: Glasgow Alcoholic Hepatitis score; INR: international normalised ratio; MDF: Maddrey’s discriminant function; MELD: Model for End-stage Liver Disease; NR: null responder to corticosteroids; PR: partial responder; PT: prothrombin time; WCC: white cell count.
More recently, baseline neutrophil-to-lymphocyte ratio (NLR) has been suggested as a measure that may help to stratify risk and likelihood of CS response in patients with AH. Patients with low (<5) and high (>8) NLR values do not appear to derive benefit from CS treatment. NLR has been incorporated into the GAHS score in place of the white cell count to create the modified GAHS (mGAHS) and this has improved the discriminatory power of the score. 16 However, validation studies are needed before NLR can be recommended for routine clinical practice.
Some histological features are also associated with severity and mortality in AH. Specifically, degree of fibrosis, neutrophil infiltration, type of bilirubinostasis and the presence of megamitochondria have been found to be independently associated with 90-day mortality and are included in the Alcoholic Hepatitis Histological Score (AHHS). 17
New markers, such as serum transferrin, have shown promise as tools for prediction of mortality in patients with severe AH. 18 However, validation studies are needed before recommending their routine use.
Due to the fact that early improvement in liver function has an important impact on short-term mortality in patients with severe AH, dynamic variables, such as those used in the Lille Model, are used as markers of therapeutic response to CS therapy.
How do we identify patients suitable for CS therapy?
The question of which patients should be treated is linked to CS treatment because severe AH was defined in studies that tested prednisolone in AH. 19 Due to the fact that patients with an MDF<32 do not derive a survival benefit from prednisolone, these patients should not be treated with CSs and should only be managed for addiction, in the absence of a specific validated pharmacological strategy. On the other hand, patients with an MDF ≥32 should be treated with a 28-day course of oral prednisolone, based on evidence from randomised controlled trials, after having undergone systematic screening for infection before steroid initiation. It has been shown that patients admitted with severe AH have a risk of being infected of 25% before the initiation of prednisolone. 20 Thus, ruling out subclinical infection is mandatory in these patients. The most prevalent infections are spontaneous bacterial peritonitis, bacteraemias, pneumonias and urinary tract infections. Interestingly, the pattern of infection is different before and after steroid initiation, with an increase in the incidence of pneumonia in patients who have received prednisolone. 20 At present, there is no definite argument for systematic antibiotic prophylaxis in patients receiving CSs for severe AH but the results of a randomised controlled trial testing amoxicillin-clavulanate are expected (NCT02281929). The onset of infection in patients receiving steroids is still a frequent problem because about 25% of them will develop sepsis in the 2 months following the initiation of prednisolone. 20 To date, no firm guidelines have been set regarding discontinuation of prednisolone. From a practical point of view, it seems reasonable to continue steroids in patients with mild sepsis (e.g. urinary tract infection, bacteraemia) and to stop CS treatment in patients with severe infection (e.g. pneumonia, septic shock). However, this strategy has never been formally evaluated.
Prednisolone is indicated in patients with severe AH because meta-analyses have demonstrated a reduction in mortality at 1 month with prednisolone over placebo. 21 , 22 Generally speaking, most trials have tested a dose of 40 mg/day of prednisolone for 1 month, but 32 mg/day of methylprednisolone can be also used. Taking these positive results into account, both the European Association for the Study of the Liver (EASL) and American Association for the Study of Liver Diseases (AASLD) recommend using prednisolone to treat severe forms of AH. 1 , 23 Of note, this treatment cannot be viewed as ideal because it does not improve 3-month or 6-month survival. Overall, about 30% of prednisolone-treated patients will die within 6 months, underlining the fact that a subgroup of these patients do not derive any survival benefit from steroid treatment. In order to identify these patients as early as possible, the Lille model can be used after 7 days of prednisolone treatment. The Lille model can be used with two different approaches: a binary prediction of survival and response to treatment according to the ideal cut-off of 0.45. Patients with a low score (<0.45) are termed responders and their survival at 6 months is good, higher than 80%. 24 On the other hand, patients with a score ≥0.45 are non-responders and their survival at 6 months is about 30%. Response to steroids can also be defined according to Lille model cut-offs of 0.16 and 0.56. 25 Complete responders are patients with a Lille score ≤0.16 and their risk of dying in the short term is minimal. Partial responders have a Lille score between 0.16–0.56 and their short-term survival is lower than that of complete responders. Null responders have a Lille score ≥0.56 and their risk of dying at 1 month is close to 50%. Comparisons to placebo-treated patients have shown these null responders have the same outcome regardless of the treatment allocated (i.e. prednisolone or placebo). Thus, prednisolone can be stopped after 7 days in the case of a Lille score ≥0.56.
Case report (cont.)
The patient was treated with prednisolone at a dose of 40 mg/day. At day 7 of therapy, total bilirubin was 22 mg/dl, creatinine 1.1 mg/dl, and INR was 2.3. The patient’s Lille score was calculated to be 0.89 and his MELD score was 28. Prednisolone was stopped at day 7. The patient had never experienced liver decompensation before the current episode. He was married, with two children, and worked actively before his admission to the hospital. Alternative therapeutic options and early liver transplantation were discussed.
Are treatments other than prednisolone useful?
Other than CSs, there are few therapeutic options that can be applied in this indication. Most molecular strategies have been tested against placebo or a control with short-term survival as the primary endpoint. While pentoxifylline was considered useful based on a small pilot study, 26 meta-analyses have demonstrated that it has no impact on survival, alone or in combination with CSs. 21 , 22 Pentoxifylline was tested because it was hypothesised to decrease Tumor necrosis factor (TNF) in the liver. Based on these results, EASL 1 and AASLD 23 do not recommend using pentoxifylline in AH.
N-acetylcysteine has also been studied because animal models have demonstrated that alcohol-related liver injury is responsible for a decrease in reduced glutathione in hepatocytes. N-acetylcysteine was tested in AH because this drug can restore glutathione homeostasis. Unfortunately, N-acetylcysteine alone does not improve survival 22 but its combination with prednisolone has suggested interesting results. However, the difference in survival associated with the combination treatment was not statistically significant in the last randomised controlled trial. 27
Due to the fact that malnutrition is frequent during AH and impacts survival in patients with decompensated cirrhosis, enteral nutrition has been tested in a recent randomised controlled trial. 28 Despite an improvement in outcomes in the per-protocol analysis (patients who received a higher number of calories had better survival), the overall conclusion of this important work was that enteral nutrition does not significantly impact mortality.
Considering that only prednisolone has been shown to be effective in patients with severe AH and that this treatment does not improve medium- or long-term survival, there is an urgent need for the development of new treatments. Several options are being evaluated but, so far, no treatment has been confirmed to be effective in a phase III trial. Recently, the results of a phase IIa trial testing DUR-928 (oxysterol sulphate, a molecule regulating genes involved in metabolism, inflammation, regeneration, and cell survival) have shown promise in 19 patients with severe AH. The treatment appears to be safe and treated patients experienced decreases in bilirubin levels, MELD scores, and MDF. 29 In addition, interleukin (IL)-22 targets hepatic inflammation and regeneration and its agonist, F-652, has shown promise in a recent phase II trial. 30 Granulocyte colony stimulating factor (GCSF) has also been evaluated in several trials with conflicting results. While Western studies do not report any survival or biological benefit, 31 data from India indicate that this treatment could be helpful. 32 At present, it is difficult to draw a firm conclusion regarding the use of G-CSF in AH. Other potential targets 33 include antibiotics (randomised controlled trial testing amoxicillin plus clavulanate vs placebo in combination with prednisolone, NCT02281929) and the IL-1R inhibitor anakinra in combination with pentoxifylline and zinc (NCT01809132). Lastly, modifying gut microbiota by faecal transplantation has been suggested to be effective in a pilot study performed in India. 34 In this study, faecal microbiota transplantation improved short-term survival and a confirmatory trial is ongoing. This approach does not selectively target bacteria involved in alcohol-related liver injury, however, in a translational study, Duan et al. have demonstrated that the use of a bacteriophage was responsible for a decrease in cytolysins produced by E. faecalis which is associated with hepatic damage during AH. 35 This strategy warrants testing in a clinical trial.
In summary, only prednisolone is available at present but there are several strategies being tested to treat severe AH. Most of them aim to modify inflammation and/or regeneration, but some also target gut microbiota. Table 3 summarises the main therapeutic strategies that are being tested in AH.
Main therapeutic strategies being tested in alcoholic hepatitis.

IL: interleukin.
Regardless of treatment, patients classified as non-responders using the Lille score have poor outcomes and are refractory to existing pharmacological strategies. With this in mind, it has been proposed that a subgroup of patients experiencing their first episode of liver decompensation related to alcohol be offered early liver transplantation. Based on a pilot study from France and Belgium, 36 this procedure has been extended to other countries, particularly the USA.37–40 Although this strategy must be confirmed with larger studies, early liver transplantation improves mortality and appears to be associated with an acceptable risk of alcohol relapse after liver transplantation. The selection of candidates for liver transplantation is extremely challenging. There is global consensus that liver transplantation should be offered with a preference for carefully selected patients who have good family support and a favourable addiction profile, and who are undergoing their first episode of liver decompensation. 1 , 23 , 36 In consideration of the fact that most of these patients have not experienced any recent sobriety period before AH, careful follow-up with regard to their addiction is required after liver transplantation. Due to the fact that a randomised controlled trial may be a questionable study design for demonstrating the benefits of early liver transplantation in patients with severe AH, Lee et al. have performed a modelling approach that tests immediate transplantation versus a delayed procedure. 41 In this study, early transplantation was associated with improved survival, despite the negative impact of alcohol relapse on outcomes. However, this strategy needs to be confirmed in prospective studies. Due to organ shortages, this treatment is only offered to a small subgroup of patients who do not respond to medical treatment and there is no salvage therapy that can be recommended as a bridge to liver transplantation. At present, no strategy has been proven effective in patients with non-response to medical treatment except liver transplantation. In these patients who cannot be transplanted, outcomes are poor. This underlines the urgent need for novel medical options to treat patients who do not improve with a course of CSs. A proposed treatment algorithm for patients with suspected AH is presented in Figure 1.
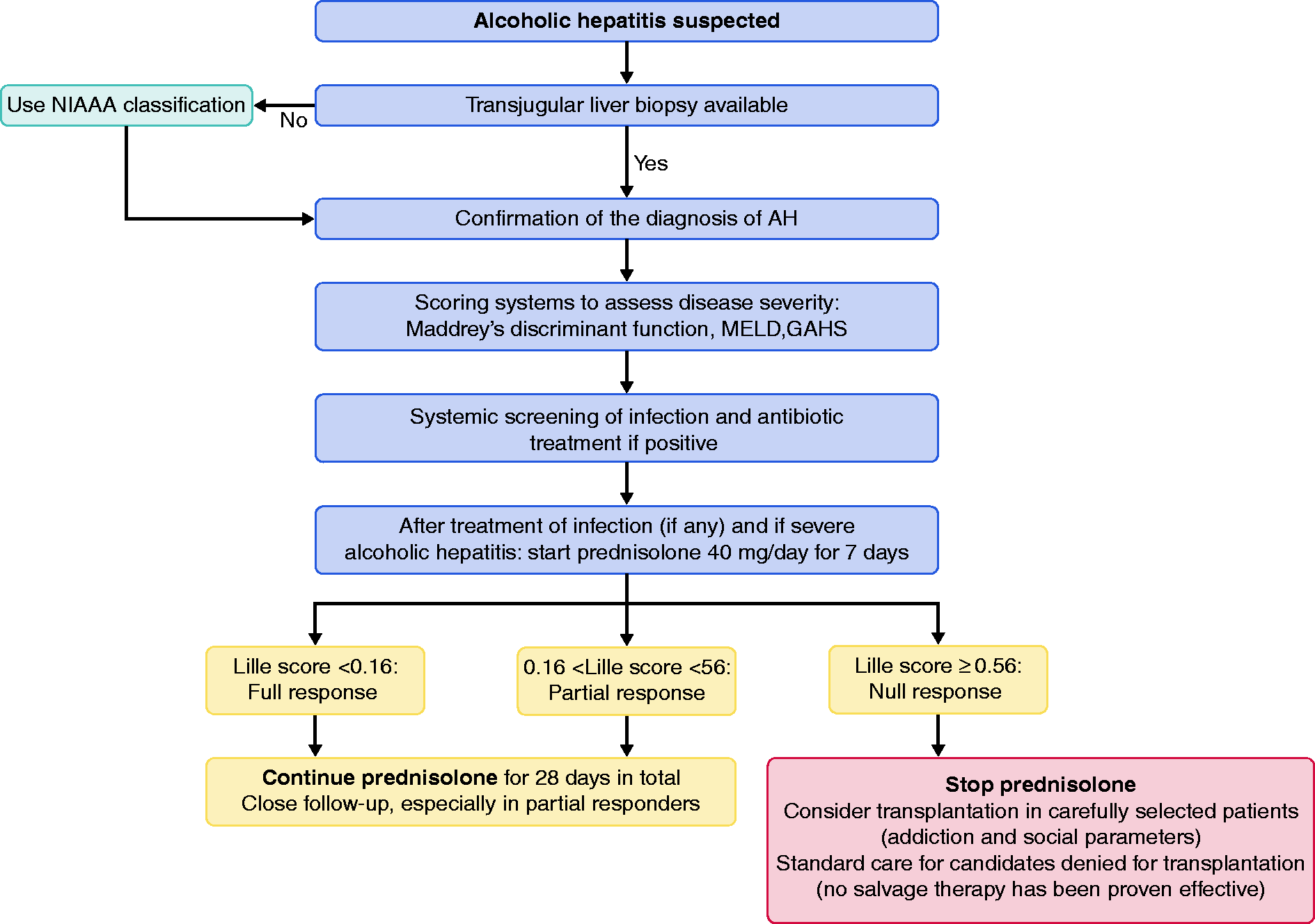
Treatment algorithm for patients with suspected alcoholic hepatitis (AH). AH is diagnosed with liver biopsy or using the National Institute on Alcohol Abuse and Alcoholism (NIAAA) classification. Scoring systems assess disease severity and patients with severe AH should be treated with corticosteroids (CSs). The Lille model for AH predicts mortality in patients with AH treated with CSs, and early liver transplantation may be considered in carefully selected non-responder patients.
Management of alcohol use disorder is a major problem because relapse drinking is the main driver of mortality after the acute episode. 42 , 43 Of note, no pharmacological intervention to treat alcohol addiction has been proven safe and effective in patients with decompensated cirrhosis, except baclofen at low doses. 44
Conclusions
In recent years, progress has been made in the management of patients with AH, including a more personalised approach for these patients. However, novel medical options are urgently needed to improve the survival of these patients, especially for non-responders to CS therapy.
Footnotes
Acknowledgements
The authors acknowledge the contribution of a medical writer, Sandy Field, for English language editing and formatting of this manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DD: has no conflicts to disclose, LCNW: has nothing to disclose, CM: has no conflicts to disclose, AL: has nothing to disclose.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
