Abstract
Background
Determining the prevalence of Barrett’s esophagus is important for defining screening strategies. We aimed to synthesize the available data, determine Barrett’s esophagus prevalence, and assess variability.
Methods
Three databases were searched. Subgroup, sensitivity, and meta-regression analyses were conducted and pooled prevalence was computed.
Results
Of 3510 studies, 103 were included. In the general population, we estimated a prevalence for endoscopic suspicion of Barrett’s esophagus of (a) any length with histologic confirmation of intestinal metaplasia as 0.96% (95% confidence interval: 0.85–1.07), (b) ≥1 cm of length with histologic confirmation of intestinal metaplasia as 0.96% (95% confidence interval: 0.75–1.18) and (c) for any length with histologic confirmation of columnar metaplasia as 3.89% (95% confidence interval: 2.25–5.54) . By excluding studies with high-risk of bias, the prevalence decreased to: (a) 0.70% (95% confidence interval: 0.61–0.79) and (b) 0.82% (95% confidence interval: 0.63–1.01). In gastroesophageal reflux disease patients, we estimated the prevalence with afore-mentioned criteria to be: (a) 7.21% (95% confidence interval: 5.61–8.81) (b) 6.72% (95% confidence interval: 3.61–9.83) and (c) 7.80% (95% confidence interval: 4.26–11.34). The Barrett’s esophagus prevalence was significantly influenced by time period, region, Barrett’s esophagus definition, Seattle protocol, and study design. There was a significant gradient East-West and North-South. There were minimal to no data available for several countries. Moreover, there was significant heterogeneity between studies.
Conclusion
There is a need to reassess the true prevalence of Barrett’s esophagus using the current guidelines in most regions. Having knowledge about the precise Barrett’s esophagus prevalence, diverse attitudes from educational to screening programs could be taken.
Introduction
The incidence of esophageal adenocarcinoma (EAC) has been increasing. 1 The poor prognosis of EAC has focused interest on Barrett’s esophagus (BE). Identification of BE with treatment of dysplasia is important to prevent invasive cancer. On the other hand, gastroesophageal reflux disease (GERD) has been associated with an increased risk of BE. Despite recommendations against population-based screening, the majority of guidelines recommend considering screening of chronic GERD patients. However, 50% of EAC patients report no previous GERD.2–5 Therefore, understanding the epidemiology of BE is difficult because the majority remain undiagnosed. Indeed, the prevalence of BE is unknown: some studies estimate a 15% prevalence in chronic GERD patients and 1–2% in the general population. 6 The BE prevalence (priori probability) is important to define and evaluate screening strategy.
BE has been an area of controversy.1–3 Its definition and practice have varied along time and across the world.7,8 The Prague classification, described in 2006, has improved BE diagnosis and increased reliability in reporting. 9 While the European, American Society for Gastrointestinal Endoscopy and American College of Gastroenterology define BE as intestinal metaplasia (IM) lining in the distal esophagus with a minimum length of 1 cm, the American Gastroenterological Association defines it as any extent of intestinal metaplasia and the British Society as any columnar metaplasia (CM) (fundic type, cardiac type, and intestinal type) lining in the distal esophagus with a minimum length of 1 cm.2–5
We aimed to synthesize the data of all studies assessing BE prevalence and determine its prevalence. We also aimed to assess the variability of BE prevalence according to the definition, geographical region, time period, and method used in order to identify the best methodology to determine it.
Methods
Search strategy
We conducted a systematic review according to the PRISMA guidelines. The research question was defined using the PICO acronym (P-population, I-intervention, C-comparator, O-outcome): what is the BE prevalence (O) in different settings (P) and which are the methodologies used (I). A sensitive search in PubMed, Scopus and Web of Knowledge was performed using the following query: “(prevalence(Title) OR epidemiolog*(Title) OR incidence(Title) OR risk(Title) OR screen*(Title)) AND (Barrett’s esophagus(Title/Abstract) OR Barrett(Title/Abstract) OR columnar-lined esophagus(Title/Abstract)).”
Selection of manuscripts
Inclusion criteria were original full-text articles published up to September 2018 and addressing BE in general and the GERD population, that met the globally accepted criteria for BE (endoscopic suspicion of BE (ESBE) with or without histologic confirmation of any CM or IM) and with a sample size of ≥100 individuals. The general population was defined as any population with or without gastrointestinal symptoms or with or without indication for esophagogastroduodenoscopy (EGD). Exclusion criteria were reviews, case-reports, case-control studies with BE cases, and non-English, non-French, non-Spanish, and non-Portuguese language. An attempt to contact corresponding authors was made when the full text was not available. When similar data was identified, we included the most recent report.
After all references (n = 3510) were imported and duplicated records were discarded, the records (n = 1531) were screened by title and abstract by two independent investigators (gastroenterology trainees). Concordance between the reviewers was measured by proportion of agreement and k-statistics achieving a good agreement (83% of agreement). Full-text eligibility was done by the same investigators. Conflicts were resolved through discussion.
Data extraction and quality assessment
A data extraction sheet was developed. Data were extracted independently by both investigators and then cross-checked by one. Studies were divided into the general and GERD population. The pooled BE prevalence was estimated according to its definition: ESBE with histologic confirmation of IM (any length or ≥1 cm of length), ESBE with histologic confirmation of CM (any length or ≥1 cm of length) and ESBE only (any length or ≥1 cm of length). The group “ESBE any length” included the studies from the group “ESBE≥1 cm of length.” For studies assessing ESBE with histologic confirmation of IM, sources of variability were evaluated.
The appraisal of study quality was done independently by both investigators using the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) checklist and the NewCastle-Ottawa Scale. Discrepancies were resolved through discussion.
Statistical analysis
The pooled prevalence was calculated using the random-effect model. Heterogeneity across studies was assessed by I2 statistic (<30%, 30–60%, 60–75% and >75% suggestive of low, moderate, substantial, and considerable heterogeneity, respectively). We explored sources of heterogeneity by subgroup analysis, sensitivity analysis, and meta-regression. We used the z-test for the logit of the prevalence to examine the impact of different factors (year of publication, geographical setting, sample size, percentage of male, type of sampling, Seattle protocol, and ESBE length) on estimates. After logit transformation, we analyzed publication bias using funnel plot and Egger’s test. Statistical significance was set at p<0.05. The STATA software was used.
Results
Study characteristics
After screening, 175 records were assessed for eligibility (Figure 1). We did not have access to 3% of these articles, with 66% unrecoverable records dating before 2006. We finally included 80 studies assessing BE prevalence in the general population (ESBE with IM: 49, ESBE with CM: 8 and ESBE only: 23) and 23 in the GERD population (ESBE with IM: 17, ESBE with CM: 1 and ESBE only: 5). Characteristics of studies are depicted in Table 1.10–112 Of the 80 studies in the general population, 12 studies performed a subgroup analysis in GERD patients: 11 studies were included in the GERD analysis,10,15,19,23,27,30,32,42,43,56,79 but one was not included due to the small sample (n = 26). There was considerable heterogeneity among studies. There was no data available regarding BE prevalence in the general and GERD populations for the majority of countries (Figures 1 and 2, Supplementary Material).
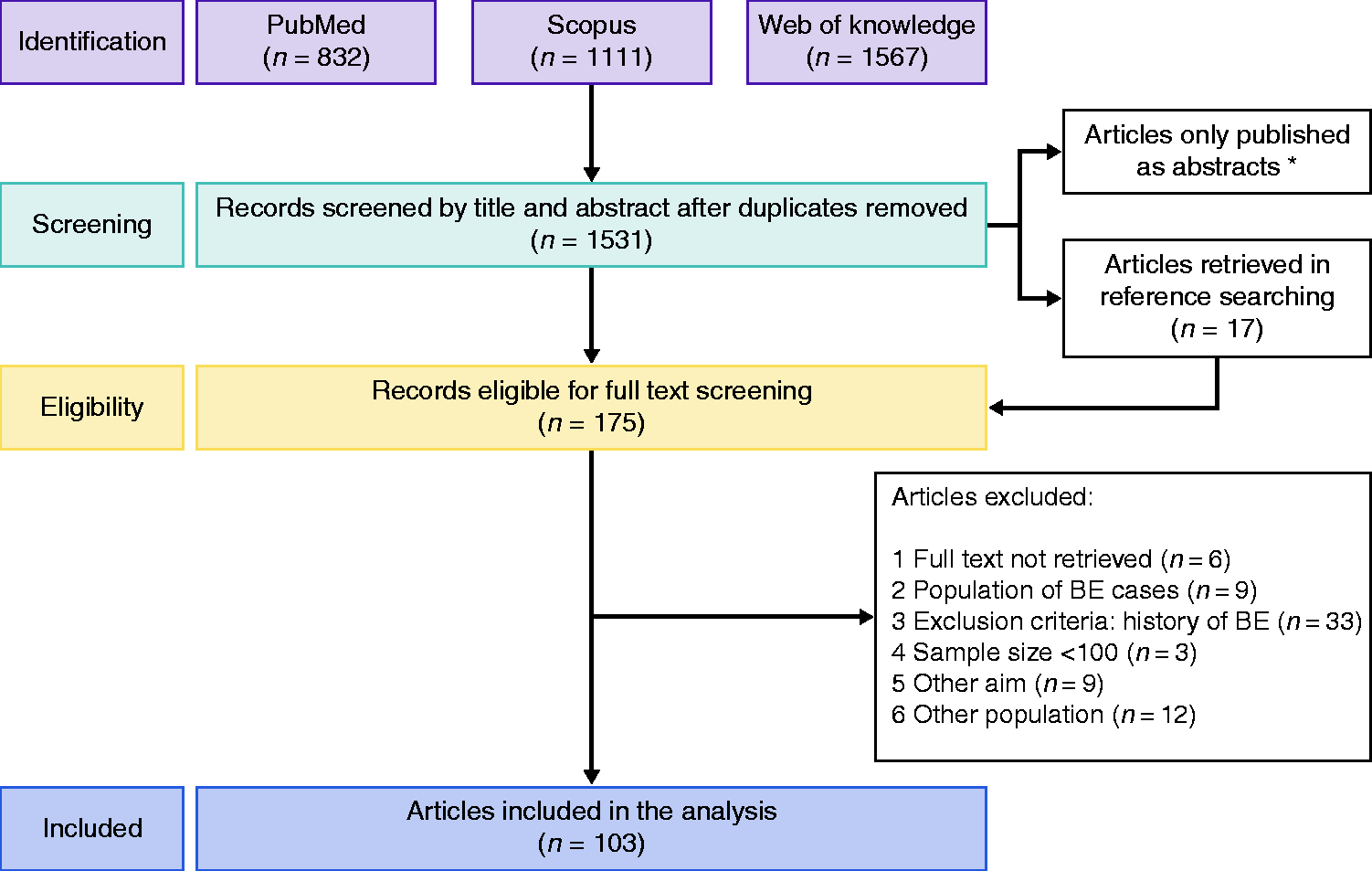
Flowchart of study selection. (*51 articles only published as abstracts). BE: Barrett’s esophagus.
Characteristics of included studies.
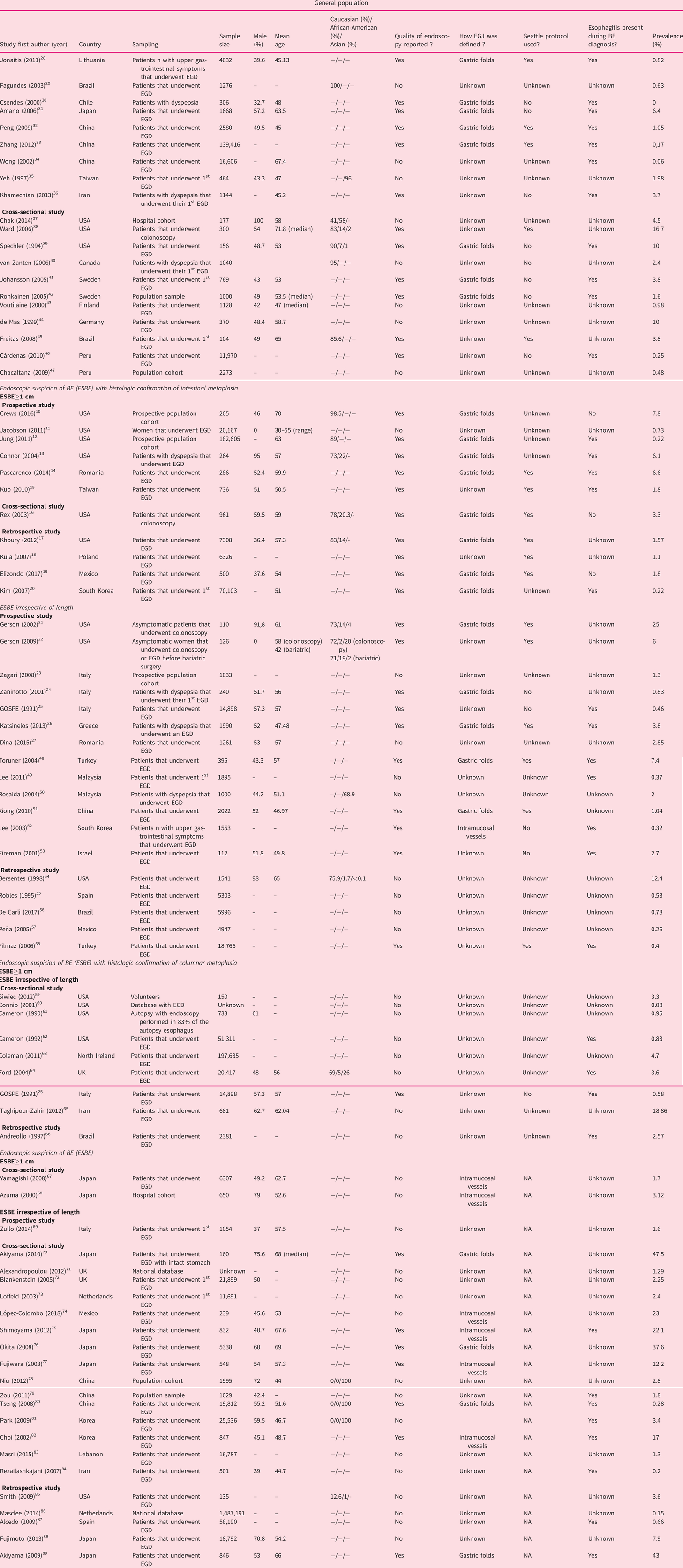
BE: Barrett’s esophagus; EGD: esophagogastroduodenoscopy; EGJ: means esophagogastric junction; ESBE: endoscopic suspicion of Barrett’s esophagus; GERD: gastroesophageal reflux disease; GOSPE: Gruppo Operativo per lo Studio delle Precancerosi dell’Esofago; NA: not applicable.
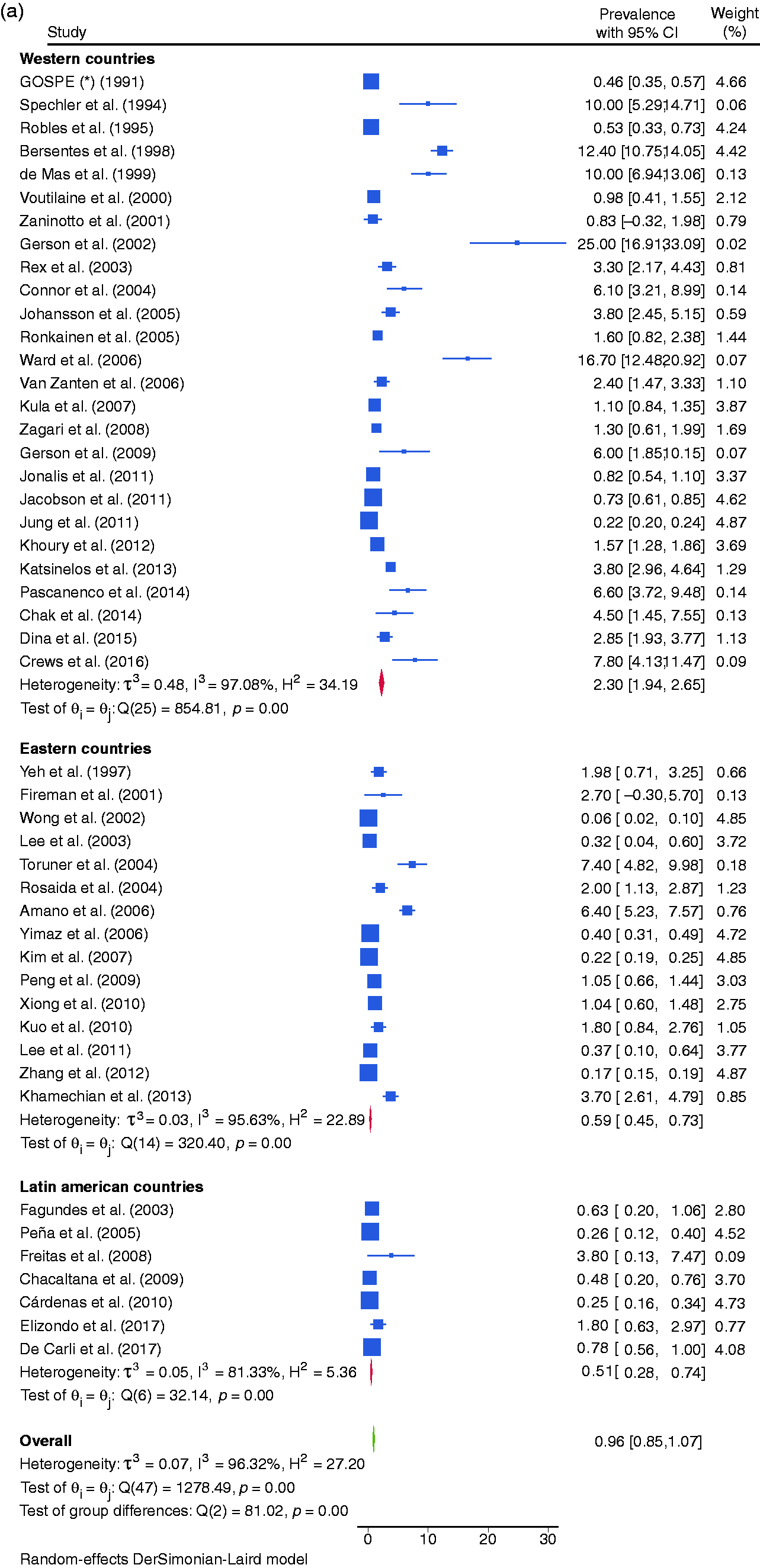
Forest plot of all studies assessing the prevalence of endoscopic suspicion of Barrett’s esophagus (ESBE) any length with histologic confirmation of intestinal metaplasia (IM) (a), of ESBE ≥1 cm of length with histologic confirmation of IM (b), and of ESBE any length with histologic confirmation of columnar metaplasia (CM) (c) in general population.
BE prevalence according to various definitions
General population
The BE prevalence varied according to definition: 0.96% (95% confidence interval (CI95%): 0.85–1.07) for ESBE any length with IM, 0.96% (CI95%: 0.75–1.18) for ESBE ≥1 cm with IM, 3.89% (CI95%: 2.25–5.54) for ESBE any length with CM, 7.04% (CI95%: 6.35–7.74) for ESBE any length and 2.26% (CI95%: 0.90–3.61) for ESBE with ≥1 cm (Figure 2).
Regarding ESBE with IM, we analyzed the statistical outliers (Figure 2). Ward et al. (16.7% of BE prevalence) and Spechler et al. (10%) conducted studies in a population with a high prevalence of GERD. In the study of Spechler et al. and Pascarenco el al. (6.6%) esophagitis was present. Bersentes et al. (12.4%), Gerson et al. (2002) (20%) and Connor et al. (6.1%) conducted studies with a high percentage of males. In the study of Gerson et al. (2009) (6%), 50% of the population was obese/overweight. Bersentes et al., De Mas et al. (10%), Amano et al. (6.4%) and Toruner et al. (7.4%) did not consider ESBE≥1 cm. Crews et al. (7.8%) estimated BE prevalence by EGD and transnasal endoscopy in an older population. By excluding these outliers, the prevalence was: BE any length with IM – 0.70% (CI95%: 0.61–0.79); BE ≥1 cm with IM – 0.82% (CI95%: 0.63–1.01).10,13,14,21,22,31,38,39,44,48,54
GERD population
The BE prevalence varied based on the definition: 7.21% (CI95%: 5.61–8.81) for ESBE any length with IM, 6.72% (CI95%: 3.61–9.83) for ESBE ≥1 cm with IM, 7.80% (CI95%: 4.26–11.34) for ESBE any length with CM and 6.51% (CI95%: 3.34–9.68) for ESBE any length.
Regarding ESBE with IM, we analyzed the statistical outliers (Figure 3). Ward et al. (19.8% of BE prevalence) conducted a study in an older population. In the study of Jobe et al. (29.8%) and Ramirez et al. (27%) >80% were men. Banki et al. (26%) assessed patients that warrant evaluation for antireflux surgery. By excluding these studies, the pooled prevalence of ESBE any length with IM was 4.53% (CI95%: 3.46–5.60).38,96,97,104
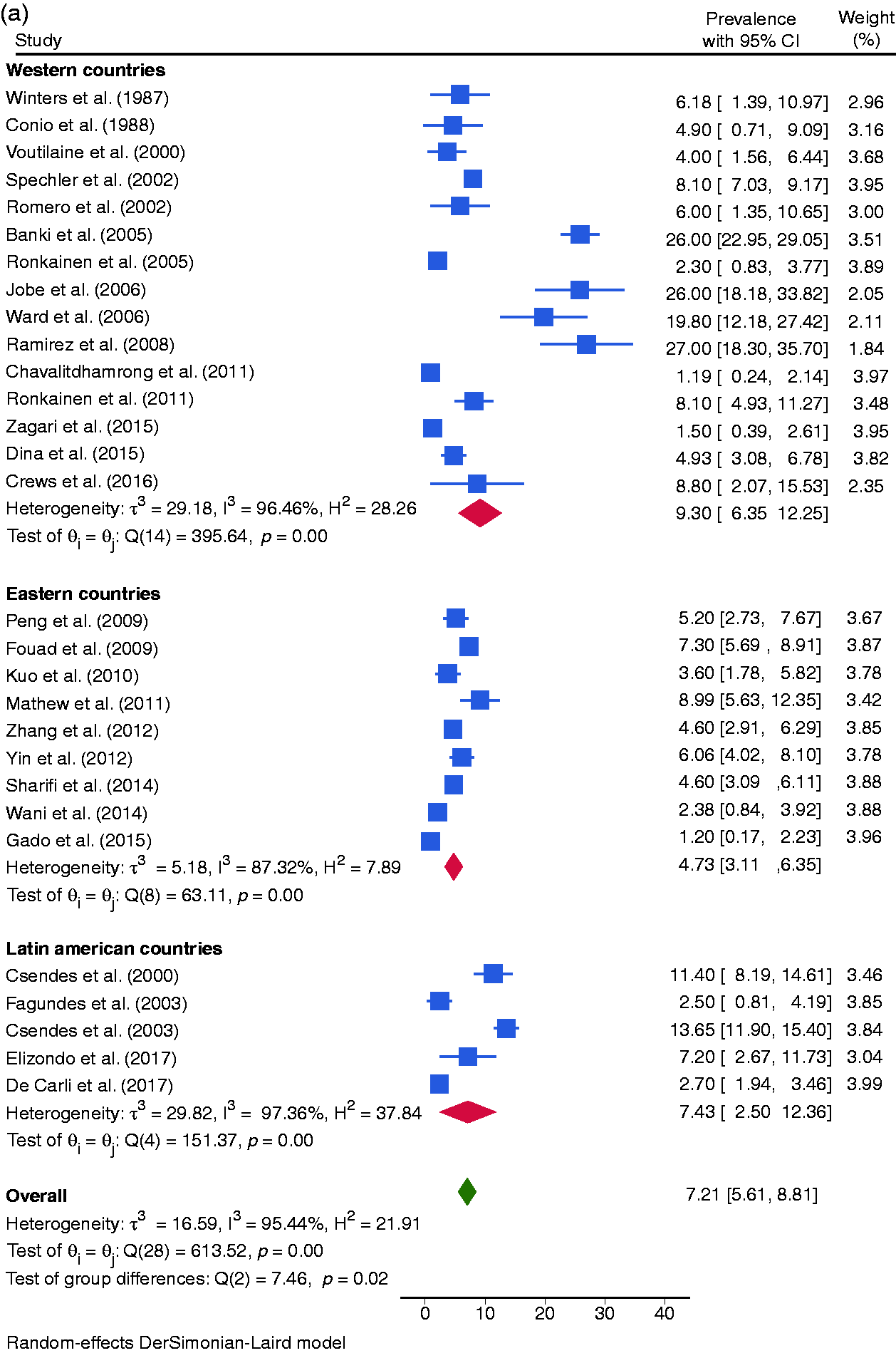
Forest plot of all studies assessing the prevalence of endoscopic suspicion of Barrett’s esophagus (ESBE) any length with histologic confirmation of intestinal metaplasia (IM) (a), of ESBE ≥1 cm of length with histologic confirmation of IM (b), and of ESBE any length with histologic confirmation of columnar metaplasia (CM) (c) in the gastroesophageal reflux disease (GERD) population.
Variability in the prevalence of BE
Geographical setting
General population
The pooled prevalence of BE in Western, Eastern and Latin American countries was 2.30% (CI95%: 1.94–2.65), 0.59% (CI95%: 0.45–0.73), and 0.51% (CI95%: 0.28–1.07) respectively (Figure 4). There was a North-South gradient in the West: in North was 2.97% (CI95%:2.44–3.50) and in South was 1.72% (CI95%:1.09–2.36).
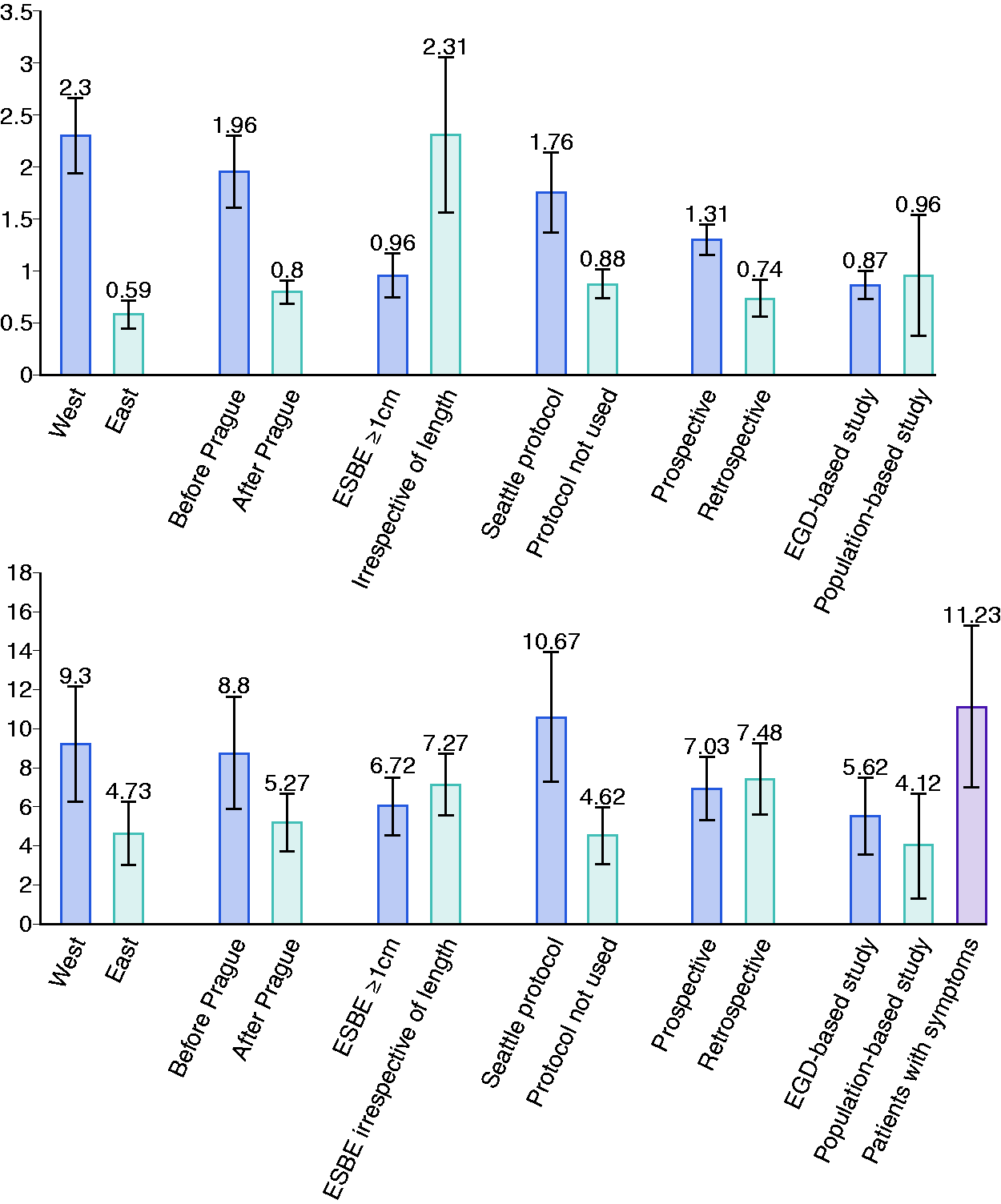
Subgroup analysis of the pooled prevalence of Barrett’s esophagus (BE) in general population (a), and in gastroesophageal reflux disease (GERD) patients (b) for geographical setting, date of publication, length of endoscopic suspicion of Barrett’s esophagus (ESBE) considered, use of Seattle protocol, study design, and type of sampling.
GERD population
The pooled prevalence of BE in Western, Eastern, and Latin American countries was 9.30% (CI95%: 6.35–12.25), 4.73% (CI95%: 3.11–6.35) and 7.43% (CI95%: 2,50–8.81) respectively (Figure 4).
Time trends
General population
There was a temporal trend with the most recent studies reporting lower estimates especially in the West. Indeed, the BE prevalence was 0.80% (CI95%: 0.69–0.92) when considering only the studies published after 2006 (date of publications of Prague classification) while it was 1.96% (CI95%: 1.62–2.30) when considering only the studies published before 2006. This difference was also found in a subgroup analysis for Western (before: 4.05% (CI95%: 3.16–4.95) vs after: 1.71% (CI95%: 1.25–2.17)) and Eastern countries (before: 1.28% (CI95%: 0.81–1.74) vs after: 0.54% (CI95%: 0.45–0.73)) (Figure 4).
GERD population
Likewise, there was a temporal trend: 5.27% (CI95%: 3.79–6.76) when considering only the studies published after 2006 vs 8.80% (CI95%: 5.91–11.69) when considering only the studies published before 2006 (Figure 4).
Length of BE
General population
When comparing studies that considered ESBE with ≥1 cm with those that did not, the prevalence of BE was significantly different (0.96% (CI95%: 0.75–1.18) vs 2.31% (CI95%: 1.56–3.05)) (Figure 4).
GERD population
The prevalence of BE was similar between studies that considered ESBE ≥1 cm and studies that did not (6.72% (CI95%: 3.61–9.83) vs 7.27% (CI95%: 5.53–8.81)) (Figure 4).
Use of the Seattle protocol
General population
When comparing studies that used Seattle protocol with those that did not, the prevalence was significantly different (1.76% (CI95%: 0.85–1.07) vs 0.88% (CI95%: 0.74–1.02)) (Figure 4).
GERD population
When comparing studies that used Seattle protocol with those that did not, the prevalence was significantly different (10.67% (CI95%: 7.31–14.02) vs 4.62% (CI95%: 3.13–6.12))(Figure 4).
Study characteristics
General population
The studies with larger sample size, lower male percentage, and lower mean/median age had the lowest BE prevalence. Prospective studies estimated a higher BE prevalence than retrospective studies (1.34% (CI95%: 1.16–1.53) vs 0.74% (CI95%: 0.56–0.91)). When comparing studies that estimated BE prevalence in patients undergoing EGD and studies that estimated BE prevalence in population cohorts/sample (these studies proposed endoscopy to individuals from a cohort conducted for unrelated reasons to BE or GERD or from a population sample) there was no difference (0.96 (CI95%: 0.85–1.07) vs 0.87 (CI95%: 0.74–1.01)) (Figure 4).
GERD population
The studies with the larger sample size, lower percentage of males, and with lower mean/median age had a lower prevalence. The prevalence was similar between prospective and retrospective studies. There was no difference between studies that estimated BE prevalence in patients undergoing EGD and studies that estimated BE prevalence in population cohorts/sample. However, the BE prevalence significantly increased to 11.23% (CI95%: 7.04–15.41) if studies assessed patients with GERD (Figure 4).
Meta-regression
General population
The univariate analysis showed a significant relationship between the BE prevalence and the geographical setting, sample size, and percentage of males. Considering the univariate and the subgroup analysis, we included publication date, geographical setting, sample size, percentage of male, and ESBE≥1 cm as independent variables in a multivariate analysis. Although only sample size had a significant impact on BE prevalence (p<0.0001), 55.63% of cross-study variance could be explained by this model (R2 = 55.63%).
GERD population
The univariate analysis showed a significant relationship between pooled prevalence and the use of Seattle protocol. Considering the univariate and the subgroup analysis, we included year of publication, geographical setting, sample size, percentage of male, type of sampling, and Seattle protocol in a multivariate analysis. Although 64.96% of the variance inter-studies was explained by this model, only year of publication (p = 0.011) and type of sampling (p = 0.016) had a significant impact on BE prevalence.
Quality assessment
Among the 103 studies, 65% were at a high risk of bias due to sample characterization and 87% due to endoscopic practice (Supplementary Material Figure 3).
Publication bias
For the general population, the funnel plot and the Egger test (p = 0.1177) indicated low risk of bias. For the GERD population, although the funnel plot indicated low risk of bias, the p-value was 0.0016 with a z-statistic of -2.06 in the Egger test (Supplementary Material Figure 4).
Discussion
The poor prognosis of EAC has been focus attention to BE. Despite BE surveillance, EAC incidence has been rising, bringing into question whether the population at risk is being correctly identified. Indeed, the precise prevalence of BE is unknown.
This study is an important review assessing global BE prevalence according to the most recent guidelines and the first study assessing variability of BE prevalence due to BE definition, geographical region, time period, and methodology. We have found that there are no data available from the majority of the countries. In some countries, the estimations are higher than expected because of BE definition and methodology used (Supplementary Material Figures 1 and 2). We also found heterogeneity among the studies.
In the general population, we estimated a pooled prevalence of 0.96% (CI95%: 0.85–1.07) for ESBE any length with histologic confirmation of IM, 0.96% (CI95%: 0.75–1.18) for ESBE ≥1 cm of length with histologic confirmation of IM and 3.89% (CI95%: 2.25–5.54) for ESBE any length with histologic confirmation of CM. By excluding studies with high risk of bias, the prevalence of ESBE any length with histologic confirmation of IM was 0.70% (CI95%: 0.61–0.79) and of ESBE ≥1 cm of length with histologic confirmation of IM was 0.82% (CI95%: 0.63–1.01). Therefore, it would be necessary to perform endoscopy in 122 individuals of the general population for the diagnosis of one patient with BE. There is no difference in the prevalence between BE any length with IM and BE ≥1 cm with IM, because the first included the studies from the last group. Indeed, when comparing studies that considered ESBE with ≥1 cm with those that did not, the prevalence was significantly different (Figure 4). In GERD, we estimated a pooled prevalence of 7.21% (CI95%: 5.61–8.81) for ESBE any length with histologic confirmation of IM, 6.72% (CI95%: 3.61–9.83) for ESBE ≥1 cm of length with histologic confirmation of IM and, 7.80% (CI95%: 4.26–11.34) for ESBE any length with histologic confirmation of CM. The prevalence was significantly influenced by the date of publication, geographical setting, BE length, the use of Seattle protocol, and the study design. There was a significant gradient East-West and North-South of BE prevalence. By including studies prior to 2006, we found a temporal trend with the most recent studies reporting lower prevalence, particularly after Prague classification. 9 Moreover, the presence of esophagitis was a source of bias, probably by making BE diagnosis difficult. The impact of ethnicity was not assessed since the studies did not estimate BE prevalence by ethnic group.
Shiota et al. 113 conducted a systematic review of BE prevalence in Asia, estimating a BE prevalence of 1.3% (0.7–2.2) in the general population with high heterogeneity. This higher value might result from studies in high-risk patients and studies that diagnosed BE without IM confirmation. They also included studies that excluded patients with known BE (that estimated incidence). No study from this review was excluded in ours. 113 Qumseya et al. 114 have conducted a systematic review of 47 studies assessing BE prevalence in different risk populations. They included studies with less than 100 patients and studies that excluded patients with known BE. Moreover, the authors differentiate the prevalence of population without any risk factor (0.8%) from the prevalence of population with ≥50 years (6.7%). However, the majority of the studies included in the first group had a mean/median age higher than 50 years and the last group included studies with mean age of 45 years. 114
The limitations of our analysis extend from the various biases within each study and the heterogeneity among studies impacting our meta-analysis. Although the BE prevalence varied based on the definition, we have attempted to provide precise estimates based on the population being evaluated (general vs GERD), BE length (any vs ≥1 cm), using Seattle protocol and geographical location.
Our findings confirm the need for well-conducted studies in order to accurately estimate BE prevalence and decrease heterogeneity. In the future, prospective population-based or EGD-based studies with a minimum sample size of 100 individuals and according to the most recent guidelines, namely using the definition of BE with ≥1 cm and histologic confirmation of IM, the Prague classification, the Seattle protocol, and in the absence of esophagitis, should be conducted.
Having knowledge about precise BE prevalence, we could draw more tailored screening strategies and ultimately assess their impact.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620939376 - Supplemental material for The global prevalence of Barrett’s esophagus: A systematic review of the published literature
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620939376 for The global prevalence of Barrett’s esophagus: A systematic review of the published literature by Inês Marques de Sá, Pedro Marcos, Prateek Sharma and Mário Dinis-Ribeiro in United European Gastroenterology Journal
Footnotes
Acknowledgements
The author contributions were as follows. IM: screening, data extraction, quality assessment, statistical analysis, and writing the manuscript; PM: screening, data extraction, and quality assessment; PS: guidance and review; MD: guidance and review.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
