Abstract
Background
Pancreatic cancer is the fourth leading cause of cancer mortality. Most patients are diagnosed with advanced pancreatic cancer, either at locally advanced or metastatic stages, and have a high rate of malnutrition and weight loss which are associated with poor outcomes. Pancreatic exocrine insufficiency is one of the causes of malnutrition and weight loss in these patients. The prevalence and clinical consequences of pancreatic exocrine insufficiency in advanced pancreatic cancer are poorly investigated with heterogeneous results. We sought to determine the prevalence and clinical consequences of pancreatic exocrine insufficiency and the effect of pancreatic enzyme replacement therapy in patients with advanced pancreatic cancer by systematic review and meta-analysis.
Methods
Scopus, Medline, and Embase were searched for cohort studies or randomised clinical trials reporting pancreatic exocrine insufficiency and/or the effect of pancreatic enzyme replacement therapy in patients with advanced pancreatic cancer. We considered pancreatic exocrine insufficiency as an abnormal result on direct and/or indirect pancreatic exocrine function tests. Pancreatic enzyme replacement therapy was evaluated by its effect on survival and quality of life in patients with advanced pancreatic cancer.
Results
A total of 11 studies were included; seven studies reported the prevalence of pancreatic exocrine insufficiency and seven the effect of pancreatic enzyme replacement therapy in advanced pancreatic cancer. The pooled prevalence of pancreatic exocrine insufficiency in advanced pancreatic cancer was 72% (95% confidence interval: 55–86%), being significantly higher when tumours were located in the pancreatic head (relative risk = 3.36, 1.07–10.54; p = 0.04) six studies investigated the impact of pancreatic enzyme replacement therapy on survival/quality of life. Pancreatic enzyme replacement therapy was associated with 3.8 months (95% confidence interval: 1.37–6.19) survival benefit. Patients receiving pancreatic enzyme replacement therapy had a trend towards a better quality of life.
Introduction
Pancreatic cancer (PC) is the ninth most common cancer in Europe and the fourth leading cause of death from malignancy. 1 PC has a very poor prognosis with only a 24% one-year survival rate and 9% at 5 years. 2 Survival in PC is mainly influenced by stage at diagnosis; in patients with an early stage (around 10% at the diagnosis), surgical resection of the tumour can be performed with a 32% 5-years survival rate. However, approximately 80% of patients are diagnosed with locally advanced or metastatic disease with a five-year survival rate of only 1% and 3% respectively. 3
Despite advances in chemotherapeutics, the five-year survival rate among patients with advanced PC (APC) has only marginally improved over the past 40 years. 4 The reasons for this poor survival are multifactorial and include biological aggressiveness, diagnosis at advanced disease stage, and the fact that due to advanced age and comorbidities a large proportion of patients do not receive any treatment. 5
The poor performance status of PC patients is also due to malnutrition. At presentation, 30% of patients are malnourished, 80% report weight loss, and over a third have lost more than 10% of their initial body weight. 6 Anorexia and cachexia are frequent features of APC and reduce the patient’s ability to receive chemotherapy and influence survival and quality of life. 7 Improving the nutritional status, either with parenteral nutrition or oral supplementation, can increase the body weight, skeletal muscle mass, fat mass and quality of life, facilitating the administration of chemotherapy without interruption. 8
Malnutrition and weight loss in PC are explained by different mechanisms such as the Warburg effect, loss of appetite, tumour-derived factors which contribute to cachexia, silent subclinical inflammation and pancreatic exocrine insufficiency (PEI). 9 PEI seems to be one of the determinant factors of malnutrition in APC due to obstruction of the main pancreatic duct, fibrosis of the gland and loss of pancreatic exocrine tissue. PEI can present with gastrointestinal symptoms such as steatorrhoea as well with signs of malabsorption and deficiency of nutrients and vitamins. 10 According to an observational study, the degree of PEI measured by faecal elastase-1 (FE-1) was strongly associated with poor survival in APC. 11
Pancreatic enzyme replacement therapy (PERT) is the standard treatment for PEI. 12 Patients with APC under PERT gain weight significantly 13 and survive longer compared with those who are not receiving PERT in observational studies.14,15 However, the rate of PERT prescription even in the presence of symptoms that could be attributable to malabsorption is low in common practice. 16
A previous meta-analysis on PEI in PC reported a prevalence of PEI of 44% before surgery, increasing to 74% six months after surgery. 17 However, data about the prevalence of PEI in APC are limited and heterogeneous and the impact of PERT has not been analysed.
This study aims to assess the prevalence and clinical consequences of PEI in patients with APC, alongside the association of PERT with survival and quality of life.
Methods
Data sources and searches
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) criteria. All studies indexed in Scopus, Embase and Medline reporting the prevalence of PEI and/or the effect of PERT in APC outcomes were retrieved up to March 2020. The search strategy is listed in Supplementary Material Methods. Two researchers (DIG and BA) scrutinised all identified studies independently and decided jointly on those for inclusion. When there was a discrepancy, the senior authors (EdM and GC) arbitrated.
Study selection
Inclusion criteria were: (a) prospective observational studies or randomised controlled trials (RCTs) that reported the rate of PEI in patients with APC (locally advanced or metastatic PC); (b) prospective observational studies or RCTs that reported the effect of PERT on outcomes of APC. PEI was defined as an abnormal result on direct and/or indirect pancreatic exocrine function tests. 18 Exclusion criteria: (a) PEI due causes other than APC, including pancreatic resection; (b) data presented only on editorials, expert opinion reports, reviews, studies published as abstracts, case reports, letters, studies based on samples lower than 10 patients.
Data extraction and quality assessment
Two authors (DIG and BA) extracted data independently from the included studies using predefined standardised forms; those two authors independently scored the included studies, and two further authors (MK and NP) resolved any disagreement. The quality of observational studies was assessed using the Newcastle-Ottawa scale 19 with a total score ≥5 indicative of high quality; the quality of RCTs was assessed using the Jadad system 20 with a total score ≥3 indicative of high quality.
Outcomes of interest
The primary outcome was the proportion of patients diagnosed with PEI following the diagnosis of APC. The level of FE-1 of 100–200 µg/g was defined as mild to moderate PEI and <100 µg/g as severe PEI.
Secondary outcomes included symptoms of PEI, coefficient of fat absorption (CFA), body weight, survival, abdominal symptoms, the proportion of prediabetes and/or diabetes mellitus (DM), serum nutritional parameters and quality of life.
APC patients were defined as those diagnosed with unresectable pancreatic cancer (either locally advanced or metastatic disease) following the consensus-based guidelines from the National Comprehensive Cancer Network. 21
Data synthesis and statistical analysis
Pooled data were expressed as prevalence with 95% confidence interval (CI). Mean and standard deviation (SD) of continuous variables were used for meta-analyses when medians were given. Data for two group comparisons were expressed as relative risk (RR) or risk difference (RD) with 95% CI. Continuous variables were expressed as mean differences (MDs). Stats Direct V3.4 (StatsDirect Ltd, Cheshire, UK) and Review Manager V.5.3 software (Cochrane Collaboration, Oxford, UK) were used to generate forest plots of pooled data using a random effects model to deliver the most conservative estimates. Heterogeneity was evaluated using χ2. A value of p<0.1 was considered significant. Statistical heterogeneity was assessed using I2 values with cut-offs of 25%, 50% and 75% to indicate low, moderate and high heterogeneity, respectively. 22 Meta-analyses compared PERT versus baseline, PERT versus placebo or PERT versus no treatment.
Subgroup analyses examined high-quality studies, studies with sample sizes ≥30, Western population. Sensitivity analyses considered studies restricted to direct PEI tests, indirect PEI tests and FE-1 tests only. Meta-regression analyses determined the impact of publication year, patient age, gender and type of PEI test on the pooled prevalence estimate using Stata SE version 14 software (StataCorp LP, College Station Texas, USA); p<0.05 was considered significant. Publication bias was assessed visually by funnel plots and using p values generated from the pooled prevalence of PEI in APC as well as by subgroups according to Begg-Mazumdar and Egger et al.
Results
Characteristics of included patients
The PRISMA flow diagram is shown in Figure 1. Eleven studies were included. Study design and characteristics are shown in Table 1. One study was conducted in USA, 23 five in Europe,11,13,14,24,25, two in Japan,26,27 one in Australia, 28 one in New Zealand 29 and one in Korea. 30 There were four RCTs,13,27,28,30 three of PERT vs placebo13,28,30 and one of PERT vs no PERT. 27 Eight studies had a prospective cohort design11,24–26,29 and one had a retrospective design. 14 All seven studies11,14,23–26,29 scored by the Newcastle-Ottawa scale with Selection, Comparability, and Outcome compositions (Supplementary Material Table 1A) were of high quality. The four RCTs13,27,28,30 were all of high quality (Supplementary Material Table 1B). Seven studies reported sufficient data to calculate the pooled prevalence of PEI11,23–27,30 and seven the effects of PERT.13,14,26–30
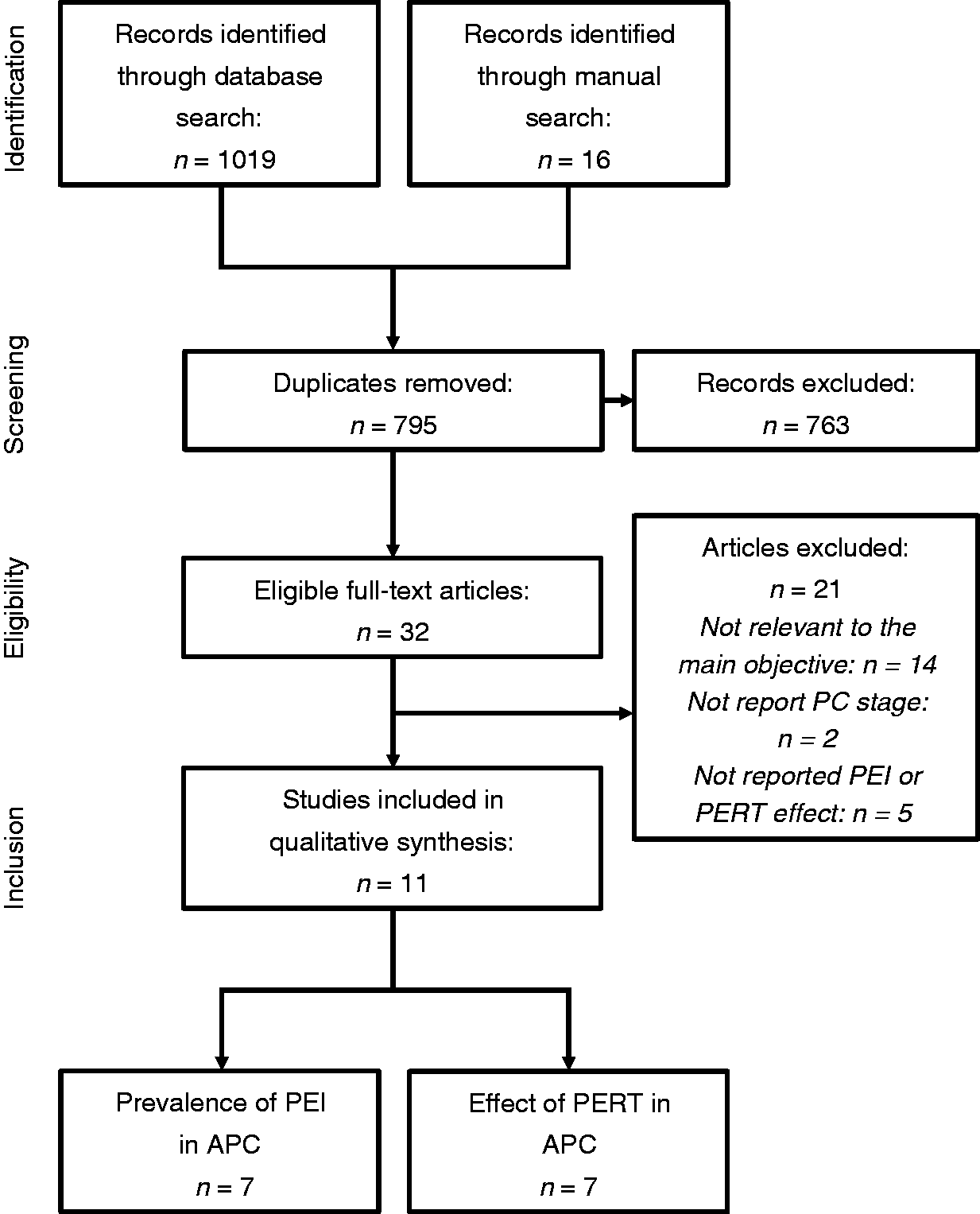
Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram. APC: advanced pancreatic cancer; PEI: pancreatic exocrine insufficiency; PERT: pancreatic enzyme replacement therapy; PC: pancreatic cancer.
Characteristic of studies and patients Included.
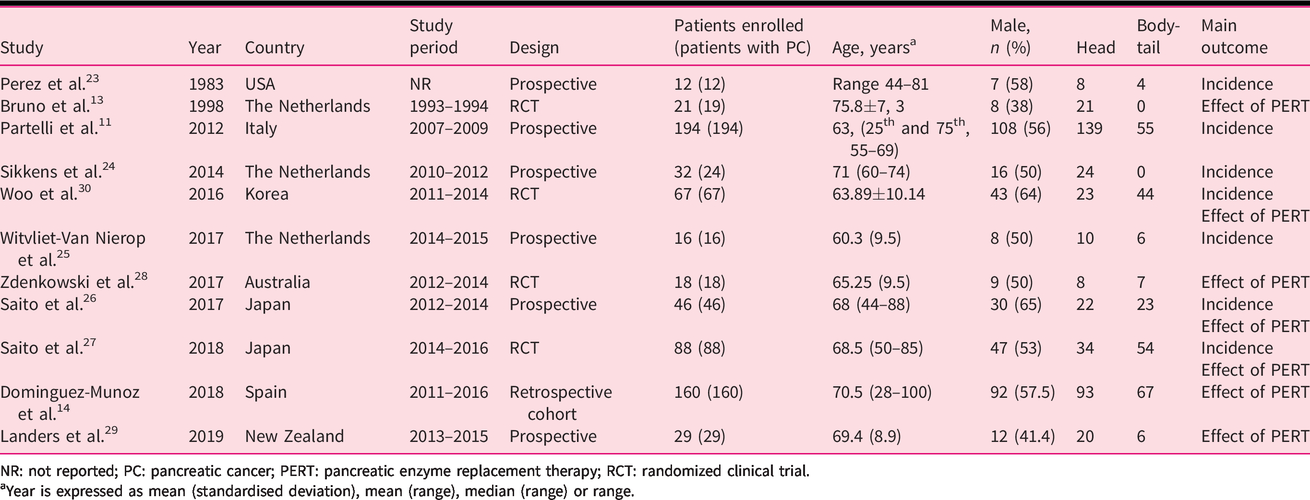
NR: not reported; PC: pancreatic cancer; PERT: pancreatic enzyme replacement therapy; RCT: randomized clinical trial.
aYear is expressed as mean (standardised deviation), mean (range), median (range) or range.
The patient’s characteristics included in the studies are shown in Table 1. Six hundred and seventy-three APC patients were included, with data on PEI available in 513 patients. The average age of included patients was 66 years and 53% were males. The primary tumour was located at the head of the pancreas in 402 patients (59.7%), at the body-tail in 266 (39.5%), while in the remaining cases the site of the primary lesion was not reported.
Pooled prevalence of pancreatic exocrine insufficiency in APC
Data on pancreatic function, PERT composition and administration schedule, and clinical outcomes are presented in Table 2. Four studies reported the rate of patients with previous history of DM.11,13,24,26 PEI was defined by FE-1 <200 μg/g in four studies,11,24,30 by N-benzoyl-L-tyrosyl-P-aminobenzoic acid (NBT-PABA) test in two studies,26,27 by triolein breath test and/or oleic acid breath test and or CFA <93% in one study, 23 by CFA<85% and/or FE-1<200 μg/g and/or faecal fat excretion >20 g/d in one study 25 and by clinical suspicion and/or weight loss >10% in two studies.14,28 Data about PERT use were reported in all 11 studies: eight studies also reported the length of follow-up11,13,24,26–30 and six presented data about health status.13,25,27–30
Pancreatic function at baseline.
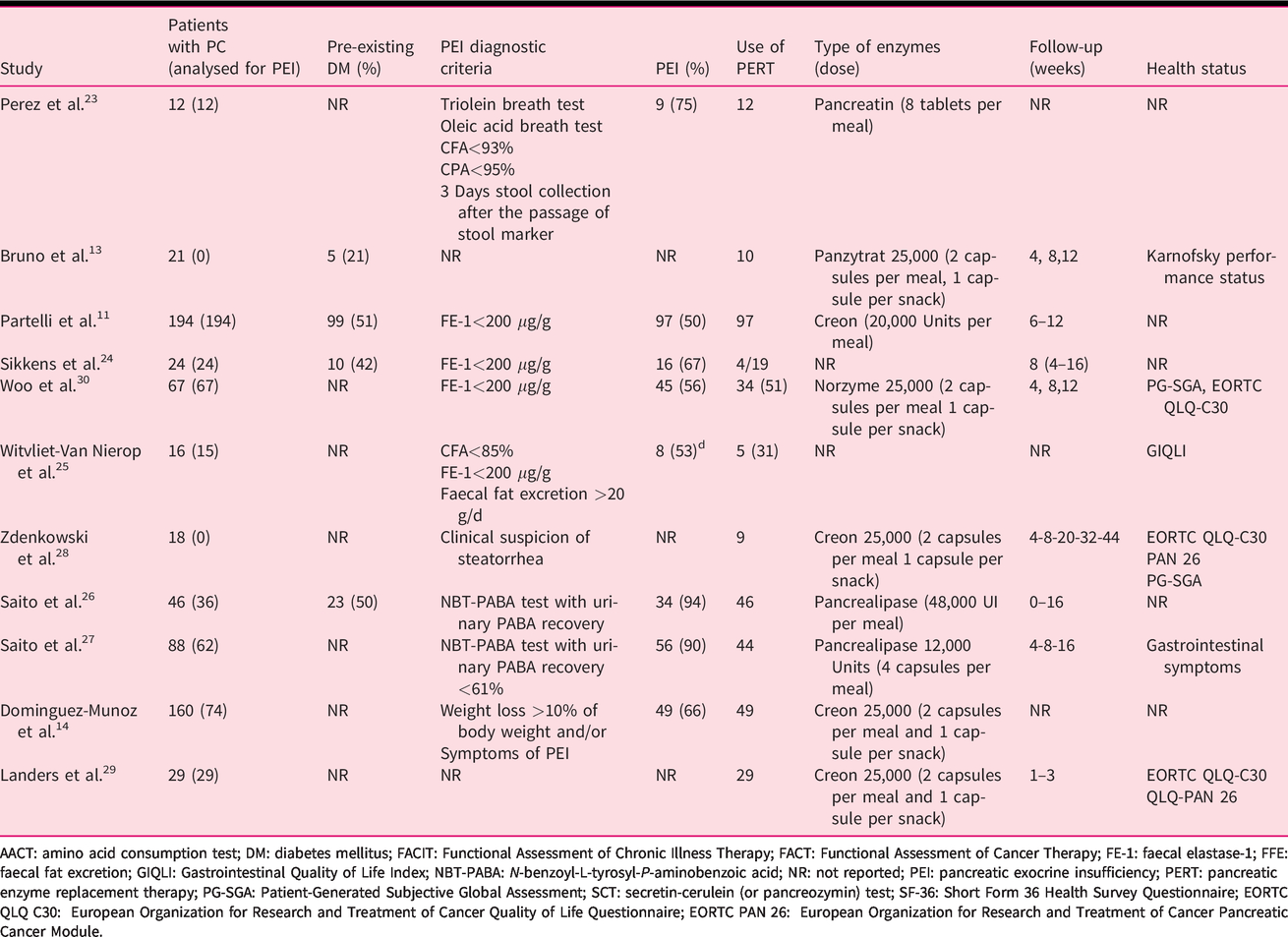
AACT: amino acid consumption test; DM: diabetes mellitus; FACIT: Functional Assessment of Chronic Illness Therapy; FACT: Functional Assessment of Cancer Therapy; FE-1: faecal elastase-1; FFE: faecal fat excretion; GIQLI: Gastrointestinal Quality of Life Index; NBT-PABA: N-benzoyl-L-tyrosyl-P-aminobenzoic acid; NR: not reported; PEI: pancreatic exocrine insufficiency; PERT: pancreatic enzyme replacement therapy; PG-SGA: Patient-Generated Subjective Global Assessment; SCT: secretin-cerulein (or pancreozymin) test; SF-36: Short Form 36 Health Survey Questionnaire; EORTC QLQ C30: European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; EORTC PAN 26: European Organization for Research and Treatment of Cancer Pancreatic Cancer Module.
A summary of the results of all the performed quantitative syntheses (meta-analyses) is shown in Table 3.
Results of meta-analysis.
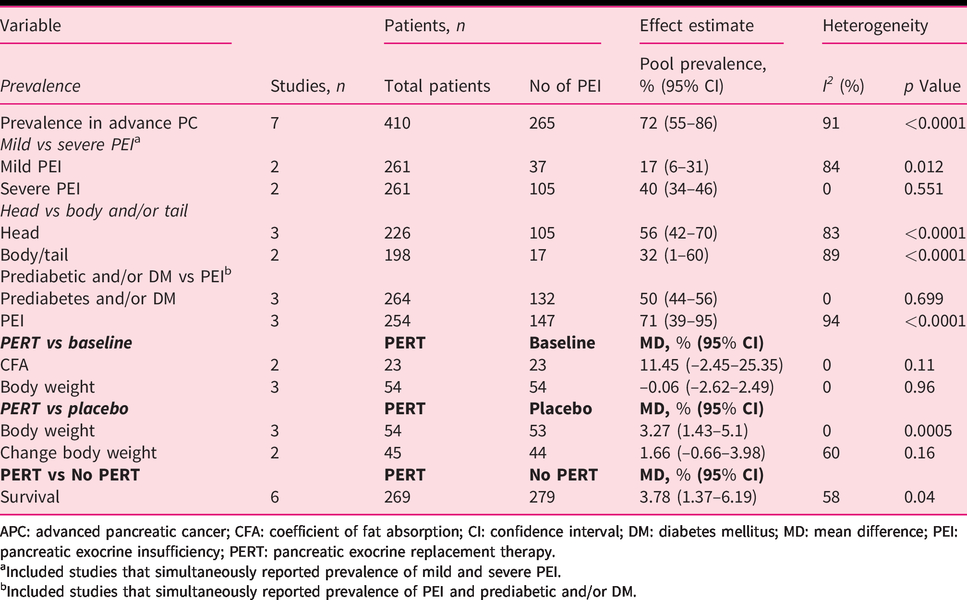
APC: advanced pancreatic cancer; CFA: coefficient of fat absorption; CI: confidence interval; DM: diabetes mellitus; MD: mean difference; PEI: pancreatic exocrine insufficiency; PERT: pancreatic exocrine replacement therapy.
aIncluded studies that simultaneously reported prevalence of mild and severe PEI.
bIncluded studies that simultaneously reported prevalence of PEI and prediabetic and/or DM.
As for the pooled prevalence of PEI, 410 patients from seven studies were included. The pooled prevalence of PEI was 72% (95% CI: 55–86%) with high statistical heterogeneity (I2 = 91%) (Figure 2(a)). Two studies reported prevalence of PEI depending on tumour location.11,23 The pooled prevalence of PEI in tumours in the pancreatic head was 56% (95% CI: 42–70%, I2 = 83%) and in the body-tail 32% (95% CI: 1–60%, I2 = 89%) with significantly higher prevalence in tumours located in the head of the pancreas (RR = 3.36, 1.07-10.54; p = 0.04). (Figure 2(b)).
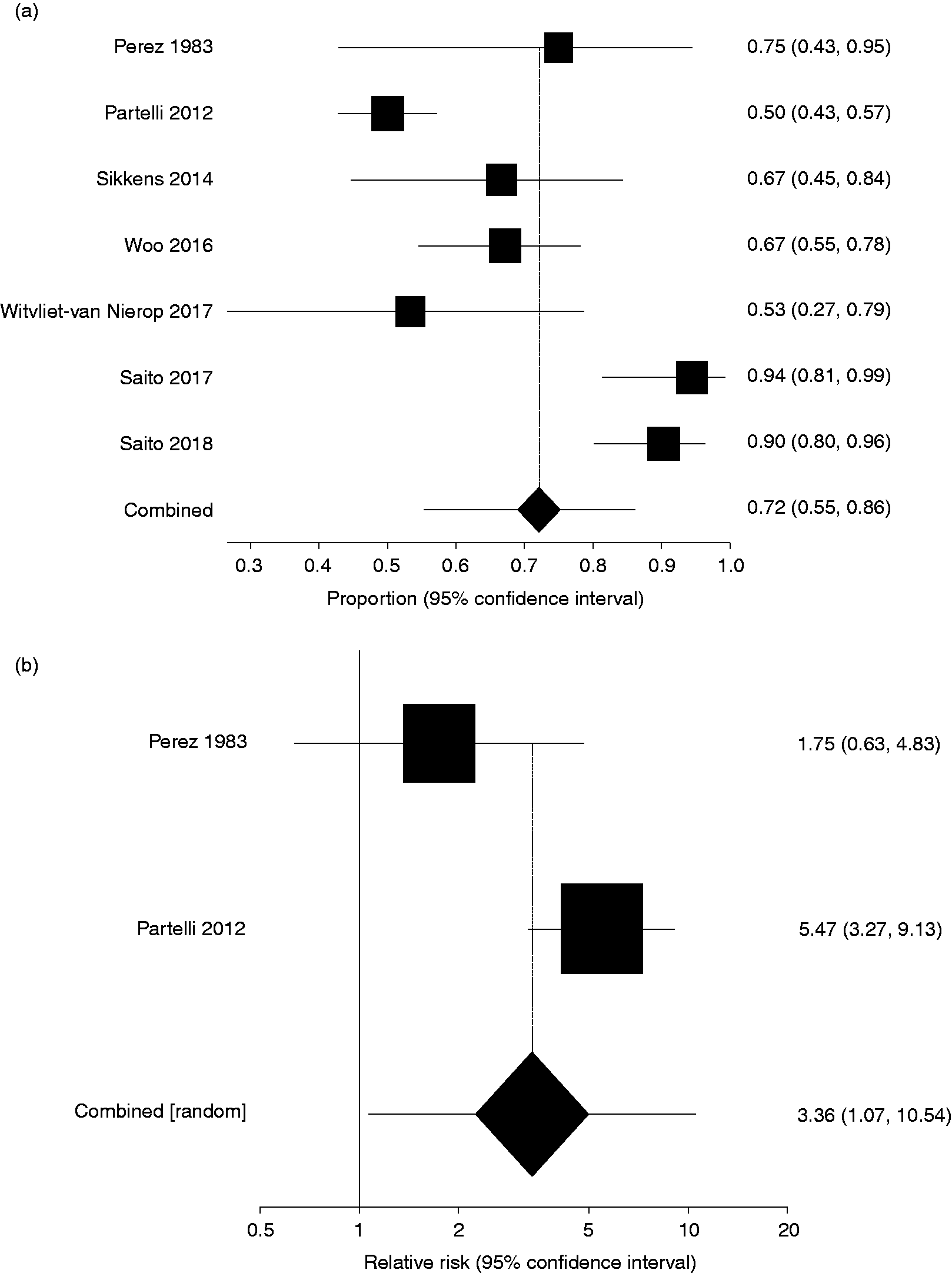
a) Pooled prevalence of pancreatic exocrine insufficiency (PEI). b) Relative risk of PEI between tumors located in the head of the pancreas and body-tail.
The pooled prevalence of pre-diabetes and/or DM was 50% (95% CI: 44–56%, I2 = 0%). In the studies reporting the prevalence of PEI and pre-diabetes and/or DM, the pooled prevalence of PEI was 71% (95% CI: 39–95%, I2 = 94%), without any statistically significant difference between PEI and pre-diabetes and/or DM (RR: 1.41, 0.86–2.30; p = 0.173, three studies)11,24,26 (see Supplementary Material Figures 2A and 2B).
Only two studies reported the severity of PEI based on FE-1.11,30 The pooled prevalence of mild to moderately severe PEI was 17% (95% CI: 6–31%, I2 = 84%) and that of severe PEI 40% (95% CI: 34–46%, I2 = 0%) (see Supplementary Material Figure 3).
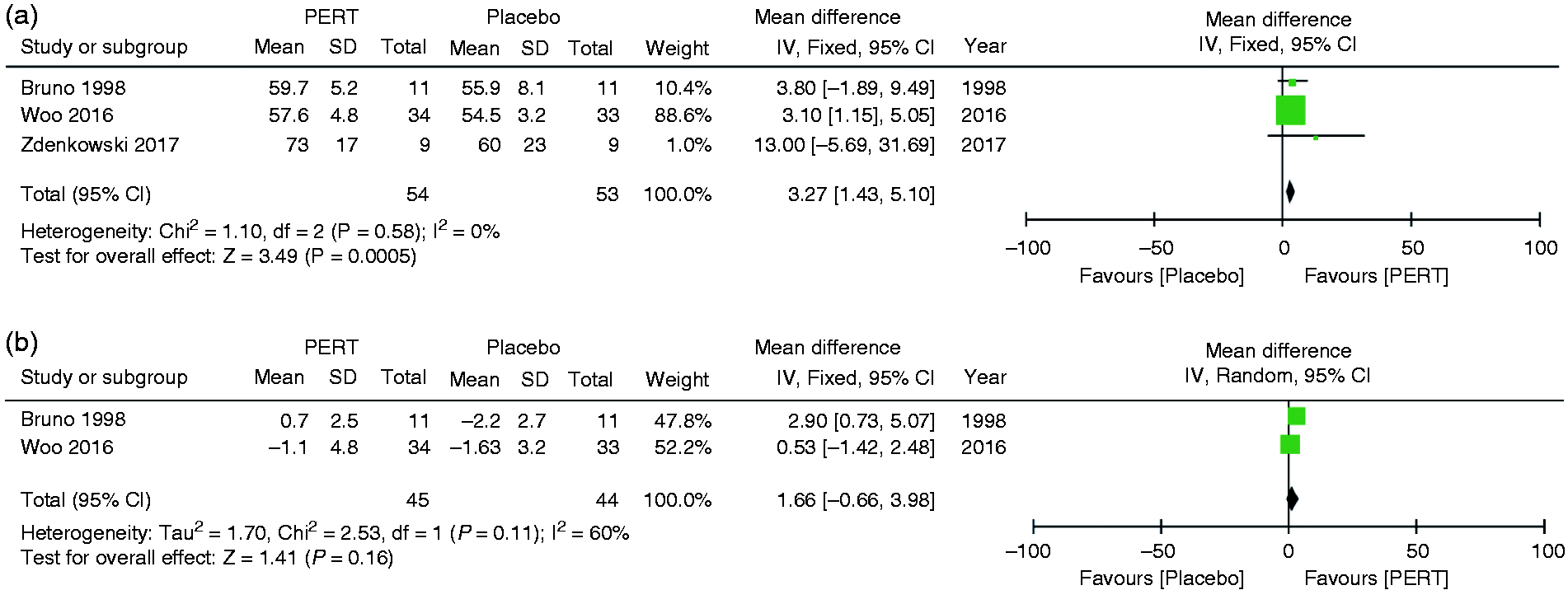
Clinical outcomes of patients under pancreatic enzyme replacement therapy (PERT) versus placebo. CI: confidence interval.
Subgroup analyses found that study quality, RCT design and sample size did not affect the prevalence of PEI in APC or cause statistical heterogeneity. Studies performed in Western countries reported a lower prevalence of PEI compared with the primary analysis 57% (95 CI: 46–67%) with lower heterogeneity (I2 = 35%) (see Supplementary Material Table 2). Sensitivity analyses showed that studies that used FE-1 had a lower pooled prevalence of PEI 60% (95% CI: 46–72%) and lower heterogeneity (I2 from 91% to 72%). The sensitivity analyses found that primary meta-analysis results were also affected by the use of indirect tests to diagnose PEI (81%, 95% CI: 64–94%; I2 = 76%) (Supplementary Material Table 3).
Meta-regression identified the type of test for the diagnosis of PEI (p = 0.044) as a contributing factor for heterogeneity. The model which included variables statistically associated with heterogeneity in the univariate analyses explained 85% of heterogeneity among studies. (Supplementary Material Table 4).
There were insufficient data for quantitative meta-analysis of the effects of PERT in quality of life, gastrointestinal symptoms and serum nutritional parameters.
Effect of PERT in patients with APC
The clinical outcomes of PERT versus baseline are presented in Table 3 and Supplementary Material Figure 1. Two studies13,23 reported the results in terms of changes of CFA with pooled results showing an increase of CFA after PERT versus baseline, albeit without statistical significance (80.45±20.8 vs 68.9±27.4; MD: 11.45, –2.45–25.35; p = 0.11) with no heterogeneity (I2 = 0%). Pooled data from three studies13,28,30 did not find any change in body weight after 8 weeks of PERT in comparison to baseline (p = 0.96). The clinical outcomes of patients under PERT versus placebo are displayed in Figure 3 and summarised in Table 3. Pooled data from three studies13,28,30 found that PERT led to a significant increase in body weight when compared to placebo after 8 weeks of treatment (MD: 3.27; 1.43–5.1; p = 0.0005). Pooled data from two studies13,30 reporting the change in body weight after PERT as compared to placebo did not show a statistically significant difference (p = 0.16).
The effect of PERT on survival is summarised in Table 3 and presented in Figure 4. Pooled data of six studies,11,14,26–28,30 demonstrated that the survival of patients who received PERT was significantly longer compared to that of those who did not (12.6±12.9 months vs 8.7±10.7 months; MD: 3.78, 1.37–6.19; p = 0.002).
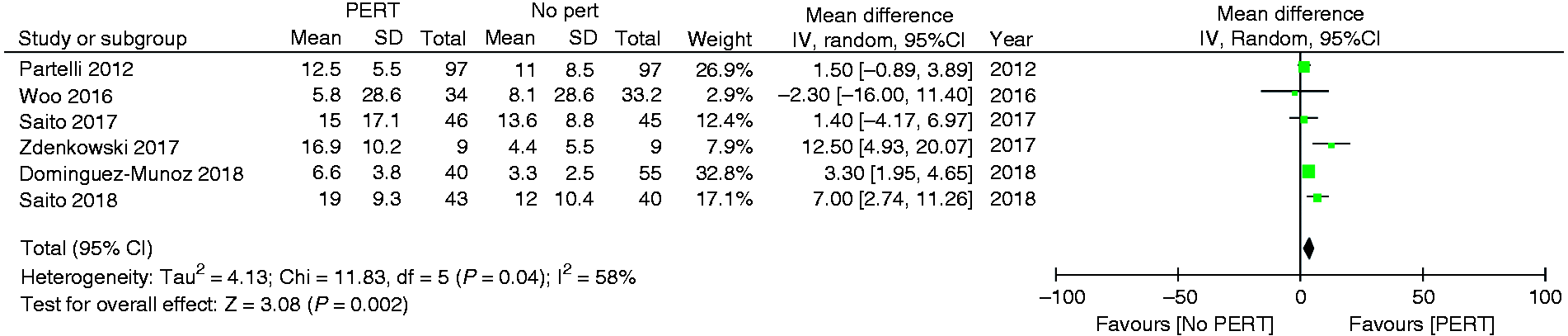
Effect of pancreatic enzyme replacement therapy (PERT) on survival. CI: confidence interval.
Publication bias
Funnel plots for publication bias are presented in Supplementary Material Figure 4. There was no evidence of publication bias either for the analysis on the prevalence of PEI in APC or for that on the effect of PERT on survival, body weight or CFA (Begg-Mazumdar and Egger tests p>0.1).
Discussion
This systematic review and meta-analysis conducted in patients with APC found that 72% of patients have PEI. PEI was not restricted to patients with the tumour in the head of the pancreas, as almost one-third (32%) of patients who had the tumour located in the body-tail were also found to have PEI. Treatment of PEI was associated with significantly longer survival compared with no treatment (12.6 vs 8.7 months, p = 0.002), and with the improvement of body weight and abdominal symptoms. Collectively, these results suggest that PEI should be carefully investigated in APC patients, as malnutrition, cachexia and weight loss have a negative impact on survival and quality of life. Indeed, the survival of patients with APC can be affected, among several other factors, by their nutritional status. Malnutrition, weight loss, sarcopenia and cachexia are related to chemotherapy-induced toxicity and worsening of quality of life and survival in several reports.7,8 Malnutrition in APC is multifactorial, but PEI leading to maldigestion and secondary malnutrition plays a major role.9,31 Patients with PEI reported the worst quality of life mainly due to difficulties managing gastrointestinal complaints, diet and digestion. 32
In the present study, PEI was diagnosed in over three out of four patients with APC, and there was a threefold increase in the prevalence of PEI in tumours located in the head of the pancreas compared to those located in the body-tail; 40% of those patients had severe PEI based on FE-1 levels.
Sensitivity and subgroup analysis showed that the prevalence of PEI decreased when FE-1 was used as well as when the studies were performed in Western countries. While FE-1 testing is easy to perform and has a rather high sensitivity, the specificity of the test in patients with APC is very low (22%), 33 and this result is therefore not unexpected. The accuracy of FE-1 is higher in other pancreatic disorders, such as chronic pancreatitis, and its use in APC needs further investigation.
PERT is the standard treatment for PEI in patients with all pancreatic diseases. Benefits of PERT in APC were reported in several studies. A population-based study from the UK 15 showed that PERT in patients with pancreatic adenocarcinoma was associated with increased survival; however, only 21% of those patients were under PERT, suggesting that awareness of the importance of such treatment is limited among physicians taking care of PC patients. In a retrospective study from Spain, 14 PERT in PC patients with PEI was associated with longer survival compared to no treatment. The present meta-analysis shows that PERT is significantly associated with longer survival in patients with APC with a difference of about four months. Notably, such differences are unusual in studies on the treatment of APC. 34 However, no data were available regarding disease stage, active treatments and performance status of patients and, therefore, residual confounding cannot be excluded and the effect of PERT on survival should be considered with caution.
While PERT has been shown to increase body weight, quality of life and survival in patients with PC,13,14,29 its use is not widespread. Forsmark et al. 35 noticed that only 21.9% of PC patients received PERT and that prescription was adequate in only 5.5%. Similar findings were reported in a study on APC patients from Australia in which only 21% of patients with malabsorption were prescribed PERT. 16 The reasons for under-treatment may be explained by lack of awareness of healthcare professionals and/or low level of evidence for a clinically relevant improvement in outcomes. Two recent randomised trials, both conducted on small cohorts, demonstrate negative results. Woo et al., 30 randomised 67 APC patients to PERT or placebo. PERT did not reduce weight loss, and it did not improve quality of life nor survival. A Japanese study 27 randomised 88 APC patients to PERT or no treatment. After eight weeks, there was no significant difference in change of BMI or in levels of serum nutritional markers. However, in both studies, the random protocol was applied regardless of PEI status and the doses of PERT per day were low based on guideline recommendations (150,000 Ph. Eur units of Norzyme – range from 25,000–257,500 Ph. Eur units per day - in the study of Woo et al. and 144,000 lipase units per day in the study of Saito et al. 27 ) A subgroup analysis among patients with tumours located in the head of the pancreas, who have a higher rate of PEI, demonstrated that there was an improvement in symptoms and nutrition in patients under PERT. 30
The main limitations of this systematic review and meta-analysis are the heterogeneity and small size of the included studies and the fact that most of them are prone to bias, as only four of them were RCTs. Another limitation is that only two studies employed the CFA, which is currently the reference method to evaluate pancreatic function, 10 allowing investigation on the effect of PERT on pancreatic function. Heterogeneity among studies also prevented the quantitative assessment of the effect of PERT on quality of life, gastrointestinal symptoms and serum nutritional markers due to different time-points and scales; therefore, no definitive conclusions can be made on those outcomes.
Large multicentre RCTs with proper sample size calculation are needed to evaluate the effect of PERT in APC with diagnosed PEI. However, until new data are published, based on the results of the present meta-analysis, PERT should be considered as an essential part of the multidisciplinary treatment of patients with APC.
In conclusion, this study shows that PEI is present in approximately 75% of patients with APC and that its treatment with PERT can prolong survival. PERT should be considered as a part of the best standard of care in patients with APC.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620938987 - Supplemental material for Pancreatic exocrine insufficiency and pancreatic enzyme replacement therapy in patients with advanced pancreatic cancer: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620938987 for Pancreatic exocrine insufficiency and pancreatic enzyme replacement therapy in patients with advanced pancreatic cancer: A systematic review and meta-analysis by Daniel de la Iglesia, Bartu Avci, Mariia Kiriukova, Nikola Panic, Maryana Bozhychko, Vasile Sandru, Enrique de-Madaria and Gabriele Capurso in United European Gastroenterology Journal
Footnotes
Acknowledgments
This study was conducted through Pancreas 2000, which is a European educational and scientific pancreatology research programme initiated by the Karolinska Institutet in Stockholm, Sweden, and the European Pancreatic Club and is supported by United European Gastroenterology.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethics approval
Not required
Informed consent
Not required
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
