Abstract
Background
Secondary sclerosing cholangitis in critically ill patients (SSC-CIP) is an emerging disease with grim prognosis.
Objective
Our aim was the analysis of prognostic factors, long-term outcome and risk of tumour development in SSC-CIP compared with primary sclerosing cholangitis (PSC) patients.
Methods
Retrospective analysis between 2008 and 2018.
Results
One hundred and eleven patients with SSC-CIP and 408 PSC patients were identified. Median orthotopic liver transplantation (OLT)-free survival was 16 months for SSC-CIP and 147 months for PSC (p < 0.001). OLT was performed in 18/111 SSC-CIP compared with 166/408 PSC patients (p < 0.001). Malignant tumours were detected in 17.9% of PSC patients (73/408) compared with 2.7% (3/111) in SSC-CIP (p < 0.001). In multivariate Cox regression analysis low levels of C-reactive protein (hazard ratio 4.687 (95% confidence interval (CI) 1.144–19.199, p = 0.032) were significantly associated with a prolonged survival whereas higher age (hazard ratio 0.488 (95% CI 0.23–1.038), p = 0.062) showed a trend for shorter survival in SSC-CIP. For PSC malignancies (hazard ratio 0.42 (95% CI 0.313–0.575), p < 0.001) and higher age (hazard ratio 0.709 (95% CI 0.544–0.922), p = 0.01) were associated with a shorter OLT-free survival.
Conclusion
SSC-CIP is characterized by acute onset of liver disease and poor prognosis but with lower tumour incidence compared with PSC.
Keywords
Key summary
Secondary sclerosing cholangitis in critically ill patients is an emerging disease with grim prognosis. Primary sclerosing cholangitis is a chronic biliary disease with similar cholangiographic findings like secondary sclerosing cholangitis. Primary sclerosing cholangitis has a high risk for hepatobiliary malignancies which is insufficiently analysed in secondary sclerosing cholangitis. Prognostic markers in secondary sclerosing cholangitis in critically ill patients have to be determined.
Secondary sclerosing cholangitis in critically ill patients is characterized by acute onset of liver disease and poor prognosis. Tumour incidence in secondary sclerosing cholangitis in critically ill patients is lower compared with primary sclerosing cholangitis. Levels of C-reactive protein and age at onset of secondary sclerosing cholangitis are prognostic factors. Liver transplant-free survival is significantly shorter in secondary sclerosing cholangitis in critically ill patients.
Introduction
Secondary sclerosing cholangitis (SSC) in critically ill patients (SSC-CIP) is a progressive and destructive cholangiopathy with a grim prognosis.1–5 The mortality rate is estimated to be higher than 50% after six months.1–6 SSC can be divided into several subgroups due to aetiology of stricture formation.7,8 Various factors, such as ischaemia, toxic, infectious, immunological processes and chronic biliary obstruction, may lead to SSC.7,8 SSC-CIP is increasingly described during or after intensive care unit (ICU) treatment of patients due to rising awareness of the disease. 1 Data on the incidence of SSC-CIP in ICU patients are still missing. From single-centre retrospective cohorts it may be assumed that the incidence ranges from 0.1% to 2% with a high number of undetected cases due to the reluctance to perform invasive diagnostics and/or poor prognosis of the critically ill patients.1,3,5 It is postulated that ischaemic injury of the bile ducts and repetitive episodes of cholangitis are the main triggers of SSC-CIP.1,2,4,9 SSC-CIP is most often diagnosed via endoscopic retrograde cholangiography (ERC) but neither diagnostic algorithms nor therapeutic approaches are sufficiently evaluated. 1
The cholangiographic features of SSC-CIP mimic the alterations which can be found in patients with primary sclerosing cholangitis (PSC). Patients with PSC have a well-described and tremendously elevated risk for hepatobiliary cancer such as cholangiocarcinoma (CCA), gall bladder cancer and colorectal cancer for patients with overlapping inflammatory bowel disease. 10 , 11 The lifetime risk for CCA is estimated to range from 5% to 20% and the gall bladder cancer risk is around 2% in PSC patients.10,11 For patients with SSC-CIP no data for tumour development are available. Therefore, our aim was the analysis of the long-term outcome, prognostic factors and risk of tumour development in a large real-life cohort of patients with SSC-CIP and PSC. Moreover, we wanted to elucidate similarities and differences between the disease entities.
Patients and methods
Patients
Patients with SSC-CIP were identified between 2008 and 2018 by retrospective chart review. SSC-CIP was diagnosed by a typical clinical course with acute deterioration of liver function tests (elevation of bilirubin (>threefold upper limit of normal (ULN)), alkaline phosphatase (>threefold ULN), gamma-glutamyl-transferase (GGT) (>threefold ULN)) during or after ICU treatment and detection of typical endoscopic findings such as strictures and dilatations of the biliary system, rarefication of the biliary tree with contrast filling defects and presence of biliary casts. A pre-existing liver disease was an exclusion criterion for patients with SSC-CIP. All patients with SSC-CIP underwent ERC and the diagnosis was established by this invasive procedure. As control group, PSC patients who presented at our hepatology unit in the same time period were analysed. PSC was diagnosed following current practice guidelines. 12 The diagnosis of PSC was based on laboratory and clinical findings or typical cholangiographic features (strictures or irregularity of intrahepatic and/or extrahepatic bile ducts) and exclusion of secondary causes for sclerosing cholangitis. Demographic characteristics, laboratory parameters at day of first diagnosis of SSC and PSC and follow-up data such as tumour development, necessity of orthotopic liver transplantation (OLT) and death were extracted from the clinical and endoscopic database. The complete follow-up of patients was included until December 2018. The patient history was analysed from time of first diagnosis. For patients with SSC-CIP follow-up started from time of first diagnosis/date of ERC. The study protocol was approved by the local institutional ethics review board and is in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients or their legal representatives.
Methods
Statistical analyses were performed using SPSS 24.0 (SPSS Inc., Chicago, Illinois, USA). Data were expressed as number/percentage or median with interquartile range (IQR). Differences between categorical variables were calculated using Pearson’s Chi-squared test. Transplant-free survival was assessed using the Kaplan–Meier estimation. Transplant-free survival was analysed from first diagnosis until last follow-up, death or OLT, whichever occurred first. Comparison was made using the log rank (Mantel–Cox) test. The hazard ratios and confidence intervals (CIs) for death/OLT were calculated using univariate and multivariate Cox’s regression analysis. A probability (p) value less than 0.05 was considered significant.
Results
We identified 111 patients with SSC-CIP between 2008 and 2018. Sepsis (n = 48/111; 43%), complicative cardiothoracic surgery interventions (n = 42/111; 38%) and polytrauma (n = 24/111; 22%) were the predominant causes for ICU admission of patients with SSC-CIP. Median ICU duration was 41.5 days (IQR 30–66.25 days) for patients with SSC-CIP. Median ventilation time was 30 days (IQR 18–51 days) and patients with SSC-CIP received at least one vasoactive drug for 12 days (7.5–27.5 days). The majority of patients with SSC-CIP had documented circulatory shock (105/111; 95%). PSC patients serving as control group were treated between 2008 and 2018 at our hepatology unit (n = 408). Demographic and laboratory parameters of the patient cohorts are given in Table 1. SSC-CIP were significantly older than PSC patients (50 (IQR 42–60) vs. 33 (IQR 25–42) years; p < 0.001) and had significantly higher cholestatic parameters (alkaline phosphatase, GGT, bilirubin), liver function tests (alanine and aspartate aminotransferase) and inflammatory parameters (white blood cells and C-reactive protein (CRP)). Patients of both groups were predominantly male (78/111; 70.3% vs. 296/408; 72.6%; p = 0.635) (Table 1). Median follow-up was 186 months (IQR 169–203) for PSC and 42 months (IQR 31–53) for SSC-CIP. OLT was performed in 18/111 SSC-CIP compared with 166/408 PSC patients (p < 0.001). Median OLT-free survival was 16 months for SSC-CIP and 147 months for PSC (p < 0.001) (Figure 1). When the survival analysis was restricted to patients who had at least a follow-up of 12 months, OLT-free survival was 92 months for SSC-CIP and 156 months for PSC patients (p < 0.001). For the SSC-CIP patients, the 30-day mortality was 20/111 (18%). Malignant tumours were detected in 17.9% of PSC patients (73/408) after a median of 33 months following diagnosis of PSC. Sixty-five patients developed a CCA (65/408; 16%), four patients developed gall bladder cancer (4/408; 1%) and four patients had other tumours (4/408; 1%). Only three malignancies (gastric cancer, hepatocellular carcinoma and renal cell carcinoma) were diagnosed in three patients with SSC-CIP (3/111; 2.7%) (p < 0.001) after a median of 77 months after diagnosis of SSC-CIP. A flow chart including both groups and their outcome is given in Figure 2.
Demographics and laboratory parameters in patients with secondary sclerosing cholangitis and primary sclerosing cholangitis.
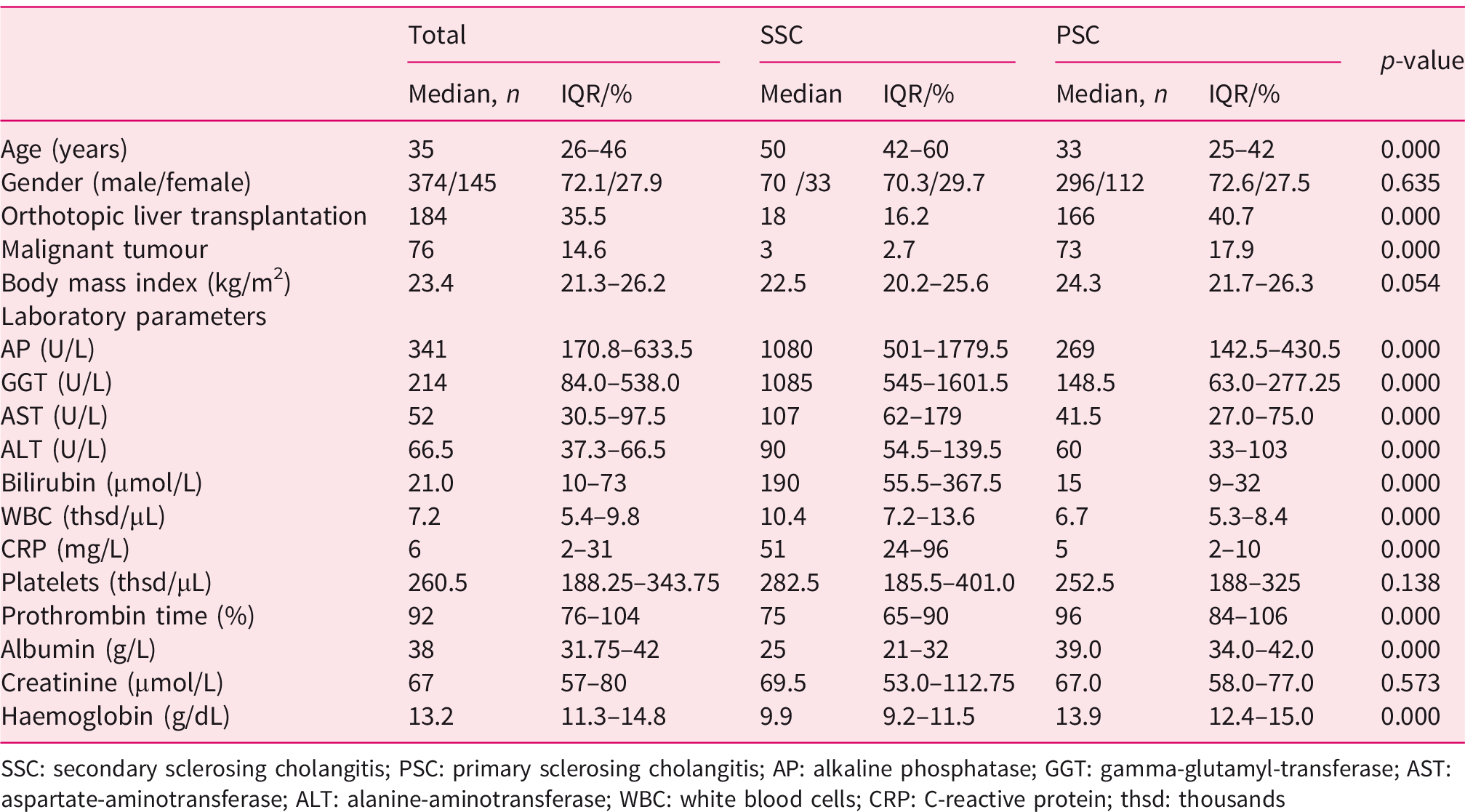
SSC: secondary sclerosing cholangitis; PSC: primary sclerosing cholangitis; AP: alkaline phosphatase; GGT: gamma-glutamyl-transferase; AST: aspartate-aminotransferase; ALT: alanine-aminotransferase; WBC: white blood cells; CRP: C-reactive protein; thsd: thousands
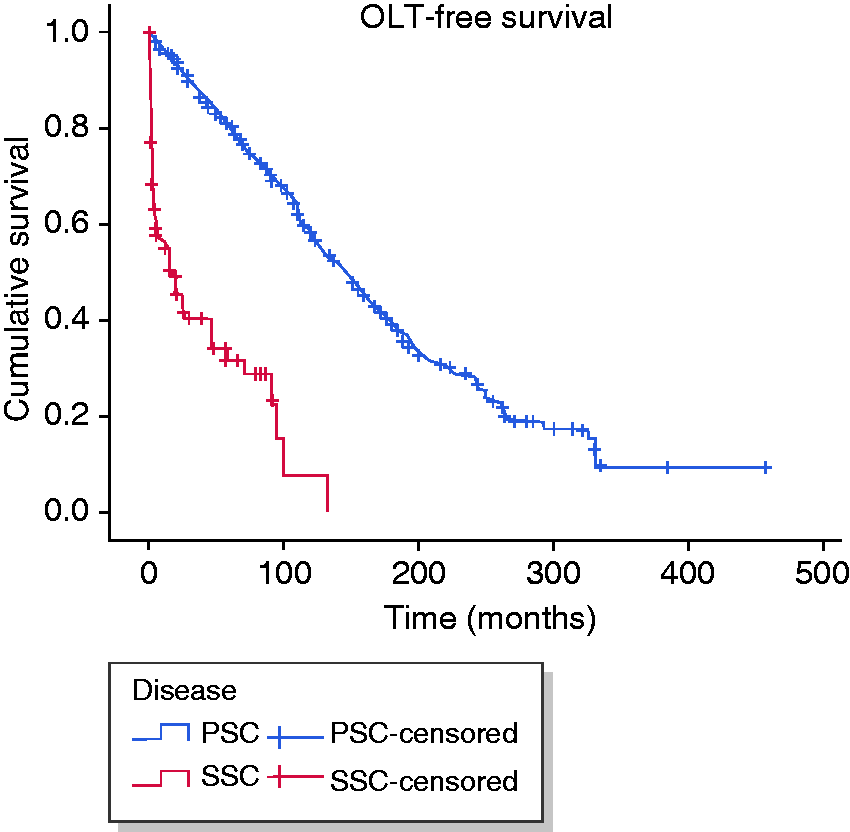
Survival analysis of patients with secondary sclerosing cholangitis (SSC) and primary sclerosing cholangitis (PSC). Median liver transplant free survival was 16 months for SSC in critically ill patients and 147 months for PSC (p < 0.001).

Flow chart including patients with primary sclerosing cholangitis (PSC) and secondary sclerosing cholangitis (SSC) in critically ill patients regarding death, orthotopic liver transplantation (OLT) and tumour development. Numbers are given as numbers and percentages.
Endoscopic findings for patients with SSC-CIP were: multiple diffuse strictures of the intrahepatic bile ducts (88 /111 (79.3 %), 7 /111 (6.3 %) with either right- or left-sided alterations alone); intra- and extrahepatic strictures (12 /111 (10.8 %)); and extrahepatic bile duct changes without intrahepatic abnormalities (4 /111 (3.6 %)). Biliary casts were present in 102 /111 patients (91.9%). Endoscopic sphincterotomy was undertaken in all patients, with three cases of mild self-limiting post-ERC pancreatitis. Endoscopic balloon dilatation and/or bougienage was performed in all patients for treatment of bile duct stenoses. Flushing of the bile ducts with saline was documented in 100/111 patients (90.1%). Endoscopic stent placement was performed in 12/111 patients (10.8%). Exemplarily, a cholangiography of SSC-CIP and PSC is given in Figure 3(a) and (b), respectively.
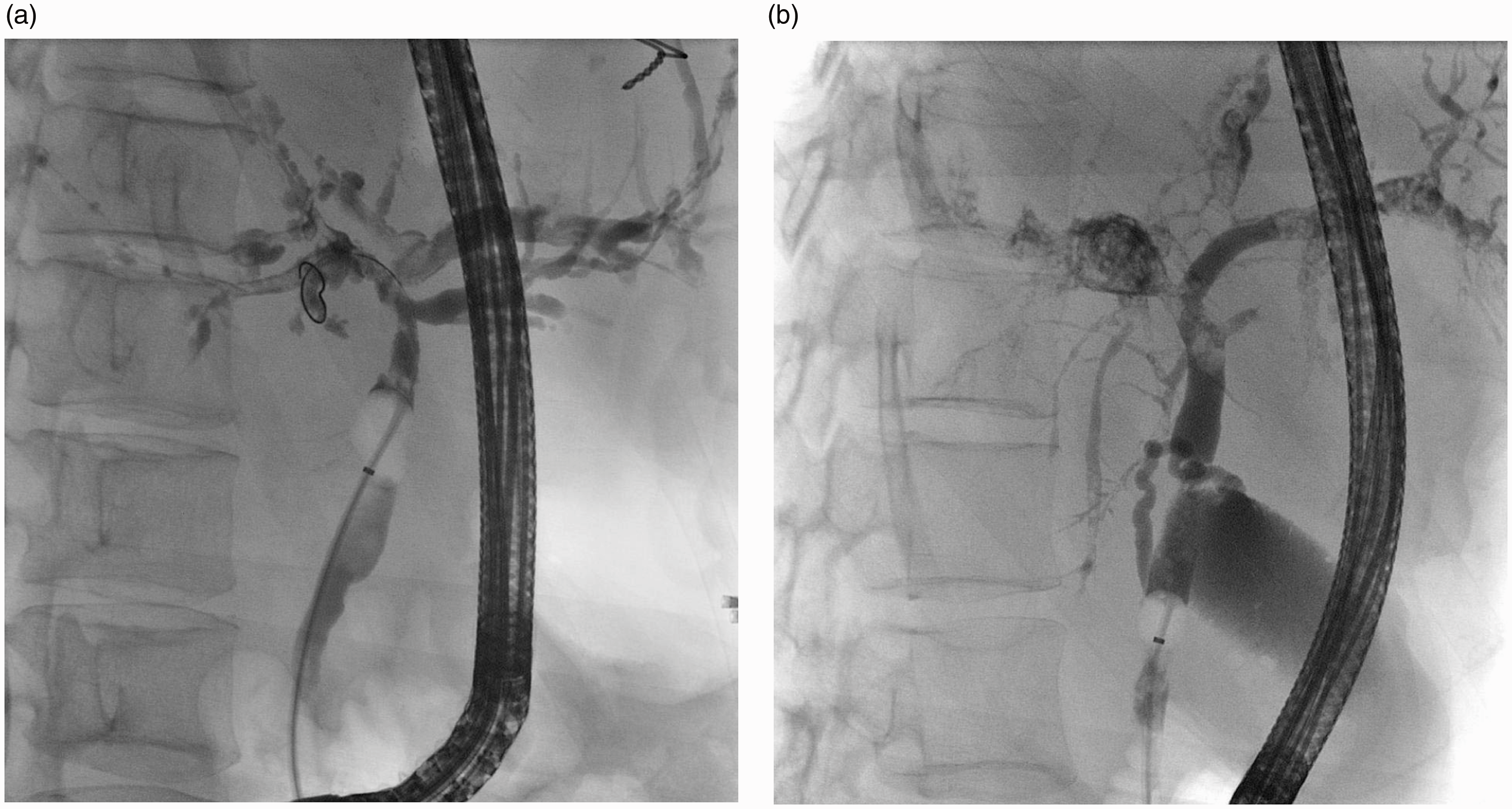
Typical endoscopic retrograde cholangiopathy imaging of a patient with secondary sclerosing cholangitis in critically ill patients (a) and primary sclerosing cholangitis (b). The intrahepatic bile ducts show signs of rarefication and contrast fillings defects.
Next, we performed univariate and multivariate survival analyses in order to analyse prognostic factors. In univariate analysis, age ≥35 years (p = 0.04), CRP > 8 mg/L (p = 0.012), albumin <35 g/L (p = 0.002) and haemoglobin < 13.5 g/dL (p = 0.028) were identified as significant negative prognostic factors for patients with SSC-CIP (Table 2). For patients with PSC, development of malignant tumours (p < 0.001), age ≥35 years (p = 0.028) and body mass index (BMI) ≤23.4 kg/m2 (p < 0.001) were identified as significant prognostic factors (Table 2). These factors were associated with a reduced transplant-free survival. Multivariate Cox regression analysis revealed that low levels of CRP (<8 mg/L, hazard ratio 4.687 (95% CI 1.144–19.199, p= 0.032) were significantly associated with a prolonged transplant-free survival whereas higher age (≥35 years, hazard ratio 0.488 (95% CI 0.23–1.038), p = 0.062) showed a statistical trend for shorter transplant-free survival in patients with SSC-CIP (Table 3). In patients with PSC, malignancies (hazard ratio 0.42 (95% CI 0.313–0.575), p < 0.001) and higher age (≥35 years, hazard ratio 0.709 (95% CI 0.544–0.922), p = 0.01) were associated with a shorter OLT-free survival whereas higher BMI (≥23.4 kg/m2, hazard ratio 1.769 (95% CI 1.277–2.451), p = 0.001) was associated with a prolonged transplant-free survival. Details are summarized in Table 3.
Univariate survival analyses for patients with secondary sclerosing cholangitis and primary sclerosing cholangitis.
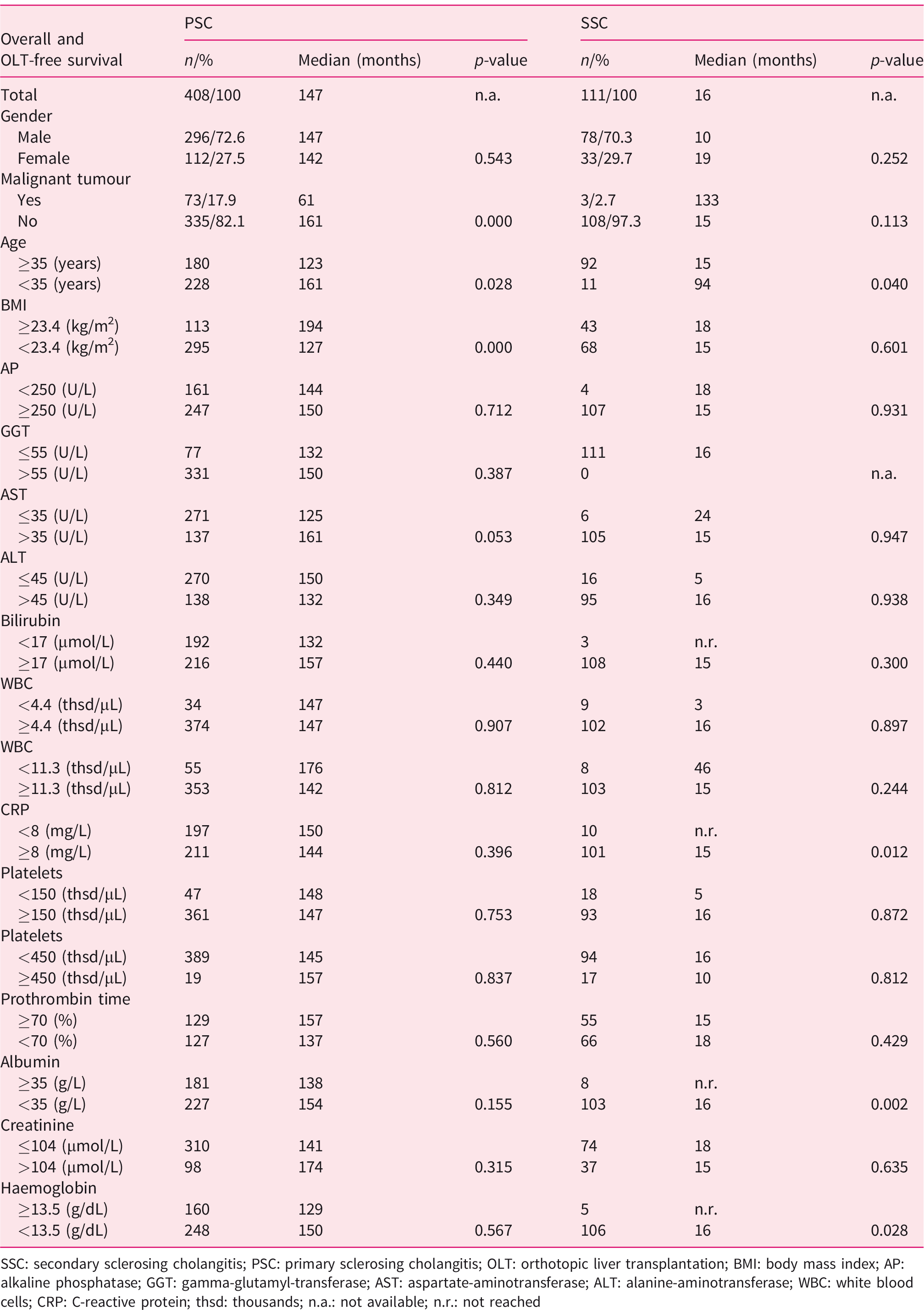
SSC: secondary sclerosing cholangitis; PSC: primary sclerosing cholangitis; OLT: orthotopic liver transplantation; BMI: body mass index; AP: alkaline phosphatase; GGT: gamma-glutamyl-transferase; AST: aspartate-aminotransferase; ALT: alanine-aminotransferase; WBC: white blood cells; CRP: C-reactive protein; thsd: thousands; n.a.: not available; n.r.: not reached
Multivariate Cox regression analysis for patients with secondary sclerosing cholangitis and primary sclerosing cholangitis.
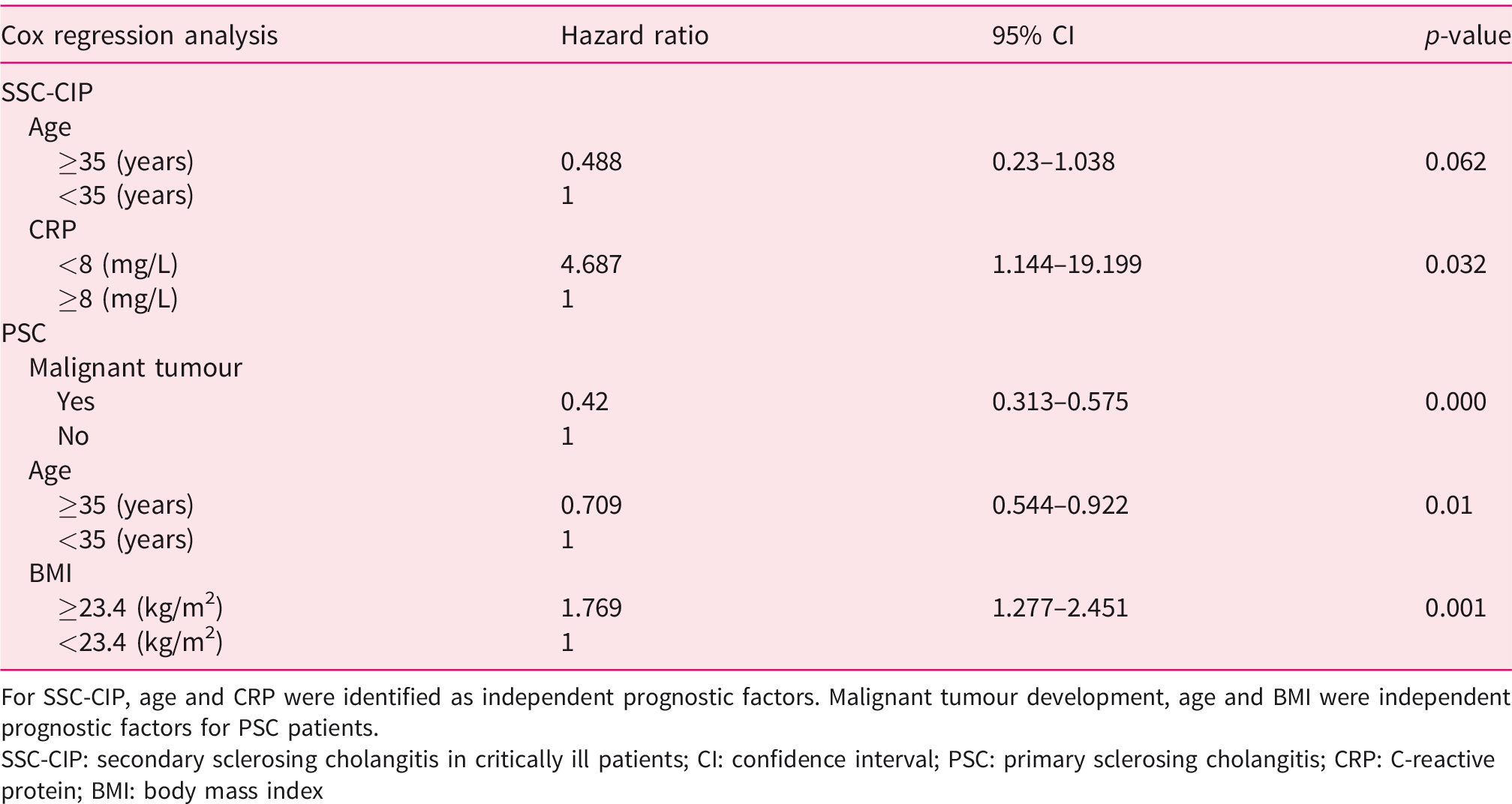
For SSC-CIP, age and CRP were identified as independent prognostic factors. Malignant tumour development, age and BMI were independent prognostic factors for PSC patients.
SSC-CIP: secondary sclerosing cholangitis in critically ill patients; CI: confidence interval; PSC: primary sclerosing cholangitis; CRP: C-reactive protein; BMI: body mass index
Discussion
In our study, we provide evidence that SSC-CIP and PSC are distinct disease entities with completely different aetiopathogenesis and prognostic factors. SSC-CIP has a grim prognosis with a median OLT-free survival of 16 months in our cohort compared with 147 months in patients with PSC. As prognostic factors in SSC-CIP, we identified that higher age was associated with a shorter OLT-free survival, whereas low levels of CRP were associated with a prolonged OLT-free survival. These findings seem reasonable as increased age is traditionally considered a risk factor for mortality in patients admitted to ICU. 13 Moreover, higher age alone is an independent risk factor for in-hospital mortality. 14 CRP as surrogate parameter of inflammation is widely used to detect infections. 15 The cut-off value of CRP to detect inflammation and/or infection varies among studies; however, its potential use in detection of immune activation is generally accepted.15,16 An elevation of CRP reflects an activation of immune responses, which are often triggered by infections in critically ill patients, leading to worse outcome.15–17 CRP values can change quite rapidly, which may have influence on our results. However, to reduce this form of bias we analysed CRP values at day of ERC before endoscopic intervention in patients with SSC-CIP. Biliary tract infection seems to play a critical role in the pathogenesis of SSC-CIP as repetitive episodes of cholangitis deteriorate the bile duct integrity. In a prior study, we could show that up to 94% of bile samples were positive for microbial growth in patients with SSC-CIP, which may be reflected by elevated CRP levels. 9 The diagnosis of SSC-CIP was made by ERC in all patients. ERC interventions include sphincterotomy and insertion of different catheters and guide wires into the normally sterile biliary system and might have the opposite effect of promoting ascending bacterial cholangitis and hence accelerate disease progression. These questions have to be addressed in a prospective study.
PSC is a complex liver disease with a heterogeneous clinical presentation. Typically, patients suffer from chronic cholestasis associated with chronic inflammation of the biliary epithelium, leading to multifocal stricture formation.18,19 This stands in harsh contrast to the acute development of liver disease in patients with SSC-CIP. Moreover, the prognosis of patients with PSC is mainly determined by the tremendously elevated risk of hepatobiliary cancer. In our analysis, approximately 18% of patients with PSC developed cancer whereas only 3% of patients with SSC-CIP developed malignant tumours. The occurrence of CCA in our cohort is in line with the published literature. Bergquist et al. reported a frequency of cholangiocarcinoma of 13% in patients with PSC. 20 Of note, no patient with SSC-CIP developed a CCA. One factor explaining these findings may be the short survival of patients with SSC-CIP after development of liver disease. The 30-day mortality for SSC-CIP was 18%, which leads to a shorter time period in which a tumour may develop. Another explanation is that the aetiopathogenesis of SSC-CIP differs completely from PSC with its smouldering bile duct inflammation over years facilitating CCA development. These assumptions are beyond the scope of our study and have to be addressed in future studies. Recently, it was shown by Rupp et al. that scheduled endoscopic balloon dilatation of dominant bile duct strictures may improve transplant-free survival in patients with PSC. 21 Lemoinne et al. found that magnetic resonance scores are associated with outcomes of patients with PSC. 22 In our study, only a small number of patients with PSC underwent ERC and data of magnetic resonance imaging and/or quality of the procedure are not sufficient for detailed analysis. Therefore we cannot address these aspects in our study.
For patients with PSC, we identified development of malignancies and higher age as prognostic factors which lead to shorter OLT-free survival. A higher BMI was associated with a prolonged survival. A low BMI is one of the manifestations of malnutrition and is associated with poor outcomes in general. 23 Additionally, sarcopenia, which is linked to low BMI among OLT recipients, is also associated with increased rates of infection and significantly higher hospital length of stay. 23 However, sarcopenia was not routinely assessed in our study. Our findings underline the importance of an adequate BMI on morbidity and mortality, especially in patients with chronic liver disease.24,25
Liver transplantation is an established therapeutic option in patients with advanced liver disease.26,27 In our cohort, 16% of patients with SSC-CIP and 41% of patients with PSC were liver transplanted. In general, OLT in SSC-CIP seems to be a good therapeutic option in selected patients after survival of the acute trigger of liver disease.28,29 However, many patients with SSC-CIP are not eligible for OLT due to their severe underlying disease with multi-organ failure.1,28,29 These findings are reflected by the harsh mortality of SSC-CIP in ICU.1,2,4 The cohort of SSC-CIP seems to consist of two distinct groups; those who survive the critical illness and those who do not. The former seem to be eligible for OLT and do not have the same tumour risk as patients with PSC. For patients with PSC, an established transplant programme using the model for end-stage liver disease score and a standard exception method is available in the Eurotransplant region facilitating OLT in patients fulfilling the transplant criteria. 30
One limitation of our study is its retrospective nature, which may lead to bias. However, we thoroughly screened our clinical and endoscopic database in order to minimize this aspect. Moreover, patients with SSC-CIP and PSC are not completely comparable. Patients with PSC have a more chronic course of disease whereas patients with SSC-CIP develop liver disease in an acute setting in ICU. Additionally, the patient follow-up differs significantly. However, due to the lack of ideal control patients and the similar cholangiographic findings in both diseases we decided to compare both patient cohorts. We believe that we provide valuable information especially for patients with SSC-CIP which may improve the clinical management of this liver disease.
In conclusion, SSC-CIP and PSC are distinct disease entities. SSC-CIP is mainly characterized by the acute onset of liver disease and the poor prognosis. PSC proceeds chronically and is complicated by a high risk of tumour development which is not seen in patients with SSC-CIP. The identified prognostic factors have to be taken into account while managing patients with sclerosing cholangitis.
Footnotes
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics approval
The study protocol was approved by the local institutional ethics review board and is in accordance with the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all patients or their legal representatives.
