Abstract
Background and aim
Probe-based confocal laser endomicroscopy (pCLE) provides real-time microscopic visualisation. Our aim was to compare the diagnostic accuracy of pCLE with standard biopsies in patients with visible oesophageal or gastric lesions.
Methods
This was a single-centre, prospective, pathologist-blinded study. Patients underwent high-resolution endoscopy, and lesions were examined by pCLE followed by standard biopsies. A definitive diagnosis was determined from resection specimen. Main outcomes were overall diagnostic accuracy, sensitivity, specificity and positive and negative predictive values.
Results
We examined 74 lesions in 67 patients. Definitive diagnoses revealed 34 malignant and 40 non-malignant lesions. pCLE diagnosis was correct in 89.2% (66/74), while diagnosis based on biopsy was correct in 85% (57/67; p = 0.6). The overall diagnostic accuracy of biopsies was 85% (76–94%) and that of pCLE was 89% (79–96%). pCLE correctly diagnosed malignant lesions, comprising oesophageal adenocarcinoma, oesophageal squamous-cell cancer or gastric adenocarcinoma, in 88.2% (30/34) of cases, while biopsy was correctly diagnosed in 75.9% (22/29; p = 0.3). Sensitivity and specificity to diagnose a malignant lesion were 75.9% (95% confidence interval (CI) 56–89%) and 100% (95% CI 90–100%) for biopsies and 88.2% (95% CI 72–97%) and 92% (95% CI 79–98%) for pCLE. No differences between biopsies and pCLE were found with regard to sensitivity, specificity to diagnose dysplastic and benign lesions (p > 0.2).
Conclusion
pCLE provides satisfactory diagnostic accuracy comparable with standard biopsies in patients with oesophageal or gastric lesions. ClinicalTrials.gov identifier: NCT0292049)
Introduction
Standard histopathological diagnosis is considered the gold standard in patients presenting with digestive pathologies, including macroscopically visible oesophageal or gastric lesions. 1 Biopsies are recommended to clarify biological behaviour of visible lesions, although in some centres, endoscopic resection (ER) may be performed without previous biopsies in presumed premalignant or malignant lesions. Specimens obtained from ER (or from endoscopic submucosal dissection (ESD)) allow accurate histopathological diagnosis and staging. However, correct diagnosis and appropriate indication for ER or ESD are often required, as endoscopic resection techniques carry a risk of adverse events, and benefits of the procedure should always outweigh the risks. An accurate histopathological diagnosis is also warranted in patients with advanced oesophageal or gastric lesions before the patients are referred for surgery or for oncological therapy. Several studies have shown that despite a general belief in their high diagnostic accuracy, standard biopsies have limitations.2,3 Probe-based confocal laser endomicroscopy (pCLE) is a relatively new tool, enabling real-time histopathological diagnostics. 4 pCLE can be used in the diagnostics through the entire gastrointestinal (GI) tract. Even though pCLE has been tested in several studies with promising results, it is not considered as a standard method for histopathological diagnosis, and its present role is rather ancillary.5–8 Several studies have shown that pCLE may provide comparable or even better diagnostic accuracy compared with standard biopsies in patients with oesophageal adenocarcinoma (AC),9–11 oesophageal squamous-cell cancer (SCC)12,13 or gastric AC.14,15 pCLE increases sensitivity and diagnostic accuracy for dysplasia or AC, if added up on high-definition endoscopy (with narrow-band imaging (NBI)) in patients with Barrett’s oesophagus (BO). 9 Moreover, the use of pCLE may allow decreased number of biopsies to detect preneoplastic or neoplastic lesions. 16 One of the reasons why pCLE has not yet been accepted as a standard method may be due to the rather weak positive predictive value (PPV; 46%) and sensitivity (18%) of pCLE to diagnose BO-related neoplasia in one prospective study. 17
The majority of studies examined the diagnostic accuracy of pCLE in a specifically defined patient population. The aim of this study was to examine the diagnostic accuracy of pCLE compared with biopsies in unselected patients presenting with visible oesophageal or gastric lesions of unknown histological diagnosis.
Methods
This was a single-centre, prospective, pathologist-blinded study. The study was approved by the Ethics Committee of the Institute for Clinical and Experimental Medicine. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. All patients signed an informed consent form. Adult patients with a visible oesophageal and/or gastric lesion (≥10 mm) of unknown histological diagnosis, referred to our department between June 2016 and September 2019, were eligible for enrolment. Exclusion criteria were absence of a visible lesion, allergy to fluorescein, already known histopathological diagnosis, pregnancy, oesophageal varices and coagulopathy. Included patients were examined by high-resolution endoscopy (with NBI), followed by pCLE and then by standard biopsies. According to the lesion’s characteristics, patients consequently underwent endoscopic treatment (endoscopic resection or endoscopic submucosal dissection), surgical treatment, oncological treatment or no treatment. Diagnoses based on pCLE and on biopsies were compared with a definitive diagnosis made from endoscopic/surgical resection specimen. In seven patients, biopsies were not taken as endoscopic resection was performed after pCLE.
Endoscopy and pCLE
Patients underwent high-definition white-light upper GI endoscopy (Endoscopes Olympus 180 or 190; Olympus Medical Systems, Tokyo, Japan). Additional imaging methods (e.g. NBI, Lugol staining, or acetic acid staining) were used at the discretion of an endoscopist. Patients were administered analgosedation with midazolam 2–5 mg if necessary. Lesions with surrounding mucosa were examined by pCLE (Cellvizio®; MaunaKea Technologies, Paris, France), and the video sequences were recorded (image acquisition 12 frames/s). We used a GastroFlexUHD probe with an imaging depth of 65 µm, lateral resolution of 1 µm and 240 µm field of view. For tissue contrast, 2.5–5 mL of 10% fluorescein (Fluorescite 100 mg/mL injected solution; Alcon Pharmaceuticals, Prague, Czech Republic) was injected intravenously a few minutes before pCLE examination. pCLE video sequences were evaluated offline by one GI pathologist who was blinded to both endoscopic and histopathologic findings. The pathologist was trained during an internship focused on the pCLE-based diagnosis and by online training portal. The validated Mainz and Miami classification systems were used for evaluation of the pCLE sequences as follows: benign, dysplastic and malignant. 18
The pathologist evaluated the quality of the pCLE videos/images (1 = excellent, 2 = satisfactory and 3 = unsatisfactory) and also made a specific histopathological diagnosis (e.g. type of cancer or polyp).
Biopsies
Standard forceps biopsies were taken by using large-capacity forceps (Radial Jaw™ 4; Boston Scientific, Natick, MA). The number of biopsies varied according to the lesion’s size, but at least two biopsies had to be taken from each lesion. The tissue specimen was put into containers with 10% neutral-buffered formalin.
Histopathology
All specimens were evaluated by an expert pathologist who specialised in GI pathology. Unclear cases with diagnostic discrepancy were solved by a consensus among three expert pathologists.
Standard biopsies
The specimens were fixed in formalin and embedded in paraffin. Slides for light microscopy were cut at serial levels 2–3 µm thick. These samples were stained with hematoxylin and eosin and periodic acid Schiff reagent.
Endoscopic resection specimen
Samples were fixed in formalin, macroscopically completely processed and stained as for standard biopsies.
Surgical resection specimen
Resected specimens including lymph nodes were fixed in formalin and cut at serial levels 2–3 µm thick.
Outcomes
The primary outcomes were overall diagnostic accuracy, sensitivity, specificity, PPV and negative predictive value (NPV) of biopsies and of pCLE to diagnose malignancy, dysplasia and/or benign lesion.
Statistical analysis
Statistical analyses were performed using standard MedCalc (MedCalc Software BV, Ostend, Belgium) and GraphPad Prism v7 (GraphPad Software, San Diego, CA). A descriptive statistic was used to calculate correct/incorrect diagnoses made with pCLE, and biopsies and differences between the two methods were tested using Fisher’s exact test. We calculated sensitivity, specificity, NPV and PPV with 95% confidence intervals (CI) of pCLE and of biopsies to diagnose or to exclude malignancy, a dysplastic lesion and/or benign lesion. We also calculated overall diagnostic accuracy of pCLE and biopsies. We planned to include at least 50 patients (crossover design, expected diagnostic accuracy of biopsies 70%, 95% CI 65–85, p = 0.05, 80% study power).
Results
Baseline characteristics
A total of 67 patients were included. The median age was 65.4 years (range 35–84 years), and the majority were male 77.6% (52/67). We examined a total of 74 lesions (53 oesophageal and 21 gastric). Definitive diagnosis was made from resection specimen in 70.2% (52/74 lesions) and from biopsy based on consensus among expert pathologists in 28.4% (21/74 lesions). In one (1.3%) patient, a definitive diagnosis was based on pCLE (the patient had advanced oesophageal SCC, and standard biopsies revealed necrotic tissue). Definitive histopathological findings are summarised in Table 1. Among 74 lesions, 28 were benign (7 non-dysplastic lesions within BE, 7 inflammatory polyps, 6 hyperplastic polyps, 2 fundic gland polyps, 2 gastric intestinal metaplasia, 2 cylindrical metaplasia, 1 apocrine metaplasia and 1 granular cell tumour; Figure 1), 12 lesions were dysplastic (BO (Figure 2), adenomatous or serrated (Figure 3) polyps, 4 low-grade dysplasia (LGD), 8 high-grade dysplasia (HGD)) and 34 lesions were malignant.
Granular cell tumour, Abrikossoff’s tumour (left: endoscopy with the lesion; middle: standard histopathology; right: pCLE). Correct diagnosis was made by both pCLE and histology. pCLE: probe-based confocal laser endomicroscopy. Agreement in the diagnosis in a patient with Barrett’s oesophagus (left: high-grade dysplasia (HGD), standard histopathology; right: HGD, pCLE). 1. Unequal luminal surface in dysplastic gland. 2. High activity of dysplastic cells (left: nucleocytoplasmic ratio increased; right: dark cells). Agreement in the diagnosis (left: standard histopathology; right: pCLE). 1. Serrated hyperplastic gastric epithelium with low-grade dysplasia; right: unequal epithelium structures of the serrated adenoma. Lesions’ characteristics.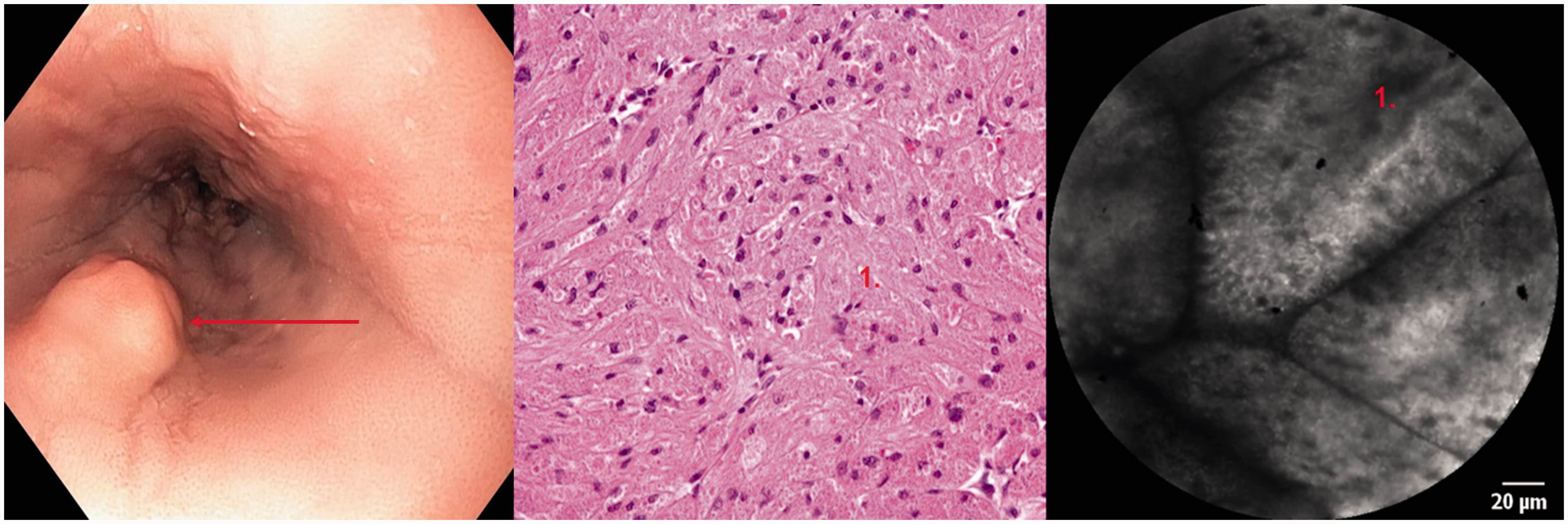
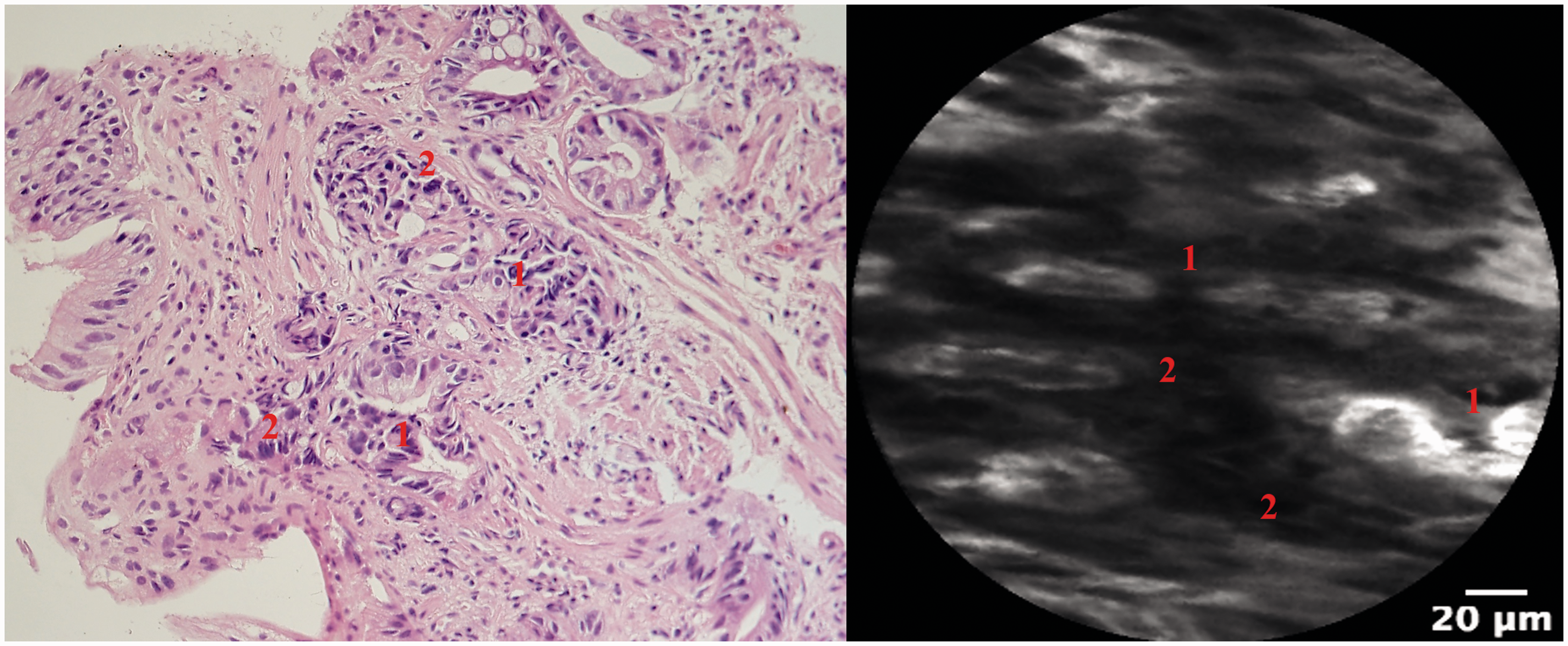
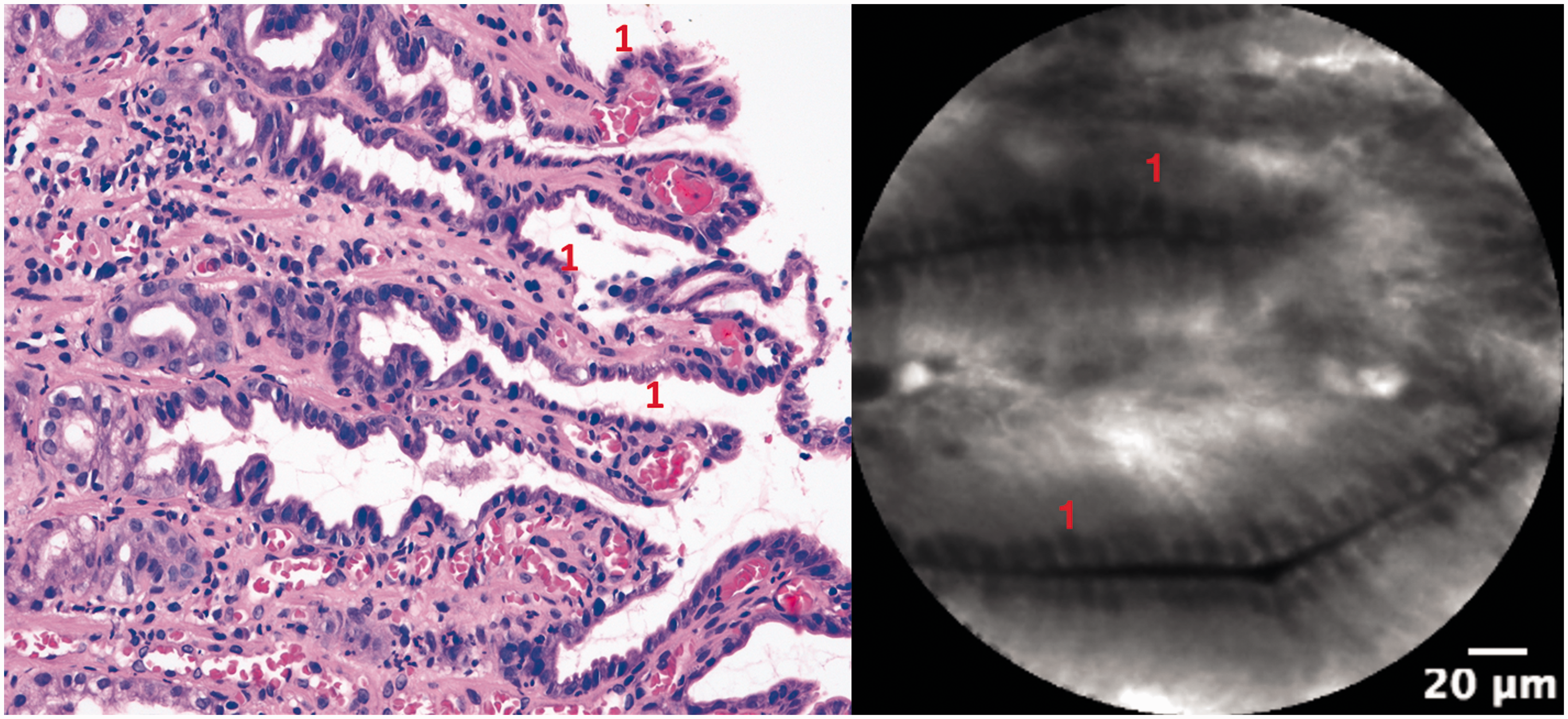
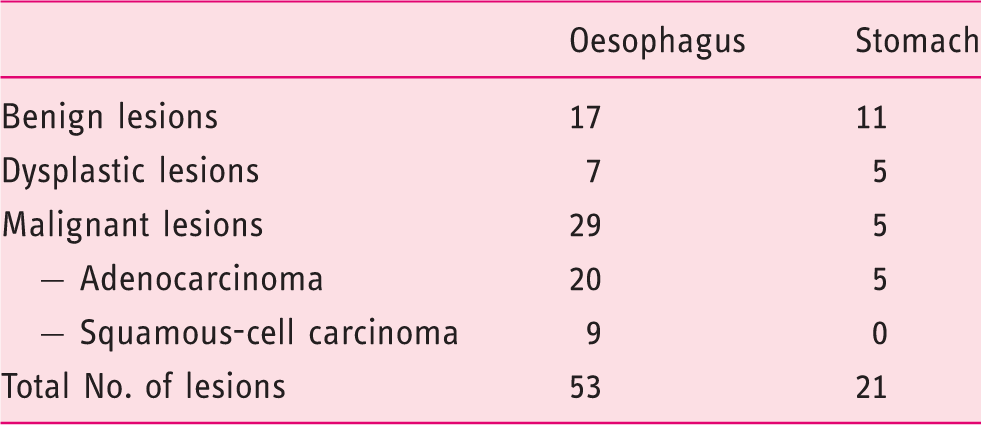
Image quality was assessed as excellent in 52 (77.6%) patients and satisfactory in 14 (20.9%) patients. In one (1.5%) patient, the image quality was unsatisfactory.
Diagnostic accuracy
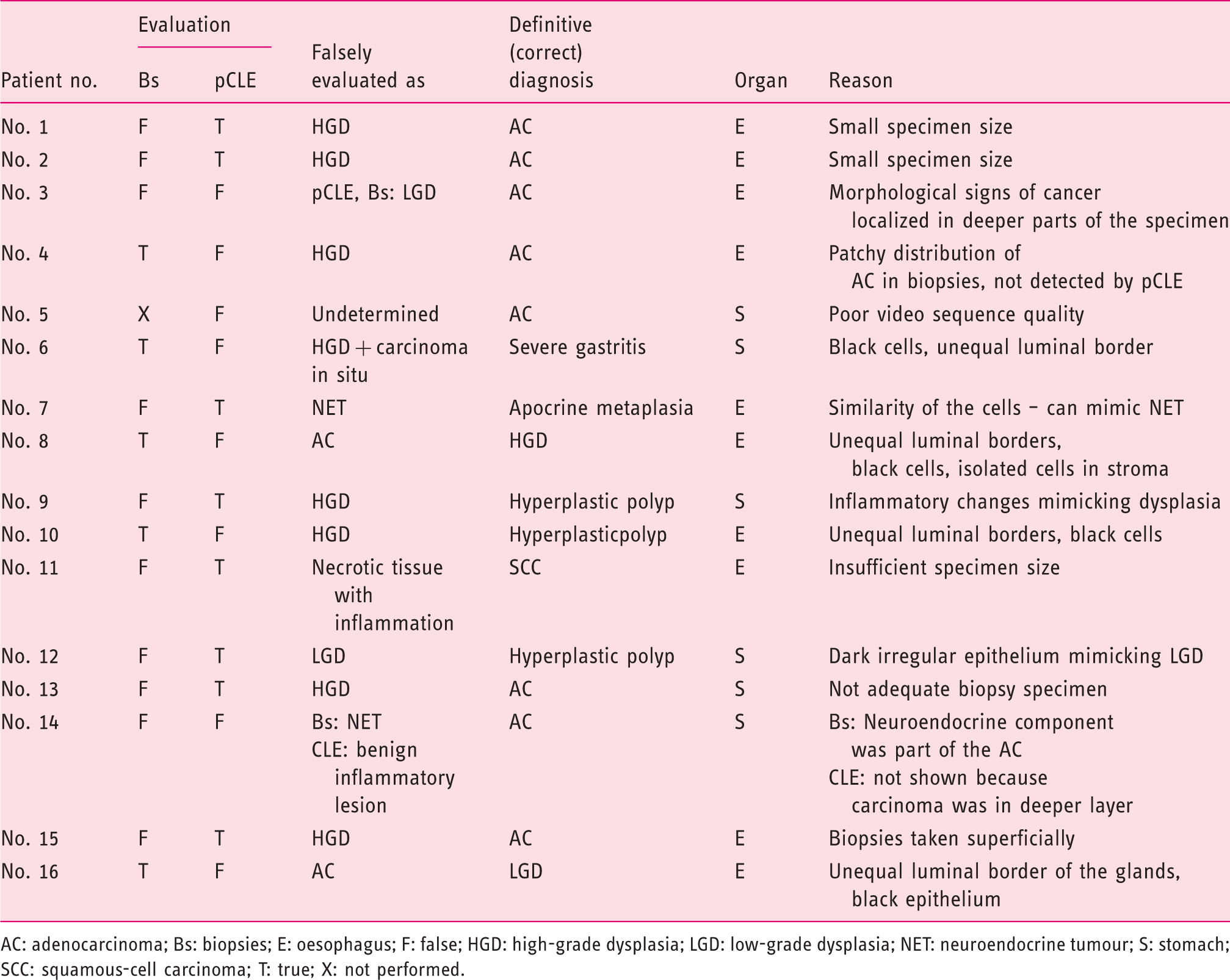
pCLE diagnosis was correct in 89.2% (66/74) and incorrect in 10.8% (8/74), while diagnosis based on forceps biopsy was correct in 85% (57/67) and incorrect in 15% (10/67; p = 0.6; Table 2). Detailed analysis of disagreement between biopsies and pCLE is shown in Table 3 (Figure 4). pCLE confirmed malignant diagnosis in 88.2% (30/34) while biopsy confirmed malignant diagnosis in 75.9% (22/29) of lesions (p = 0.3). Dysplastic lesions were correctly diagnosed by pCLE in 83% (10/12) and by biopsies in 100% (10/10; p = 0.48). Diagnosis of benign lesions was accurate with pCLE in 92.8% (26/28), while diagnosis based on forceps biopsy was accurate in 89.2% (25/28) of lesions (p = 1). pCLE allowed a specific histopathological diagnosis to be made in the majority of patients with a ‘correct’ diagnosis (90.9%; 60/66; Figures 1 and 2).
Incorrect diagnosis by pCLE (left: gastritis, standard histopathology; right: dysplastic appearance, pCLE); correct diagnosis was gastritis. 1. Unequal luminal surface in dysplastic gland. 2. High activity of cells influenced by inflammation (left: nucleocytoplasmic ratio increased; right: dark cells). Diagnostic accuracy of pCLE compared with standard biopsies and sensitivity, specificity, PPV and NPV to diagnose (or exclude a diagnosis of) a malignant lesion. Values are percentages with 95% confidence intervals. Differences between biopsies and pCLE were not significant. pCLE: probe-based confocal laser endomicroscopy; NPV: negative predictive value; PPV: positive predictive value. Detailed analysis of incorrect diagnoses with biopsies and/or pCLE. AC: adenocarcinoma; Bs: biopsies; E: oesophagus; F: false; HGD: high-grade dysplasia; LGD: low-grade dysplasia; NET: neuroendocrine tumour; S: stomach; SCC: squamous-cell carcinoma; T: true; X: not performed.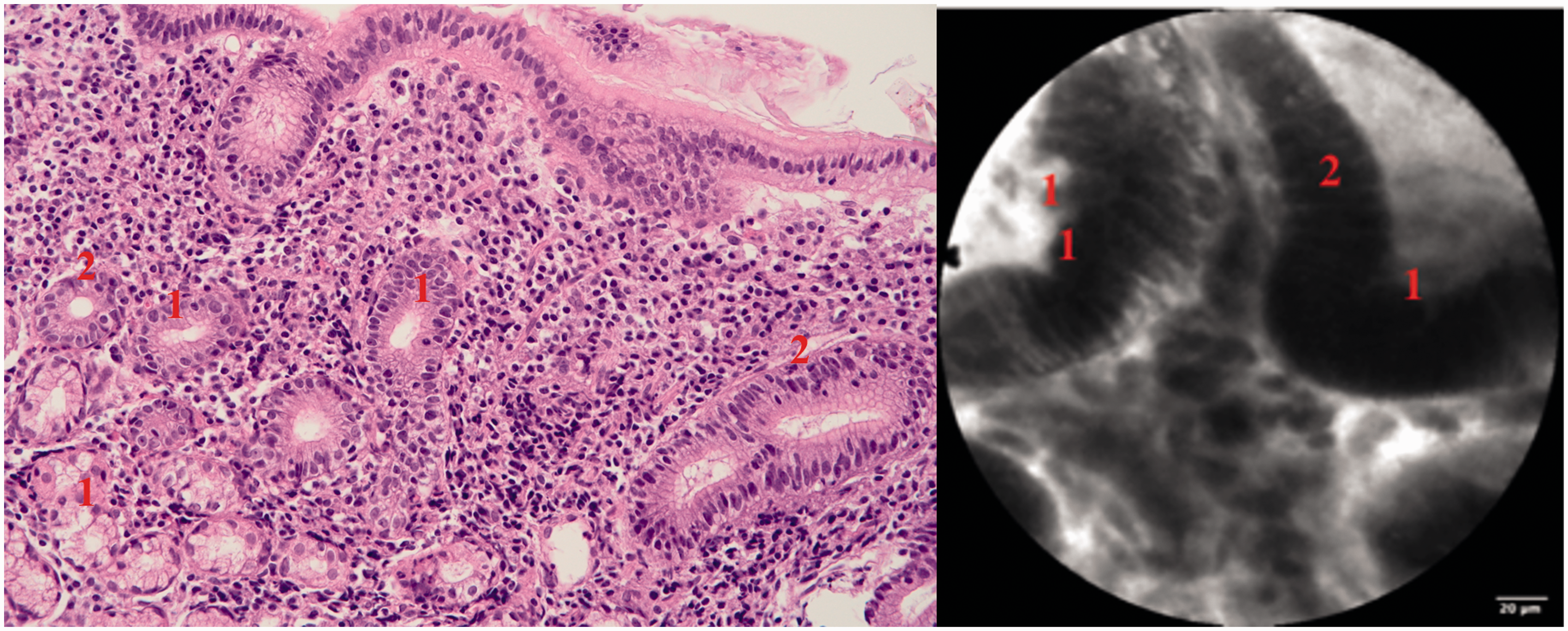
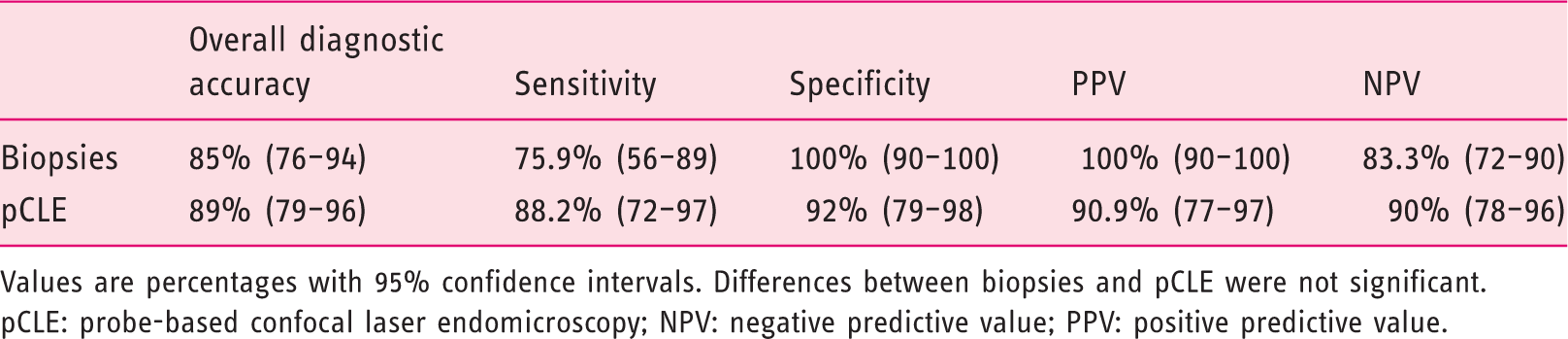
Overall diagnostic accuracy of biopsies was 85% (95% CI 76–94%) and that of pCLE was 89% (95% CI 79–96%; p = 0.9). Sensitivity and specificity to diagnose malignant lesion were 75.9% (95% CI 56–89%) and 100% (95% CI 90–100%) for biopsies and 88.2% (95% CI 72–97%) and 92% (95% CI 79–98%) for pCLE (p > 0.3). PPV and NPV to diagnose (or to exclude) malignancy were 100% (95% CI 90–100%) and 83.3% (95% CI 72–90%) for biopsies and 90.9% (95% CI 77–97%) and 90% (95% CI 78–96%) for pCLE (p > 0.1). Sensitivity and specificity to diagnose dysplasia were 100% (95% CI 91–100%) and 88.7% (95% CI 73–93%) for biopsies and 83.3% (95% CI 81–98%) and 93.5% (95% CI 89–100%) for pCLE (p > 0.2). Finally, sensitivity and specificity to diagnose benign lesion were 89.3% (95% CI 72–97.7%) and 94.9% (95% CI 83–99.4%) for biopsies and 92.9% (95% 76.5–99.1%) and 97.8% (95% CI 88.5–99.9%) for pCLE (p > 0.2).
Discussion
Our prospective study compared diagnostic accuracy of pCLE with biopsies in patients with visible oesophageal or gastric lesions. We found that both methods have a high diagnostic accuracy (85% for biopsies, 89% for pCLE) with satisfactory sensitivity and specificity to diagnose malignant, dysplastic and benign lesions. Biopsies are still considered the gold standard for histopathological diagnosis in patients with digestive pathologies. Diagnosis based on biopsies is considered reliable but not perfect. For example, there is ongoing controversy about the diagnostic accuracy of biopsies to detect low-grade dysplasia in patients with BO (LGD is often over-diagnosed). 19 Even biopsies from a visible lesion are not accurate enough to provide a reliable diagnosis. Two studies showed that a definitive diagnosis of a considerable number of visible neoplastic lesions had to be changed based on the assessment of a specimen from ER.2,3 In the present study, biopsies showed a trend for decreased sensitivity to diagnose a malignancy (76%). In the majority of incorrectly diagnosed cases, biopsies revealed dysplasia or necrotic tissue. Thus, novel diagnostic approaches have been developed and are being tested with the aim of improving both a lesion’s detection and diagnostic accuracy. pCLE represents a relatively new method enabling ‘virtual’ microscopic diagnosis. There are several reasons why pCLE may be a useful tool which might, in some circumstances, replace standard tissue-sampling techniques. First, it may provide diagnosis quickly. Second, no tissue sampling is necessary (and there is therefore no risk of adverse events such as bleeding in patients with anticoagulants). Third, a probe may examine a significantly larger area compared with biopsies. Fourth, pCLE may be assessed by a trained endoscopist. Last, no scarring occurs after pCLE, and it is well accepted that biopsies should not be taken from flat colorectal lesions (scarring makes subsequent endoscopic resection difficult). The use of pCLE may prevent an endoscopic resection of lesions which are not indicated for endoscopic removal (e.g. tuberculosis, lymphoma).
pCLE may be accepted as a standard method if it fulfils certain criteria. The ASGE proposed the performance thresholds in the Preservation and Incorporation of Valuable endoscopic Innovations (PIVI) document, which are defined as (a) imaging technology with targeted biopsies should have a per-patient sensitivity of ≥90% and a NPV of ≥98% for detecting HGD or early AC in patients with BO compared with the current standard protocol, and (b) the imaging technology should have a specificity that is sufficiently high (80%) to allow a reduction in the number of biopsies. 20 In our study, PIVI criteria for pCLE were not entirely fulfilled, as the sensitivity was 88% and the NPV was 92%. Nevertheless, the values found in our study may be considered satisfactory because standard biopsies did not reach the thresholds either, and our lesions included both types of oesophageal cancers and also gastric cancer. Several studies showed that pCLE may provide comparable or even better diagnostic accuracy compared with standard biopsies in patients with oesophageal AC,9–11 SCC12,13 or gastric AC. 14
The majority of studies assessing the role of pCLE in the detection of HGD/AC in patients with BO examined if adding on pCLE to other diagnostic methods may decrease the number of random biopsies. Sharma et al. 9 found that combining NBI with high-definition white-light endoscopy and with pCLE resulted in excellent sensitivity (100%) in diagnosing oesophageal AC and/or HGD, and therefore biopsies could have been avoided in 60% of patients when all three methods were negative. Bertani et al. 21 demonstrated the sensitivity, specificity, PPV and NPV of pCLE to be 100%, 83%, 67% and 100%, respectively, for dysplasia detection in patients with BO.
One important prospective study investigated whether pCLE could replace biopsy in the endoscopic evaluation of patients with BO. 17 A total of 670 pairs of biopsies and pCLE video sequences were analysed, with neoplasia being histologically diagnosed in 8.3%. The specificity and NPV of pCLE in excluding neoplasia were 97% and 93%, respectively. Both PPV and sensitivity were rather poor (46% and 28%, respectively). The authors concluded that pCLE may be regarded as non-inferior to endoscopic biopsy in excluding neoplasia, but it cannot replace standard biopsies because of the low PPV and sensitivity.
Our study demonstrated that in patients with BO and with a visible lesion, pCLE provides a reliable diagnosis of HGD/AC in the majority of cases. pCLE did not diagnose oesophageal AC correctly in 2/20 patients, while the corresponding rate for biopsies was 6/20 patients. In patients with BO (without a visible lesion), the potential usefulness of pCLE possibly lies in improving the diagnostic yield for dysplasia and in reducing the total number of biopsies. In patients with a visible lesion, pCLE provides comparable results with biopsies.
pCLE seems reliable in diagnosing oesophageal SCC. Pech et al. 22 demonstrated excellent accuracy of pCLE in the diagnosis of histologically proven SCC in 27 patients, as all lesions were correctly diagnosed by two endoscopists. In another study, 23 the sensitivity, specificity and diagnostic accuracy of pCLE in patients with oesophageal SCC were >90%. In our study, we examined nine patients with oesophageal SCC, and all of them were correctly diagnosed with pCLE, while biopsies failed to diagnose SCC correctly in one patient (biopsies showed necrotic tissue). The following successful chemoradiotherapy was pursued based only on the results of pCLE.
Finally, pCLE also seems reliable in diagnosing gastric lesions. In a meta-analysis of 23 studies, 24 the pooled sensitivity and specificity for the correct diagnosis of gastric AC were 91% (95% CI 88–94%) and 99% (95% CI 99–99%). In our study, five patients presented with gastric AC. Both biopsies and pCLE did not correctly diagnose gastric cancer in two patients (Table 3). One case of incorrect pCLE diagnosis was due to poor image quality.
Our study differed from previous studies by including ‘unselected’ patients with a lesion with unknown histopathology. This design mirrors more realistically clinical practice, as an endoscopist usually examines a whole spectrum of patients. The overall diagnostic accuracy of pCLE in diagnosing oesophageal and gastric lesions was comparable with biopsies. Thus, we believe that pCLE might replace standard biopsies in patients with visible oesophageal/gastric lesions. The major drawback of pCLE is its high cost, which is significantly higher compared with histopathology (US$500 for pCLE vs. US$150 for five biopsies), and unfortunately, pCLE is usually not reimbursed.
Our study has several limitations. First, we included 67 patients with 74 lesions, and even though the minimum number of planned patients was 50, our sample size is still limited. Second, all sequences were evaluated offline, and real-time assessment was not performed. Finally, we did not examine the ability of pCLE to assess other important characteristics such as grading, tumour-cell dissociation and so on.
The strength of this study is its atypical design and the fact that a definitive diagnosis was confirmed (in the majority of cases) from resection specimen.
In conclusion, our study showed that pCLE is comparable with standard biopsies in terms of overall diagnostic accuracy to diagnose oesophageal/gastric lesions correctly. Our results suggest that in patients with a visible oesophageal/gastric lesion, pCLE could be recognised as a standard method) (providing an accurate histopathological diagnosis.
Footnotes
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by a grant from the Czech Ministry of Health (Grant: 16-27648A; ClinicalTrials.gov identifier: NCT0292049).
Ethics approval
The study protocol was in accordance with the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the local ethics committee.
Informed consent
Written informed consent was obtained from all patients..
