Abstract
Background and aims
Sessile serrated adenomas/polyps (SSA/Ps) are difficult to differentiate from non-neoplastic tissue on white-light endoscopy. Confocal laser endomicroscopy (CLE) provides subcellular imaging and real-time “optical biopsy”. The aim of this study was to prospectively describe CLE features of SSA/Ps.
Patients and methods
Consecutive patients with SSA/Ps were prospectively evaluated with probe-based CLE imaging. CLE images and polyp histology were independently reviewed by three endoscopists and an expert gastrointestinal (GI) pathologist. Distinguishing CLE features of SSA/Ps were identified in conjunction with pathologic correlation.
Results
In total, 260 CLE images were generated from nine SSA/Ps evaluated in seven patients. Four consensus CLE features of SSA/P were identified: (1) a mucus cap with a bright, cloud-like appearance; (2) thin, branching crypts; (3) increased number of goblet cells and microvesicular mucin-containing cells; and (4) architectural disarray, with dystrophic goblet cells and lack of regular circular crypts
Conclusion
This is a novel description of characteristic CLE features of SSA/Ps. The four features we identified are easy to detect and may allow for CLE to serve as a diagnostic modality.
Introduction
Sessile serrated adenomas/polyps (SSA/Ps) are often flat, located in the proximal colon, and estimated to contribute to up to 30% of all colorectal cancer (CRC).1–3 They may also be responsible for interval cancers as they are the precursor lesions for rapidly developing carcinoma via the microsatellite instability and CpG island methylator phenotype pathways. 1
Given their subtle morphologic features on white-light endoscopy (WLE), SSA/Ps are difficult to detect and differentiate from non-neoplastic polyps and are incompletely resected in up to one-third of cases.1,2,4 Improper endoscopic characterization could have significant consequences if resect and discard or diagnose and leave in situ strategies are to be implemented. Image-enhanced endoscopy and dye-based chromoendoscopy (with or without magnification) has a steep learning curve, and the evidence to support its use in SSA/P detection and classification to date has been limited.5–7
Confocal laser endomicroscopy (CLE) allows for in vivo image magnification of 1000-fold and thus can provide subcellular imaging of the gastrointestinal (GI) mucosa, providing real-time “optical biopsy”. 8 CLE has shown the ability to detect dysplasia and neoplasia in Barrett’s esophagus and differentiate between hyperplastic and adenomatous colorectal polyps.8–10
Given the importance of SSA/Ps in the development of right-sided and interval CRC and the current difficulty of WLE to correctly characterize these lesions, the aim of this study was to prospectively describe CLE features of SSA/Ps.
Methods
Endoscopic procedure
The study was approved by the Institutional Review Board at the Yale University School of Medicine. It was a prospective proof-of-concept cohort study at one tertiary care academic center. Consecutive patients with known SSA/Ps referred for endoscopic mucosal resection were prospectively included in the study after informed consent was obtained. Colonoscopy using standard high-definition WLE (Olympus GIF-H180, Japan) was first performed on all patients to detect the SSA/P. In order to illuminate the extracellular matrix of the mucosal epithelium and lamina propria 5 ml of 10% fluorescein sodium was given intravenously. 11 The high-definition confocal imaging probe (pCLE), (Mauna Kea Technologies, France) was passed through the biopsy channel of the endoscope and positioned onto the mucosal surface of the SSA/P. Multiple dynamic images of each lesion were collected at an average rate of 12 frames per second. Probe-based CLE allows for a 240 µm field of view, 55 to 65 µm imaging depth, and 1 µm resolution. 12 On average, 30 CLE images were acquired per lesion. Images where greater than 70% of field of view was obscured by artifact (motion or debris) or poor tissue apposition were excluded. All other acquired images were used for review. Following CLE imaging, the SSA/P was endoscopically resected and submitted for standard pathologic examination.
Image review and histologic correlation
Prior to patient enrollment, an exploratory meeting with three endoscopists experienced in CLE imaging and an expert GI pathologist was arranged to discuss CLE technology, review prior CLE literature for colorectal polyps, and list potential CLE features of SSA/Ps. All resected lesions were initially evaluated by a GI pathologist in a blinded and prospective manner for patient diagnosis. The histologic slides of each patient were re-reviewed by a second pathologist with expertise in GI pathology (JG) to confirm the diagnosis of SSA/P according to WHO histologic guidelines. 13 The set of CLE images associated with every SSA/P was then independently reviewed by the three endoscopists and GI pathologist and assessed for the postulated CLE features. Following independent analysis, a collaborative review of each CLE image set in conjunction with pathologic correlation was conducted. Only CLE features present in every SSA/Ps were described as distinguishing CLE features of SSA/Ps.
Results
Seven patients with known SSA/Ps referred to our academic center for endoscopic mucosal resection were prospectively included in this proof-of-concept study. Nine SSA/Ps were identified from the seven patients and 260 CLE images were generated. The median size of the polyps was 14 mm (range 7–17 mm). Five polyps (55%) were located in the right colon, proximal to the splenic flexure. In all of the cases, CLE imaging was initially limited by the presence of a mucus cap on the SSA/P. The mucus cap was rinsed with water to achieve cellular imaging.
CLE Features of SSA/Ps.

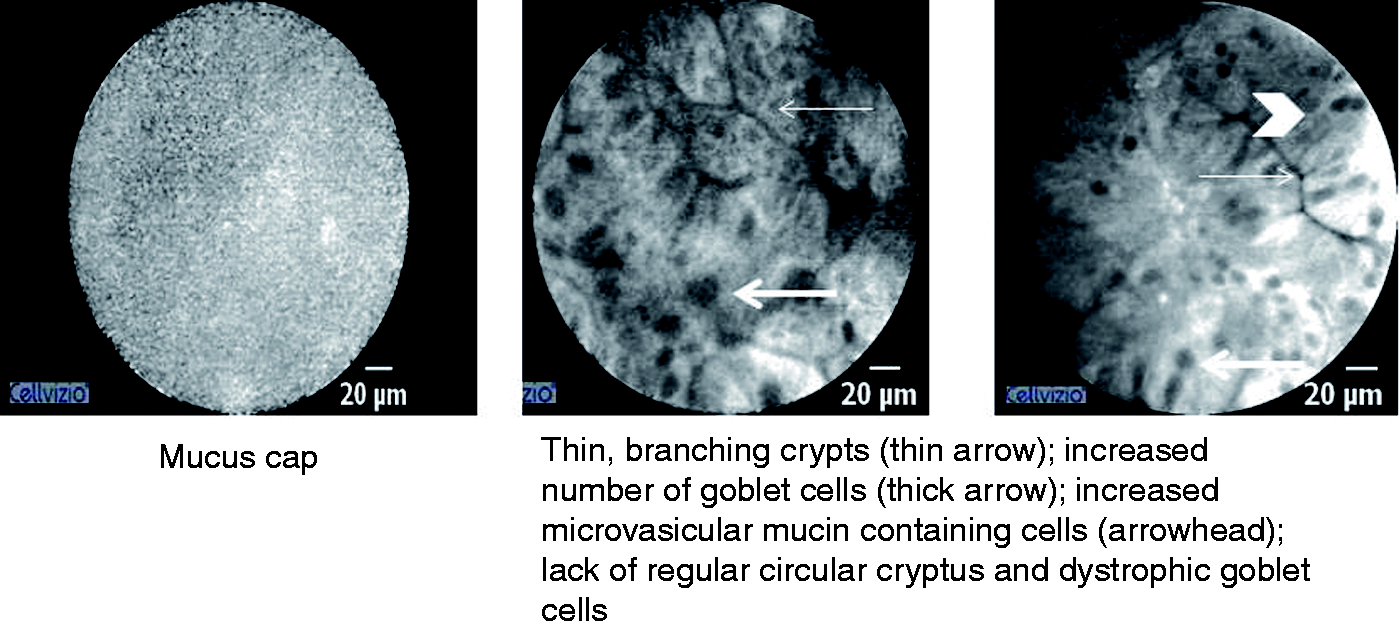
CLE features of SSA/Ps.
Discussion
This is the first published description of characteristic CLE features of SSA/Ps. The four features we identified are easy to detect and could allow for CLE to serve as a new modality to differentiate SSA/Ps from normal colonic mucosa and other polyps.
The most distinguishing histologic feature of SSA/P, which differentiates them from hyperplastic polyps, is the presence of crypt architectural distortion, most often manifesting as dilation, branching or horizontal extension at the base of crypts.
14
This architectural change is best assessed by examining well-oriented, full thickness histologic sections of the mucosa and cannot be visualized with the more superficial, cross-sectional CLE imaging. In the CLE images, we identified fern-like thin and branching crypts, which correlate with the complex and prominent serrated luminal edges of the superficial crypts in the SSA/Ps, pathologically referred to as “hyperserration” (Figure 2(a)).
Histologic images of SSA/Ps.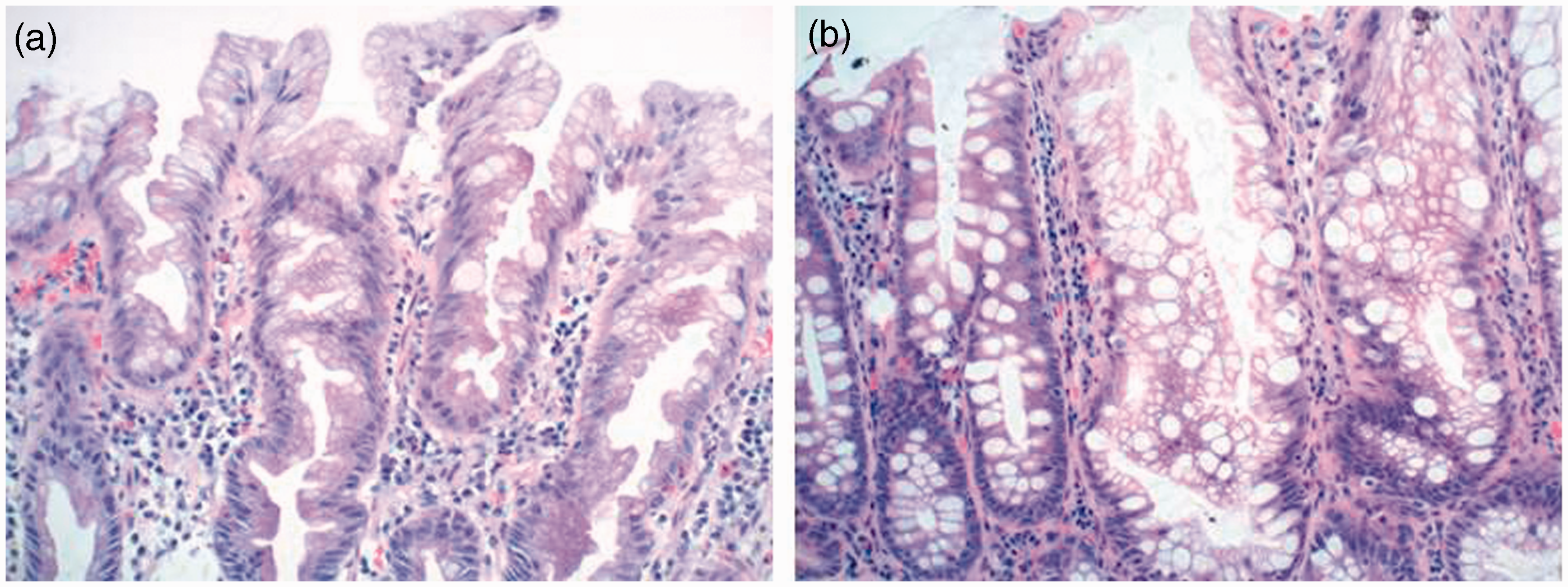
An increase in overall mucin was also seen on CLE with an increased number of goblet cells (large, well-defined black spheres) and cells with microvesicular mucin (smaller gray spheres). These mucin-containing cells were found in variably thick epithelial layers around the thin branching crypts. Compared with previously published reports about hyperplastic polyps, SSA/Ps have more mature goblet cells and mucinous cells within the bases of their crypts (Figure 2(b)). 14 These cells contribute to excess mucin production within the lumen of crypts, resulting in both crypt dilation at the base as well as the “mucus caps” seen on the surface of 65–100% of SSA/Ps with WLE. 2 CLE visualized the mucus cap as a bright, featureless cloud containing debris. While CLE effectively visualized the mucus caps and the overall increase in mucin with goblet cells and cells with microvesicular mucin, the orientation of crypts was not as easily recognized. Therefore, while CLE identified thin and branching crypts, differentiating the apex from the base of the crypt was not as facile as it is on standard histologic transverse cross-sections.
CLE imaging of goblet cells within SSA/P did not show continuity of the goblet cell apex with the crypt, as is seen in normal colonic epithelium. We interpreted this observation as analogous to the histologic finding of dystrophic goblet cells; i.e. goblet cells that have lost polarity within the epithelium. On the CLE images, the dystrophic goblet cells were seen as dark ovoid/rounded structures, not in contact with the thin branch-like lumina. These appeared to be randomly distributed among other columnar to ovoid cells that had dark gray flocculent cytoplasmic features. These cells were the microvesicular epithelial cells of the SSP. A normal epithelial cell on CLE appears to have a relatively bright and uniform cytoplasm and will be very columnar.
The final feature we identified is likely to be the most sensitive but least specific for SSA/Ps. All CLE images of SSA/Ps in this study demonstrated a lack of normal colonic cellular and glandular architecture, which are crypts with round regular luminal openings with a uniform layer of epithelial cells (Figure 3). The absence of circular luminal openings of the crypts on CLE correlates well with the histologic features of serrated lesions, including the disorganized architectural features and distorted crypt growth patterns of SSA/P.
Normal colonic mucosa on CLE.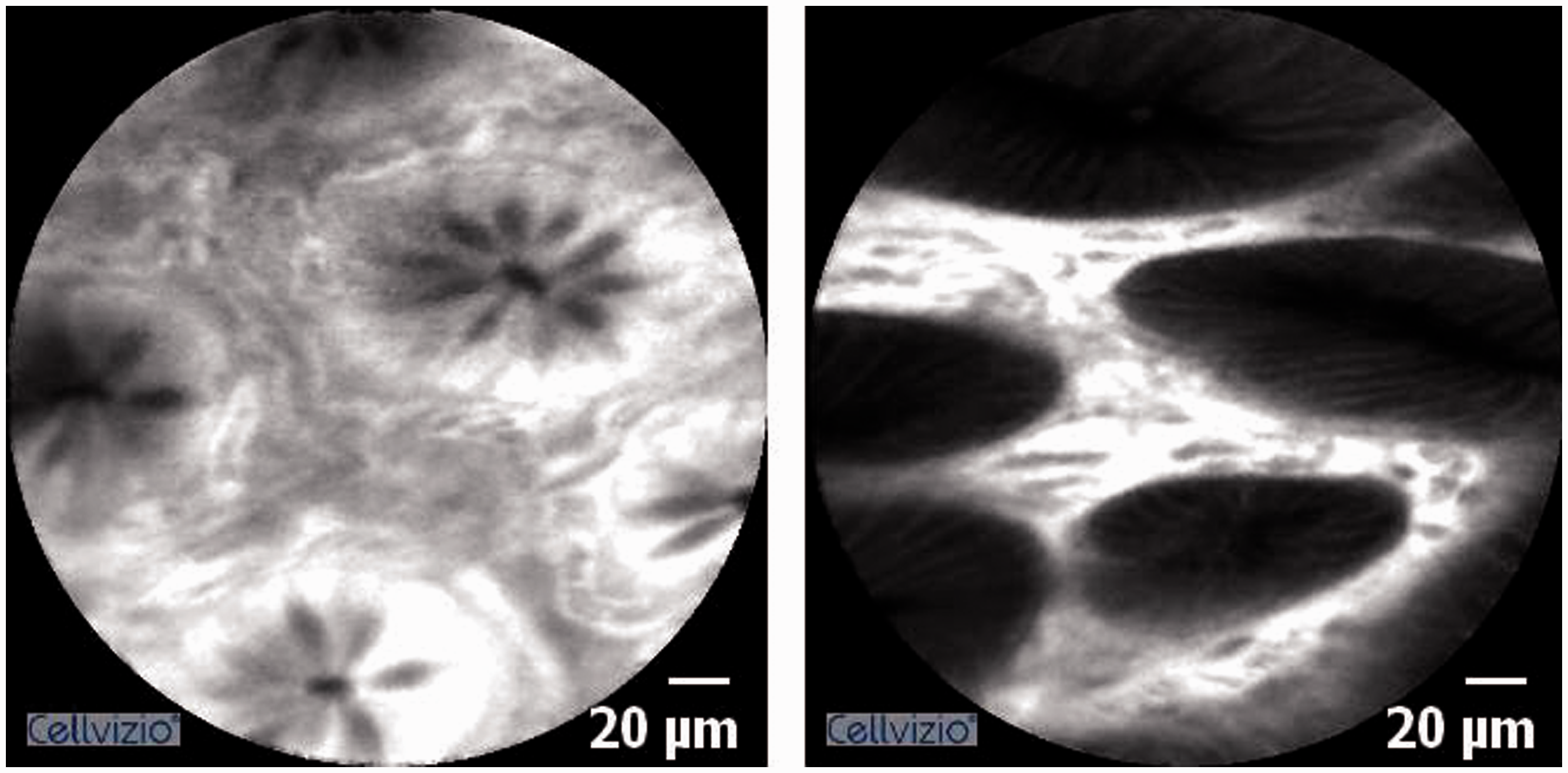
CLE classification systems of colorectal polyps have been established for probe-based CLE (Miami classification) and endoscope-based CLE (Mainz classification); however, this report is the first description of SSA/P features.8,15 Adenomas in both the Miami and Mainz classification demonstrate a villiform transformation of the crypts, which is in stark contrast to the elongated, thin branching crypts we identified in SSA/Ps. In the Miami classification, the crypts of hyperplastic polyps appear as thin slits or stellate openings. While the elongated nature of crypts seen on CLE for SSA/Ps in this study is similar, the significant branching of the crypts is likely more specific to SSA/Ps. However, it may be possible that this irregular or fern-like thin branching may be a feature that distinguishes all serrated polyps from adenomatous polyps as opposed to distinguishing between serrated subtypes. We identified an increase in overall mucin and goblet cells in SSA/Ps, in contrast to the depletion of goblet cells described in hyperplastic polyps (Mainz criteria) and adenomas (Miami classification). Given the clear visibility of goblet cells on CLE this is an easily applied feature for in vivo analysis. Our finding of architectural disarray in SSA/Ps is consistent with the CLE criteria for adenomatous polyps (Mainz classification).
While certain features of SSA/Ps such as the mucus cap and often larger size compared with hyperplastic polyps make them more visible on WLE, the overall detection and diagnosis rate for SSA/Ps varies significantly among endoscopists, ranging from 0% to 13%. Dye-based chromoendoscopy has shown some promise but remains cumbersome, and image-enhanced endoscopy has not yet been successful in either detection or accurate characterization of these lesions. 5 While CLE has its limitations (intravenous contrast agent, cost, learning curve), our pilot study suggests that it has the potential to enhance our ability to differentiate SSA/Ps from normal colonic mucosa, and we describe features distinguishing them from normal tissue. Such differentiation may allow for better characterization of SSA/P borders and allow for more precise and complete resections, which is a deficiency in our current clinical practice. In the right colon, extensive areas of hyperplastic change may occasionally be identified which can be confused for SSA/Ps and may lead to large resections which may not be beneficial. Dysplasia is rarely present in SSA/Ps and was not present in our study lesions. CLE has demonstrated the ability to detect dysplasia in other settings such as Barrett’s esophagus; however, further study is required.
This is a proof-of-concept study limited by a small sample size, and findings need to be validated in larger studies. However, evaluation of nine SSA/Ps generated 260 high-quality CLE images, and the features reported here were consistently present in all polyps and were correlated with histologic findings. Prospective in vivo assessment of SSA/Ps versus non-SSA/P lesions would be the next step in further assessing CLE’s role for SSA/Ps.
As they may play a vital role in the development of interval cancers, SSA/Ps have recently become an important factor in the effort to improve CRC screening. Increased detection, in vivo differentiation, and complete resection of these lesions is essential. Moreover, it is often difficult to differentiate SSA/Ps from thickened folds or peri-appendiceal lymphoglandular complexes in the proximal colon. Prospective in vivo assessment of SSA/Ps versus non-SSA/P lesions, including hyperplastic polyps, would be the next step in further assessing CLE’s role for SSA/Ps. While this is a proof-of-concept study that requires prospective validation with comparison to hyperplastic polyps and tubular adenomas, CLE characteristics of SSA/Ps were identified that may facilitate real-time endoscopic optical diagnosis and guide endoscopic therapy.
Footnotes
Conflicts of Interest
None declared.
Funding
This study was supported by NIH T32 DK007356-36 (to N.D. Parikh)
