Abstract
Introduction
Achalasia is a primary motor disorder of the oesophagus characterised by absence of peristalsis and insufficient lower oesophageal sphincter relaxation. With new advances and developments in achalasia management, there is an increasing demand for comprehensive evidence-based guidelines to assist clinicians in achalasia patient care.
Methods
Guidelines were established by a working group of representatives from United European Gastroenterology, European Society of Neurogastroenterology and Motility,
Results
These guidelines focus on the definition of achalasia, treatment aims, diagnostic tests, medical, endoscopic and surgical therapy, management of treatment failure, follow-up and oesophageal cancer risk.
Conclusion
These multidisciplinary guidelines provide a comprehensive evidence-based framework with recommendations on the diagnosis, treatment and follow-up of adult achalasia patients.
Abbreviations
Appraisal of Guidelines for Research and Evaluation body mass index botulinum toxin oesophageal adenocarcinoma European Association of Endoscopic Surgery European Society of Gastrointestinal and Abdominal Radiology European Society of Neurogastroenterology and Motility gastro-oesophageal reflux disease Grading of Recommendations Assessment, Development, and Evaluation high-resolution manometry impedance planimetry integrated relaxation pressure lower oesophageal sphincter laparoscopic heller myotomy oesophago-gastric junction PD, pneumatic dilation patient, intervention, control, outcome peroral endoscopic myotomy proton pump inhibitor randomised controlled trial squamous cell carcinoma timed barium oesophagram United European Gastroenterology.
Introduction
Achalasia is a primary motility disorder in which insufficient relaxation of the lower oesophageal sphincter (LOS) and absent peristalsis result in stasis of ingested foods, subsequently leading to oesophageal symptoms of dysphagia, regurgitation, chest pain or weight loss. 1 Achalasia occurs as an effect of the destruction of enteric neurons controlling the LOS and oesophageal body musculature by an unknown cause, most likely inflammatory. Idiopathic achalasia is a rare disease and affects individuals of both sexes and all ages. The annual incidence is estimated between 1.07 and 2.2 cases per 100,000 individuals, with prevalence rates estimated between 10 and 15.7 per 100,000 individuals.2–4
Manometric subtypes of achalasia.
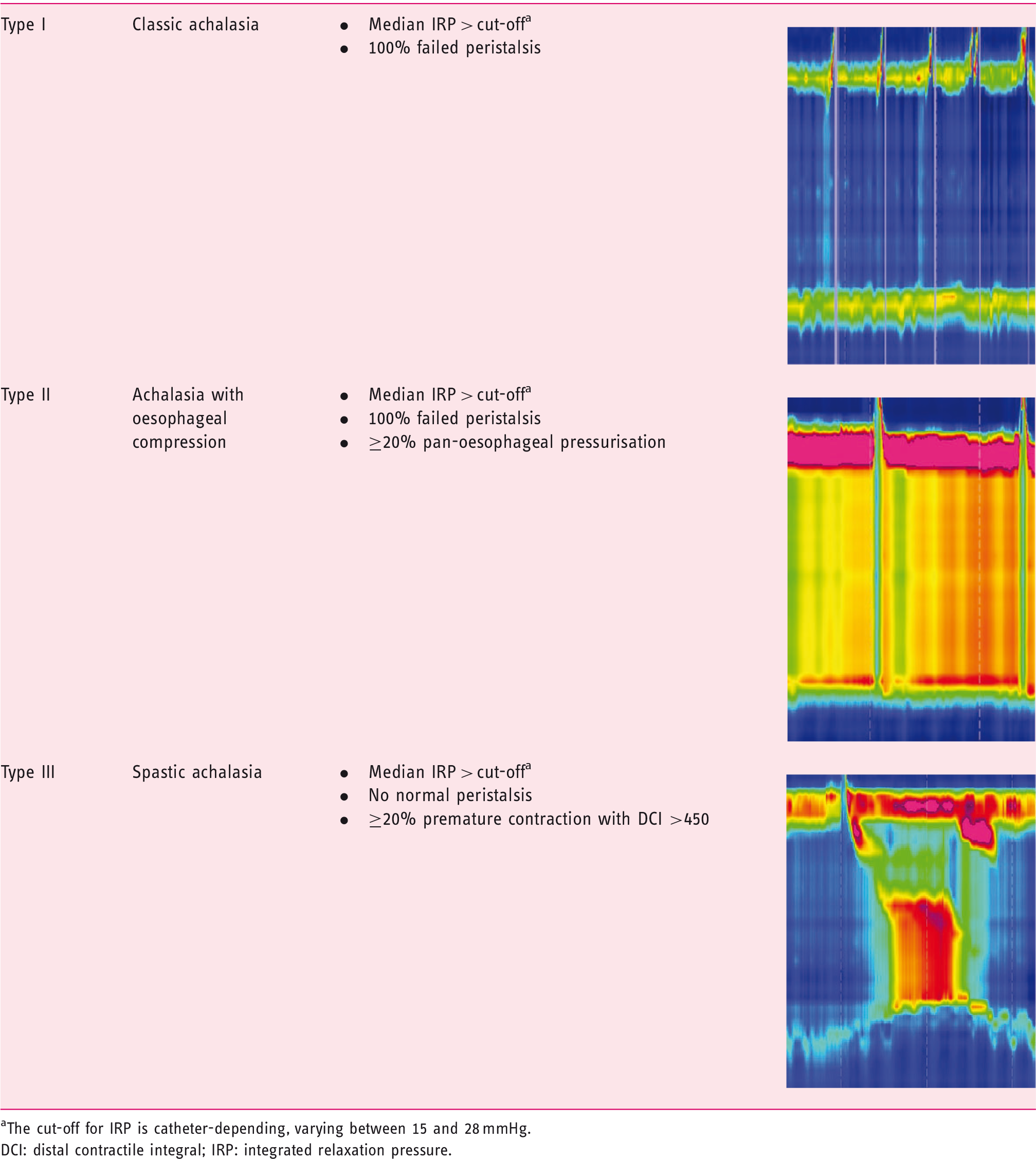
The cut-off for IRP is catheter-depending, varying between 15 and 28 mmHg.
DCI: distal contractile integral; IRP: integrated relaxation pressure.
The clinical care of patients with achalasia has changed significantly in the past decade under the influence of new developments such as HRM, per-oral endoscopic myotomy (POEM) and studies providing new insights regarding achalasia subtypes, cancer risk and follow-up. Given the substantial growth of knowledge in past years, there is need for comprehensive, evidence-based European guidelines covering all aspects of the disease. These multidisciplinary guidelines aim to provide an evidence-based framework with recommendations on the diagnosis, treatment and follow-up of adult achalasia patients. Chagas disease and achalasia secondary to other disorders, as can be seen after fundoplication, bariatric surgery, sarcoid infiltration, opiate usage or malignancy, are not covered by these guidelines. These guidelines are intended for clinicians involved in their management, including gastroenterologists, endoscopists, radiologists, gastrointestinal (GI) surgeons, dietitians and primary-care practitioners.
Methodology
The achalasia guidelines working group
Ten researchers and clinicians with recognised expertise in the field of clinical achalasia management were gathered (A.B., G.B., P.F., A.P., S.R., A.S., A.T., E.T., B.W. and G.Z.) on behalf of United European Gastroenterology (UEG), the European Society of Neurogastroenterology and Motility (ESNM), the
From assessment of evidence to recommendation
Grading of Recommendations Assessment, Development, and Evaluation (GRADE) definitions of quality and certainty of the evidence.

GRADE on strength of recommendation and guide to interpretation.

Consensus process
In order to establish consensus-based recommendations, a second physical meeting was organised in Amsterdam, the Netherlands, on 11 April 2019. GRADE assessments and recommendations were presented and discussed. Voting was conducted according to the nominal group technique and based upon a six-point Likert scale (1 = strongly disagree; 2 = mostly disagree; 3 = somewhat disagree; 4 = somewhat agree; 5 = mostly agree; 6 = strongly agree). A recommendation was approved if >75% of the members agreed (reflected by a Likert score of 4–6).
Recommendations
Summary of recommendations of the United European Gastroenterology Clinical Guidelines Committee for the diagnosis, management and follow-up of achalasia.

LOS: lower oesophageal sphincter; TBO: timed barium oesophagram; OGJ: oesophago-gastric junction; CT: computed tomography; BTX: botulinum toxin; POEM: per-oral endoscopic myotomy; LHM: laparoscopic Heller myotomy; PD: pneumatic dilation: PPI: proton pump inhibitors; GORD: Gastro-oesophageal reflux disease; GI: gastrointestinal.
1. Achalasia diagnosis
1.1. What is the current definition of achalasia?

1.2. What is the value of HRM and conventional manometry in achalasia diagnosis?
The diagnosis of achalasia requires not only impaired oesophago-gastric junction (OGJ) relaxation, but also absent or abnormal peristalsis. Therefore, oesophageal manometry is considered as being the gold standard for the diagnosis of achalasia, as it evaluates both pressures of the LOS and contractility of the oesophageal body. Worldwide, HRM, usually defined as manometry carried out with a catheter with at least 21 pressure sensors spaced at 1-cm intervals, 5 is rapidly replacing conventional manometry. The generally perceived advantages of HRM over conventional manometry are that positioning of the catheter is less critical and that interpretation of the recorded pressures, displayed in the form of topographical colour-coded plots, is more intuitive.
In four of the five included studies, the diagnosis of achalasia was made with HRM more often than with conventional manometry.6–9 However, one may argue that a higher rate of achalasia diagnosis with HRM does not prove that HRM is better than conventional manometry; HRM might also lead to more false-positive findings. The only prospective randomised trial that compared HRM and conventional manometry 9 had the additional advantage of defining the clinical outcome after six months as the gold standard, and found a superior sensitivity of HRM for the diagnosis of achalasia to that of conventional manometry (93% vs. 78%). The specificities of both tests were equal (100%). 9
1.3. What is the value of (timed) barium swallow studies in achalasia diagnosis?
The barium oesophagram is generally seen as a valuable and complementary, but relatively insensitive, diagnostic test. One study evaluated the diagnostic value of barium oesophagraphy in comparison to HRM and found a high sensitivity but poor specificity for detecting dysmotility. The authors concluded that barium swallow studies accurately rule out achalasia-related dysmotility but are not very helpful in diagnosing other causes of dysmotility.
12
Two studies comparing barium oesophagraphy with conventional manometry found sensitivities for achalasia diagnosis between 58% and 75%.11,13 However, as the positive predictive accuracy was 96%, the authors concluded that the barium oesophagram is a useful tool in achalasia diagnosis.
11
Similar sensitivity and specificity rates were obtained in another study comparing barium swallow studies with HRM; the diagnostic sensitivity, specificity and accuracy of the barium oesophagram were 78.3%, 88.0% and 83.0%, respectively.
14
Consequently, it may be concluded that diagnosing achalasia by using barium oesophagram alone has a limited yield. The technique of timed barium oesophagram (TBO) is similar to the usual barium swallow study but uses set time intervals (one, two and five minutes) after ingestion of a fixed barium suspension to measure the height and width of the barium column in order to assess oesophageal emptying more objectively (Figure 1).
15
Because of this advantage, TBO is generally preferred over a standard barium oesophagram. One study compared TBO to HRM, and found a sensitivity of 85% and specificity of 86%.
15
Interpretation of timed barium esophagram. Radiographs taken 0, 1, 2 and 5 minutes in left posterior oblique position after ingestion of 100 to 200 mL low-density barium suspension in an achalasia patient. Measurement of height and width of barium column, measured from the OGJ to the barium-foam interface. Barium height of >5 cm at 1 min and >2 cm at 5 min are suggestive of achalasia.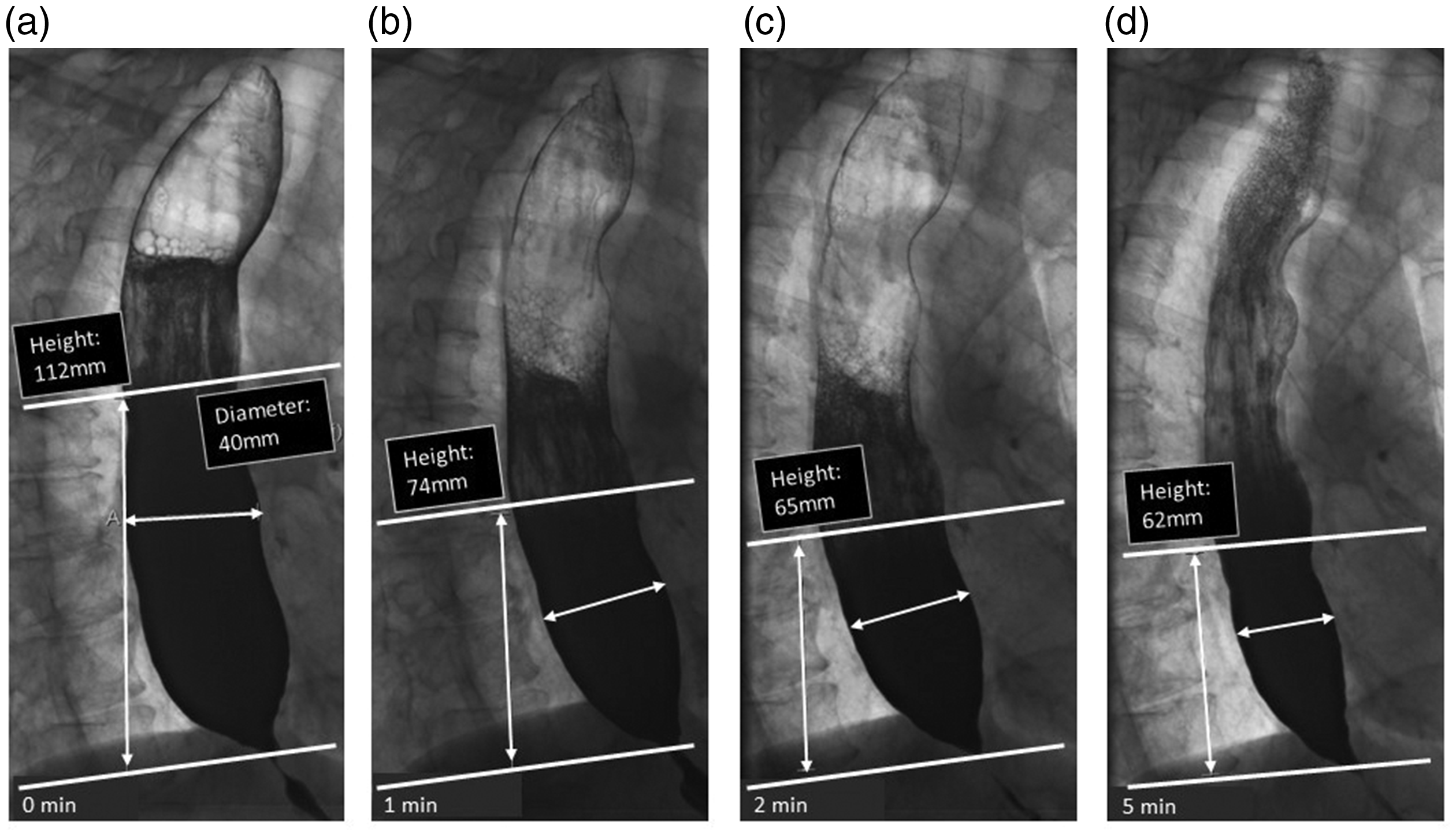

1.4. What is the value of oesophageal impedance planimetry in the diagnosis of achalasia?
Oesophageal impedance planimetry is a technique in which the cross-sectional area of the oesophagus is simultaneously measured at multiple levels using a saline-filled cylindrical bag containing an array of impedance electrodes. 6 The commercially available device for endoluminal impedance planimetry is known as Endoflip®.
Studies using impedance planimetry have consistently demonstrated that the distensibility of the OGJ is reduced in untreated achalasia compared to healthy controls.16–19 A systematic review identified six studies with data on OGJ distensibility in untreated achalasia patients (
However, in order to distinguish achalasia from OGJ outflow obstruction, information about the motility of the tubular oesophagus is required, which is not provided by impedance planimetry measurement. Recent studies indicate that dynamic impedance planimetry can also provide information on peristalsis.21,22 However, this technique assesses distension- rather than swallow-induced contractions, and requires sedation. Furthermore, high-quality diagnostic studies comparing impedance planimetry with the gold standard HRM are not available yet. In line with this, one recommendation from a recent American Gastroenterological Association clinical practice update on functional lumen imaging is that clinicians should not make a diagnosis of achalasia based on impedance planimetry alone. 23

1.5. What is the value of endoscopy in achalasia diagnosis?

1.6. In which patients should additional diagnostic tests be performed in order to exclude pseudo-achalasia?

1.7. What information should the newly diagnosed patient receive?
Information the newly diagnosed achalasia patient should receive.

2. Achalasia treatment
2.1. What should we aim for when treating achalasia patients?

2.2. What is the role of oral pharmacological therapy in achalasia?

2.3. What is the comparative therapeutic efficacy and safety of endoscopic botulinum toxin injection in the treatment of achalasia?

2.4. What is the comparative therapeutic efficacy and safety of endoscopic dilation?

2.5. What is the comparative therapeutic efficacy and safety of POEM?

2.6. What is the comparative therapeutic efficacy and safety of surgical myotomy?

2.7. What are predictors of treatment outcome? How to choose initial treatment

2.8. Overall recommendations on treatment (comparative effectiveness and safety)

2.9. How to treat recurrence post LHM

2.10. How to treat recurrence post POEM

2.11. What are the indications for oesophagectomy?

2.12. What is the role of alternative therapies in the treatment of achalasia?

3. Achalasia follow-up
3.1. How to diagnose and manage recurrent or persistent dysphagia after treatment
Potential causes for persistent and recurrent dysphagia after initial treatment.

Given the wide variety of potential causes of recurrent dysphagia, it is critical to undertake a comprehensive evaluation using objective testing in order to determine the pathophysiology underpinning the recurrent symptoms, and thus select the appropriate treatment. Conversely, in selected cases of persistent dysphagia, where the diagnosis of achalasia is beyond doubt, it may be appropriate to proceed immediately to further treatment without repeat testing (e.g. POEM after failure to improve with PD).

3.2. How to diagnose and manage recurrent or persistent chest pain after treatment
Although chest pain is one of the main presenting symptoms of achalasia, its response to treatment is less well studied and remarkably underreported, most likely as dysphagia is considered the leading and most relevant symptom. Nevertheless, up to 64% of patients report chest pain, often occurring in the middle of the night (in 47% of patients with chest pain) and lasting from a few minutes to almost 24 hours. 111 In contrast to dysphagia, chest pain is more challenging to treat and represents a risk factor for unsatisfactory treatment results for both PD and LHM.37,54,112 In approximately 19% of patients, chest pain is completely relieved following LHM, but in the remainder, chest pain persists, with an intensity that is less (73%), similar (21%) or even more severe (4%) than before surgery. 113 Comparable results have been reported for PD. 111 Of note, chest pain persists in these patients, even though dysphagia was successfully treated. In general, achalasia-associated chest pain seems to decrease with time, but complete disappearance is rather exceptional. 111
The exact cause underlying (non-cardiac) chest pain remains unknown, and can be attributed to acid reflux, oesophageal motor abnormalities or visceral hypersensitivity. However, as chest pain is also considered to result from oesophageal distension as a result of incomplete emptying, treatment failure should first be excluded in patients with persistent or recurrent chest pain by performing oesophageal manometry and TBO.
If manometry (IRP above cut-off; catheter-depending, varying between 15 and 28 mmHg) 114 or TBO barium column height of >5cm after 5 minutes are abnormal, 115 treatment should aim to normalise oesophageal emptying. HRM also serves to exclude spastic contractions as cause of the pain. If there is no evidence indicating insufficient treatment, one can consider investigation for GORD as the trigger of chest pain using 24-hour pH (impedance) monitoring and treat accordingly. 116 Data demonstrating the effect of PPI on chest pain in achalasia are, however, lacking, and anecdotally the response to PPI is poor if there is chest pain without heartburn.

3.3. How to manage reflux disease after treatment
As the aim of achalasia treatment is to alleviate the OGJ obstruction, an expected side effect of treatment is the occurrence of GORD, usually defined in achalasia as the presence of reflux oesophagitis or pathological acid exposure. Indeed, GORD is frequently observed after treatment (10–31% of cases after PD,51–53,55,58,121 5–35% after LHM52,53,55,121–123 and up to 60% of patients after POEM50,60,61,124–126). GORD complications, including peptic stricture, Barrett’s mucosa and oesophageal adenocarcinoma (OA), have been reported after achalasia treatment.124,126–130 Comparative studies demonstrated that the rate of GORD was similar after PD and LHM with fundoplication. 121 One study showed that LHM without lateral and posterior dissection might also achieve sufficient reflux control. 131 However, in other studies, the prevalence of GORD was significantly higher after POEM or LHM without fundoplication than after PD or LHM with fundoplication.50,60,62,132 Therefore, systematic screening for GORD after achalasia treatment should be recommended if the risk for GORD is high. Moreover, due to the different GORD rates, the choice of achalasia treatment should take into account the risk of iatrogenic reflux disease. In line with this, empiric PPI therapy might be considered in patients who undergoing myotomy without an anti-reflux procedure.
GORD symptoms such as heartburn and regurgitation are not reliable to diagnose GORD in achalasia patients, especially as regurgitation is also a hallmark of achalasia and poor oesophageal emptying. An upper endoscopy can reveal oesophagitis and Barrett’s mucosa as proof of GORD. Another way to diagnose GORD is 24-hour oesophageal pH monitoring. The interpretation of this examination requires a careful review of pH tracings to eliminate periods of oesophageal fermentation. 53 The correlation between oesophageal symptoms and objective diagnosis of GORD (including oesophagitis and oesophageal acid exposure) is poor.62,123,133–135 Upper GI endoscopy, TBO and 24-hour pH monitoring might be complementary.

3.4. Is surveillance endoscopy for dysplasia needed?
What is the incidence of oesophageal cancer in achalasia patients?
Achalasia is a risk factor for oesophageal cancer. Poor oesophageal clearance increases bacterial growth, chemical irritation and mucosal inflammation that can facilitate dysplastic changes of oesophageal epithelial cells and result in squamous-cell carcinoma (SCC). 141 Furthermore, acid exposure secondary to reduction of OGJ pressure as a consequence of achalasia treatment may lead to Barrett’s mucosa and OA. 142
The exact level of risk for oesophageal cancer (SCC and OA) is controversial. Differences in study design (retrospective or prospective, length of Follow-up, number of patients, countries) might explain some of the observed differences. While the absolute risk of oesophageal cancer is quite low in achalasia, the relative risk of cancer is higher in achalasia patients than in the general population (risk ratio to develop OA and SCC in achalasia patients is 6.63 and 72.65, respectively).143,144 Most of the cases of carcinoma are observed more than 10 years after symptom onset.144,145 The type of treatment does not influence the risk of cancer,130,146 but to date there are no long-term data following POEM. Cancer risk might be higher in males and in patients with Chagas disease.130,146,147
Screening practices differ among geographic regions (routine endoscopy vs. no endoscopy, screening intervals).92,148 Chromoendoscopy with lugol was proposed to improve the detection rate of dysplastic lesion, but the yield was low and hampered by stratification risk. 145

Conclusions and future perspectives
The ESNM/UEG guidelines on the management of achalasia are the result of an evidence-based approach and international and multidisciplinary efforts. These guidelines provide recommendations for key aspects of the diagnosis and management of achalasia, combined with comments based on the best available literature and the opinions of leading European achalasia experts. The main objectives of these guidelines are to reduce variation in practice and to improve patient outcomes across Europe. Consequently, thorough and extensive dissemination of these guidelines is needed to assure high compliance in clinical practice. Promotion of these guidelines as well as education play a key role in this regard. Future well-designed clinical trials should address the knowledge gaps and unmet needs that have arisen during the development of these guidelines.
Supplemental Material
UEG903213 Supplemental Table - Supplemental material for European Guideline on Achalasia – UEG and ESNM recommendations
Supplemental material, UEG903213 Supplemental Table for European Guideline on Achalasia – UEG and ESNM recommendations by RAB Oude Nijhuis, G Zaninotto, S Roman, GE Boeckxstaens, P Fockens, MW Langendam, AA Plumb, AJPM Smout, EM Targarona, AS Trukhmanov, BLAM Weusten and AJ Bredenoord in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
Research support: Bayer (A.B.), Crospon (S.R.), Diversatek (S.R.), Laborie (A.B.), Medtronic (S.R.), Nutricia (A.B.), Norgine (A.B.). Advisory, honoraria or consultation: Calypso (A.B.), Celgene (A.B.), Cook (P.F.), Diversatek (A.B.), EsoCap (A.B.), Ethicon (P.F.), Falk (A.B.), Fujifilm (P.F.), Laborie (A.B.), Medtronic (P.F., S.R.), Olympus (P.F.), Regeneron (A.B.). Speaker’s bureau: Actavis (A.P.), Falk (A.B.), Janssen-Cilag (A.P.), Laborie (A.B.), Mayoly Spindler (S.R.), Medtronic (A.B.), Takeda (A.P.). No disclosures or conflicts of interest: G.B., M.L., R.O.N., A.T., E.T., A.S., B.W., G.Z.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: These guidelines have been developed and funded within the United European Gastroenterology.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

