Abstract
Background
Acute pancreatitis (AP) is a frequent disorder with considerable morbidity and mortality. Obesity has previously been reported to influence disease severity.
Objective
The aim of this study was to investigate the association of adipose and muscle parameters with the severity grade of AP.
Methods
In total 454 patients were recruited. The first contrast-enhanced computed tomography of each patient was reviewed for adipose and muscle tissue parameters at L3 level. Associations with disease severity were analysed through logistic regression analysis. The predictive capacity of the parameters was investigated using receiver operating characteristic (ROC) curves.
Results
No distinct variation was found between the AP severity groups in either adipose tissue parameters (visceral adipose tissue and subcutaneous adipose tissue) or visceral muscle ratio. However, muscle mass and mean muscle attenuation differed significantly with p-values of 0.037 and 0.003 respectively. In multivariate analysis, low muscle attenuation was associated with severe AP with an odds ratio of 4.09 (95% confidence intervals: 1.61–10.36, p-value 0.003). No body parameter presented sufficient predictive capability in ROC-curve analysis.
Conclusions
Our results demonstrate that a low muscle attenuation level is associated with an increased risk of severe AP. Future prospective studies will help identify the underlying mechanisms and characterise the influence of body composition parameters on AP.
Key summary
Established knowledge
Acute pancreatitis is one of the most frequent gastrointestinal disorders implicating patient suffering and economical burden on healthcare systems; Studies on the association between obesity and severity of acute pancreatitis have yielded heterogeneous results.
New findings
In this large European cohort, muscle mass and muscle attenuation are important parameters for the outcome of acute pancreatitis; No association between visceral fat parameters and severity of acute pancreatitis was found in multivariate analysis; None of the body parameters are useful as predictive markers for severe disease.
Introduction
Acute pancreatitis (AP) is one of the most frequent diagnoses among gastrointestinal disorders requiring hospitalisation. 1 The course of the disease varies substantially, from self-restricting without complications to multi-organ failure and considerable risk of death. Multiple prognostic scoring systems, both clinical and radiological, have been developed, however none with sufficient predictive capacity. 2 Furthermore, the key steps of the pathophysiological mechanisms underlying AP are to a large extent unknown.
Multiple reports have demonstrated an association between obesity and worse outcome of AP.3–5 Body mass index (BMI) has been used as a surrogate for obesity, with high BMI considered an independent and established risk factor for severe AP.6,7 BMI has also been proposed as a prognostic variable to be included in scoring systems. 8 However, in more recent studies the impact of BMI has not been confirmed. 9 Rather, it seems to be the individual distribution of corporal fat that influences the outcome in AP.10–12
The pancreas is surrounded by visceral adipose tissue (VAT). As pro-inflammatory cytokines are secreted by VAT during AP, VAT has been suggested to be a major factor in the devolvement of peripancreatic necrosis. 13 Release of unsaturated fatty acids induce necrosis, systemic inflammation and organ failure. 14 The secretion is significantly greater in the VAT than in the subcutaneous adipose tissue (SAT). 15 Nevertheless, there are several reports where no association between VAT, SAT and severity of AP was found.5,16–18
It is well known that sarcopenia, regardless of patient BMI, has been associated with poor prognosis and increased risk of complications and morbidity in cancer patients. 19 The influence of muscle parameters such as muscle mass (MM), mean muscle attenuation (MMA) and visceral muscle ratio (VMR) on disease outcome has not been thoroughly analysed in AP with only a few papers published.16,17
In summary the results of previous works are heterogeneous and for certain parameters the number of studies is insufficient for making general conclusions.
Through this large European multicentre study we aimed to assess the significance of adipose and muscle parameters in the severity development of AP.
Materials and methods
Patients
Patients for this study were identified through a post hoc analysis of an established European multicentre cohort (involving six centres: Lund, Sweden; Helsinki, Finland; Barcelona, Spain; Tallinn, Estonia; Kaunas, Lithuania and Sofia, Bulgaria) as well as additional patients from two German centres (Halle and Leipzig). 20 In all centres, patients older than 18 years with a first episode of AP were retrospectively and consecutively enrolled. In the original six European centres patients were identified from January 2012 to January 2013, and in the two German centres the inclusion period was from January 2010 to January 2016. AP was defined by fulfilment of at least two out of three criteria: acute upper abdominal pain, serum amylase ≥3 times the upper limit, or characteristic findings on computed tomography (CT). At each centre in the European cohort 50 AP patients in whom at least one contrast-enhanced CT (CECT) was performed were included. In the German centres all available AP patients with at least one CECT within the investigated time frame were recruited. Severity of AP was assessed according to the revised Atlanta criteria. 21 Patients were classified as mild (no complications), moderately severe (organ failure for less than 48 h, defined morphological findings on CT or deterioration of a previous comorbidity) and severe AP (organ failure for more than 48 h).
Patients with known chronic pancreatitis, signs of chronic pancreatitis on CECT or with a CECT of insufficient quality were excluded. Ethical approval was obtained at the Martin Luther University of Halle-Wittenberg (date 21 July 2018, Bearbeitungsnummer 2017-96, Ethik-Kommission Medizinsche Fakultät, Martin Luther Universität Halle-Wittenberg). Due to the retrospective nature of the study and anonymisation of all data no informed consent was requested. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki (6th revision, 2008).
Body composition parameters
The first CECT performed after onset of AP was retrieved for all patients. Adipose tissue and muscle parameters were measured using DCMtool and OsiriXLite v9.0 (Pixmeo SARL, Geneva, Switzerland) for which excellent inter-observer agreement has previously been found.
22
A cross-sectional slice at the level of the L3 vertebra body was selected, and regions of interest (ROI) including the compartments of muscles and adipose tissue were drawn manually (see Figure 1). Visible peripancreatic necrosis and stranding extending to this area were present only in rare cases and thus not excluded from the measurements. Within the ROIs tissue was selected by the specific ranges −190 to −30 Hounsfield units (HU) for adipose tissue and −30 to 150 for muscle tissue, as reported earlier.
23
For visualisation of the separate ROIs see Supplementary Figure 1, A–E. VMR equals MM/VAT. The mean muscle attenuation (MMA) was extracted from the histogram in the range of −29 to +150 HU (see illustrative histogram in Supplementary Figure 1, A–E). As the measurement of myosteatosis is influenced by the CT phase, solely CTs performed in the native or venous phases were used for this analysis.
24
All analyses were performed by two independent investigators (MJM and LEK) together with a radiologist specialised in gastroenterological radiology (NL). To test the inter-observer variability of the analyses, a randomised sub selection of 35 CECTs (7.7% of the total cohort) was analysed a second time by MJM in a blinded manner. Inter-observer agreement coefficients were between 0.873 and 1.0 for all parameters, with a mean coefficient value of 0.954.
Regions of interest (ROI) of the distinct body compartments were visualised and defined by DCMTool. In (a) compartments for fat measurements are shown. The subcutaneous compartment is located between the blue lines. The visceral compartment resides within the green line. In (b) muscle compartments are demonstrated. Abdominal muscles are highlighted in blue, the Psoas muscle in red, and the muscles of the back in green. For more details of the measurements see 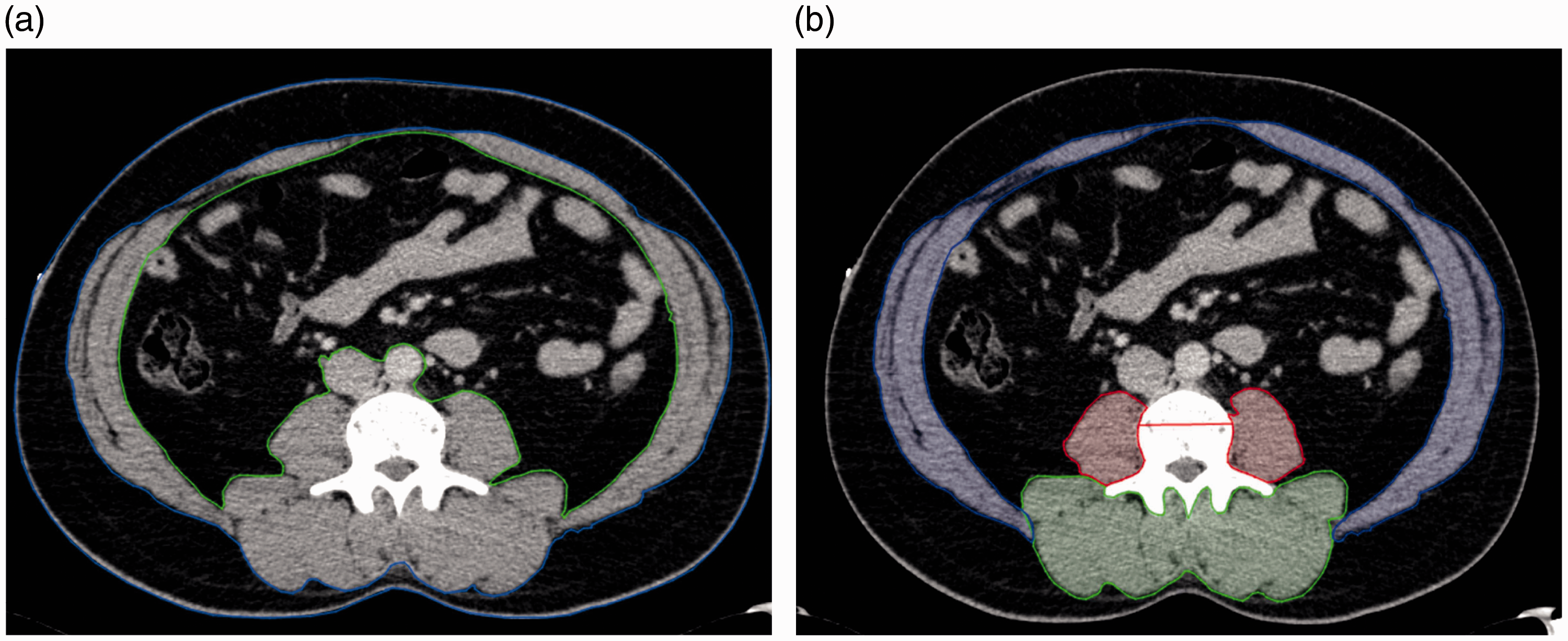
Statistical analysis
Data is presented as absolute numbers and percentages for categorical variables, and as median and interquartile range (IQR) for continuous variables. Categorical variables were compared using the chi-square test and Mann–Whitney U for continuous data. As normality was not assumed, the Mann–Whitney U or the Kruskal–Wallis tests were performed for comparison between groups. Possible associations between measured parameters and severe AP were investigated through univariate and multivariate logistic regression analysis. In multivariate analysis the data was adjusted for gender and age, where the latter was dichotomised at the upper IQR level (73 years). In addition, in both univariate and multivariate analysis the body composition parameters were separated into three groups (tertiles) based on the lowest, middle and highest IQR level of each parameter. Results of the regression analysis are presented as odds ratios (ORs) and adjusted odds ratios (aORs) with corresponding 95% confidence intervals (CIs) and p-values. To test the predictive capacity of the measured parameters receiver operating characteristic (ROC) curves with corresponding areas under the curves were performed. In all analysis a p-value of ≤0.05 was considered statistically significant.
All calculations were performed using the SPSS statistics software version 24 (IBM Corp., Armonk, NY, USA).
Results
Patients
Patient characteristics.
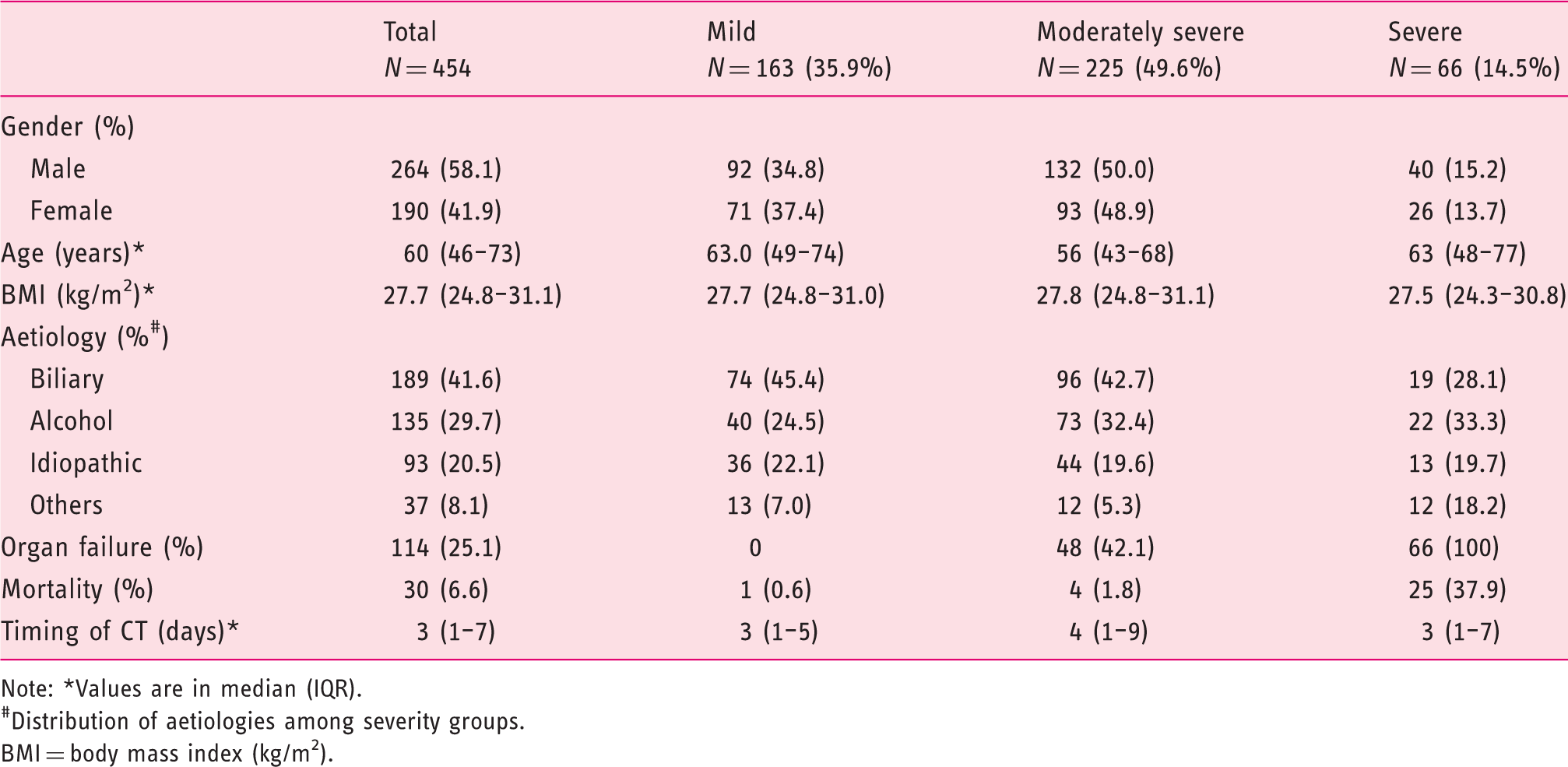
Note: *Values are in median (IQR).
#Distribution of aetiologies among severity groups.
BMI = body mass index (kg/m2).
Muscle and adipose parameters
Among the 454 CTs analysed, 183 were performed in the native-, 243 in the venous- and 28 in the arterial phase. No significant difference was found between the phases either for the two adipose tissue parameters (VAT and SAT) or two of the three muscle tissue parameters (MM and VMR). As described in the Methods section, the analysis of MMA solely included native and venous CTs. Median time from admission to CT was 3 days (IQR 1–7) with no significant difference between severity groups.
Association between body composition parameters and severity of acute pancreatitis.
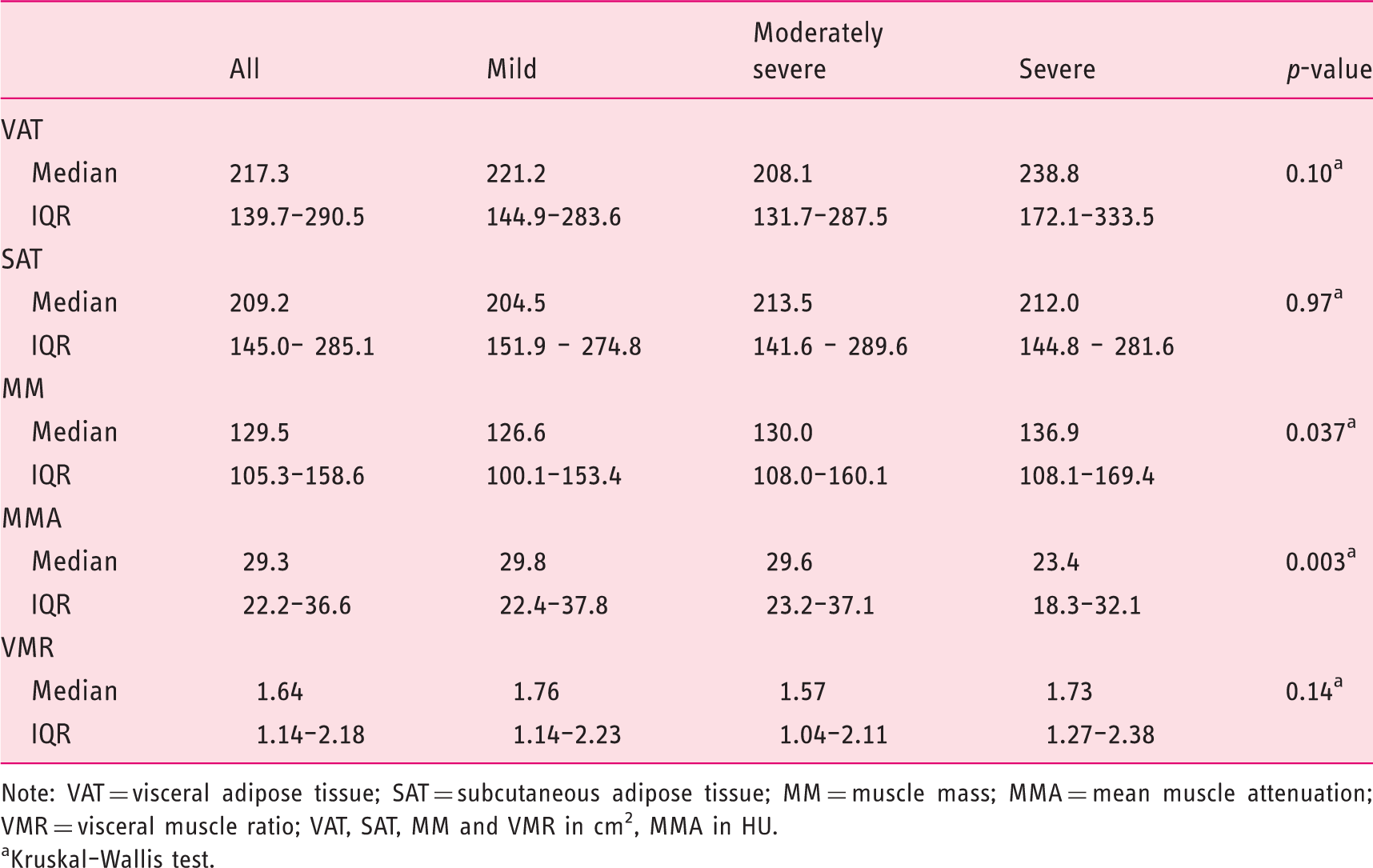
Note: VAT = visceral adipose tissue; SAT = subcutaneous adipose tissue; MM = muscle mass; MMA = mean muscle attenuation; VMR = visceral muscle ratio; VAT, SAT, MM and VMR in cm2, MMA in HU.
Kruskal–Wallis test.
Additional analysis was performed for the temporal impact of AP on the corporal parameters. For 41 consecutive CTs (first CT versus second CT) body parameter measurements were compared. Significant differences were found in the moderately severe group for VAT (VATearly mean = 240.1 cm2 vs. VATlate mean = 220.5 cm2; p-value 0.0083) and MM (MMearly mean = 131.2 cm2 vs. MMlate mean = 123.8 cm2; p-value 0.0003); see Supplementary Table 1.
Regression analysis
Univariate and multivariate regression analysis of risk factors for severe acute pancreatitis. Age was dichotomised at 73 years (highest tertile in the IQR).
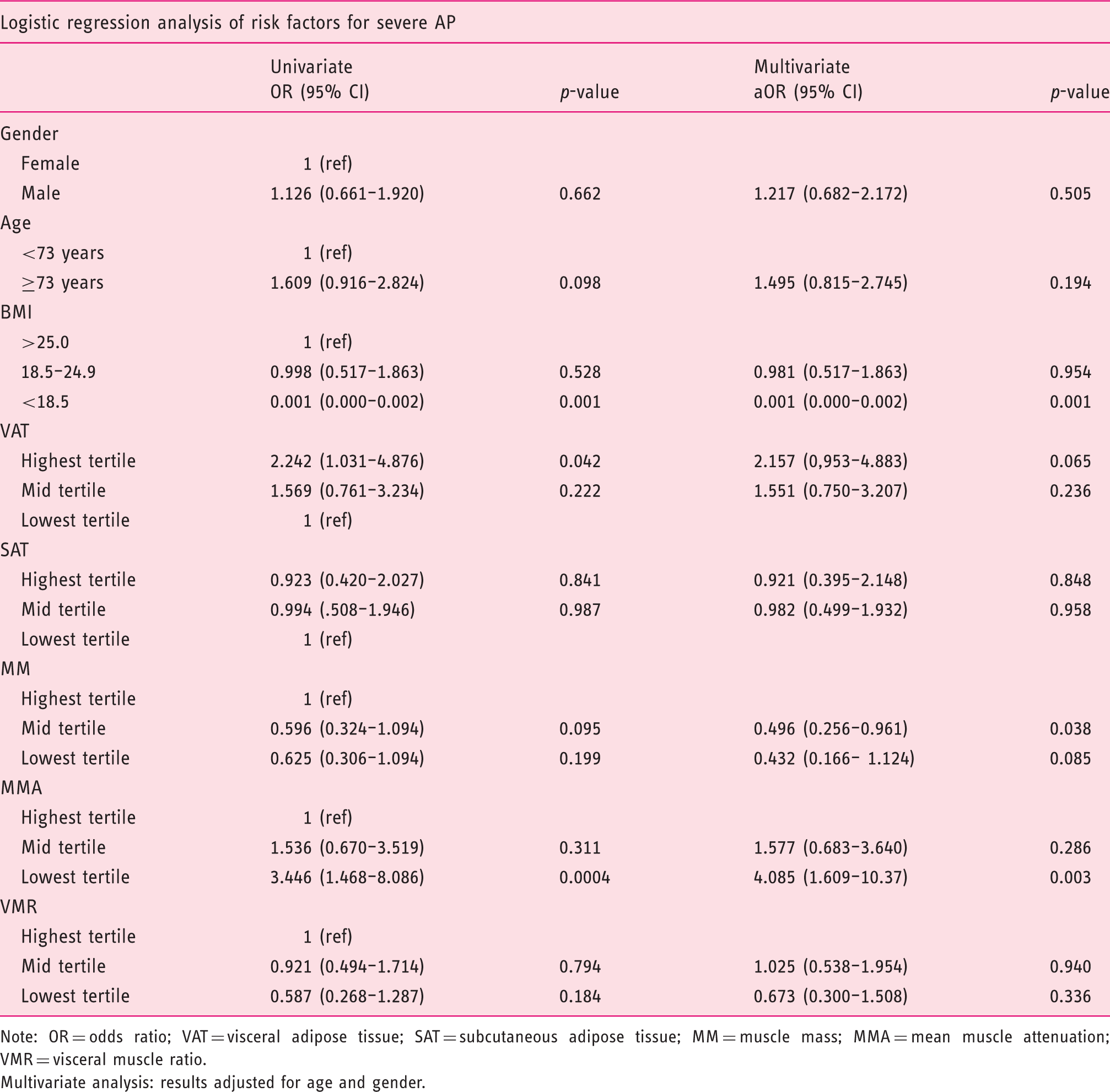
Note: OR = odds ratio; VAT = visceral adipose tissue; SAT = subcutaneous adipose tissue; MM = muscle mass; MMA = mean muscle attenuation; VMR = visceral muscle ratio.
Multivariate analysis: results adjusted for age and gender.
ROC curves
For each parameter a ROC curve was performed (see Supplementary Figure 2, A–E) to analyse their predictive capacity. In summary, all areas under the curves were small (range 0.424–0.571) and no cut-off value for severe AP could be retrieved.
Discussion
As obesity increasingly affects healthcare systems worldwide we aimed to investigate possible associations between severe AP and body composition parameters. Our results indicate that MM and muscle attenuation are important parameters for the outcome of AP. We were not able to demonstrate any association between either BMI level or corporal fat parameters and severe AP. Moreover, none of the investigated markers showed any prognostic capability in ROC-curve analysis.
Former studies have, using BMI as a surrogate marker, presented evidence that obesity has a negative impact on the disease course in AP.3–7 More recent works have investigated other parameters such as VAT and SAT.10–12 However, results have been contradictory and a correlation between these markers with AP severity is still unclear.5,18–20,25 Regarding BMI, our data was in line with that of several other studies, demonstrating that BMI in itself does not influence the grade of severity in AP.3,9,26 In our cohort the very small subgroup (n = 3) of underweight patients (BMI <18 kg/m2) demonstrated a significant association with decreased risk of severe AP (OR 0.001, 95% CI 0–0.002, p-value 0.001). These results, however, lack reliability due to the small sample size. The influence of a very low BMI on the outcome of AP patients needs further investigation.
The impact and relevance of intra-corporal adipose tissue distribution on AP severity has been debated. 3 When reviewing previous investigations of VAT and SAT, considerable differences were found compared with our data. Herein, the median value of VAT was 217.3 cm2 and 209.2 cm2 for SAT. Several studies that demonstrated a correlation between adipose tissue parameters and severe AP also reported lower levels of VAT and SAT in the groups with mild (median levels 80–105 cm2) and moderately severe AP (median levels 89–150 cm2).10–12 This evident variation compared with our data is interesting, though difficult to explain. Referred works investigated different populations (one American and two Asian) with diverse aetiologies, used various software programs in analyses and, finally, not all followed the same classification of AP. Our results were consistent throughout the European countries and also comparable with a recent publication from the Netherlands investigating the association between CT-assessed body composition with mortality in patients with necrotising pancreatitis, as well as an Australian study on intensive care patients.16,17 As our methodology is similar to the one in the Dutch study and the median values of VAT and SAT vary considerably between the American and Asian studies, we speculate that the observed variations are based on geographical or ethnical differences. Regarding adipose tissue parameters, several works report similar conclusions to ours5,16–18; however, these studies differ in both investigated outcomes and set-up, making direct comparison with our findings difficult.
As we observed a correlation of VAT with severity in univariate- (OR 2.24; 95% CI 1.03–4.88; p-value 0.04) but not in multivariate analysis (p-value 0.07) we wondered whether AP itself might influence VAT measurement. The results presented in Supplementary Table 1 and Supplementary Figure 3 demonstrate a significant difference in VAT and MM for the moderately severe group. The decrease in VAT over time in both the moderately severe and severe groups might mirror a general disease-induced reduction in adipose tissue as there is a similar tendency in the SAT group. A direct comparison of the first and second CTs revealed that the measurement of VAT was influenced by parameters like bowel distension, the volume of the thoracic cavity and by fluid collections due to AP. Furthermore, the MM demonstrated a general decline, which might be due to patients being ill and hospitalised and thus in a state of muscle reduction and general oedema. The previously mentioned work from the Netherlands found a correlation with MM depletion during the disease course, which we were able to partially replicate in our consecutive CTs. 16 Nonetheless, it is difficult to make general conclusions in such cases based on retrospective data, and prospective trials with predefined protocols are needed.
The influence of muscle parameter levels has not been extensively investigated in AP. Our analyses depicted a higher MM in severe AP (136.9 cm2) compared with moderately severe (130 cm2) or mild (126.6 cm2) cases (p-value 0.037). These numbers surprised us as low MM generally correlated with worse outcome in diseases. A hypothesis is that the severely ill patients already at a very early stage of the disease demonstrated more fluid sequestration and that oedema consequently influenced the measuring, giving falsely high values. Similarly, in multivariate analysis, lower levels of MM were unexpectedly associated with less severe AP, however, this was significant for the mid-tertile only (p-value 0.038). The lowest tertile for MM presented a p-value of 0.085, which might be due to a lower number of patients creating a type 2 error in this subgroup. The highest tertile of MM was set as reference as we assumed this to be favourable with regard to severity outcome. With our results demonstrating the opposite, despite being adjusted for age and gender, we again think oedema might be an influencing factor. This theory can neither be confirmed in this study, nor have we found any other to compare it with. In the work by Brewster et al., only patients with severe AP were investigated and their level of MM was 167.8 (144.2–203.7) cm2, which is in line with our figures. 17 However, patients in the Australian study were younger and no data was provided regarding on what day during the course of the disease the investigated CT was performed.
In our work, a low level of MMA, indicating myosteatosis, demonstrated a strong association with severity (aOR 4.1, 95% CI 1.6–10.4, p-value 0.003). In addition, its measurement was not influenced by the disease course (Supplementary Table 1; p-value 0.215). This observation is, again, partly supported by the study from van Grinsven et al. who analysed the correlation of MMA with mortality in AP patients and found a distinct association in the univariate analysis (OR 2.37, 95% CI 1.43–3.92, p-value 0.001) that, however, was not significant in the multivariate analysis (OR 1.13, 95% CI 0.62–2.08, p-value 0.69). 16 MMA characterises the intramuscular, and not the intermuscular, fat content. Low muscle attenuation has previously been related to poor prognosis and increased risk of complications and morbidity in cancer patients. 19 It has been proposed that fat infiltration underlies reduced muscle attenuation, whereas the composition of the intramuscular accumulated fat is largely unknown. Although myosteatosis has been associated with different pathological conditions, the underlying mechanisms are poorly elucidated making it impossible to explain its impact on severity in AP.
Our study has several limitations. The retrospective nature of the study implicated that there was no exact predefined study protocol for the CECTs, although all centres used equal standard protocols when investigating the pancreas. Moreover, only patients who had a CECT performed were included, potentially introducing selection bias, as patients with a mild disease course but no CECT performed were not enrolled. According to AP guidelines, patients with mild disease do not require a CECT unless another diagnosis needs to be excluded. 27 As a result, the group of patients with mild AP is likely to be smaller in radiology studies, as well as those with moderately severe diseases are more numerous compared with studies with consecutive inclusion of all AP patients. 28 We also acknowledge that the impact of oedema on our results, as well as those of all previous studies on body parameters in AP, remains largely unknown. However, herein we collated a large, well-characterised AP cohort from centres all over Europe, affording our study a robust research methodology. The pan-European multicentre setting additionally provides less risk of disproportion regarding basal patient characteristics and cultural variations.
In conclusion, our results cannot confirm prior findings that obesity per se or distinct visceral fat parameters are associated with severity in AP. The data presented indicates that patients with low MM have a better outcome, while a low MMA correlates with severe disease. Both observations are however in need of further investigation to understand the underlying mechanisms. As our results, as well as those from previous studies, to a large extent stem from retrospective analyses, a similar but prospective study including patients from different geographical regions would seem pertinent to address the questions that remain unanswered. Lastly, it is our opinion that understanding the function and influence of adipose tissue within the abdominal cavity is one of several key requirements for a profound knowledge of the pathophysiological processes in severe AP.
Supplemental Material
UEG882520 Supplemental Material - Supplemental material for Mean muscle attenuation correlates with severe acute pancreatitis unlike visceral adipose tissue and subcutaneous adipose tissue
Supplemental material, UEG882520 Supplemental Material for Mean muscle attenuation correlates with severe acute pancreatitis unlike visceral adipose tissue and subcutaneous adipose tissue by Hanna Sternby, Mariella Mahle, Nicolas Linder, Laureen Erichson-Kirst, Robert C Verdonk, Alexandra Dimova, Povilas Ignatavicius, Lucas Ilzarbe, Peeter Koiva, Anne Penttilä, Sara Regnér, Thomas L Bollen, Richard Brill, Franz Stangl, Walter A Wohlgemuth, Vijay Singh, Harald Busse, Patrick Michl, Sebastian Beer and Jonas Rosendahl in United European Gastroenterology Journal
Footnotes
Acknowledgements
Author contribution
The first two authors contributed equally to this work. The last two authors jointly supervised the work. HS, MM, VS, SB and JR conceived, designed and directed the study. MM, LEK, RB, FS and NL performed the analyses of the CT scans. HS performed the statistical analysis. All other co-authors recruited study subjects and collected clinical data. HS and JR drafted the manuscript with substantial help from VS and RCV. All authors contributed to the intellectual content with critical revisions and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval
Ethical approval was obtained at the Martin-Luther University of Halle-Wittenberg (date 21.07.2018, Bearbeitungsnummer 2017-96, Ethik- Kommission Medizinsche Fakultät Martin-Luther Universität Halle- Wittenberg).
Funding
National Institutes of Health (NIH) from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (grant no. RO1DK092460) and from the Department of Army to VS (grant no. PR110417).
Informed consent
Due to the retrospective nature of the study and anonymisation of all data, no informed consent was requested.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
