Abstract
Autoimmune hepatitis is a rare and chronic liver disease that is characterised by increased serum transaminases and immunoglobulin G, inflammatory liver histology and presence of circulating autoantibodies. An autoimmune hepatitis diagnosis justifies life-long treatment in most patients in order to prevent development of cirrhosis and end-stage liver disease. The cornerstone of treatment is steroid induction therapy followed by maintenance therapy with azathioprine, which is effective in most cases. For patients who do not respond to standard treatment, second-line treatment with other immunosuppressants can be effective. Treatment should be aimed at biochemical remission of the disease, which is defined as normalization of transaminases and immunoglobulin G. Patients should be monitored intensively during the first months of treatment in order to monitor side-effects, assess symptoms and individualise treatment. Specialist consultation should be sought in difficult-to-treat patients. Future studies and networking initiatives should result in optimization of current treatment strategies in autoimmune hepatitis.
Keywords
Introduction
Simplified diagnostic criteria for the diagnosis of autoimmune hepatitis (AIH).
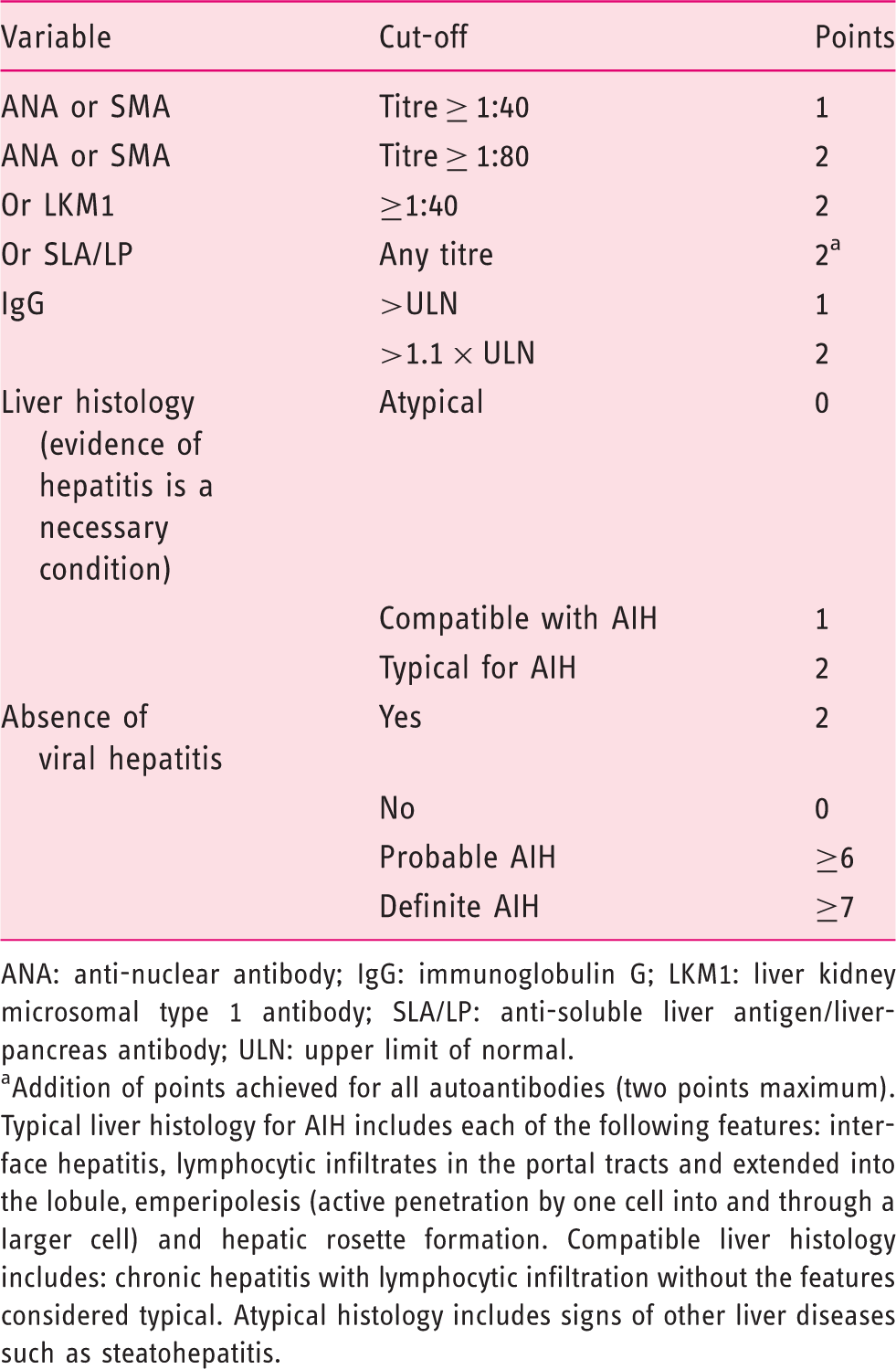
ANA: anti-nuclear antibody; IgG: immunoglobulin G; LKM1: liver kidney microsomal type 1 antibody; SLA/LP: anti-soluble liver antigen/liver-pancreas antibody; ULN: upper limit of normal.
Addition of points achieved for all autoantibodies (two points maximum). Typical liver histology for AIH includes each of the following features: interface hepatitis, lymphocytic infiltrates in the portal tracts and extended into the lobule, emperipolesis (active penetration by one cell into and through a larger cell) and hepatic rosette formation. Compatible liver histology includes: chronic hepatitis with lymphocytic infiltration without the features considered typical. Atypical histology includes signs of other liver diseases such as steatohepatitis.
Even though establishing the diagnosis of AIH is sometimes complex and cumbersome, clinical management of AIH can also be a challenging journey, given the lifelong therapy and potential side-effects. In this review article, we will discuss the clinical management of adult AIH patients and its latest developments, based on recent literature. Our aim is to assist the general gastroenterologist and hepatologist in the management of AIH, once an AIH diagnosis has been confirmed.
Literature search
We performed a PubMed search with the MeSH term ‘autoimmune hepatitis’ and ‘autoimmune hepatitis’ in the title field. All searches were limited to the English language and publication date within the last five years at time of search (May 2019). For the purpose of this review, we primarily selected articles that focus on clinical management of AIH. For a comprehensive review on mechanisms and diagnosis of AIH we refer to another article. 1 We identified a total of 114 articles that met our inclusion criteria.
Why should we treat an AIH patient?
Typically, the first question of AIH patients after hearing their diagnosis is: ‘Do I need treatment?’. Untreated AIH leads to progression of fibrosis to cirrhosis and, eventually, end-stage liver disease. Older studies showed that immunosuppressive treatment with steroids in AIH patients not only improved liver function tests, but also improved symptoms and prolonged survival.4–6 More recent studies have shown that treatment also leads to regression of liver fibrosis, even at the cirrhotic stage of disease. 7 This data indicates that treatment is warranted in patients with AIH.
Treatment for everyone?
It is unknown whether patients with mild disease (ALT < 3 times upper limit of normal, histological activity index (HAI) < 3 and no advanced fibrosis) will benefit from treatment, since most studies only included patients with moderate to severe disease activity. A decision not to treat mild AIH can be deemed as a possible option, especially in patients of older age or with severe comorbidities. However, AIH has a fluctuating disease course and patients who present asymptomatically may develop symptoms or elevation of transaminases that warrant treatment. 8 Therefore, we recommend treatment in every AIH patient, unless there are compelling reasons not to treat. Without treatment, close monitoring of transaminases and immunoglobulin G (IgG) should occur every 3–6 months in order to detect a possible flare of the disease and non-invasive measures of liver fibrosis such as transient elastography (TE) can be used to monitor for disease progression.
Is AIH treatment lifelong?
Immunosuppressive therapy should be continued for at least two years following complete normalization of transaminases and IgG. One study found that relapse of the disease occurs in up to 90% of patients once treatment is stopped. 9 In patients eligible for a trial of drug withdrawal, liver enzymes should be monitored closely. Using this approach, one study reported a long-term remission rate of 54% after drug withdrawal. 10 Preferably, a liver biopsy should be performed prior to stopping of therapy. If there is histological disease activity (HAI > 3) present, immunosuppressive treatment should not be stopped. The HAI is a histological scoring tool that rates the hepatitis components of periportal necrosis, intralobular degeneration and portal inflammation on a scale from 1–18. A HAI score from 1–3 indicates minimal hepatitis (Supplementary Material Table 1). 11
How should we treat an AIH patient?
Steroid induction therapy
Steroid therapy is the mainstay for inducing remission in AIH: studies with azathioprine induction therapy alone showed low remission rates and high mortality. 12 Steroid treatment, predniso(lo)ne in most cases, can be initiated as monotherapy or in combination with azathioprine. 13 Most guidelines advise an initial predniso(lo)ne dose between 0.50–1.00 mg/kg per day, although some centres start with a high initial dose of 1.00 mg/kg with rapid tapering within the following months.14,15 A recent retrospective study showed that patients who were treated with a predniso(lo)ne dose below 0.50 mg/kg/day, achieved similar remission rates when compared to patients who were treated with higher dosages. 16 Given the considerable side-effects of steroid therapy, physicians should prescribe a predniso(lo)ne dose that provides ample suppression of inflammatory activity and that is acceptable to the individual patient in terms of tolerability. We prefer tapering of steroids to be response-guided and tailored to the individual patient, in contrast to a fixed steroid-dosing schedule, although both methods have never been compared in AIH.
Budesonide at a dose of 9 mg/day provides an alternative induction agent in AIH and is associated with less steroid-related side-effects, but is contraindicated in patients with cirrhosis due to increased systemic side-effects as a result of portosystemic shunting. 17
Maintenance therapy
Azathioprine is the first drug of choice for maintenance therapy in AIH.
18
Azathioprine is ideally introduced 2–4 weeks after initiation of steroid treatment, in order to anticipate possible hepatotoxicity. To minimise side-effects, azathioprine is started at a dose of 50 mg/day, which can be increased to 1–2 mg/kg/day, depending on individual treatment response (Table 2). Both a combination of predniso(lo)ne and azathioprine and azathioprine alone are effective in maintaining remission,
6
although tapering steroids should occur as soon as maintenance therapy is initiated. With this regimen, 75–80% of patients will achieve normalization of transaminases.
19
Steroid-free therapy should be a treatment goal in every AIH patient to prevent steroid-related complications and should be aimed for within the first year of treatment. There is not sufficient data to recommend an alternative to azathioprine as first-line therapy. Mycophenolate mofetil (MMF) has proven to be effective and safe as first-line treatment in uncontrolled studies, with remission rates up to 88% and dosages from 1000–2000 mg/day.20,21 A simple algorithm for AIH treatment is presented in Figure 1.
Treatment algorithm for an adult patient with a first presentation of autoimmune hepatitis (AIH). The mainstay of treatment is steroid induction therapy followed by maintenance therapy with azathioprine (AZA). AIH treatment should always be individualised. 6-MP: 6-mercaptopurine; MMF: mycophenolate mofetil; PBC: primary biliary cholangitis; PSC: primary sclerosing cholangitis. Key recommendations for treatment of an adult autoimmune hepatitis (AIH) patient. 6-MP: 6-mercaptopurine; IgG: immunoglobulin G; MMF: mycophenolate mofetil.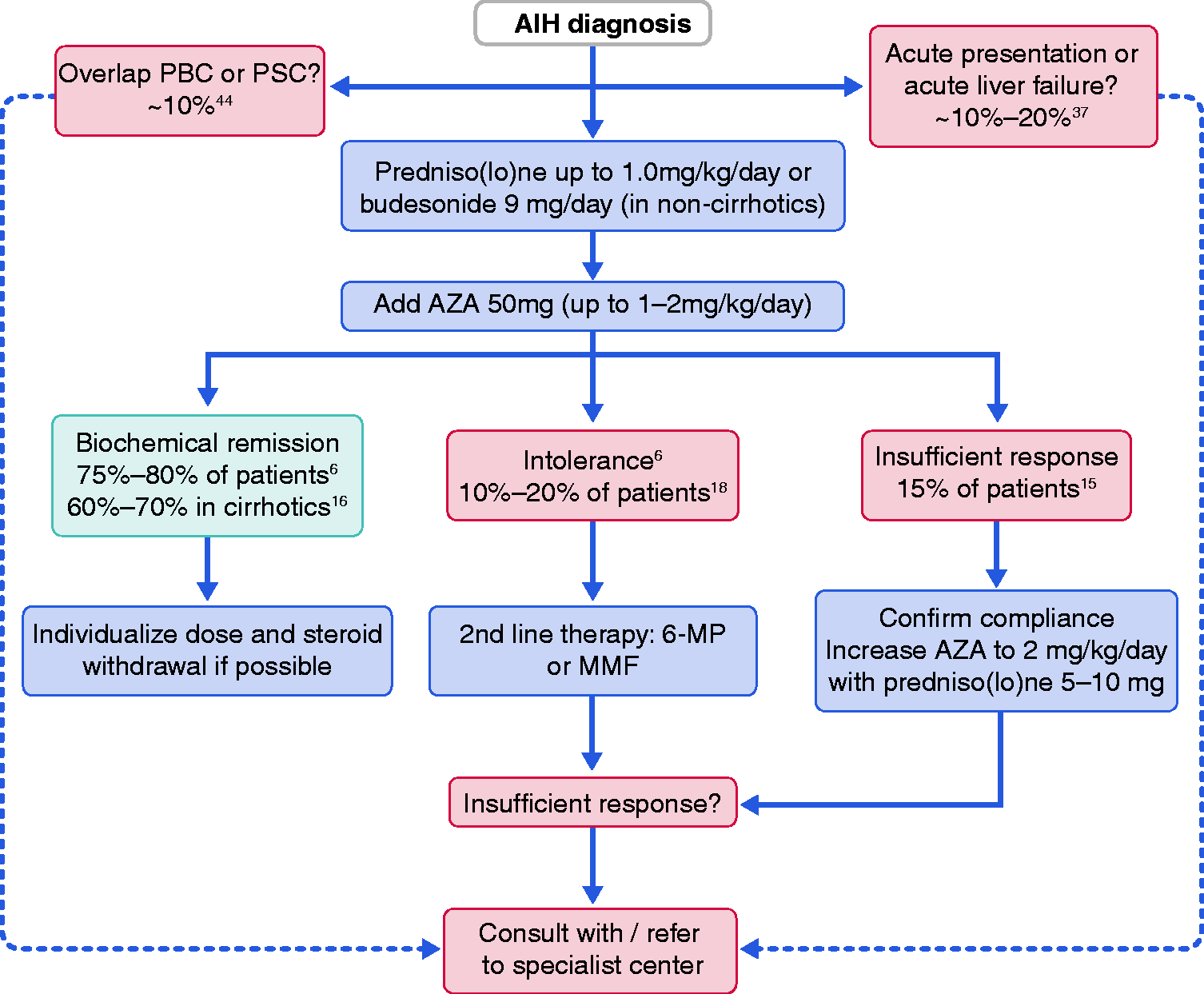

What is a satisfactory response during treatment?
Remission of disease
Histological and biochemical remission of AIH should be the fundamental treatment goal in every patient. A HAI score of <4/18 is used to define histological remission. Since frequent biopsies are an unattractive option, the surrogate endpoint of biochemical remission is used, which is defined as repeatedly normal serum transaminases and normal serum IgG. This endpoint is incorporated in most international guidelines.13,22 A recent study demonstrated that biochemical remission is associated with regression of fibrosis and low histological disease activity. 7 A different study showed that histological hepatitis activity still exists in approximately 50% of patients even though they had achieved biochemical remission. 23 Additionally, AIH is a disease with an unpredictable course and frequent relapses. Therefore, AIH patients should be monitored for transaminases and IgG at six-month intervals at least even if they are in prolonged biochemical remission. Use of TE may be helpful for the detection of fibrosis progression during the disease course. 24
What problems do we encounter during AIH treatment?
Side-effects
Management of medication and side-effects.
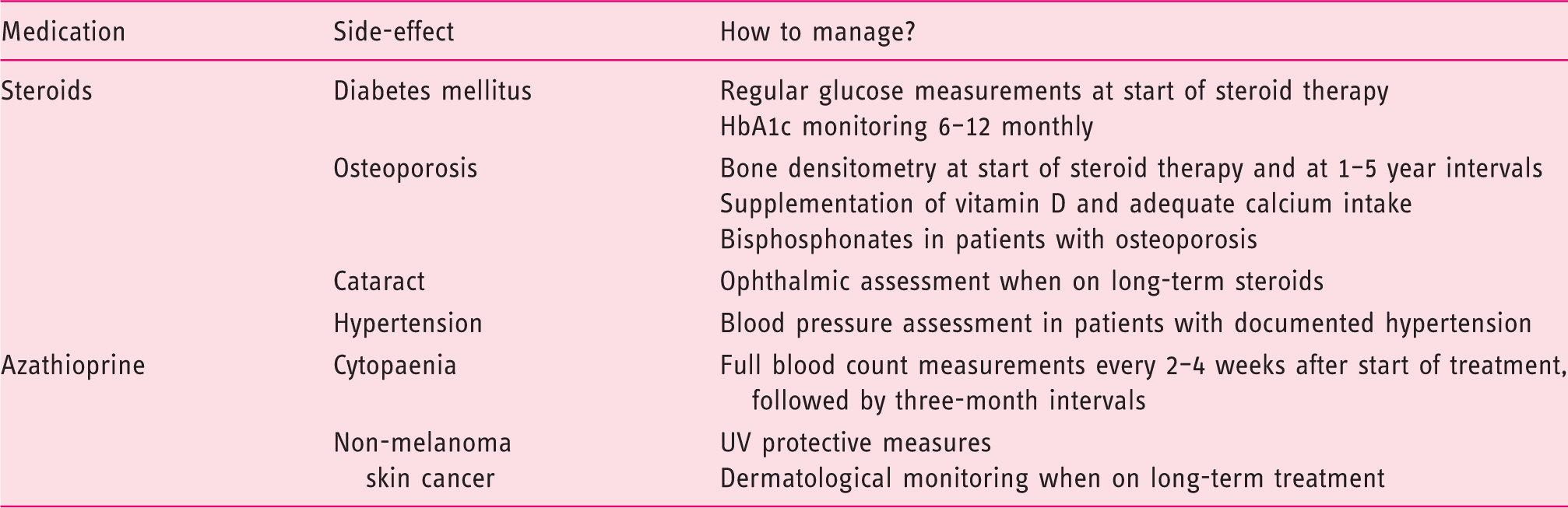
The exact number of AIH patients that experience azathioprine intolerance is unknown in AIH, but old series report rates of 10–20%, although this may be higher in real-world practice.13,25 Most side-effects are limited to nausea and gastrointestinal discomfort but in some cases may result in rash, arthralgia or pancreatitis which warrants discontinuation. 15 Split-dose administration of azathioprine is an easy option to diminish mild side-effects. 26 Full blood counts should be monitored to detect possible bone marrow toxicity. Some patients might benefit from dose reduction, although close monitoring of transaminases and IgG is recommended to avert a possible flare of the disease. In case of intolerable side-effects, alternative immunosuppression in recommended.
Insufficient response to treatment
Approximately 15% of patients may experience an insufficient response to therapy by demonstrating failure to normalise biochemical or histological parameters. A first step should always be to reconfirm the AIH diagnosis, preferably with help of an experienced liver pathologist. Second, it is important to ensure treatment adherence, given that adherence rates can drop to 65% after six months of chronic drug therapy in other chronic diseases. 27 Measuring 6-thioguanine (6-TG) concentrations during treatment may help to identify those patients who are non-adherent. Little is known about optimal 6-TG concentrations, although one study showed that 6-TG concentrations above 220 pmol/8 × 108 RBC are associated with remission. 28 Patients with an insufficient response to standard therapy might benefit from increased dosages of azathioprine up to 2 mg/kg/day, together with 5–10 mg predniso(lo)ne.
Alternative treatment options
There is no consensus on the best second-line treatment options in AIH. Intolerance and insufficient response are two different scenarios. In general, intolerance to treatment is manageable with 6-mercaptopurine (6-MP) or MMF, while an insufficient response to first-line treatment is more difficult to deal with.
6-MP is widely used as an alternative to azathioprine in inflammatory bowel disease (IBD) and has better tolerability. 29 Although it is used in clinical management of AIH, data on 6-MP efficacy is limited to two case series with a dosage ranging from 25–75 mg/day.30,31 It shows a favourable response especially in azathioprine-intolerant patients, and acceptable tolerance rates. 6-TG can also be used as an alternative to either azathioprine or 6-MP but has been associated with development of nodular regenerative hyperplasia in patients with IBD. 32 A recent study in AIH patients showed that 6-TG treatment with a dose of 20 mg/day is well-tolerated and leads to complete biochemical remission in patients with prior insufficient response to thiopurines. 33
The most studied second-line drug in AIH is MMF at a dose of 1000–2000 mg/day. Reduction of serum transaminases occurs in 33–100% of patients and histological remission occurs in 73% of patients. 34 A recent study showed that patients on MMF with an initial non-response to first-line treatment have lower remission rates (34–57%) than patients with azathioprine intolerance (62–91%). 35 MMF has a favourable safety profile but has teratogenic effects, which makes the drug less useful in women in childbearing age. We recommend that treatment with MMF should be provided by physicians who have experience with the drug.
The calcineurin inhibitors tacrolimus and cyclosporine are options for second-line therapy in AIH. Reports on these drugs demonstrate response rates ranging from 27–94%. 36 Calcineurin inhibitors can have considerable side effects with nephrotoxicity and hypertension being the most prominent. Prescription should therefore only be carried out by physicians with ample experience concerning these drugs.
How to manage difficult-to-treat AIH patients?
Acute presentation
There are no validated definitions for AIH with acute presentation. 37 Patients who present with acute, icteric AIH and concomitant coagulopathy (INR≥1.5) respond to oral or intravenous steroids (1 mg/kg) in the majority of cases, although in a number of patients transplantation might be the only treatment option.38–40
However, steroid use might not be beneficial in the setting of acute liver failure with hepatic encephalopathy, one retrospective study found that corticosteroid use in AIH with acute liver failure was associated with increased mortality in patients with the highest MELD score. 41 Timely consultation with a transplant centre is strongly recommended to arrange rapid referral when necessary.
Patients with cirrhosis
Up to 30% of patients have confirmed cirrhosis at diagnosis, which is a sign of subclinical disease course months or even years prior to diagnosis and is associated with poorer outcomes.8,19,42 Patients who present with decompensated cirrhosis should be treated in close collaboration with a transplant centre. It is advised to offer ultrasound surveillance to cirrhotic patients. 43
Variant syndromes with primary biliary cholangitis (PBC) and primary sclerosing cholangitis (PSC)
AIH patients might present with additional features of PBC or PSC, or develop these features at a certain time during their disease course. Standardised definitions of these variant syndromes are currently lacking and, given their rarity, evidence-based recommendations for treatment are lacking as well. In general, it is important to treat patients with autoimmune liver disease according to their predominant phenotype. 44 Consultation with an expert centre is advised in order to prevent overtreatment.
Mood disorders
Improvement in mood disorders should be an important treatment goal in AIH. Depressive symptoms and anxiety are often present in AIH patients and results in lower health-related quality of life (HRQoL), even when in biochemical remission. 45 Since occurrence of depressive and anxiety symptoms in AIH may be associated with non-adherence to treatment, 46 physicians should actively ask patients about possible symptoms of depression and anxiety and refer them to mental healthcare providers if necessary. Although improvement of HRQoL should be a desirable treatment goal in AIH, validated questionnaires that assess the wide range of problems in AIH are currently lacking.
When should we refer an AIH patient to a specialist centre?
Scenarios in which consultation with a specialist centre is recommended.

MMF: mycophenolate mofetil; PBC: primary biliary cholangitis; PSC: primary sclerosing cholangitis.
Consultation with transplant centre is strongly recommended.
The European Reference Network for Hepatological Diseases (ERN RARE-LIVER) was launched in March 2017 and is a European Union credited joint venture between expert centres across Europe, linking professional societies and patients' groups with the main objective to improve the care of patients with rare liver diseases. 47 Aside from conducting prospective high-quality registries, ERN RARE-LIVER offers expert advice on difficult AIH cases using an online clinical patient management system established by EU. These developments will undoubtedly lead to optimization of current treatment strategies and to reduce discrepancies in AIH care delivery.
Future prospects
Drugs under investigation
Ongoing trials with new drugs in autoimmune hepatitis (AIH).

Conclusion
AIH treatment involves steroid induction therapy followed by maintenance therapy with azathioprine with biochemical remission of disease as primary treatment goal, which is achievable for the majority of patients. Caution should be exercised in patients with an acute, fulminant presentation of the disease, in cirrhotics, pregnancy and in patients with prolonged insufficient response to standard therapy. Early access to specialist consultation should improve AIH healthcare delivery and outcome. Referral should take place for difficult cases. International networks such as the ERN RARE-LIVER will provide groundwork for registries and studies that will provide more insight into optimal management strategies in AIH.
Supplemental Material
Supplemental material for Clinical management of autoimmune hepatitis
Supplemental Material for Clinical management of autoimmune hepatitis by Simon Pape, Christoph Schramm and Tom JG Gevers in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
