Abstract
Background
Cortiment®MMX® (budesonide MMX®) is currently approved for the induction of remission in mild-to-moderate ulcerative colitis (UC) patients when 5-ASA treatment is not sufficient. Data in real-life settings are lacking.
Methods
This was a multicentre observational prospective cohort study conducted in Europe and Canada. Effectiveness, safety, and tolerability of Cortiment®MMX® in a real-life setting of patients treated for mild-to-moderate UC was investigated. Patients were prescribed Cortiment®MMX® in accordance with the Summary of the Product Characteristics (SmPC).
The primary endpoint was the clinical benefit of Cortiment® MMX® in routine practice (improvement ≥ 3 points in the clinical sub-scores of the Ulcerative Colitis Disease Activity Index, UCDAI).
Results
Data from 326 patients with mild-to-moderate UC were analysed for the primary endpoint. Clinical benefit was achieved in 60.1% (196/326) of patients at the end of Cortiment®MMX® treatment. Clinical remission (UCDAI clinical sub-score ≤ 1), full symptoms resolution (rectal bleeding (RB) = 0 and stool frequency (SF) = 0) and symptoms resolution (RB = 0 + SF ≤ 1) at the end of the Cortiment®MMX® treatment were achieved in 51.8%, 45.1% and 63.2% of patients, respectively. The median time to symptoms resolution was 30 days (range 29.0–36.0 days). Fifty patients (14.3%) had to discontinue Cortiment®MMX® due to adverse events; 17.5% of patients (n = 61) reported at least one adverse event related to the study drug.
Conclusions
This was the first time that a large cohort study was conducted with Cortiment®MMX® in a real-life setting. It demonstrated that Cortiment®MMX® is effective, safe and well tolerated in about 60% of UC patients.
Key summary
Established knowledge on this subject
Budesonide MMX® is approved for the treatment of active mild-to-moderate ulcerative colitis; Budesonide MMX® represents a valid and safer alternative to systemic steroids in patients with ulcerative colitis who failed 5-ASA treatment; There are no large-scale real-life data showing the best therapeutic approach in patients with indication to treatment with budesonide MMX® (monotherapy vs. combination with 5-ASA).
What are the significant and/or new findings of this study?
Our real-life data confirm the effectiveness and safety of budesonide MMX® in patients with mild-to-moderate active ulcerative colitis in terms of clinical benefits and quality of life. Our data suggest that adding budesonide MMX® to 5-ASA treatment might be the best therapeutic approach to induce clinical remission compared to budesonide MMX® monotherapy.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory bowel disease (IBD) that involves the colon and the rectum, with continuous inflammation, erosions and/or ulcers affecting the colonic mucosa.1,4 Usually, the course of the disease is characterised by flares of active disease with diarrhoea, rectal bleeding (RB) and rectal urgency, alternating with periods of remission. 2 The main therapeutic goals to treat active flares are the control of symptoms, normalisation of bowel movements, disappearance of bleeding and rectal urgency, combined with the normalisation of inflammatory biomarkers, such as C-reactive protein and/or faecal calprotectin (FC), and the disappearance of mucosal lesions.3,4
Systemic corticosteroids, such as prednisolone or equivalents, are effective therapies in active UC of any severity, but their use is limited by their adverse effects. 5 In order to minimise the side effects of systemic steroids, budesonide, a locally acting corticosteroid with low bioavailability and little systemic absorption, represents a valid alternative in IBD patients.6,7 However, the pH-dependent release mechanism of budesonide may limit effectiveness along the colonic mucosa. 8 In order to address this unmet need, a colonic release system (MMX®, Multi-Matrix System) that provides targeted drug delivery to the entire colon has been used successfully in conjunction with both mesalazine and budesonide to allow a continuous and uniform release of the active principles along the colon.9–13
In the phase 2 trial, the first data in patients with left-sided UC showed that 47.1% of patients treated with budesonide MMX® 9 mg once daily achieved clinical remission compared to 33.0% of placebo-treated patients. 14 More recently, the CORE-II trial, a phase 3, randomised, double-blind, double-dummy, placebo-controlled, parallel-group trial was conducted on 512 patients with active mild-to-moderate UC, mostly in European countries. 9 Patients were randomised 1:1:1:1 to receive budesonide MMX® 9 mg/day, budesonide MMX® 6 mg/day, controlled ileal-release budesonide 9 mg/day once daily), or a placebo for 8 weeks. 9 Combined clinical and endoscopic remission at week 8 was achieved in 17.4% of patients treated with budesonide MMX® 9 mg compared to 4.5% of patients in the placebo group (p = 0.0047). 9 As a secondary study objective, more patients achieved histological healing (16.5% vs. 6.7%, p = 0.036) and complete symptoms resolution (23.9% vs. 11.2%, p = 0.022) when treated with budesonide MMX® 9 mg compared with the placebo. 9 Similar results were found in the CORE-I trial conducted in the United States. 15 In the pooled analysis, budesonide MMX® safety and tolerability was comparable to the placebo in terms of adverse events (AEs) and mean cortisol serum levels. 16
Currently, budesonide MMX® 9 mg/day is approved for the induction of remission in patients with mild-to-moderate UC where 5-ASA treatment is not sufficient. The aim of this study was to evaluate the effectiveness and tolerability of budesonide MMX® (Cortiment®MMX®) in mild-to-moderate UC in a real-life setting.
Methods
This was a multicentre observational prospective cohort study conducted in IBD referral centres in Poland, Italy, Germany, the United Kingdom, the Netherlands, Sweden, Ireland and Canada. The main aim of this study was to collect and analyse data on the effectiveness, safety and tolerability of Cortiment®MMX® in a real-life setting of patients treated for mild-to-moderate UC.
Inclusion and exclusion criteria
Eligible patients were adult (aged ≥ 18 years), of either gender, who had been prescribed Cortiment®MMX® for the treatment of mild-to-moderate active UC within a 5-day window prior to being included in the study. Exclusion criteria were severe active/fulminant UC, use of concomitant antibiotics or systemic corticosteroids for the current flare, history of total/sub-total colectomy, known hypersensitivity to the active substance or to any of the excipients.
Study endpoints and outcome measures
Primary endpoint
The primary endpoint of this study was to assess the clinical benefit of Cortiment®MMX® in routine practice, defined as the percentage of patients achieving improvement ≥3 points in the clinical sub-scores of the UCDAI (Ulcerative Colitis Disease Activity Index) at the end of Cortiment®MMX® induction treatment. 17
Secondary endpoints
Secondary effectiveness endpoints assessed at the end of Cortiment®MMX® induction treatment were:
the percentage of patients with clinical remission defined as UCDAI clinical sub-score ≤ 1; the percentage of patients with symptoms resolution (RB = 0 + stool frequency (SF) ≤ 1, urgency = 0); the percentage of patients with full symptoms resolution (RB = 0 + SF = 0, urgency = 0); time to symptoms resolution (RB = 0, SF ≤ 1, urgency = 0); the change in health-related quality of life (Short Inflammatory Bowel Disease Questionnaire, SIBDQ)18,19; the change in health economic parameters (Work Productivity and Activity Impairment, WPAI)
20
; the percentage of patients with FC within normal range (when FC tests were performed); the percentage of patients with endoscopic healing (UCDAI endoscopic sub-score = 0); and the percentage of patient in remission (UCDAI endoscopic sub-score ≤ 1) (when endoscopies performed).
Additional study endpoints
As additional study endpoints, tolerability of Cortiment®MMX® in a real-life setting (AEs) and adverse drug reactions (ADRs) were assessed from 2 weeks up to 6 months after the last Cortiment®MMX® dose for prolonged tapering off. Moreover, to determine how Cortiment®MMX® is prescribed and used by gastroenterologists in routine clinical practice, the study evaluated the percentage of patients given Cortiment®MMX® as monotherapy or as an add-on therapy to 5-ASA and the relative time frames, rates of Cortiment®MMX® treatment discontinuation/end of treatment (rate, reason, time frame, tapering off), and follow-up UC treatments (5-ASA, corticosteroids, immunosuppressants or biologics) were evaluated.
Safety
For safety analyses, AEs and concomitant diseases were recorded, by assigned preferred terms (PTs) and categorisation into the Primary System Organ Class according to the MedDRA thesaurus version 20.1. Patients reporting more than one AE or concomitant disease with the same PTs were counted once. The AEs were reported as per national safety reporting requirements.
Study time points
Effectiveness was evaluated at the end of Cortiment®MMX® induction treatment. The tolerability and use of Cortiment®MMX® were assessed from 2 weeks up to a maximum of 6 months (in particular circumstances) after the last Cortiment®MMX® dose.
Study groups
The primary endpoint, clinical benefit, (improvement ≥ 3 points in the UCDAI clinical sub-scores at end of treatment) was compared between three cohorts of patients. The cohorts were defined as:
Cortiment®MMX® added to 5-ASA at least 14 days after increased/optimised 5-ASA dose for the treatment of flare (late add-on) (Cohort 1); Cortiment®MMX® added to 5-ASA within 14 days since 5-ASA increased/optimised for the treatment of flare or without 5-ASA dose modification (early add-on) (Cohort 2); and Cortiment®MMX® as monotherapy for the treatment of flare (mono) (Cohort 3).
Statistical analyses
Descriptive statistics were used to analyse the study data. Continuous variables were summarised using the number of patients, mean, standard deviation, minimum, first quartile, median, third quartile and maximum. For categorical variables, data were summarised by the number and percentage of patients in each category; 95% confidence intervals (CIs) were provided to evaluate the level of precision of the calculated estimates.
Incidence percentages were calculated as follows: number of patients with one or more occurrences of the item divided by the number of patients in the analysis set, times 100.
Missing data were not imputed. When summarising categorical variables, in case of any missing responses, these were shown as a separate category. For continuous variables, the number of non-missing observations was displayed.
All discontinuations after enrolment were summarised by time of, and reason for, discontinuation.
Tests were carried out with a two-sided significance level of 0.05. CIs were set at 95%.
The association between the explanatory clinical variables and the outcome (response) variable was tested in univariable analyses with the chi-square test. Then, all selected variables (using p < 0.20 as selection criterion) were included in the multivariable logistic regression analysis, and a backwards stepwise selection process was followed. 23 Odds ratios with 95% CIs derived from the final multivariable logistic regression model are presented.
No formal hypothesis was tested in this observational, prospective cohort study. All enrolled patients who received at least one dose of Cortiment®MMX® and met the inclusion or exclusion criteria were included in the analyses.
The estimated sample size was 350 patients. 23
In order to perform a comparison among the three cohorts, patients were matched to adjust for the effects of covariates. Matching was based on the propensity score method. The following covariates were considered for the calculation of the propensity score: gender (male, female), age (18–34; 35–59; 60 + years), number of UC flares in the last year, baseline extension of disease, baseline endoscopic sub-score, baseline score for stool frequency during the last 3 days, baseline score for RB during the last 3 days, baseline response for urgency episodes during the last week, baseline UC-related extra-intestinal symptoms during the last week, Physician's Global Assessment at baseline. The propensity score was calculated by means of a multivariable logistic regression analysis. The absolute difference among cohorts’ patients’ matched propensity scores was set to 0.01.
Ethical considerations
This study was conducted according to the 1975 Declaration of Helsinki and the European Medicines Agency Guidelines for Good Clinical Practice. All ethical review boards from each participating centre approved this study. This study was registered on clinicaltrials.gov (ClinicalTrials.gov Identifier: NCT02586259). Written, informed consent was obtained from each patient included in the study prior to the start of any study procedure.
Results
Study population
Baseline characteristics of the study population.
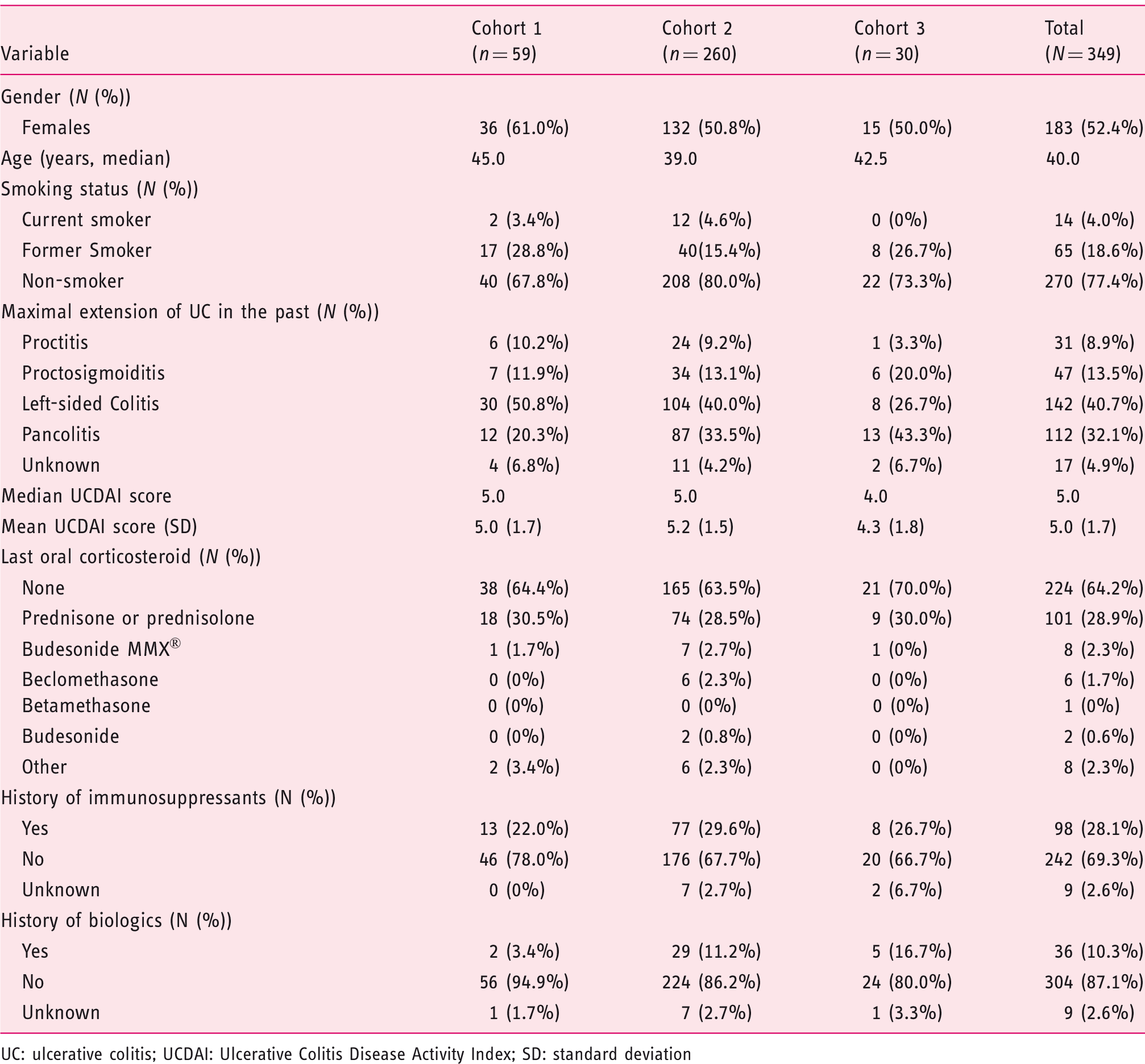
UC: ulcerative colitis; UCDAI: Ulcerative Colitis Disease Activity Index; SD: standard deviation
Effectiveness on symptoms
Clinical effectiveness endpoints at the end of treatment.
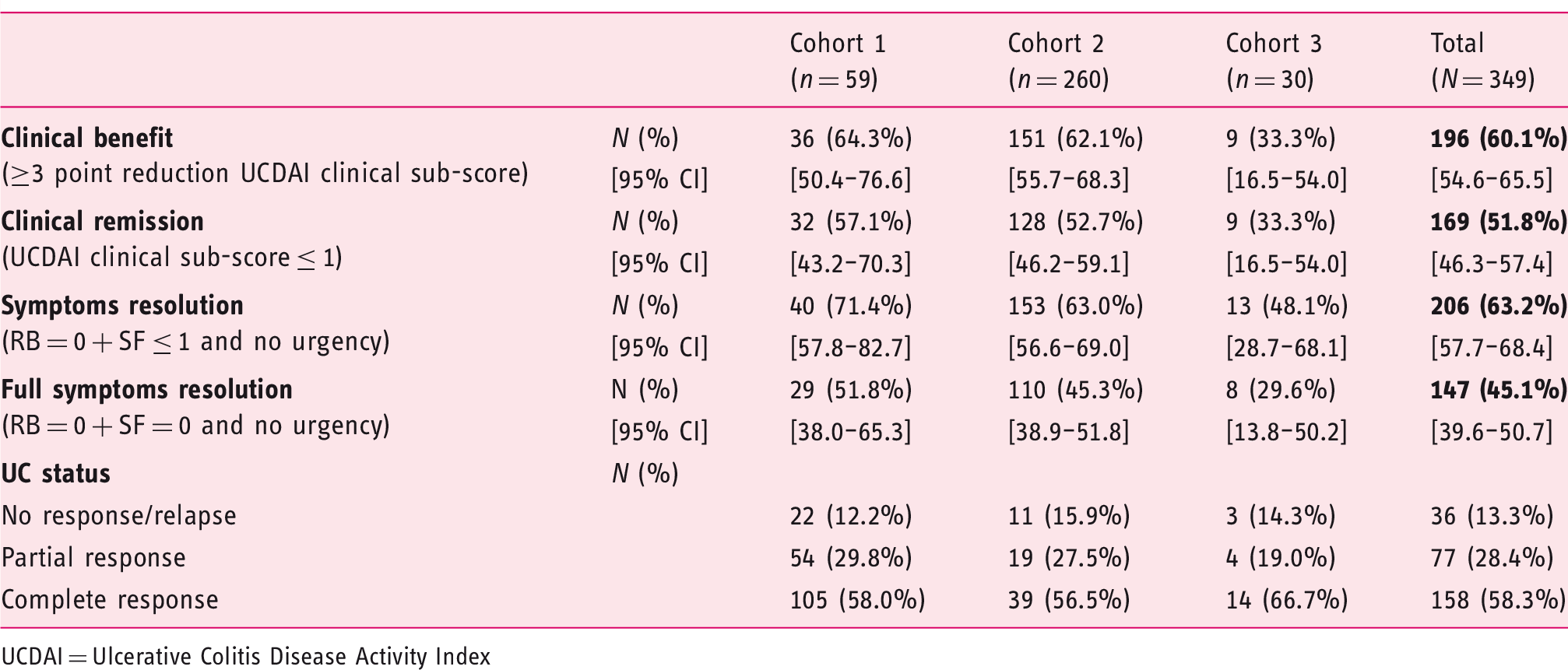
UCDAI = Ulcerative Colitis Disease Activity Index
Clinical remission, defined as UCDAI clinical sub-score ≤ 1, was achieved in 169 (51.8%) patients in the overall evaluable set (57.1%, 52.7% and 33.3% patients in cohorts 1, 2 and 3 respectively) (Table 2).
Full resolution of symptoms at the end of the Cortiment®MMX® treatment, defined as the absence of rectal bleeding (RB = 0), normalisation of bowel habit (SF = 0) and no urgency was achieved in 147 patients (45.1%) (51.8%, 45.3% and 29.6% in cohorts 1, 2 and 3 respectively). Symptoms resolution (RB = 0, SF ≤ 1 and no urgency) was achieved in 206 (63.2%) patients from the entire study population (71.4%, 63.0%, and 48.1% in the three cohorts, respectively). The median time to symptoms resolution was 30 days (range 29.0–36.0 days) (Table 2).
After propensity score matching, the overall difference was statistically significant for clinical benefit (p = 0.0101), clinical remission (p = 0.0216) and symptoms resolution (p = 0.0366), whereas it was not statistically significant for full symptoms resolution (p = 0.0752).
Effectiveness on objective signs of inflammation
Data about objective measures of inflammation, such as FC and endoscopy, at baseline and at the end of induction were available for 14 and 32 patients, respectively. Due to the small number of patients who underwent dosing of FC at both baseline and end of Cortiment®MMX® induction treatment, no clear tendency can be drawn. Mucosal healing (defined as a Mayo endoscopic sub-score equal to 0) was achieved in 5/32 patients (15.6%) at the end of treatment, whereas endoscopic remission (Mayo endoscopic sub-score ≤ 1) was achieved in 16/32 patients (50.0%).
Patient-reported outcomes
The median increase in the SIBDQ was 10.0 points for the whole cohort compared to baseline assessment (p < 0.001). Improvement in quality of life was observed in all three cohorts (median SIBDQ increase of 11.0, 14.0 and 7.0 respectively) with a significant improvement in quality of life (p < 0.001) only in cohorts 1 and 2 (Figure 1). Additionally, patient satisfaction of Cortiment®MMX® treatment was high (Visual Analogue Scale (VAS) scale: rating of 7- to 10/10) in 61.3% of patients, with similar rates in the three cohorts (71.3%, 60.4% and 60.4% respectively).
Median improvement of quality of life (SIBDQ scores) and decrease of work impairment scores (WPAI) compared to baseline among the study cohorts and in the overall populations.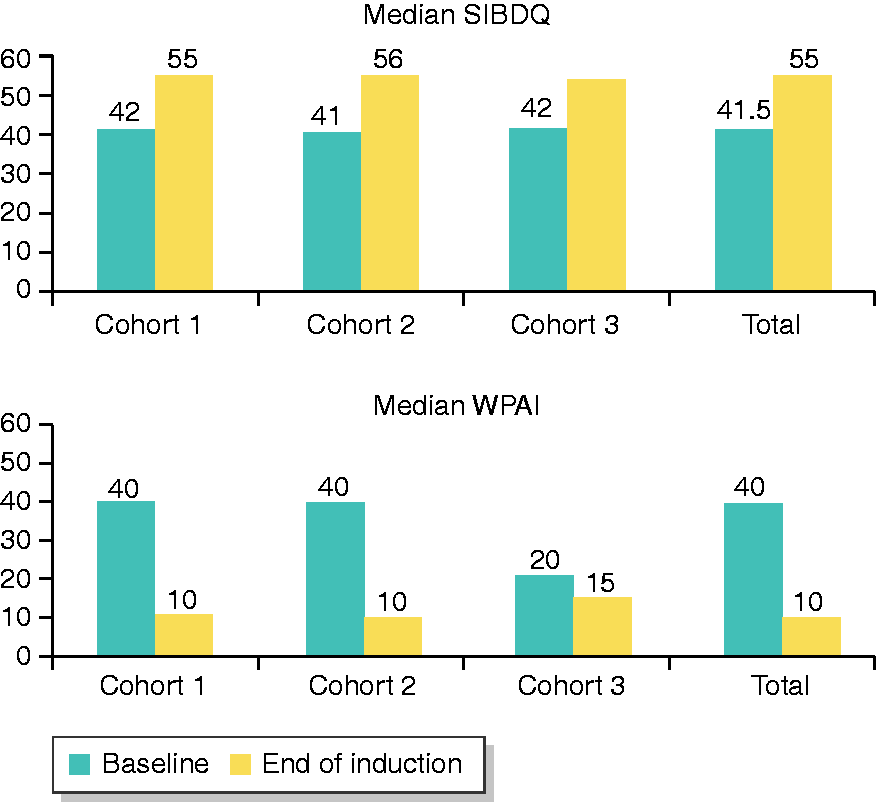
Regarding work productivity, the mean percent work time missed due to ill health, the mean percent impairment while working due to ill health and the mean percent overall work impairment due to ill health were all reduced in the whole population by 8.9%, 21.3% and 23.7% respectively (p < 0.0001 in both the paired t-test and the non-parametric signed rank sum test for all variables, Figure 1).
Predictors of clinical benefit
Final multivariable logistic regression model for predictors of clinical benefit in the study cohort.
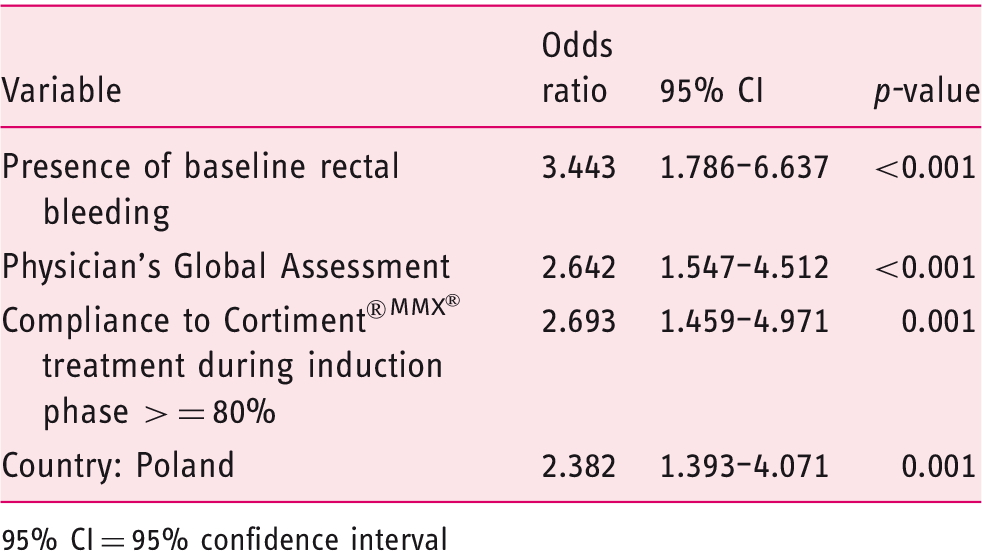
95% CI = 95% confidence interval
Safety
Patients with adverse events.
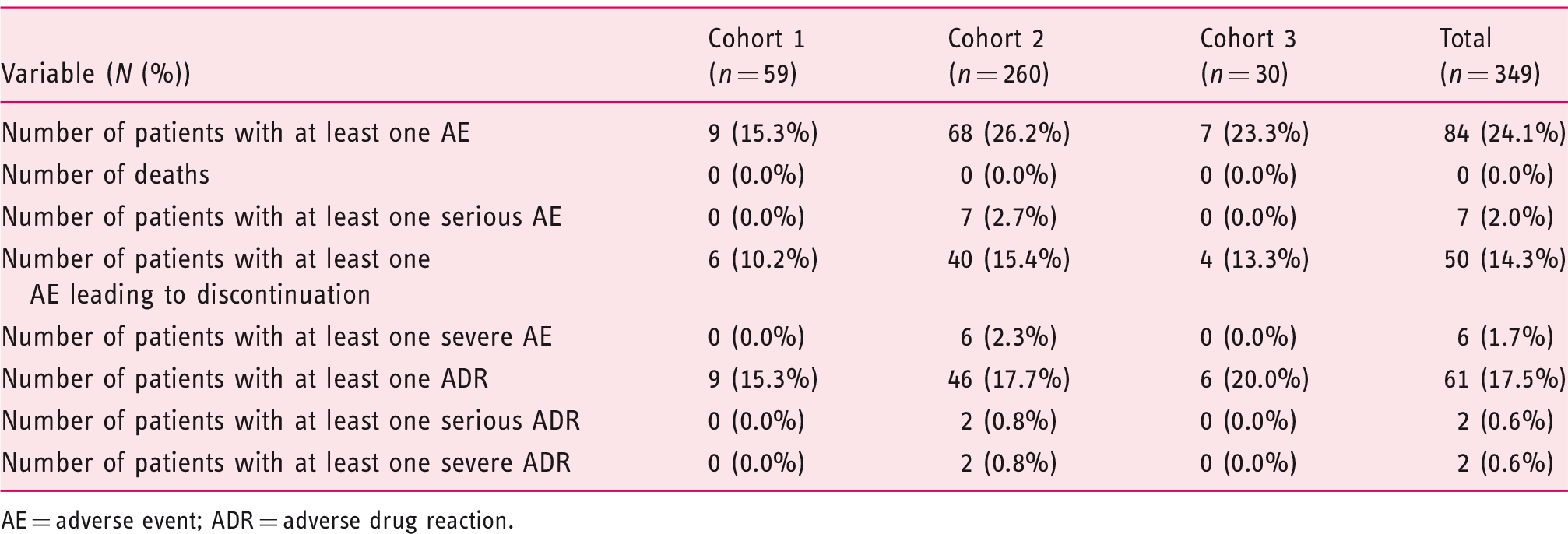
AE = adverse event; ADR = adverse drug reaction.
Prescribing patterns
The study showed that Cortiment®MMX® was mainly prescribed as an add-on therapy to 5-ASA (91.4% vs. 8.6% as monotherapy). In this patient population, 5-ASA dose was optimised before Cortiment®MMX® induction treatment in 41.0% of patients (143). The 5-ASA dose optimisation was performed within 14 days for 24.1% (84) of patients with a median time of 0.0 days (95% CI [29.0, 53.0]), and after a minimum of 14 days for 16.9% (59) of patients with a median time of 43 days (95% CI [0.0, 1.0]).
At the end of Cortiment®MMX® induction treatment, Cortiment®MMX® treatment was stopped for the majority of patients (83.8%, n = 274) and was modified for tapering in 16.2% (53) of patients. The majority of patients with tapering were taking 9mg/day of Cortiment®MMX® every 2 days (77.4%, n = 41).
The median duration of Cortiment®MMX® treatment was 57.0 days. The mean (standard deviation, SD) time from the end of Cortiment®MMX® induction treatment to follow-up visit was 29.3 (24.8) days, and was performed in 270 (77.4%) patients. The most common UC treatment at follow-up was oral 5-ASA, which was taken by 230 patients (85.2% of patients participated in the follow-up assessment), whereas systemic corticosteroids were taken by 38 (14.1%) patients, biological drugs were taken by 21 (7.8%) patients and immunosuppressants were taken by 32 (11.9%) patients.
Discussion
Budesonide MMX® is currently approved in Europe for the treatment of active mild-to-moderate UC, for patients refractory or intolerant to mesalazine. Data from the CORE trials8,15,18 showed that one-third of patients receiving Cortiment®MMX® 9 mg/day for 8 weeks as monotherapy achieved clinical improvement (reduction of ≥3 points in UCDAI sub-score), and about one-quarter achieved full symptoms resolution (RB = 0 + SF = 0). However, data from real-life clinical practice were lacking.
Our cohort study included 349 patients who had been prescribed Cortiment®MMX® by their treating physician according to the local SmPC, and independently of their participation in the study. Of these patients, 23 were lost at follow-up before Visit 2, and were therefore excluded from the primary endpoint analysis. When Cortiment®MMX® was used as an add-on to 5-ASA therapy, we found that 64.3% (Cohort 1) and 62.1% (Cohort 2) of patients achieved clinical benefit (improvement ≥3 points in the UCDAI clinical sub-score) with no differences in the timing of administration (less or more than 14 days since 5-ASA was started). Moreover, in both add-on cohorts, 51.8% (Cohort 1) and 45.3% (Cohort 2) of patients achieved full symptoms resolution, defined as the absence of RB, normalisation of bowel habits and no urgency, compared to 29.6% receiving Cortiment®MMX® as monotherapy (Cohort 3). Absence of RB and normalisation of bowel habits are currently considered as the most valuable patient-reported outcomes in evaluating treatment success in UC. 5 The time to symptoms resolution (RB = 0 + SF ≤ 1) ranged from 29 to 36 days. Outcomes related to a patient's daily life were improved significantly following Cortiment®MMX® treatment. Health-related quality of life measures (according to the SIBDQ) increased and WPAI scores reduced. Moreover, Cortiment®MMX® treatment satisfaction, as reported by the patients, was generally high. These data might suggest that there is greater benefit from being treated with Cortiment®MMX® in combination with 5-ASA compounds rather than as a monotherapy. Regarding the objective signs of inflammation, the number of patients where data were available both at baseline and end of treatment was small. Thus, no clear conclusion on the effect of Cortiment®MMX® treatment on endoscopic appearance and FC levels can be given.
Baseline RB, baseline Physician's Global Assessment, compliance with the treatment during induction ≥80% and being treated in Poland were factors associated with a higher probability of responding to budesonide MMX®.
Budesonide was well tolerated, with 24.0% of patients reporting at least one AE, compared to 56.5% of patients in the CORE pooled analysis. No major AEs that are usually seen with systemic steroids were observed, and the most common AEs related to drug inefficacy, gastrointestinal disorders or product use issues. These data confirm that budesonide MMX® acts locally with negligible systemic effects and can be considered a safe drug in the mild-to-moderate UC population.
Our study has some limitations. First, there was a different proportion of patients enrolled in the add-on cohorts, 1 and 2, compared to Cohort 3 (monotherapy). Patients were not stratified for strategy chosen, however, our cohort was large enough to capture significant differences among the cohorts and our data are not different from previous Randomized Controlled Trial (RCT) data,8,15,18 supporting the validity of our results. Second, the difference in response rates among countries (i.e. Poland vs. other countries) reflects the heterogeneity of the study population, which may have had an impact on these findings (Polish patients represent 51.2% of the population). However, this is expected in a real-life cohort, which usually enrols patients that are different from RCT study populations.8,15,18 Third, data on objective measures of inflammation are limited, as few patients underwent endoscopies and FC tests at both baseline and end of Cortiment®MMX® treatment.
However, this study has also several strengths. This is the largest real-life cohort study in mild-to-moderate UC patients treated with Cortiment®MMX®, who were mainly treated in third-level referral centres with experienced IBD clinicians. Our findings demonstrate good effectiveness in real-life practice and help to understand the best patient profile and the best strategies to adopt for the use of Cortiment®MMX® in daily practice. The cohort affiliation of patients also indicated that in about half of the cases, 5-ASA dose optimisation is not carried out according to mild-to-moderate UC treatment guidelines. 6
In conclusion, in this large real-life UC cohort, Cortiment®MMX® was confirmed to be effective, safe and well tolerated.
Footnotes
Acknowledgments
Ferring and the authors would like to thank all study investigators and their teams for their contributions. (Participating sites and principal study investigators listing in the Appendix.) The principal investigators in this study were Ardizzone Sandro, Astegiano Marco, Augustyn Monika, Baluta Małgorzata, Barczyk Wojciech, Bendia Emanuele, Binkowska-Borgosz Izabela, Błaszczyńska Małgorzata, Bresso Francesca, Brymora Małgorzata, Castiglione Fabiana, Comberlato Michele, Cosintino Rocco, Costa Francesco, Detka-Kowalska Iga, Drobińska Anna, Dudkowiak Robert, Eberhardson Michael, Frączek Grzegorz, Geccherle Andrea, Gionchetti Paolo, Halfvarson Jonas, Hayee Bu, Hertervig Erik, Hjortswang Henrik, Janiak Maria, Jansen Jeroen, Jasiński Bolesław, Jastrzębska Marta, Jessen Petra, Jonas Maurycy, Kaczmarzyk Paweł, Kaniewska Magdalena, Kędzierska Lidia, Kempiński Radosław, Klincewicz Beata, Klugmann Tobias, Kosik-Warzyńska Romana, Krause Thomas, Krawczyk Marek, Krela-Kaźmierczak Iwona, Łapiński Janusz, Latos Wojciech, Łykowska-Szuber Liliana, Maciejewska Katarzyna, Majowski Jarosław, Malewski Waldemar, Małuch Piotr, Mamos Arkadiusz, Manerowski Marcin, Markowski Adam, Mazurek Tadusz, Miehlke Stefan, Mowat Craig, Mularczyk Aldona, Mulcahy Hugh, Nijhawan Pardep, Orlando Ambrogio, Orłowski Marcin, Pastorelli Luca, Penpicki Andrzej, Pilecka Dorota, Piotrowski Wojciech, Prasad Neeraj, Reddy Jagan, Rocca Rodolfo, Romatowski Jacek, Rydzewska Grażyna, Sangfelt Per, Schubert Stefan, Seenan John Paul, Selinger Christian, Singh Andrew, Strózik Agnieszka, Świątkowski Tomasz, Tomecki Roman, Walentek Tomasz, Waśko-Czopnik Dorota, West Rachel, Wierzbicka Katarzyna, Wiśniewska Jarosińśka Maria, Wiśniowski Marek, Wojnarowska Renata, Wójtowicz Henryk, Wontor-Buksińska Aleksandra, Worobiec Jacek and Wyszkowski Mariusz.
Conflict of interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SD has served as a speaker, consultant and advisory board member for Schering-Plough, Abbott (AbbVie) Laboratories, Merck and Co, UCB Pharma, Ferring, Cellerix, Millenium Takeda, Nycomed, Pharmacosmos, Actelion, α Wasserman, Genentech, Grunenthal, Pfizer, Astra Zeneca, Novo Nordisk, Cosmo Pharmaceuticals, Vifor, and Johnson and Johnson. AH has lectured/been on advisory boards for Atlantic, BMS, Falk Pharma, MSD, AbbVie, Takeda, Hospira, Napp Pharmaceuticals, Ferring, Yakult, Pfizer, Janssen, Warner Chillcott, and Genentech. AD has served as a speaker and consultant for Ferring, Falk Pharma, Mundipharma, Hospira, Sandoz, Otsuka, AbbVie, Janssen, Takeda, MSD, Vifor, and Pharmacosmos. GF received consultancy fees from Ferring, MSD, AbbVie, Takeda, Janssen, Amgen, Sandoz, Samsung Bioepis, Celltrion. EL has received fees for: Educational Grant: fees for Educational activities paid by the Company, MSD, AbbVie; speaker fees: AbbVie, Ferring, MSD, Chiesi, Mitsubishi Pharma, Hospira, Janssen, Takeda; serving on an advisory board: AbbVie, Ferring, MSD, Takeda, Mitsubishi Pharma, Celltrion, Prometheus. BS is supported by FIRMAD and has served as advisor for Ferring. GD’H has served as advisor for AbbVie, Ablynx, Amakem, AM Pharma, Avaxia, Biogen, Bristol Meiers Squibb, Boerhinger Ingelheim, Celgene, Celltrion, Cosmo, Covidien, Ferring, DrFALK Pharma, Engene, Ferring, Galapagos, Gilead, Glaxo Smith Kline, Hospira, Johnson and Johnson, Medimetrics, Millenium/Takeda, Mitsubishi Pharma, Merck Sharp Dome, Mundipharma, Novonordisk, Pfizer, Prometheus laboratories/Nestle, Receptos, Robarts Clinical Trials, Salix, Sandoz, Setpoint, Shire, Teva, Tigenix, Tillotts, Topivert, Versant and Vifor, and received speaker fees from AbbVie, Ferring, Johnson and Johnson, MSD, Mundipharma, Norgine, Pfizer, Shire, Millenium/Takeda, Tillotts, and Vifor. ID has served as a speaker and/or consultant for Ferring, Falk Pharma, Rafa laboratories, AbbVie, Janssen, Takeda, Genentech, Pfizer, Protalix, Arena, Gilead, Celltrion, MSD, and Given Imaging. GR has served as a speaker and consultant for AbbVie, Ardeypharm, AstraZeneca, Boehringer, Celgene, Ferring, Falk Pharma, Genentech, Janssen, Novartis, Merck, MSD, Pfizer, Pharmabiome, Roche, Takeda, Tillots, UCB, Vital Solutions, and Vifor, Zeller.KP is an employee of Ferring. LPB has received consulting fees from Merck, AbbVie, Janssen, Genentech, Mitsubishi, Ferring, Norgine, Tillots, Vifor, Therakos, Pharmacosmos, Pilège, BMS, UCB Pharma, Hospira, Celltrion, Takeda, Biogaran, Boerhinger Ingelheim, Lilly, Pfizer, HAC-Pharma, Index Pharmaceuticals, Amgen, and Sandoz; and lecture fees from Merck, AbbVie, Takeda, Janssen, Takeda, Ferring, Norgine, Tillots, Vifor, Therakos, Mitsubishi, and HAC-Pharma.
Funding
Ferring Pharmaceuticals funded the study and participated in the study design, interpretation of data, review, and approval of the publication.
Ethics approval
The first Ethical Committee approval has been obtained on 22/12/2015 in Ethikkommission der Ärztekammer Hamburg, Germany. Then, the protocol was approved as follows: Ireland 25/10/2016 Ethics and Medical Research Committee The Netherlands 26/09/2016 ACWO OLVG (Notification only) Sweden 08/06/2016 Regionala etikprövningsnämnden i Stockholm Canada 05/12/2016 IRB Services Italia 13/12/2016 Comitato Etico Azienda Ospedaliera Universitaria Policlinico “Sant'Orsola – Malpighi Great Britain 20/07/2017 South East Scotland Research Ethics Committee Poland 18/03/2016 Komisja Bioetyczna Okregowej Izby Lekarskie (Notification only).
Informed consent
An informed consent form in the patients' native language was used. Two versions of the informed consent form were reviewed and approved by the local Ethical Committees, and used during the study (version 1.0, Sep 8th, 2015, and version 2.0, July 4th, 2017).
Participating sites listing
Country
Principal investigator
Institution name
City
Canada
Nijhawan, Pardep
Digestive Health Clinic
Richmond Hill
Reddy, Jagan
Sudbury Endoscopy Centre
Sudbury
Singh, Andrew
PerCuro Clinical Research
Victoria
Germany
Miehlke, Stefan
Magen-Darm-Zentrum, Facharztzentrum Eppendorf
Hamburg
Krause, Thomas
Gastroenterologie Opernstraße
Kassel
Schubert, Stefan
Gastroenterologie am Bayerischen Platz
Berlin
Klugmann, Tobias
Internistische Gemeinschaftspraxis
Leipzig
Jessen, Petra
Gemeinschaftspraxis
Altenholz
United Kingdom
Prasad, Neeraj
Royal Albert Edward Infirmary
Wigan
Hayee, Bu
Kings College Hospital
London
Selinger, Christian
St. James's University Hospital
Leeds
Seenan, John Paul
Queen Elizabeth University Hospital
Glasgow
Mowat, Craig
Tayside University Hospitals NHS Trust, Ninewells Hospital and Medical School
Dundee
Ireland
Mulcahy, Hugh
St. Vincent's University Hospital
Dublin
Italy
Gionchetti, Paolo
A.O.U. Policlinico S. Orsola Malpighi
Bologna
Pastorelli, Luca
Policlinico S. Donato
San Donato Milanese
Astegiano, Marco
A.O.U. Città della Salute e della Scienza
Torino
Rocca, Rodolfo
A.O. Ordine Mauriziano P.O. Umberto I
Torino
Cosintino, Rocco
A.O. San Camillo Forlanini
Roma
Orlando, Ambrogio
A.O. Ospedali Riuniti Villa Sofia - Cervello
Palermo
Castiglione, Fabiana
A.O.U. Federico II
Napoli
Geccherle, Andrea
A.O. Ospedale Sacro Cuore Don Calabria
Negrar
Bendia, Emanuele
A.O.U. Ospedali Riuniti Umberto I, G.M. Lancisi, G. Salesi
Ancona
Comberlato, Michele
A.S.A.A. Ospedale di Bolzano
Bolzano
Costa, Francesco
A.O.U. Pisana Presidio di Cisanello
Pisa
Ardizzone, Sandro
A.S.S.T. Fatebenefratelli Sacco
Milano
Netherlands
Jansen, Jeroen
OLVG – Oost
Amsterdam
West, Rachel
Fransiscus Gasthuis
Rotterdam
Poland
Wiśniewska Jarosińśka, Maria
Centrum Medyczne Św Rodziny Poradnia Gastrologiczna
Łódź
Brymora, Małgorzata
Poradnia Gastroenterologiczna-Szpital Uniwersytecki nr 2
Bydgoszcz
Manerowski, Marcin
Poradnia Chorób Jelitowych-Szpital Uniwersytecki nr 2
Bydgoszcz
Błaszczyńska, Małgorzata
SP ZOZ ZESPÓŁ SZPITALI MIEJSKICH
Chorzów
Binkowska-Borgosz, Izabela
SPSK 1 PUM w Szczecinie
Szczecin
Wontor-Buksińska, Aleksandra
Indywidualna Praktyka Gastrologiczna
Częstochowa
Dudkowiak, Robert
Uniwersytecki Szpital Kliniczny
Wrocław
Kędzierska, Lidia
SPSK 2 PUM w Szczecinie
Szczecin
Kempiński, Radosław
Uniwersytecki Szpital Kliniczny Wrocław
Wrocław
Malewski, Waldemar
NZOZ Termedica
Poznań
Mamos, Arkadiusz
NZOZ Med-Gastr
Łódź
Mazurek, Tadusz
Centrum Medyczne Medicor
Rzeszów
Mularczyk, Aldona
Specjalistyczna Praktyka Lekarska
Katowice
Pilecka, Dorota
SPWSZ Szczecin
Szczecin
Piotrowski, Wojciech
Centrum Medyczne Poradnia Gastrologiczna
Łódź
Walentek, Tomasz
Szpital Specjalistyczny 1
Bytom
Kosik-Warzyńska, Romana
SPWSZ w Szczecinie
Szczecin
Wojnarowska, Renata
Klinika Gastroenterologii USK nr 1
Łódź
Markowski, Adam
Humana Media Omeda
Białystok
Romatowski, Jacek
NZOZ Specjalistyczne Centrun Gastrologii GASTROMED
Białystok
Tomecki, Roman
Przychodnia Fundacji Gastroenterologicznej
Warszawa
Wierzbicka, Katarzyna
SPZOZ Szpital Bielański
Warszawa
Wiśniowski, Marek
Centrum Medyczne Medyk
Rzeszów
Baluta, Małgorzata
Centrum Medyczne Dąbrowa - Dąbrówka
Gdynia
Detka-Kowalska, Iga
Oddział Gastroenterologiczny Szpital Specjalistyczny
Końskie
Drobińska, Anna
Klinika Gastroenterologii
Gdańsk
Frączek, Grzegorz
Centrum Medyczne Starówka
Sokołów Podlaski
Janiak, Maria
Klinika Gastroenterologii I Hepatologii
Gdańsk
Jasiński, Bolesław
Klinika Gastroenterologii i Hepatologii/Gabinet Prywatny
Wojkowice
Jastrzębska, Marta
Oddział Gastroenterologiczny Szpital Specjalistyczny im. Św. Łukasza
Końskie
Kaczmarzyk, Paweł
Szpital Kielecki św. Aleksandra Sp. z o.o.
Kielce
Krawczyk, Marek
Szpital Miejski
Tychy
Krela-Kaźmierczak, Iwona
Szpital Kliniczny UM
Poznań
Latos, Wojciech
Gabinet
Gliwice
Łykowska-Szuber, Liliana
Szpital Kliniczny z przychodnia im. H.Święcickiego UM
Poznań
Małuch, Piotr
Specjalistyczna Praktyka
Dąbrowa Górnicza
Penpicki, Andrzej
NZOZ GASTROMED
Białystok
Świątkowski, Tomasz
Przychodnia Specjalistyczna
Kędzierzyn - Koźle
Wójtowicz, Henryk
Przychodnia Sucholeska
Suchy Las
Strózik, Agnieszka
Gabinet Internistyczny
Częstochowa
Augustyn, Monika
Indywidualna Praktyka Lekarska
Kraków
Barczyk, Wojciech
105 Szpital Wojskowy SPZOZ Żary
Żary
Jonas, Maurycy
Spółdzielnia Pracy Specjalistów Rentgenologów
Warszawa
Łapiński, Janusz
Centrum Gastrologiczno Hepatologiczne
Wrocław
Majowski, Jarosław
Centrum Medyczne Medita
Jelenia Góra
Worobiec, Jacek
Specjalistyczna Poradnia Lekarska i Szpital CDT MEDICUS
Lubin
Orłowski, Marcin
Przychodnia Lekarska Nowy Chełm
Gdańsk
Waśko-Czopnik, Dorota
USK Wrocław
Wrocław
Klincewicz, Beata
Szpital Kliniczny im. Karola Jonschera Uniwersytetu Medycznego im. Karola Marcinkowskiego w Poznaniu
Poznań
Rydzewska, Grażyna
CSK MSW 1
Warszawa
Wyszkowski, Mariusz
Zespół Przychodni Specjalistycznych PRIMA
Warszawa
Maciejewska, Katarzyna
CSK MSWiA
Warszawa
Kaniewska, Magdalena
Szpital MSWiA
Warszawa
Sweden
Bresso, Francesca
Karolinska University Hospital
Stockholm
Halfvarson, Jonas
Örebro University Hospital
Örebro
Sangfelt, Per
Uppsala University Hospital
Uppsala
Hjortswang, Henrik
Linköping University Hospital
Linköping
Hertervig, Erik
Skåne University Hospital
Lund
Eberhardson, Michael
Danderyds sjukhus
Stockholm
