Abstract
Background
Data on the natural history of acute lower gastrointestinal bleeding (ALGIB) are lacking. We evaluated five-year bleeding risk and mortality in ALGIB patients and controls. Furthermore, we aimed to find predictors of rebleeding.
Methods
This was a population-based retrospective case-control study conducted at the National University Hospital of Iceland, and included every individual who underwent endoscopy in 2010–2011. ALGIB was defined as rectal bleeding leading to hospitalisation or occurring in a hospitalised patient. Controls were randomly selected from those who underwent endoscopy in the same time period but who did not have GIB, and were matched for sex and age. Patients were followed up five years after index bleeding. Rebleeding was defined as ALGIB >14 days after index bleeding.
Results
In total, 2294 patients underwent 2602 colonoscopies in 2010–2011. Of those, 319 (14%) had ALGIB. The mean age for cases and controls was 64 and 65 years (±19.3–20.7), respectively, and females accounted for 51–52% of the study population. For ALGIB patients, the five-year risk of a bleeding was 20% (95% confidence interval (CI) 15–24%) compared to 3% (95% CI 1–5%) in controls (log rank < 0.0001; co-morbidity-adjusted hazard ratio (HR) 6.9 (95% CI 3.4–14)). Only 37% of bleeders had the same cause of index bleeding and rebleeding. In ALGIB patients, age and inflammatory bowel disease (IBD) were predictors of rebleeding, with odds ratios per 10 years of 1.3 (95% CI 1.1–1.6) and 4.3 (95% CI 1.5–12), respectively. Bleeders did not have a higher risk of five-year mortality compared to controls (HR = 1.2; 95% CI 0.87–1.6).
Conclusions
One fifth of ALGIB patients had rebleeding during follow-up. Age and IBD were independent predictors of rebleeding. ALGIB was not associated with lower five-year survival.
Keywords
Introduction
Acute lower gastrointestinal bleeding (ALGIB) is a common cause of hospital admission, with studies showing incidence ranging from 21 to 87/100,000 inhabitants per year.1–4 Furthermore, the incidence of ALGIB has been reported to rise with age3,4 and to be increasing overall. 2 The increase in ALGIB, in addition to increasing life expectancy in the Western world, 5 indicates that ALGIB will become a greater health concern in the near future. In spite of this, studies on the natural history of patients with ALGIB are scarce, and guidelines mention that ALGIB rebleeding rates are poorly characterised. 6 The few studies that exist on the long-term rebleeding rates and mortality of ALGIB patients have reported a three-year cumulative rebleeding rate of 18–35%3,7,8 and a three-year survival of 69–94%.1,9,10 Most of these studies had a relatively short follow-up period and were not population based,7,8 and none compared their bleeder cohort to controls.3,7,8
As ALGIB is a growing issue, identifying predictors of rebleeding is of major importance potentially to reduce its magnitude. One study found being older than 65 years of age, non-steroidal anti-inflammatory drugs (NSAIDs) and the use of non-aspirin anti-platelets were risk factors of rebleeding. 8 However, data on this subject are very limited.
We aimed to evaluate the five-year cumulative rebleeding rate and mortality in patients with ALGIB and to compare their prognosis to a control group. Secondary aims were to compare ALGIB patients who had a rebleed and those who did not in an attempt to identify independent predictors of rebleeding.
Methods
Study design and patient finding
This was a population-based retrospective study conducted at the National University Hospital of Iceland, and it included all those undergoing oesophagogastroduodenoscopy (OGD) and/or colonoscopy in 2010–2011. Patients were recruited by reviewing every OGD and colonoscopy report from 2010–2011 electronically stored in the hospital (n = 8018). Cases included patients (≥18 years) who had ALGIB, defined as rectal bleeding/melena located distal to the ligament of Treitz2,3,9 that lead to hospitalisation or occurred in a hospitalised patient. Patients undergoing outpatient endoscopies were not included. If the cause of bleeding in patients with rectal bleeding was not found on colonoscopy, those patients had to have a negative OGD to be included in the study cohort. Patients with diverticulosis and no other apparent cause of bleeding were assumed to have diverticular bleeding. Controls were randomly selected from among those who underwent endoscopy in the same time period but were not suspected of having GIB and were matched for sex and age. Cases and controls were then followed up exactly five years after index bleeding by examining their electronic medical records. A rebleed was defined as another ALGIB event that led to hospitalisation or occurred in a hospitalised patient >14 days after index bleeding – an interval chosen to minimise the risk of including patients with continued bleeding.
Medical records at the University Hospital are electronically connected to different registers kept by the Directorate of Health (www.landlaeknir.is) in Iceland. These registers hold data on date of death as well as the place of residence for every individual in Iceland.
Patients
The National University Hospital of Iceland is one of two tertiary care hospitals in Iceland. In 2010, it had 677 beds and was bound to serve a population of 151,000 individuals aged ≥ 18 years in the greater metropolitan area of Reykjavík. 11 The hospital also receives patients residing in the western and southern part of Iceland outside of Reykjavík – a population of 50,000 individuals (≥18 years). 11
Data collection and variables
In February to March 2017, electronic medical records of cases and controls were retrospectively reviewed, and data were collected on co-morbidities according to the Charlson Comorbidity Index, 12 rebleeding and its cause as well as date of death. Additionally, the following laboratory values were collected for bleeders at the index bleeding: haemoglobin, haematocrit, platelets, creatinine and international normalised ratio (INR). Pharmaceuticals noted were NSAIDs, low-dose aspirin (LDA), anticoagulants, anti-platelets and proton-pump inhibitors (PPIs). Anticoagulants included warfarin, dabigatran, apixaban, rivaroxaban, heparin, low-molecular-weight heparins and fondaparinux. Anti-platelets included LDA, clopidogrel, dipyridamole, prasugrel, ticagrelor, ticlopidine, eptfibatide and anagrelide. PPIs included esomeprazole, omeprazole, rabeprazole and lansoprazole. Bleeding was considered clinically significant if the patients required a blood transfusion (Hb < 100 g/L), became hemodynamically unstable, were admitted to intensive care, required surgery or died.
Statistics
All data were processed using R vXX Version 3.5.3 (The R Project for Statistical Computing, Vienna, Austria) and Microsoft Excel 2010 (Microsoft, Redmond, WA). The Mann–Whitney U-test was used to test for differences in continuous variables between groups, and Fisher's exact test was used to test for differences in dichotomous variables between groups. The Charlson Comorbidity Index is displayed as a categorical variable. In statistical calculations, it was handled as a continuous variable. The Kaplan–Meier method was used to evaluate the cumulative probability of a bleeding event and survival after five years of follow-up. Each individual in the study contributed exactly five person-years to the calculations, aside from those who experienced an event (rebleeding or death) or were censored (death in the rebleeding analysis). Rebleeding and survival in cases and controls were compared with Cox's proportional hazard model. Stepwise logistic regression analysis was used in an attempt to find independent risk factors of rebleeding. All tests were two-tailed and were carried out at a 5% significance level. The results are presented as means and standard deviations (SD) or medians and interquartile ranges (IQR). All confidence intervals (CI) are 95%.
Access to data and statistical calculations is available on request from the corresponding author.
Results
Baseline characteristics of bleeders and controls.
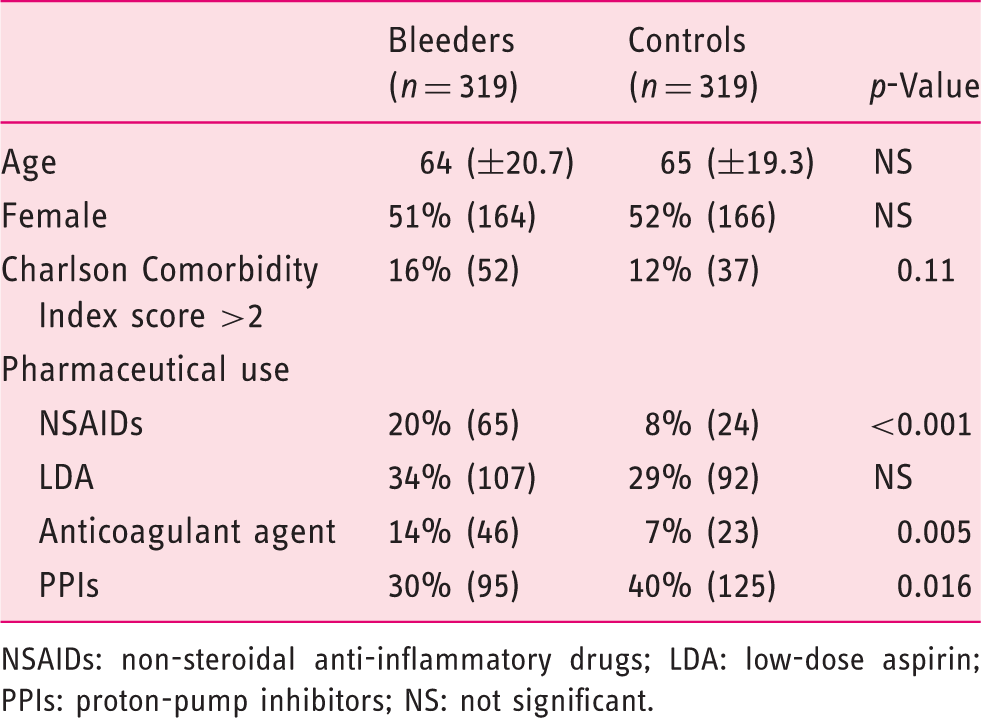
NSAIDs: non-steroidal anti-inflammatory drugs; LDA: low-dose aspirin; PPIs: proton-pump inhibitors; NS: not significant.
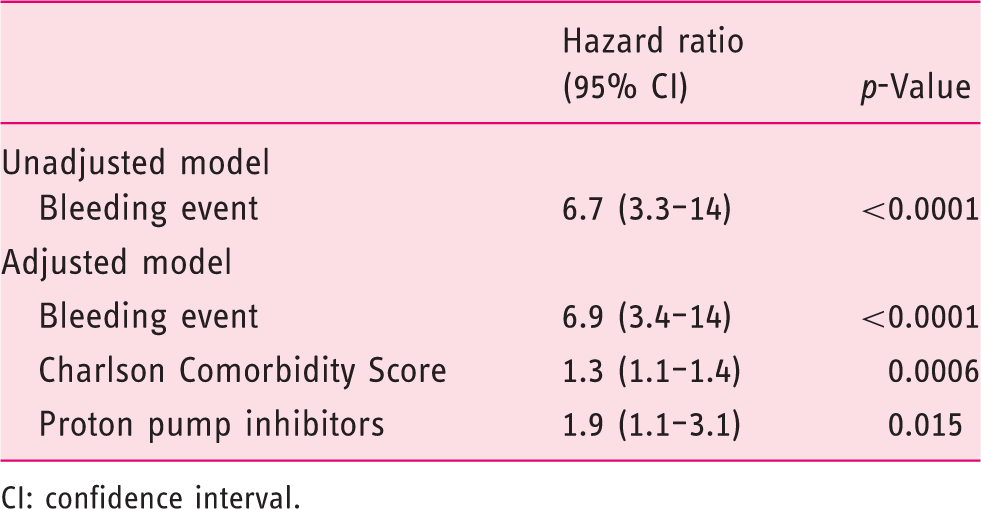
ALGIB patients had a 20% (95% CI 15–24%) cumulative rebleeding rate at the end of the five-year follow-up, with the greatest risk during the first year (7%; 95% CI 4–10%) and then gradually increasing over the last four years of follow-up (Figure 1). Controls had a much lower probability of a bleeding event during follow-up, with a cumulative event rate of 3% (95% CI 1–5%; log rank <0.001; Figure 1). Patients with ALGIB and controls were compared using Cox's proportional hazard model. Bleeders were more likely to experience another bleeding event when compared to controls (hazard ratio (HR) = 6.7; 95% CI 3.3–14). When correcting for possible confounders, the HR was very similar (6.9; 95% CI 3.4–14; Table 2).
The cumulative probability of a bleeding event among ALGIB patients and controls. A comparison of hazard for bleeding events between cases and controls. CI: confidence interval.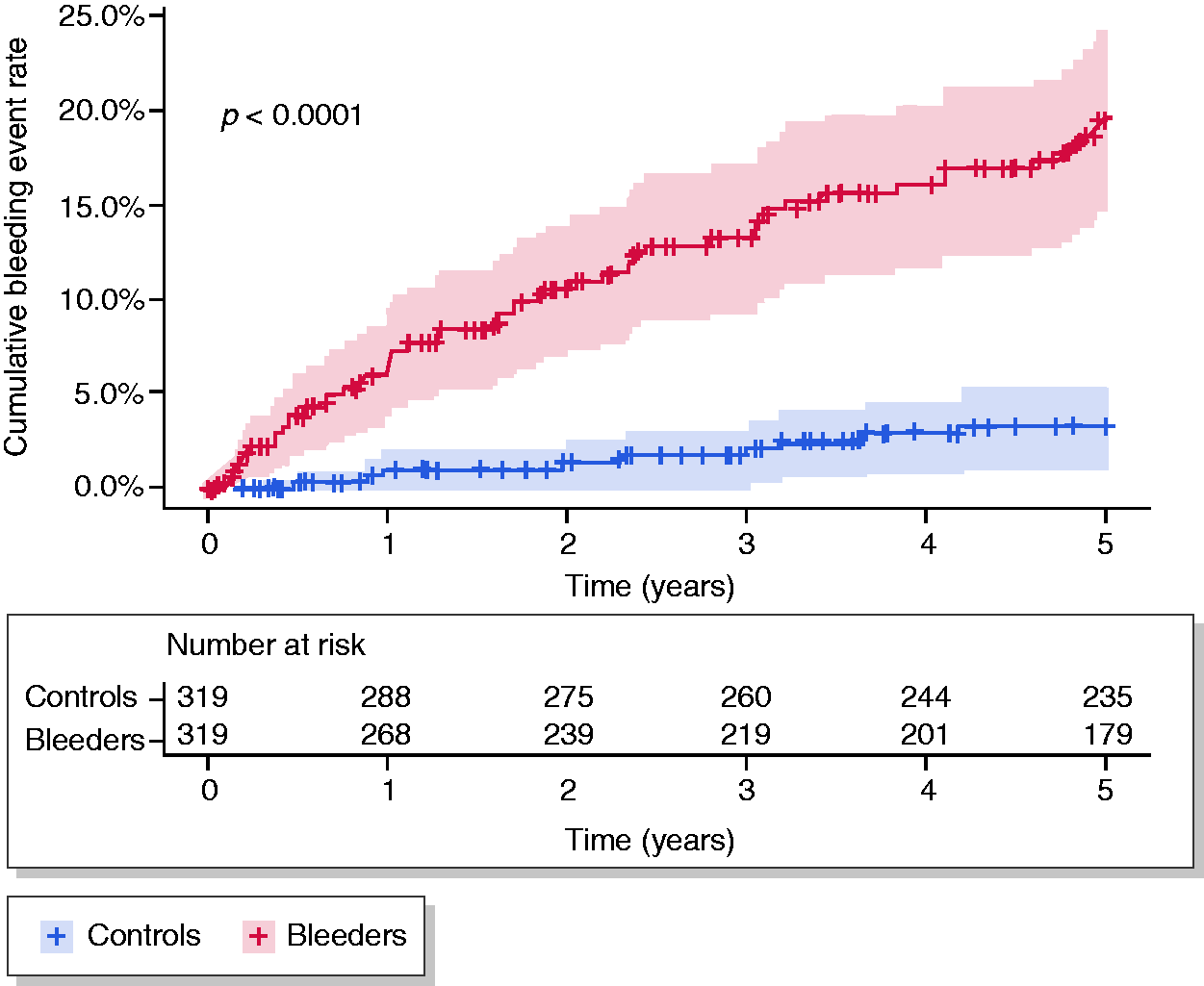
A comparison of those ALGIB patients with and without a rebleed.
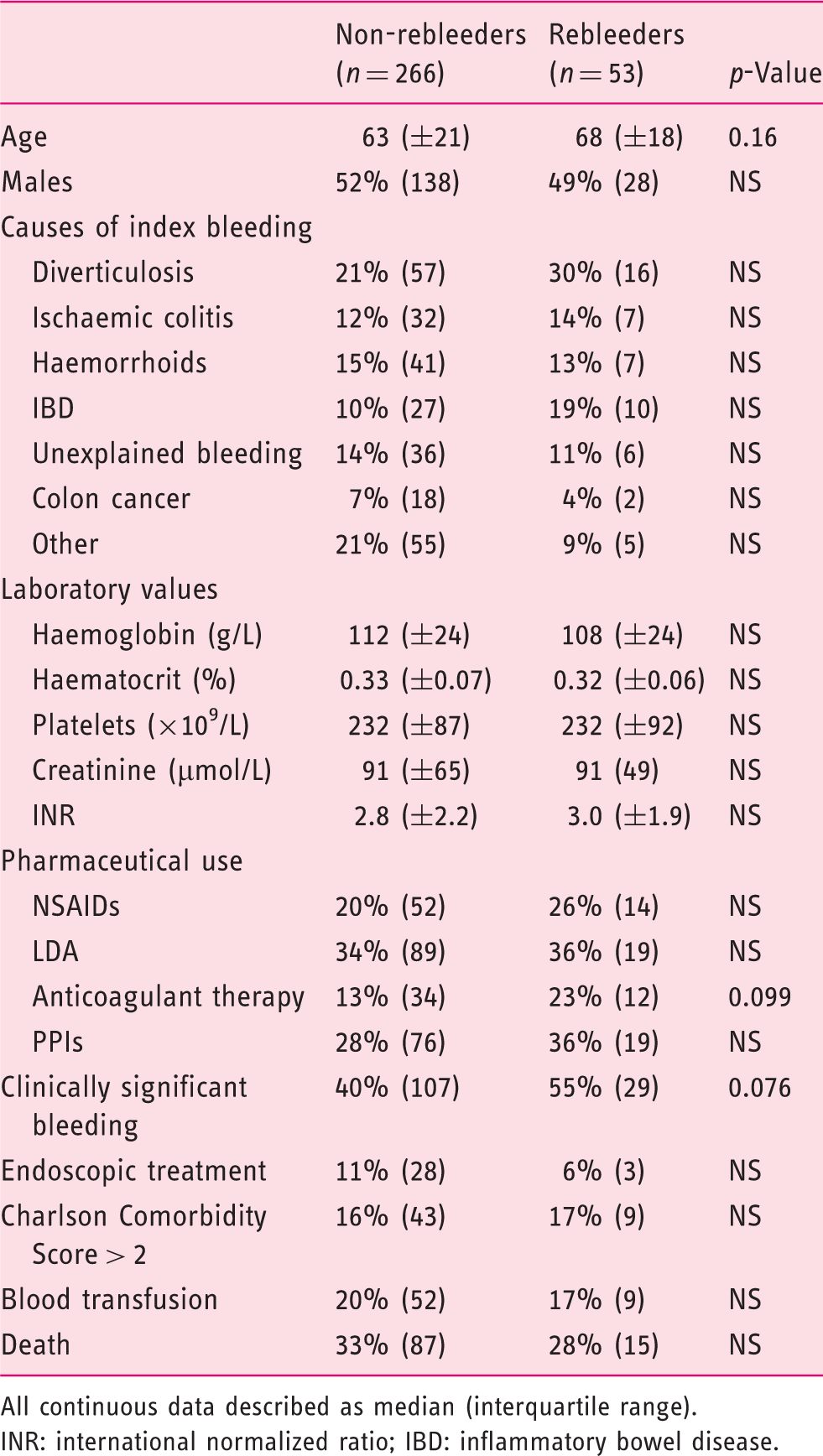
All continuous data described as median (interquartile range).
INR: international normalized ratio; IBD: inflammatory bowel disease.
In stepwise logistic regression, age and having IBD as a cause of index bleeding were associated with an increased risk of a rebleed (odds ratio (OR) = 1.3 (95% CI 1.1–1.6) per 10 years and 4.3 (95% CI 1.5–12), respectively). A sub-analysis where all IBD patients were excluded was performed, and the rebleeding rate was similar (18%; 95% CI 13–23%). Furthermore, age was still a statistically significant risk factor for rebleeding, with a similar OR of 1.2 (95% CI 1.0–1.5) when compared to the analysis that included IBD patients (1.3; 95% CI 1.1–1.6).
Out of all ALGIB patients included in the follow-up, 1% (3/319) had rectal bleeding from an upper GI source. One had a peptic ulcer, one ab ulcer on a Billroth II anastomosis and one gastric antral vascular ectasia. Of controls, 2% (7/319) had rectal bleeding and/or melena from an upper GI source, five had a peptic ulcer, one had variceal bleeding and one had oesophagitis.
The survival of bleeders at the end of follow-up was 70% (95% CI 65–75%) compared to 75% (95% CI 71–80%) in controls (log rank = 0.12; Figure 2). In Cox regression in which the Charlson Comorbidity Index score was corrected for, there was no difference in survival between bleeders and controls (HR = 1.1; 95% CI 0.85–1.5). Three patients with ALGIB, but none of the controls, had a rebleeding-related death.
The cumulative probability of survival for bleeders and controls.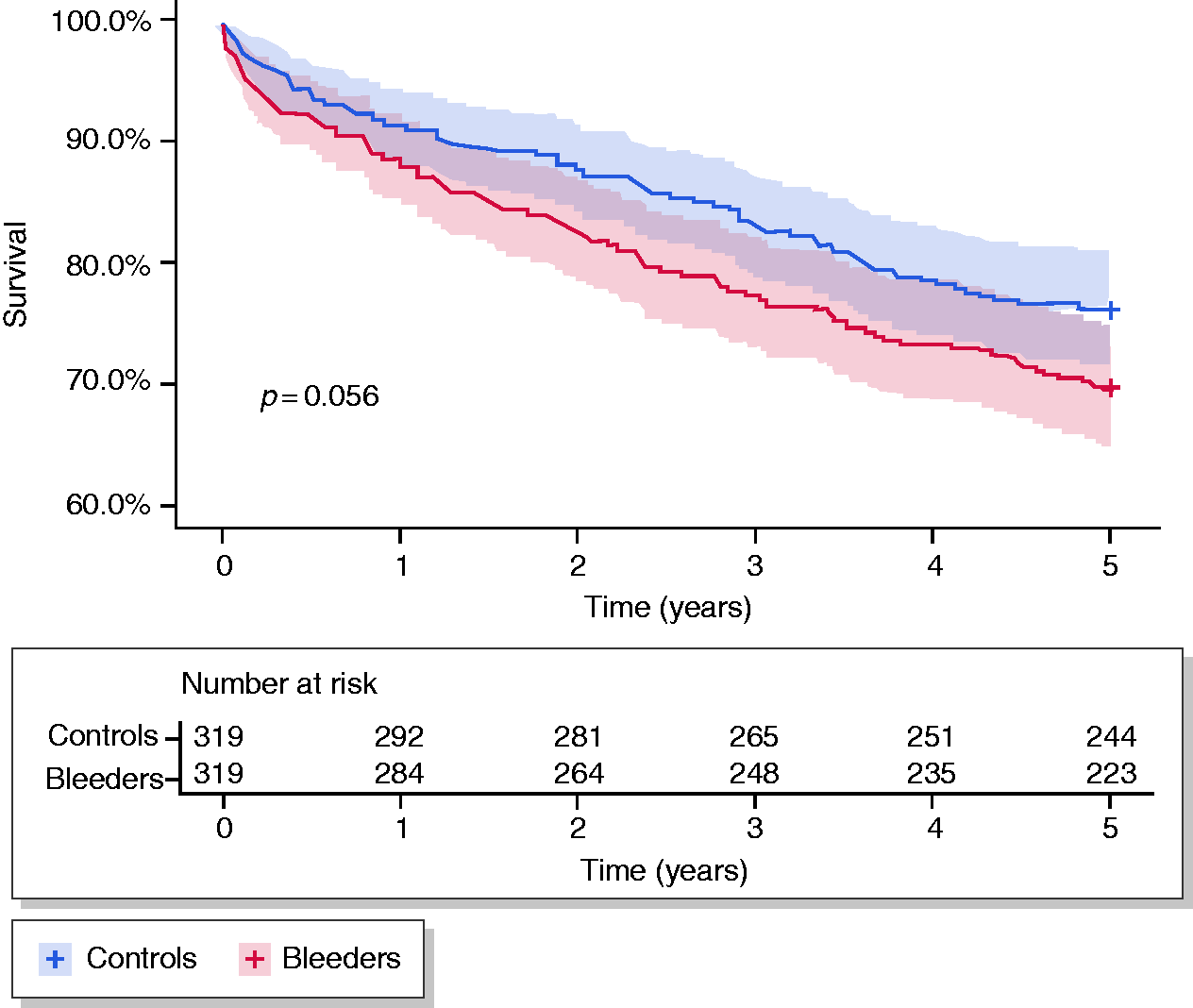
Discussion
In the current study, we found that the cumulative probability of a rebleed was 20% for ALGIB patients during five years of follow-up. When compared to controls, patients had more than seven times the hazard of another bleeding event during follow-up. Higher age and IBD as a cause of index bleeding were found to be independent predictors of a rebleed. Having ALGIB was not associated with a lower five-year survival when compared to controls.
To our knowledge, this study is the first to compare the rebleeding rates of patients with ALGIB and bleeding events in controls. With regard to rebleeding rates in ALGIB patients alone, our results indicate that the probability is the highest in the first year of follow-up (7%) and then increases evenly after that. This is in line with previous studies showing the cumulative rebleeding rate at one year to be 9–19%.3,7,8 The two- and three-year cumulative rebleeding rates in the current study were 11% and 13% – comparable to two of the aforementioned studies with 13–19% rebleeding rates.3,7,13,14 However, others have reported a higher rate of 35%. 8 Although studies with follow-up times longer than two to three years are lacking, there was one randomised controlled trial that compared urgent colonoscopy to standard care in 50 ALGIB patients. Overall rebleeding was 15% during a mean follow-up of about five years 15 – marginally lower than in the current study. Differences between the current and aforementioned studies may be explained by the fact that all but one 3 were not population based and the mean follow-up time in some of these studies did not exceed two years, varying greatly between individuals.7,8 Furthermore, in a study from a surgical unit where ALGIB patients were identified by records of technetium-labelled red blood cell scan, 30-day mortality was found to be 18% 6 – considerably higher than in the current and other studies (0–7%)9,10,16 – indicating patient selection was biased towards more serious bleeds in that study. In addition, differences in the definition of rebleeding and differences in aetiology may skew the comparison between studies.
One study previously showed that being older than 65 years of age, NSAIDs and use of non-aspirin anti-platelet drugs are independent risk factors for a rebleed. 8 In the current study, we identified age and IBD as independent risk factors for a rebleed. Interestingly, the use of anticoagulants and anti-platelets at index bleeding were not found to predict rebleeding. Although the data are not available in the present study, current clinical practice in Iceland is to restart antithrombotic therapy as soon as possible after a GIB event, as recommended in the guidelines. 6 It is therefore reassuring that antithrombotic therapy is not clearly associated with long-term rebleeding.
With regard to endoscopic haemostasis, it was lower in the current study when compared to some 9 but not all studies. 10 A possible explanation for lower endoscopic rates in the current study may be the population-based setting which eliminates the risk of case selection.
Having an episode of ALGIB did not seem to be associated with a lower survival when compared to controls. The one- and three-year survival rates in ALGIB patients have been reported to be greater (96% and 94–95%),8,13 similar (89% and 82%) 3 and lower (75% and 69%) 7 than the current study's report of 89% and 78%. Apart from previously explained differences in these studies, we can see no apparent reason for these diverse numbers of survival. It could be speculated that the high life expectancy of 84 years in Japan may partly explain the discrepancies of our results and that of Aoki et al. 8 However, the life expectancy is also high in Iceland (82 years). 17
The population-based design of this present study, its complete follow-up of five years and comparison of ALGIB cases to a control group are the main strengths of this study. This makes the current study an important addition to the literature, as similar studies have been lacking in these aspects. The study does, however, have some limitations. First, we may have missed some cases of ALGIB in rural areas, as indicated in the Methods, by comparing the incidence of ALGIB with and without individuals from rural areas. Another cause of concern is control selection bias, as controls were selected from among those who underwent colonoscopies in the same time period not the general population. This may have led to an underestimate of cumulative probability of a bleeding event and an overestimate in the probability of death in five years if we assume the controls had a lower overall health status when compared to individuals of the same age and sex in the general population. Finally, the study was retrospective and somewhat lacking in power, especially in the comparison of non-rebleeders and rebleeders, which was aimed at finding predictors of rebleeding.
In summary, the probability of ALGIB patients having a bleeding episode in five years is 20%, and their hazard of bleeding is approximately seven times greater than in controls. ALGIB does not seem to be associated with worse survival when compared to those who do not bleed. Higher age and IBD appear to be risk factors for a rebleed.
Footnotes
Declaration of conflicting interests
None declared.
Ethics approval
The study was approved by the Bioethics Committee of Iceland (10-014-V3).
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Informed consent
Icelandic legislation on studies of anonymised retrospective data does not require informed consent.
