Abstract
Background and Objectives
Corticosteroids are often empirically used to treat idiosyncratic hepatotoxicity with severe features. Interestingly, intravenous methylprednisolone (MP) is increasingly being recognized as being responsible for liver injury. We aimed to characterize MP-induced liver injury by analyzing demographical, clinical, laboratory and outcome data of three MP-induced hepatotoxicity cases and compared this information with that of previously published cases.
Case series
Three females with multiple sclerosis (MS) were treated intravenously with MP, mean daily dose 767 mg. Liver damage occurred 2 to 6 weeks after exposure. Severity was mild to moderate. Two patients suffered positive rechallenge.
Literature review
We identified 50 published cases of MP hepatotoxicity. Most of these cases were female (86%) and main treatment indications were MS (29 cases) and Graves' ophthalmopathy (13 cases). Hepatocellular damage predominated and mean time to onset was 6 weeks. Four patients died and rechallenge occurred in 19 cases.
Conclusion
MP pulses can induce severe liver injury, often with an autoimmune phenotype, particularly in patients with MS and Graves' ophthalmopathy. Consequently, these patient groups should have liver tests monitored when treated with MP to provide safer patient care.
Keywords
Key summary
Corticosteroids, which are used in severe forms of acute liver injury due to their potent immunomodulatory activity, are considered non-hazardous agents to the liver, and indeed they are underrepresented in drug-induced liver injury (DILI) databases. However, cases of methylprednisolone (MP)-DILI have been increasingly reported in recent years.
Main findings of our study
Liver injury associated with MP typically appears during the first weeks after receiving high doses of the agent, manifesting with a hepatocellular pattern of damage, and commonly goes unnoticed favouring rechallenge with the drug. MP-induced liver injury frequently presents with an autoimmune phenotype, particularly in patients with multiple sclerosis and Graves' ophthalmopathy, which suggests that the drug may unmask AIH in susceptible subjects. Liver tests in patients with autoimmune disorders should be monitored when treated with high-dose courses of MP in order to diagnose liver injury early and to provide a better management of these patients.
Introduction
Methylprednisolone (MP), a synthetic glucocorticoid drug, exerts potent anti-inflammatory and immunomodulatory effects, therefore it is prescribed to treat severe inflammatory, autoimmune and neoplastic disorders, as well as in the setting of transplant rejection, among other uses. High-dose intravenous MP is the standard therapy for relapsing autoimmune disorders such as multiple sclerosis, where doses ranging from 500 mg/day for 5 days to 1 g/day for 3 days are commonly used.
Corticosteroids are considered to be safe, in terms of hepatotoxicity, thus, they are underrepresented in hepatotoxicity databases. 1 However, a number of liver injury cases, particularly those related to high-dose methylprednisolone, have been reported in recent years. This situation has triggered controversy, in terms of the attribution of causality since corticosteroids have been empirically advocated for the management of severe forms of idiosyncratic drug-induced liver injury (DILI), as well as in cases of hepatotoxicity presenting with autoimmunity and systemic hypersensitivity features.
In the present study, we aimed to characterize the clinical phenotype of MP-induced liver injury (DILI) by analyzing three cases included in Spanish (from 1994 to 2018) and Latin-American (from 2013 to 2018) DILI registries. Both databases share the same standardized operating procedures, which have been published elsewhere. 2 Information from newly reported cases was then compared with that of 50 previously published cases retrieved from an extensive literature review.
Case series
Patient 1
A 31-year-old female with a 3-year history of multiple sclerosis (MS) under treatment with interferon beta (IFN-β) and glatiramer acetate for the last 9 and 3 months, respectively, was admitted to the hospital due to acute relapsing disease.
The patient received a 3-day course of 300 mg/day intravenous pulse MP. Two weeks later, weakness, nausea, weight loss and jaundice ensued. Total bilirubin was 3.6 mg/dL (TBL < 1.0 mg/dL), aspartate aminotransferase 1420 U/L (AST < 31 U/L), alanine aminotransferase 2194 U/L (ALT < 31 U/L) and alkaline phosphatase 229 U/L (ALP < 129 U/L), indicating hepatocellular injury. All medications were stopped at this point (Figure 1(a)). The patient's liver tests at the time of starting MP therapy were normal (Figure 1(a)). Serology tests for viral hepatitis A (HAV), B (HBV), C (HCV), cytomegalovirus (CMV), Epstein–Barr virus (EBV) and human immunodeficiency virus (HIV) were negative. Immunoglobulins, ceruloplasmin, copper, iron and transferrin serum levels were within normal ranges. Antinuclear (ANA), antimitochondrial (AMA) and anti-smooth muscle antibodies (ASMA) were negative. However, positive ANA titers of 1/320 were found at the time of biochemistry normalization. Liver imaging (CT scan, cholangiography) were normal. Liver biopsy performed 1 month after initiating MP treatment showed centrizonal necrosis with portal fibrosis and mixed inflammatory infiltrates. A maximum TBL value of 12.6 mg/dL was reached 5 weeks after onset.
Temporal relationship between courses of intravenous MP pulse and liver biochemistry in four patients. The data shown correspond to patient 1 with a single episode after exposure to MP (a), patient 2 who developed after a first episode up to three more events of MP hepatotoxicity (b) and patient 3 who after a first event of MP-induced liver injury suffered from rechallenge due to MP (c).Concomitant treatments present at onset of each liver dysfunction episode are displayed. Abbreviations: MP: methylprednisolone; TBL: serum total bilirubin (mg/dl); ALT: alanine aminotransferase (U/L); ALP: alkaline phosphatase (U/L); interferon-β: interferon beta; Bx: biopsy; g: gram; d: day; i.v.: intravenous; RC: rechallenge; CaC: calcium carbonate; CaG: calcium gluconate; MCP: metoclopramide; prn: pro re nata.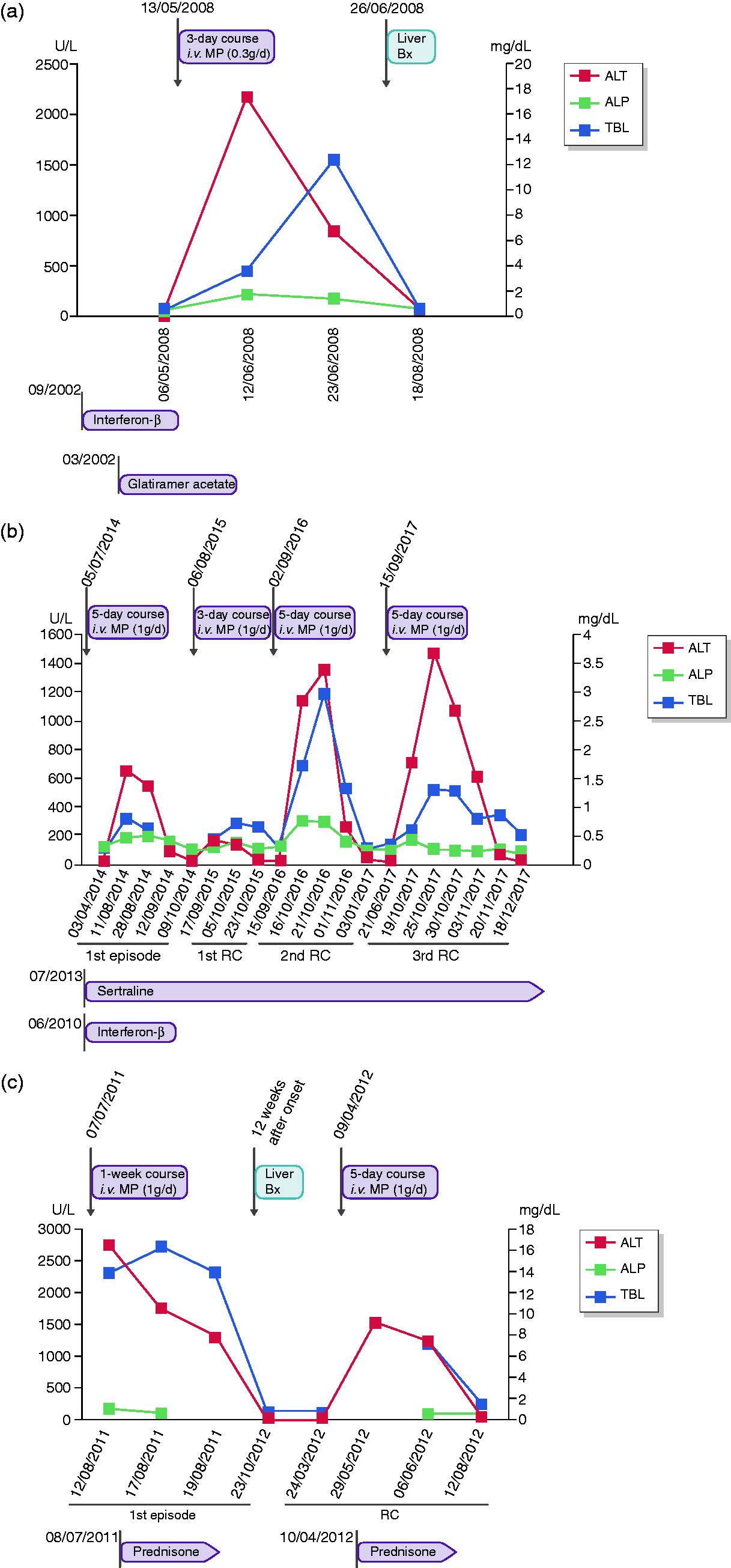
Liver tests returned to normal within 70 days. Causality assessment using the Council for International Organizations of Medical Sciences (CIOMS) scale 3 scored 7 points (probable) for MP.
Patient 2
A 36-year-old female with a previous history of MS, depressive disorder and non-alcoholic fatty liver disease and with normal liver tests (Figure 1(b)) and stable body weight (60 kg) was treated with IFN-β for 4 years and sertraline for the last year. She presented with relapsing MS and received a 5-day course of high-dose (1 g/day) intravenous methylprednisolone and then prednisone 60 mg/day, which was gradually reduced and ultimately withdrawn in 3 weeks. Laboratory tests 5 weeks after starting the MP showed acute hepatocellular injury: TBL 0.8 mg/dL ( < 1.1 mg/dL), AST 419 U/L ( < 27 U/L), ALT 630 U/L ( < 33 U/L), and ALP 193 U/L ( < 270 U/L). Serology for HAV, HBV, HCV, CMV and EBV was negative. ANA, AMA, ASMA and anti-liver/kidney microsomal antibody type 1 ([anti-LKM-1]) were all negative. An abdominal ultrasound showed normal liver parenchyma and biliary ducts and a thin-walled gallbladder containing biliary sludge. Laboratory chemistries were normal 8 weeks later; the patient remained on sertraline, however IFN-β was discontinued. Several months later during follow-up, liver tests remained normal.
One year later, a similar episode occurred following MS exacerbation and administration of intravenous MP (1 g/day for 3 days) and the same course of dose titration. Two weeks after MP cessation, abnormal liver parameters were found: TBL 0.46 mg/dL, AST 122 U/L, ALT 165 U/L and ALP 125 U/L (Figure 1(b)). Viral hepatitis was excluded, and autoantibodies were negative. Eleven weeks later, liver tests were normal.
Over the next 2 years, due to relapsing–remitting MS, she received subsequent 5-day courses of high intravenous dose of MP, which were followed by flares of aminotransferases (first year: TBL 1.75 mg/dL, AST 896 U/L, ALT 1151 U/L and ALP 314 U/L; second year: TBL 0.57 mg/dL, AST 485 U/L, ALT 696 U/L, ALP 172 U/L) (Figure 1(b)). Screening for viral hepatitis was again negative.
CIOMS scale yielded a score of 9 (highly probable) for MP hepatotoxicity with three positive rechallenges.
Patient 3
A 33-year-old female with a 12-year history of MS was referred to the clinic due to marked jaundice, pruritus and elevated transaminases. The patient had up to six MS exacerbations in the past and had received bolus, high-dose (1 g/day) MP for 3 to 7 days, followed by oral prednisone, with complete neurological remission. Routine biochemistry after each treatment was normal.
Following a new MS relapse and administration of intravenous MP (1 g/day for 1 week) and 12 days of oral prednisone, laboratory tests were performed 6 weeks later, indicating severe hepatocellular injury: TBL 13.9 mg/dL ( < 1.0 mg/dL), AST 1823 U/L ( < 104 U/L), ALT 2737 U/L ( < 52 U/L) and ALP 169 U/L ( < 104 U/L). Serology ruled out viral causes. Screening for autoantibodies was negative, serum ceruloplasmin; globulin levels were normal, as was abdominal ultrasonography. Clinical and laboratory findings 2 weeks later showed spontaneous improvement. Three months later, a liver biopsy specimen showed minimal residual hepatitis.
Ten months later, similar acute hepatitis-like symptoms occurred following MS exacerbation and intravenous MP administration (1 g/day for 5 days). Laboratory tests showed the following results: TBL 2.47 mg/dL, AST 1113 U/L, ALT 1500 U/L and INR 1.31, which returned to normal several weeks later. The CIOMS scale provided a score of 7 (probable) for MP with positive re-exposure.
Literature review
Summarized demographic and clinical information of methylprednisolone-induced liver injury events reviewed in literature (n=50).
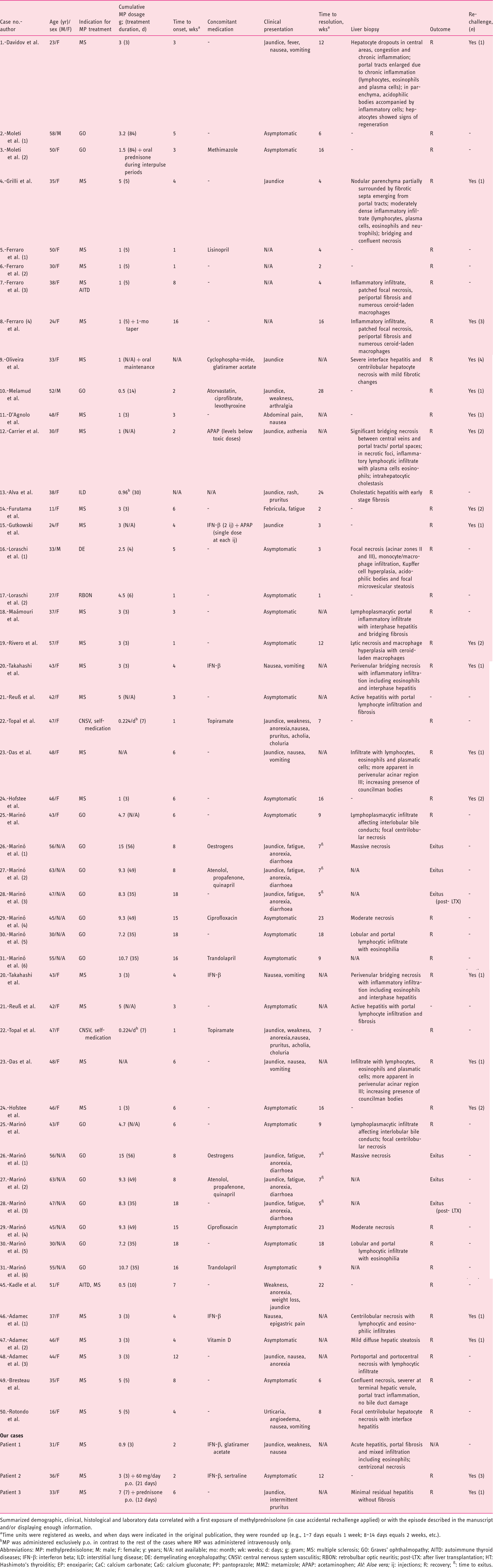
Summarized demographic, clinical, histological and laboratory data correlated with a first exposure of methylprednisolone (in case accidental rechallenge applied) or with the episode described in the manuscript and/or displaying enough information.
Time units were registered as weeks, and when days were indicated in the original publication, they were rounded up (e.g., 1–7 days equals 1 week; 8–14 days equals 2 weeks, etc.).
MP was administered exclusively p.o. in contrast to the rest of the cases where MP was administered intravenously only.
Abbreviations: MP: methylprednisolone; M: male; F: female; y: years; N/A: not available; mo: month; wk: weeks; d: days; g: gram; MS: multiple sclerosis; GO: Graves' ophthalmopathy; AITD: autoimmune thyroid diseases; IFN-β: interferon beta; ILD: interstitial lung disease; DE: demyelinating encephalopathy; CNSV: central nervous system vasculitis; RBON: retrobulbar optic neuritis; post-LTX: after liver transplantation; HT: Hashimoto's thyroiditis; EP: enoxiparin; CaC: calcium carbonate; CaG: calcium gluconate; PP: pantoprazole; MMZ: metamizole; APAP: acetaminophen; AV: Aloe vera; ij: injections; R: recovery; ß: time to exitus.
Summarized biochemical, liver injury pattern and autoimmunity information of methylprednisolone-induced liver injury events at peak reviewed from literature (n = 50) and new reported cases (n = 3).
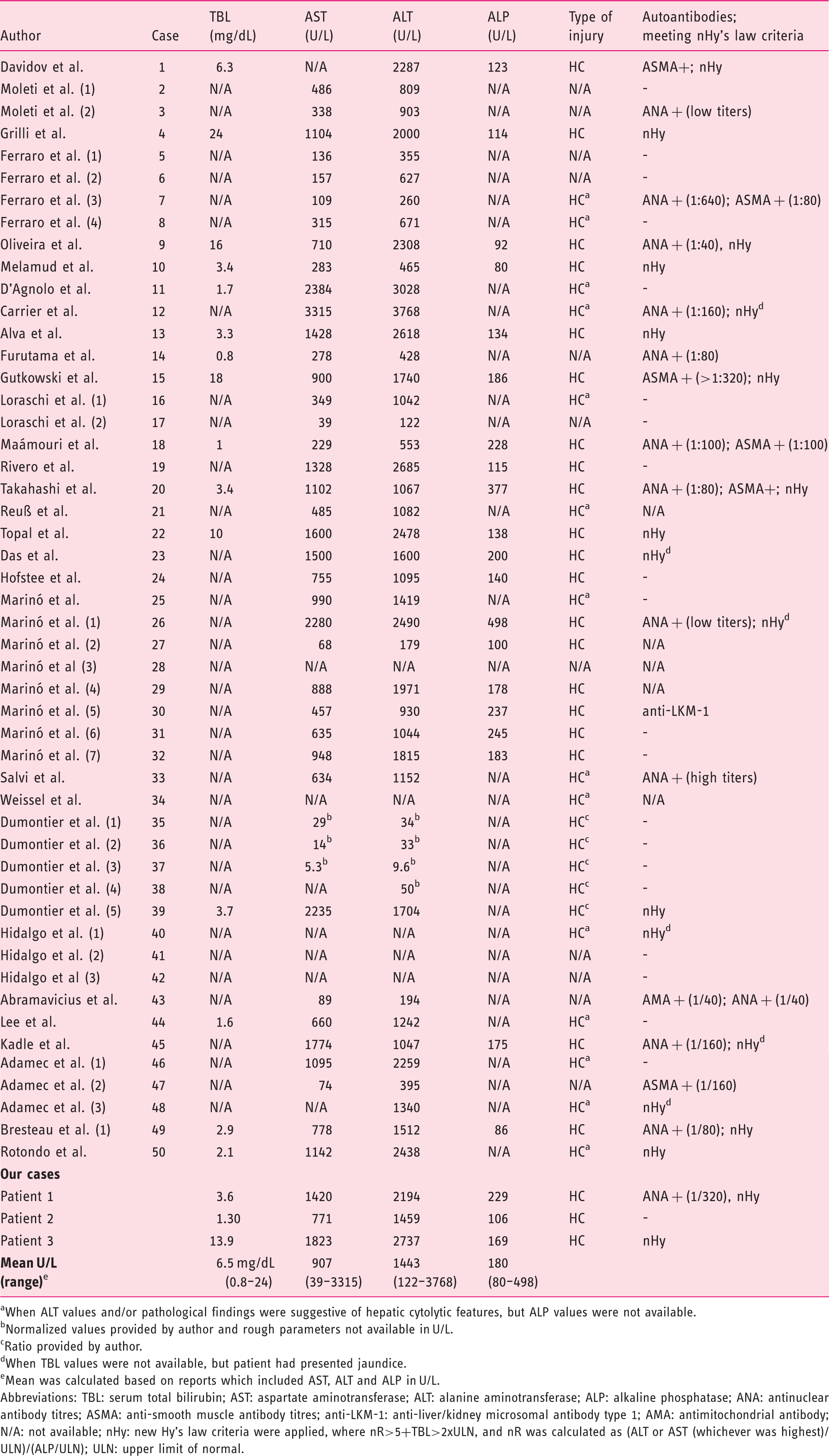
When ALT values and/or pathological findings were suggestive of hepatic cytolytic features, but ALP values were not available.
Normalized values provided by author and rough parameters not available in U/L.
Ratio provided by author.
When TBL values were not available, but patient had presented jaundice.
Mean was calculated based on reports which included AST, ALT and ALP in U/L.
Abbreviations: TBL: serum total bilirubin; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; ANA: antinuclear antibody titres; ASMA: anti-smooth muscle antibody titres; anti-LKM-1: anti-liver/kidney microsomal antibody type 1; AMA: antimitochondrial antibody; N/A: not available; nHy: new Hy's law criteria were applied, where nR>5+TBL>2xULN, and nR was calculated as (ALT or AST (whichever was highest)/ULN)/(ALP/ULN); ULN: upper limit of normal.
Most patients were female (86%), with a mean age of 40 years (range 11–74 years). The primary indications for MP therapy were exacerbated episodes of demyelinating diseases (29 cases of MS), followed by Graves' ophthalmopathy (GO) (13 cases).
MP was mainly administered intravenously (96%). The cumulative MP doses ranged from 0.2 to 15 g, and the average MP treatment duration was 22 days (range 3–136 days), with a mean duration of 5 days for demyelinating disorders and 47 days for GO.
Twenty-one patients presented weakness, generalised malaise, nausea, abdominal pain and pruritus. Jaundice was a common feature in symptomatic patients (76%). In addition, 21 patients were asymptomatic, and the diagnosis of liver injury was based on a routine blood test. No information regarding symptoms was available for the remaining eight cases. The mean time to symptoms onset was 6 weeks (range 1–20 weeks) (Table 1).
The mean values for peak liver tests were as follows: TBL 6.5 mg/dL (range, 0.8–24 mg/dL), AST 874 U/L (range, 39–3315 U/L), ALT 1393 U/L (range, 122–3768 U/L) and ALP 181 U/L (range, 80–498 U/L). Sixteen cases presented with positive autoantibody titers (36%). Of those cases, eight had ANA titers, three cases ASMA titers and one case had anti-LKM-1 titers. Three additional cases presented simultaneously with ANA and ASMA titers, and one case had positive AMA and ANA autoantibodies. In 29 cases autoimmunity titers were negative, while autoantibody information was not available in five cases (Table 2).
Liver biopsy information was available for 32 (64%) patients. The primary findings were necrosis in 22 cases and apoptotic changes in two cases (Table 1). Interestingly, the pattern of liver injury, based on biochemical parameters, was hepatocellular in all cases with available information (n = 39). Seventeen of 22 patients for whom sufficient information was available met new Hy's (nHy) law criteria (Table 2).
Most MP-DILI patients recovered (94%), but four patients died. The mean time to resolution was 16 weeks (range, 1–154 weeks), and in the four fatal cases, the mean time to exitus was 5 weeks (range, 2–7 weeks). Rechallenge was observed in 19 cases, and in eight of those re-exposure to MP occurred more than two times. MP was the only administered drug in 26 cases; 18 cases had other associated medications on long-term use or with a low hepatotoxic potential. In six cases no information was provided (Table 1).
Discussion
MP is an unexpected cause of DILI, in fact, it is used as treatment of choice for severe hepatitis, as well as liver disease with autoimmune features, due to its anti-inflammatory and immunosuppressive activities. 20 However, our case series and review of the literature indicate that intravenous high-dose courses of MP can induce severe hepatitis that recurs with repeated administration of the drug and, occasionally, can lead to a fatal progression.24,26
Our analysis shows that MP hepatotoxicity can be severe. All four fatal cases were diagnosed with GO relapse (31% lethality within the GO subgroup) and were exposed to higher cumulative MP doses (mean 12 g) compared to the remaining patients (mean cumulative MP dose 2 g (range, 0.18–10.7 g)). Seventeen of the earlier reported events (81%), as well as two of our cases (patients 1, 3) met nHy's law criteria, defined as hepatocellular injury (new ratio, nR ≥ 5) calculated as ALT or AST (whichever was highest/upper limit of normal, ULN)/(ALP/ULN) accompanied by total bilirubin > 2 x ULN. 2 In the majority of cases analyzed in this study, MP was administered to treat exacerbations of an underlying autoimmune disease. Although MP is more commonly used in patients with indications other than autoimmune disorders (e.g., asthma, COPD exacerbations and medullar compression), there are no reported cases of MP-DILI when used in these indications, which suggests that patients with an underlying autoimmune disease, particularly MS or GO, have a higher risk of developing MP-DILI. Indeed, a recent prospective observational single-centre study by Nociti et al. (2018)37 conducted in 175 MS patients treated with i.v. MP pulses showed an 8.6% prevalence of liver injury. Together, these observations highlight that MP-DILI may be a growing concern in the management of patients with autoimmune disorders and a new challenge in the field of hepatotoxicity.
It is worth noting that patients 11, 14, 43 and 48 responded favourably to dexamethasone therapy, with no subsequent flares in transaminases or only mild liver dysfunction (case 43). Thus, dexamethasone might be a safe glucocorticoid alternative in patients with a previous episode of MP-DILI.13,32
Out of the 50 reported cases, 16 patients had positive autoantibody titers as did one of our cases. However, most likely positive autoantibodies in these cases were one of the features of the underlying autoimmune disease rather than of the DILI episode. 35
In five of the reviewed MP-DILI cases, a diagnosis of AIH triggered by MP was established (cases 18, 20, 21, 25 and 33). In cases 20, 25 and 33, baseline liver tests were within normal ranges, making less probable an underlying non-diagnosed AIH.
The adaptive immune response is believed to play an important role in the mechanism of DILI. 35 Steroids exert a potent immunosuppressive effect and have often been empirically used in severe forms of liver injury. Hence, one may hypothesize that the MP-induced transient immunosuppression, followed by an immune reconstitution, could awake an autoimmune-like reaction in a susceptible host. This condition may then unmask AIH in predisposed patients.
Once a diagnosis of liver damage was made in cases 20, 25 and 33, therapy was switched to oral corticoids (prednisolone in case 20 and prednisone in the other cases). In case 18, treatment with azathioprine and oral prednisone followed the AIH diagnosis. However, not all cases had this type of progression.
Furthermore, the time to resolution lasted 9 and 18 weeks for cases 25 and 33, respectively, and remained unknown in case 18, although the authors described transaminase normalization within 2 weeks after starting immunosuppressant treatment. Similarly, for the remaining MP-induced liver injury events, the mean time to spontaneous recovery was 15 weeks (range, 1–154 weeks). Thus, the profile of serum liver enzyme recovery and the time to resolution suggest an independent course from the immunosuppressive therapy administered. It is difficult to distinguish MP-induced liver injury from AIH unmasked by MP. Nevertheless, there is a well-defined timeline between the treatment initiation with MP and the development of liver injury, with a mean time to onset of 6 weeks (range, 1–20 weeks). Hence, the relationship between liver injury and intake of MP is undeniable.
Another aspect of interest in MP-DILI is the high rate of rechallenge episodes. It is already known that positive rechallenge due to a suspected causative agent strengthens causality in DILI. 36 In the published cases 38% had a positive rechallenge. Most of our patients had positive re-exposure to MP, which led to a new ALT flare. Most likely, the frequent rechallenge to MP occurs due to lack of awareness of MP hepatotoxicity potential. The elevated numbers of re-exposure, as well as the fact that fatal progression has been described in MP-DILI, are concerning. Thus, an accurate diagnosis of MP-DILI is key for an early discontinuation of the drug and to prevent rechallenge.
In conclusion, MP-induced liver injury, which remains a rare cause of hepatotoxicity, mainly presenting with hepatocellular injury, should be considered in patients having an underlying autoimmune disorder, especially MS and GO, who receive high pulsed i.v. doses of the drug in a short period of time. Liver injury appears frequently during the first weeks after treatment initiation and the severity ranges from asymptomatic or mild to severe and even fatal. Patients presenting an underlying autoimmune disease require a closer follow-up and liver monitoring during and after high-dose MP therapy.
Footnotes
Author contributions
Conceptualization: RJA, MRD; investigation and data analysis: MEZ, JPB, MRD, AOA, NH, JC, FC, IMC, JSC, RSJ, AGJ, MGC; writing: MEZ, JPB, MRD, MIL, RJA.
Declaration of conflicting interests
None declared.
Ethics approval
This study was reviewed and approved by the Ethical Review Board H U Virgen de la Victoria 9 July 2010.
Funding
This work was supported by ISCiii-Fondos FEDER (PI18-01804, PI18-00901, PT17/0017/0020, JR16/00015, CM17/00243); CS-SAS (PI-0274/2016, PI-0285-2016); and AEMPS.
Informed consent
All three DILI patients provided informed consent prior to study enrolment.
