Abstract
Introduction
Only a minority of patients with a positive fecal occult blood test (FOBT) undergo a follow-up second diagnostic procedure, thus minimizing its contribution for colorectal cancer (CRC) prevention. We aimed to obtain a precise estimation of this problem and also assess the diagnostic yield of CRC and adenomas by colonoscopy in these patients.
Methods
Literature searches were conducted for “compliance” OR “adherence” AND “fecal occult blood test” OR “fecal immunohistochemical test” AND “colonoscopy.” Comprehensive meta-analysis software was used.
Results
The search resulted in 42 studies (512,496 patients with positive FOBT), published through December 31, 2017. A funnel plot demonstrates a moderate publication bias. Compliance with any second procedure, colonoscopy, or combination of double-contrast barium enema with or without sigmoidoscopy in patients with a positive FOBT was 0.725 with 95% confidence interval (CI) 0.649–0.790 (
Discussion
Compliance with diagnostic evaluation after a positive FOBT is still suboptimal. Therefore, measures to increase compliance need to be taken given the increased risk of CRC in these patients.
Current knowledge
Worldwide, colorectal cancer (CRC) screening programs have been implemented and resulted in CRC incidence and mortality reduction. Choosing fecal occult blood test (FOBT) as a screening strategy requires subsequent intervention, usually by full colonoscopy examination. Only a minority of positive FOBTs are followed by an adequate evaluation, a suboptimal condition that undermines the benefits of undergoing the screening test itself.
New findings in this study
Compliance rates with any second procedure, colonoscopy, or combination of double-contrast barium enema with or without sigmoidoscopy in patients with a positive fecal occult blood test (FOBT) are 0.725, 0.804 and 0.197, respectively. Worldwide, over the past 25 years, the compliance rate for positive FOBT has improved. Still, incomplete follow-up after positive FOBT has been and remains a major problem. Measures need to be taken in each national screening program to increase this specific compliance rate thus reducing the incidence and mortality rates from colorectal cancer.
Introduction
Colorectal cancer (CRC) is the second leading cause of death from malignancies in developed countries. In the United States (US), about 50,000 people die from the disease each year. 1 Fortunately, however, in contrast to other malignancies, CRC can be prevented when premalignant polyps are removed following their detection by an efficient screening. Therefore, CRC screening programs have been implemented worldwide in the last two decades, 2 resulting in a steady reduction in the incidence and mortality from CRC. 3 Despite these beneficial effects, only 60% to 65% of people older than 50 years actually participate in CRC screening. 4 There are multiple screening modalities. As opposed to the European guidelines on CRC screening that recommend screening by fecal immunochemical test (FIT), recently, based on the results of a modeling study the US Preventive Services Task Force updated its guidelines and listed eight optional screening strategies. The purpose of this update was to encourage testing, assuming that offering a variety of screening programs would make it easier for patients to choose their preferred program, thus improving compliance rates. The problem is that improvement in participation rate of CRC screening is not enough. Choosing fecal occult blood test (FOBT) as a screening strategy requires subsequent intervention, usually by full colon examination. 3 However, according to several studies, only 25% 5 to 55% 6 of positive FOBTs are followed by an adequate evaluation, suggesting a suboptimal condition. Failure to complete an appropriate evaluation after a positive result can undermine the benefits of undergoing the screening test itself and minimize its potential contribution to the reduction of incidence and mortality. While many studies had already investigated the compliance rate for colorectal screening with FOBT, the second compliance rate, for the colonoscopy itself, in patients with a positive FOBT, has never been thoroughly studied. Therefore, to obtain a precise estimation of this problem, our study represents the first quantitative meta-analysis of the compliance rate for the second diagnostic evaluation after a positive FOBT (primary aim). In addition, we explored the diagnostic yield of CRC and adenomas in patients with a positive FOBT (intention-to-screen analysis) by colonoscopy (per protocol analysis) (secondary aim).
Methods
Preferred reporting items for systematic reviews and meta-analyses guidelines were strictly followed. 7 A local protocol for conducting a meta-analysis was applied based on previous descriptions. 8 A data-extraction table was used (available on request).
Data sources and searches
We conducted a comprehensive search of PubMed, MEDLINE, Scopus, EMBASE, and CENTRAL through December 31, 2017. We used the following keywords and medical heading terms: “compliance” OR “adherence” AND “fecal occult blood test” OR “fecal immunohistochemical test” AND “colonoscopy.” Manual searches of articles’ bibliographies were also performed. Only fully published, controlled studies in English were included. The exclusion criteria were duplications, review articles, mathematical models, opinion papers and editorials, high-risk patients or screening of other malignancies, data presented in percentages, and articles based on questionnaires or not related to screening.
Study selection
Three authors (R.G.B., H.L. and D.B.) independently reviewed the pertinent studies to determine their eligibility. Only studies that met all the following criteria were included: reported the precise compliance rate of second diagnostic evaluation of patients with a positive FOBT, reported which diagnostic evaluation was applied, evaluated population without risk factors for CRC, and evaluated adult patients older than 18 years.
Data extraction and quality assessment
Summary of included studies.
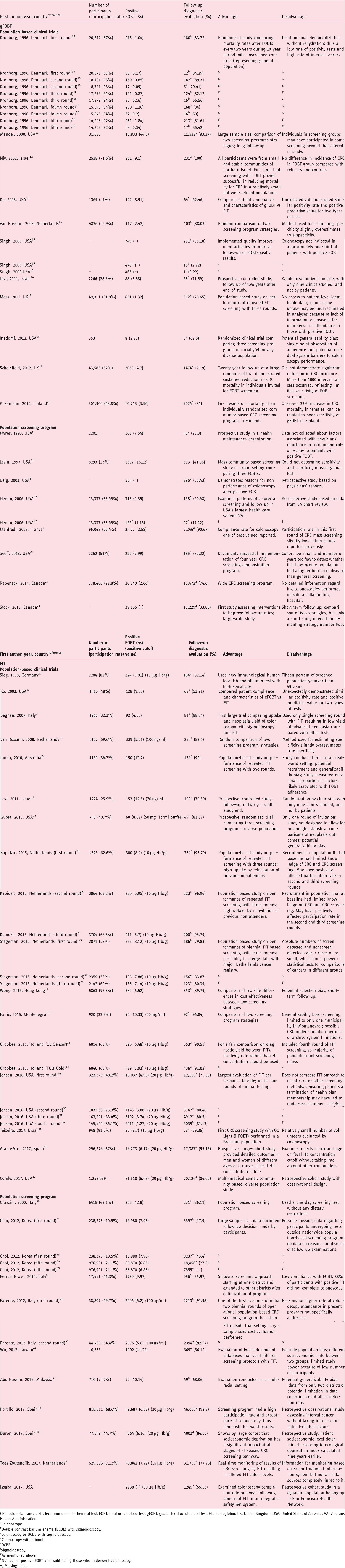
CRC: colorectal cancer; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; Hb: hemoglobin; UK: United Kingdom; USA: United States of America; VA: Veterans Health Administration.
Colonoscopy.
Double-contrast barium enema (DCBE) with sigmoidoscopy.
Colonoscopy or DCBE with sigmoidoscopy.
Colonoscopy with albumin.
DCBE.
Sigmoidoscopy.
As mentioned above.
Number of positive FOBT after subtracting those who underwent colonoscopy.
, Missing data.
Data synthesis and analysis
Meta-analysis was performed by using Comprehensive meta-analysis software (Version 3, BioStat Inc, Englewood, NJ, USA). Pooled odds ratios (ORs) and 95% confidence intervals (CIs) were calculated for the number of patients with each pathological diagnosis. Heterogeneity between studies was evaluated using the Cochran
Our secondary outcome measures were the diagnostic yield of CRC, advanced adenoma and simple adenoma, defined as the proportion of participants undergoing colonoscopy after a positive FOBT and diagnosed with CRC, advanced adenoma or simple adenoma, relative to the number of participants with a positive test result, respectively.
Results
Search strategy and selection studies are shown in Figure 1. Database search identified 245 studies. Ten additional studies were identified after manual searches of articles’ bibliographies. We identified 255 articles. Fourteen articles were excluded because they were studies not in full text, not in English or duplications. A total of 199 studies were excluded based on abstract text: review articles, mathematical models, high-risk patients, data in percentage, opinion papers and editorials, screening of other malignancies, not related to screening, or based on questionnaires.
Flowchart of articles identified for the meta-analysis.
In total, we were left with 42 studies that fulfilled the inclusion criteria and were published through December 31, 2017 (Figure 1). The analysis itself included 69 substudies/data sets, which included 512,496 patients from 17 countries (US: 13, Spain: 3, Brazil: 1, Canada: 2, Denmark: 1, Germany: 1, Italy: 4, Israel: 2, France: 1, Netherland: 5, Finland: 2, Australia: 1, United Kingdom: 2, Korea: 1, Taiwan: 1, Montenegro: 1, and Malaysia: 1). Funnel plot demonstrates a moderate publication bias. Egger test Funnel plot for publication bias.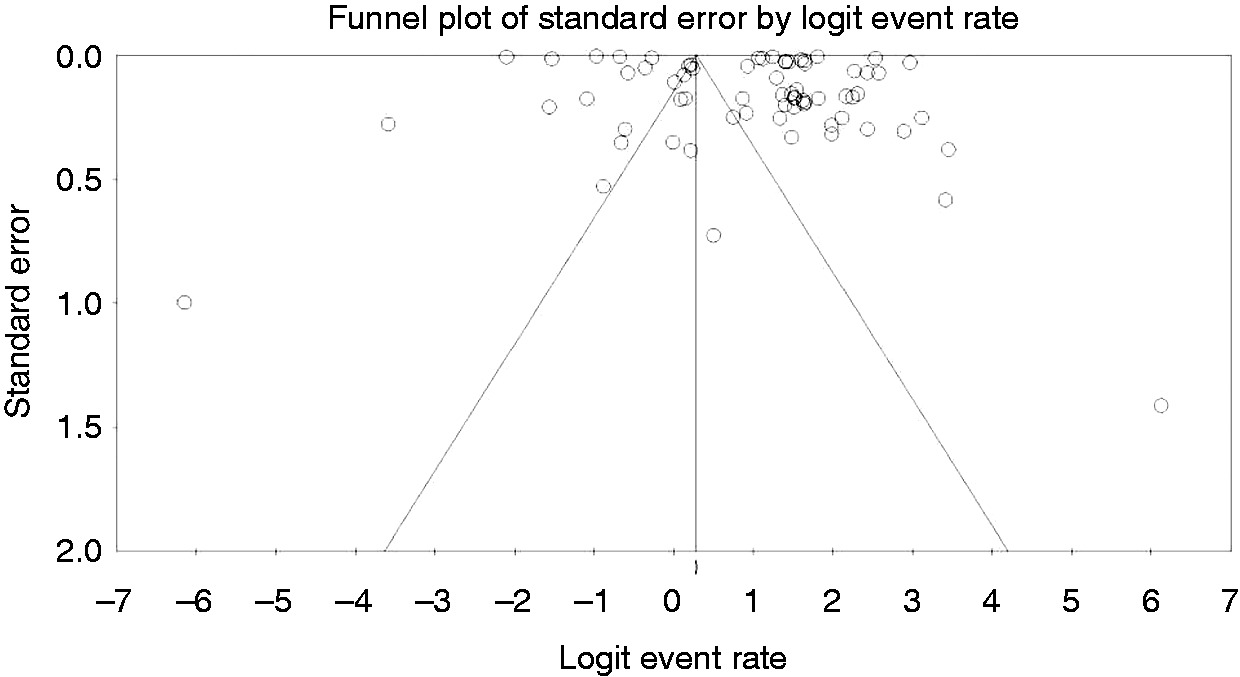
Therefore, a random-effects model was used in our meta-analysis. The event rate for the number of patients with a positive FOBT out of all screening participants was 0.0591 with 95% CI 0.059–0.059 (
The compliance rate for any second procedure, for colonoscopy, or for combination of DCBE with or without sigmoidoscopy in patients with a positive FOBT was 0.725 with 95% CI 0.649–0.790 ( Meta-analysis of descriptive studies looking at the compliance of patients with a positive FOBT for second procedure (all types): 42 studies (69 substudies/data sets); 302,197 procedures performed in 512,496 patients with a positive FOBT. CI: confidence interval; DCBE: double-contrast barium enema; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; UK: United Kingdom; USA: United States of America.
The compliance rate for any second procedure in patients with a positive FOBT after classification of the studies according to their publication year (at intervals of 10 years) was 0.701 with 95% CI 0.562–0.810 ( (a) Meta-analysis of descriptive studies published 1993–2002, looking at the compliance of patients with a positive FOBT for second procedure: seven studies (16 substudies/data sets); 13,665 procedures performed in 17,204 patients with a positive FOBT. (b) Meta-analysis of descriptive studies published 2003–2012, looking at the compliance of patients with a positive FOBT for second procedure: 15 studies (25 substudies/data sets); 48,917 procedures performed in 187,509 patients with a positive FOBT. (c) Meta-analysis of descriptive studies published 2013–2017, looking at the compliance of patients with a positive FOBT for second procedure: 20 studies (28 substudies/data sets); 239,615 procedures performed in 307,783 patients with a positive FOBT. CI: confidence interval; DCBE: double-contrast barium enema; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; USA: United States of America.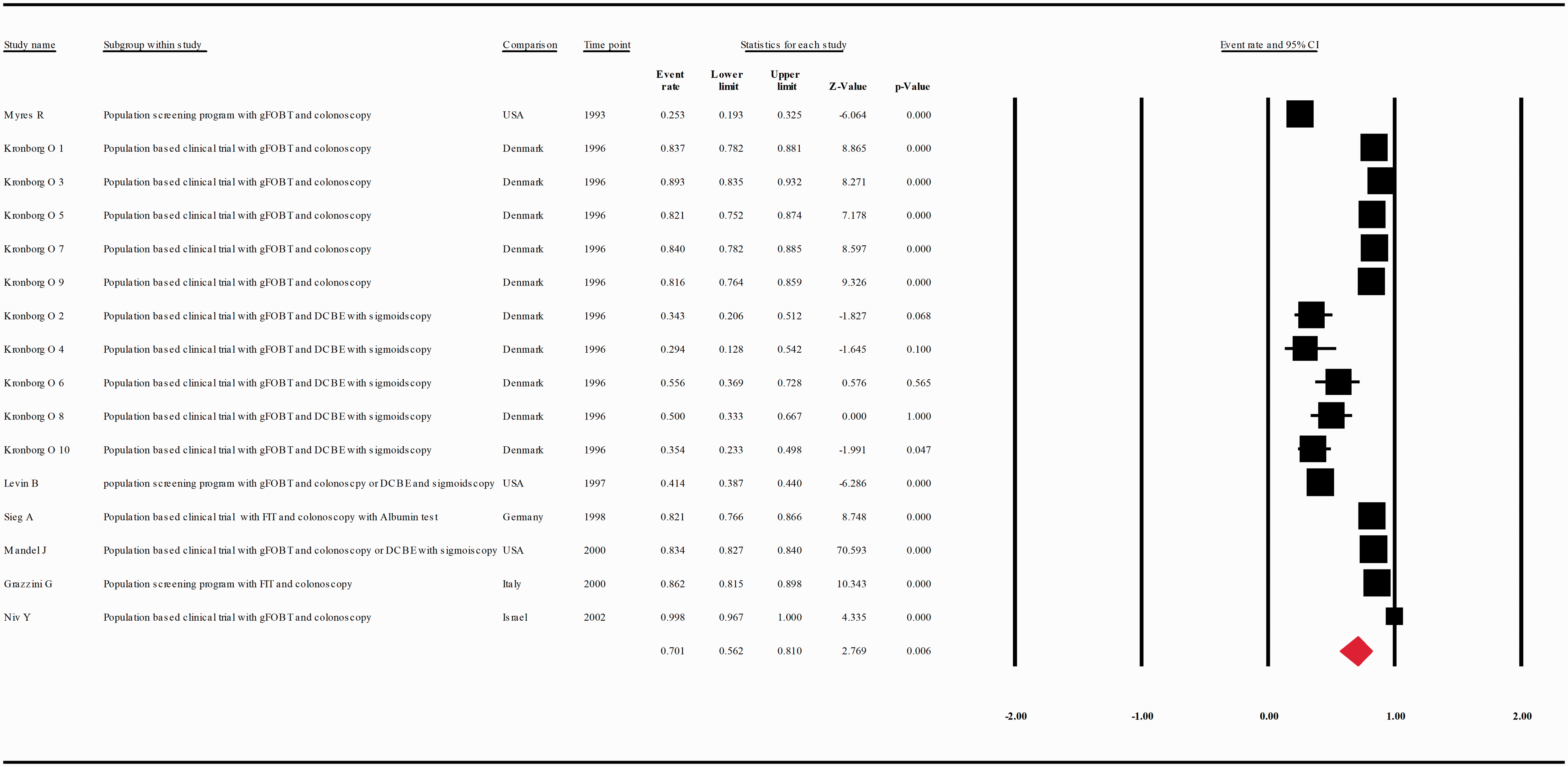
When data were analyzed according to the screening test used, the compliance rate for any second procedure in patients with a positive guaiac FOBT (gFOBT) was 0.599 with 95% CI 0.489–0.701 ( (a) Meta-analysis of descriptive studies, looking at the compliance of patients with a positive gFOBT for second procedure: 19 studies (31 substudies/data sets); 56,393 procedures performed in 95,752 patients with a positive FOBT. (b) Meta-analysis of descriptive studies, looking at the compliance of patients with a positive FIT for second procedure: 26 studies (38 substudies/data sets); 245,804 procedures performed in 416,744 patients with a positive FOBT. (c) Meta-analysis of population-based clinical trials studies, looking at the compliance of patients with a positive FOBT for second procedure: 25 studies (46 substudies/data sets); 142,965 procedures performed in 169,941 patients with a positive FOBT. (d) Meta-analysis of population screening programs studies, looking at the compliance of patients with a positive FOBT for second procedure: 18 studies (23 substudies/data sets); 159,232 procedures performed in 342,555 patients with a positive FOBT. CI: confidence interval; DCBE: double-contrast barium enema; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; UK: United Kingdom; USA: United States of America.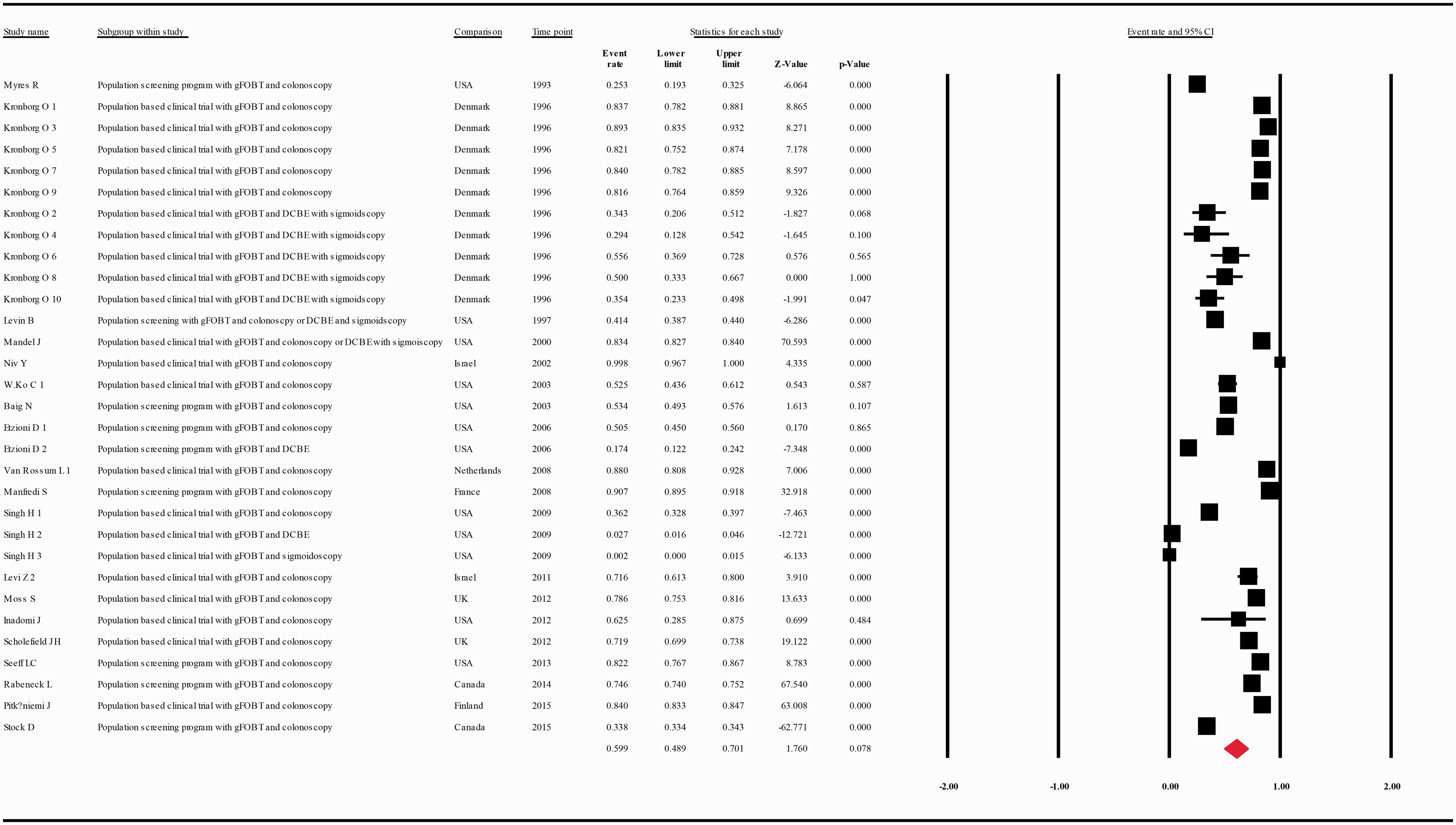
When we analyzed the results of population-based clinical trials studies separately from the results of population screening programs studies, the compliance rate for any second procedure in patients with a positive FOBT was 0.836 with 95% CI 0.834–0.838 (
Heterogeneity (the proportion of inconsistency in individual studies) between studies was significant with
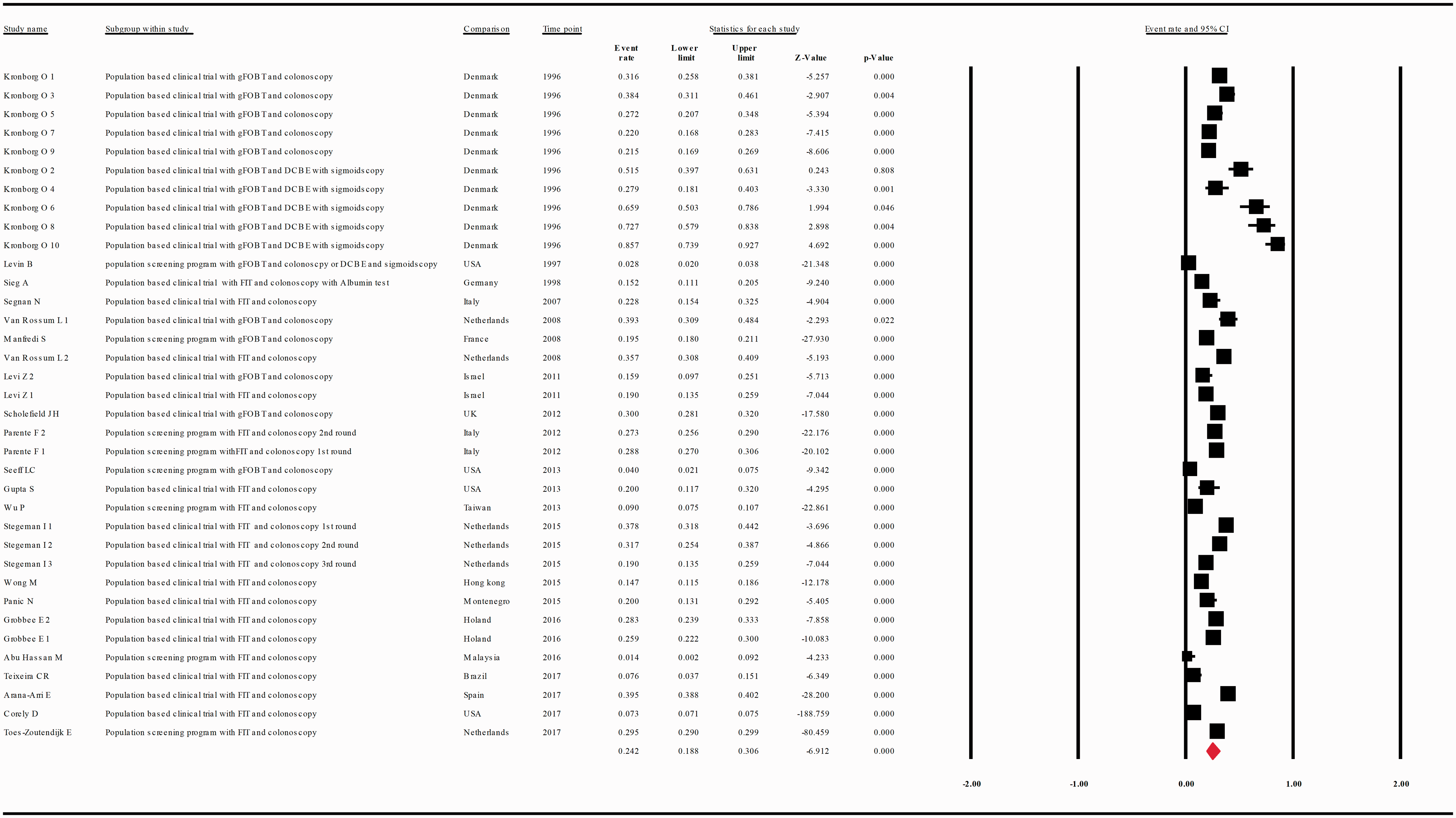
The diagnostic yield of CRC, advanced adenoma and simple adenoma, by colonoscopy, was provided in 30, 21 and 15 studies, respectively, and in 50, 36 and 18 substudies, respectively. Thus, the diagnostic yield for CRC, advanced adenoma and simple adenoma in patients with a positive FOBT (intention-to-screen analysis) was 0.058 with 95% CI 0.050–0.068( Meta-analysis of descriptive studies looking at the diagnostic yield of CRC: 30 studies (50 substudies/data sets); 11,591 cases in 269,149 patients with a positive FOBT. CI: confidence interval; DCBE: double-contrast barium enema; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; USA: United States of America. (a) Meta-analysis of descriptive studies looking at the diagnostic yield of advanced adenoma: 21 studies (36 substudies/data sets) 29,140 cases in 157,158 patients with a positive FOBT. (b) Meta-analysis of descriptive studies looking at the diagnostic yield of simple adenoma: 15 studies (18 substudies/data sets); 9716 cases in 51,913 patients with a positive FOBT. CI: confidence interval; DCBE: double-contrast barium enema; FIT: fecal immunohistochemical test; FOBT: fecal occult blood test; gFOBT: guaiac fecal occult blood test; UK: United Kingdom; USA: United States of America.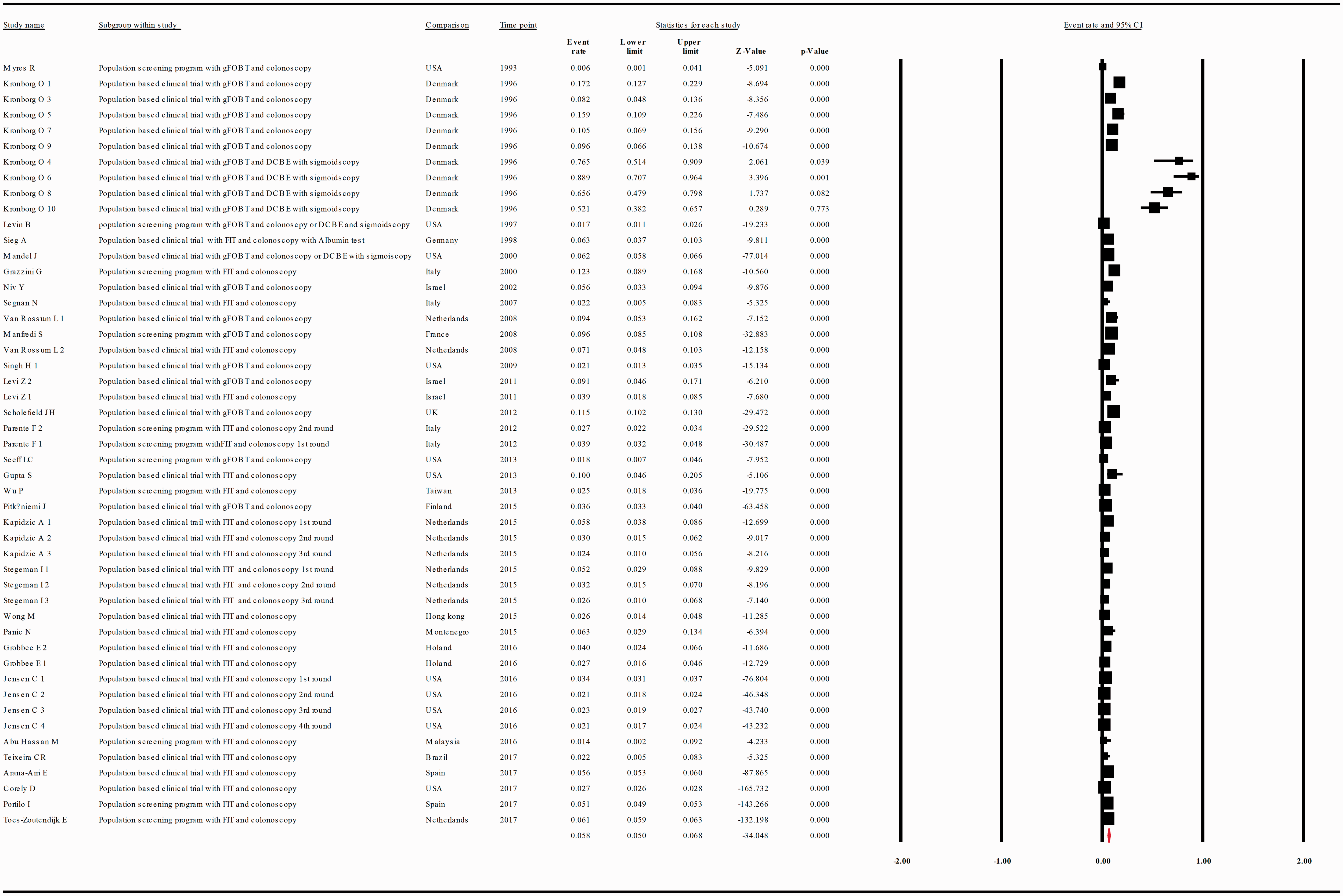
When the compliance rate for colonoscopy and for DCBE, in patients with a positive FOBT, was compared, we found an OR of 2.996, in favor of colonoscopy, with 95% CI 0.571–15.729, (
Discussion
There has been continuous debate over many years among gastroenterologists and public health experts regarding the best way to screen the average-risk population for prevention and early detection of CRC. Following the first randomized, controlled studies of screening with FOBT and many cost-effectiveness studies lacking studies reporting on screening with colonoscopy, the scientific community was convinced that annual FOBT during 25 years is comparable to colonoscopy every 10 years. 47 There is no debate, though, that colonoscopy should be the second procedure in patients with a positive FOBT. Many studies examined the compliance rate for screening with FOBT or colonoscopy as the first procedure, but the second compliance rate for the colonoscopy itself in patients with a positive FOBT has never been thoroughly studied. Therefore, our systematic review and meta-analysis is the first that provides a precise, quantitative analysis of the compliance rate for colonoscopy, or for DCBE with or without sigmoidoscopy, after a positive result from fecal CRC screening tests.
In our meta-analysis, a positive FOBT was found in 5.91% of the participants. The compliance rate for colonoscopy, or for DCBE with or without sigmoidoscopy, after a positive test result was 80.4% and 19.7%, respectively. In addition, consistent with the reported rate in a study by Yabroff et al., 48 we have found that the calculated compliance rate for any second diagnostic evaluation was 72.5%. Thus, about 30% of participants with a positive FOBT did not undergo a second diagnostic evaluation and were at risk of having CRC. For example, Meester and colleagues, by using a microsimulation model of an average-risk screening cohort, estimated that a delay of 12 months in performing colonoscopy after a positive FIT can produce an increase of 4% in incidence of CRC and 16% in mortality. 49 They did not include patients who refused colonoscopy, who are probably at higher risk. Meester’s estimation is supported by the work of Corley et al., which is based on a retrospective cohort study of Kaiser Permanente Northern California and Southern California health plan members showing that, compared with colonoscopy performed during the first 30 days, after a positive FIT result, each additional 30-day delay is associated with an average increased risk of approximately 3% for any CRC, and approximately 5% of advanced-stage disease, excluding follow-up examinations performed, within six months. 37
When we calculated the compliance rate for any second procedure in patients with a positive FOBT, after classification of the studies according to their publication year (at intervals of 10 years) we found that between the first 10 years (1993–2002), it was 70.1%, while between the last five years (2013–2017) it was 84.1%, a result that can point at an improving trend in the last years, maybe due to an increase in the awareness of patients and clinicians of the importance of adherence to positive screening tests. 1
When we separated the studies according to the screening test used, the calculated compliance rate for any second diagnostic evaluation was 59.9% for gFOBT and 80.6% for FIT.
In our study, meta-analysis of the studies according to type revealed that the calculated compliance rate for any second diagnostic evaluation of the population-based clinical trials studies was 22% higher than that of the population screening programs studies (83.6% vs 61.6%). This result can be explained by situations that can happen more often in screening programs and less frequently in controlled clinical trials, for example, absence of physician recommendation for further follow-up, incomplete communication of results between providers. 48
It should be emphasized that the problem of incomplete diagnostic evaluation after screening programs has also been reported in screening for other types of cancer. 48 In our meta-analysis, the diagnostic yield of CRC in patients with a positive FOBT was 5.8%. This result is 10-fold higher than that of screening colonoscopy among the average-risk population 50 and reflects a significant number of patients who are at risk. Interestingly, in our meta-analysis, contrary to the adenoma diagnostic yield reported in screening colonoscopies (average-risk population),23,50 the diagnostic yield of advanced adenoma (containing severe/high-grade dysplasia, more than 20% villous component, or larger than 10 mm) was higher than that of simple adenoma (24.2% vs 14.7%). Since an advanced adenoma is associated with bleeding tendency more than a simple adenoma, this finding is expected and supports the effectiveness of screening by FOBT. The compliance rate for colonoscopy in patients with a positive FOBT was found to be almost 300% higher than that for DCBE.
Strengths of this meta-analysis reside in the inclusion of diverse studies from different countries and during a period of more than 20 years (1993–2017). Thus, our study emphasizes that incomplete follow-up after abnormal stool-based tests has been and remains a major worldwide problem that should be addressed.
The limitations of our study are the moderate heterogeneity among studies, the different FOBT brands used over the years and their performance’s heterogeneity, and the different approaches to second diagnostic evaluation. Nevertheless, our findings should alert health care providers to patients with a positive FOBT who do not undergo colonoscopy and are at risk of an undiagnosed CRC.
In conclusion, the second diagnostic evaluation after a positive FOBT is about 70%. Since at least 90% of the patients with a positive test should undergo diagnostic test to obtain an effective mass screening program, 21 an effort should be made in each national screening program to increase this specific compliance rate, thus maximizing the positional contribution to reduce the incidence and mortality rates of CRC.
Supplemental Material
Supplemental material for The compliance rate for the second diagnostic evaluation after a positive fecal occult blood test: A systematic review and meta-analysis
Supplemental Material for The compliance rate for the second diagnostic evaluation after a positive fecal occult blood test: A systematic review and meta-analysis by Rachel Gingold-Belfer, Haim Leibovitzh, Doron Boltin, Nidal Issa, Tsachi Tsadok Perets, Ram Dickman and Yaron Niv in United European Gastroenterology Journal
Footnotes
Acknowledgment
This study was presented as a poster at Digestive Disease Week, May 2018, in Washington, DC, USA, and was published as an abstract in
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
Approval from ethics committee was not required for the study.
Informed consent
Informed consent was not required for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
