Abstract
Objective
Fecal transferrin has been suggested as a complementary or even superior marker for early detection of colorectal cancer (CRC) besides fecal hemoglobin. We aimed to evaluate both markers individually and in combination in a large cohort of participants of screening colonoscopy.
Methods
Precolonoscopy stool samples were obtained from participants of screening colonoscopy and frozen at −80℃ until blinded analysis, using a dual-quantitative fecal immunochemical test (FIT) for hemoglobin and transferrin. Sensitivity, specificity and area under the curve (AUC) were calculated for CRC and advanced adenoma (AA).
Results
A total of 1667 participants fulfilled our inclusion criteria. All individuals with advanced neoplasm (AN) (16 CRC, 200 AA) and 300 randomly selected participants without AN were included. Mean age was 63 years and 56% were male. The AUC for CRC and AA was 92% and 68%, respectively, for hemoglobin vs. 79% and 58%, respectively for transferrin. Combination of both markers yielded an AUC for CRC and AA of 92% and 68%, respectively.
Conclusion
FIT for hemoglobin shows better diagnostic performance than FIT for transferrin for the detection of ANs (both proximal and distal neoplasms), and a combination of both markers does not improve the diagnostic performance.
Keywords
Key summary
Fecal immunochemical tests (FITs) for hemoglobin are widely recommended and used for colorectal cancer (CRC) screening. The sensitivity for detection for premalignant lesions, however, especially in the proximal part of the colon, is rather low. Transferrin, which is released together with hemoglobin through bleeding into the digestive tract, has been reported to be more stable in the fecal matrix than hemoglobin, and therefore was suggested to be a potentially more sensitive marker for early detection of colorectal neoplasms. It has furthermore been suggested that combinations of FITs with fecal transferrin might increase the sensitivity for detecting colorectal neoplasms, but evaluations of diagnostic performance among average-risk screening participants were not performed. FIT for hemoglobin showed better diagnostic performance than fecal transferrin, and a combination of both markers did not improve the diagnostic performance for the detection of advanced neoplasms in an average-risk screening population. FIT for hemoglobin showed substantially higher sensitivities than fecal transferrin for detecting both proximal and distal neoplasms.
Introduction
Colorectal cancer (CRC) is the third most common cancer globally, causing approximately 1.4 million new cases and 700,000 deaths per year. 1 Randomized trials have demonstrated that screening with guaiac-based fecal occult blood tests can reduce CRC mortality by up to 30%.2–4 Even larger effects should be possible with fecal immunochemical tests (FITs) for hemoglobin, which show better diagnostic performance5–7 and higher screening adherence rates in routine practice.8,9 Therefore, FITs are meanwhile widely recommended and used for CRC screening in many countries.10–12
However, to improve the sensitivity of FITs, especially for the detection of premalignant lesions of CRC, new approaches like the combination of FITs with other stool-based markers are under investigation. 13 Previous studies suggested that combining FITs for hemoglobin with fecal transferrin might increase sensitivity among symptomatic individuals.14–16 For example, Sheng et al. reported an increase of sensitivity for CRC and advanced adenoma (AA) from 75% to 90% and from 44% to 78%, respectively. 14 Transferrin is an iron-binding blood plasma protein that is released together with hemoglobin through bleeding into the digestive tract. Furthermore, transferrin has been reported to be more stable in the fecal matrix than hemoglobin, 17 and was suggested to be a more sensitive marker for the detection of colorectal neoplasms, especially in the proximal colon.
We aimed to evaluate and combine the diagnostic performance of hemoglobin and transferrin, and to assess the test performance regarding the detection of proximal and distal advanced neoplasms (ANs) in a large cohort of participants of screening colonoscopy.
Materials and methods
This article follows the Standards for Reporting of Diagnostic Accuracy statement 18 and the Fecal Immunochemical Tests for Hemoglobin Evaluation Reporting checklist. 19
Study design and study population
This study was conducted in parallel with a direct comparison of diagnostic performance of nine quantitative FITs, which has been reported in detail elsewhere. 20 Briefly, this study is mainly based on the Begleitende Evaluierung innovativer Testverfahren zur Darmkrebsfrüherkennung (BliTz) study, an ongoing prospective study among participants of the German screening colonoscopy program. Participants of screening colonoscopy are informed and recruited at a preparatory visit in one of 20 cooperating gastroenterology practices, typically one week before colonoscopy. Because of the low number of CRC cases in a true screening setting, an additional group of CRC patients was included who were recruited in the DACHSplus satellite substudy of the Darmkrebs: Chancen der Verhütung durch Screening (DACHS) study. In the DACHSplus substudy CRC patients were referred for surgery to one of four collaborating hospitals, where the patients were informed and recruited prior to initiation of any therapy.
Between 2005 and 2010, the participants of both studies received a 60 ml container for stool collection. These individuals were considered for this study.
Both studies have been approved by the ethics committee of the University of Heidelberg (BliTz study (178/2005): June 13, 2005 and DACHSplus study (310/2001): March 27, 2006) and by the State Chambers of Physicians of Baden-Wuerttemberg, Rhineland-Palatinate and Hesse. Both study protocols conform to the ethical guidelines of the 1975 Declaration of Helsinki (revised version from October 2000, Edinburgh) as reflected in a prior approval by each institution’s human research committee. Written, informed consent was obtained from each participant included in the study.
Figure 1 shows exclusion criteria and flow diagrams of the study participants. From the screening setting (BliTz study), all eligible 216 cases with AN (i.e. CRC or AA, defined as adenoma with high-grade dysplasia, tubulovillous or villous components, or adenoma ≥1 cm) and 300 randomly selected participants without AN (a mixed group of participants with nonadvanced adenoma, hyperplastic polyps and no findings at colonoscopy) were included. In addition, 50 patients with CRC from the DACHSplus study (clinical setting) were included. In total, 566 stool samples were analyzed in this study.
Flow diagram of selection of study participants of the screening setting (a) and of the clinical setting (b). AA: advanced adenoma; CRC: colorectal cancer.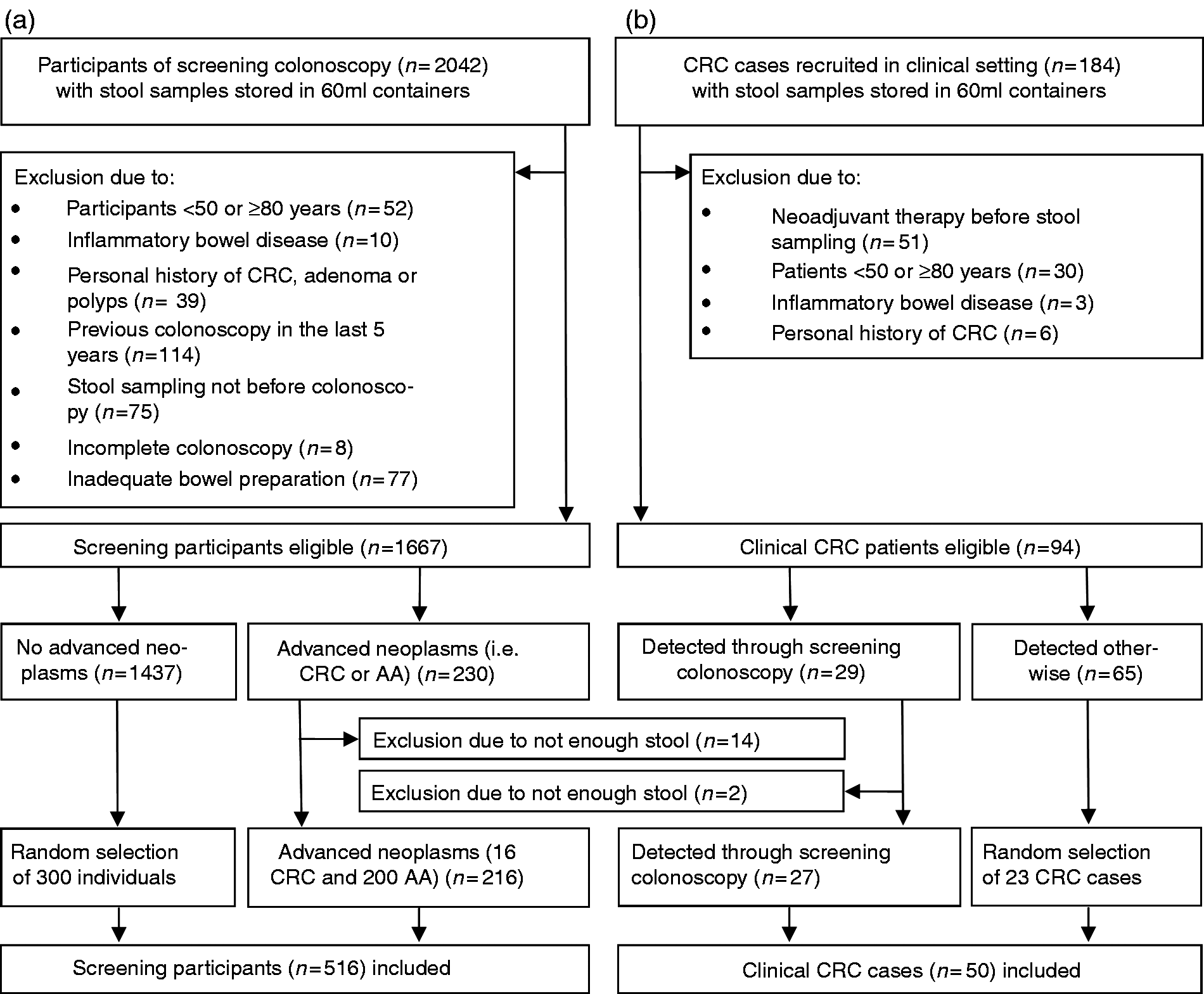
Sample and data collection
Study participants were asked to collect one stool sample from a single bowel movement, without any specific recommendations for dietary or medicinal restrictions, before starting bowel preparation for colonoscopy (screening setting) or initiation of therapy (clinical setting). Participants were asked to keep the stool-filled container frozen or, if not possible, refrigerated until their colonoscopy appointment (screening setting) or hospital admission (clinical setting). Upon receipt the containers were immediately frozen at −20℃ in the practice (screening setting) or in the hospital (clinical setting), then shipped on dry ice to a central laboratory and finally stored at −80℃ at the German Cancer Research Center (DKFZ).
Colonoscopists were blinded with respect to any test results. Colonoscopy and histology reports were collected from all participants of the screening colonoscopy. After surgery, medical reports on the clinical CRC patients were collected from the hospitals. Relevant information was extracted by two independent, trained research assistants who were blinded to the test results.
Laboratory analyses
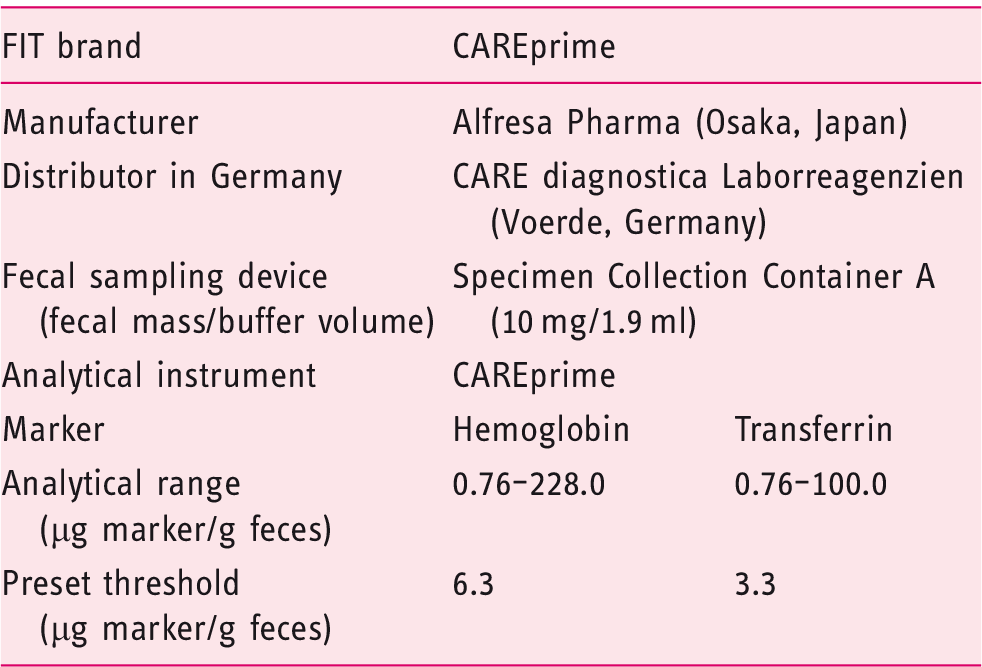
The stool samples were thawed overnight in a refrigerator at the DKFZ and homogenized before collecting a defined amount of stool using the specific fecal sampling device (FSD) of the manufacturer. This FSD was a small buffer-filled vial containing a serrated plastic stick for stool collection. After stabbing the collection stick into three different areas of the stool sample, it was checked if all serrations on the stick were completely filled with stool. Then the stick was inserted back into the vial. The vial had a tight entrance that removed excess stool, leaving only a defined mass of stool in the serrations of the stick.
Afterward, the stool-filled FSDs were mixed on a vortexer and shipped without cooling to the distributing cooperating company (CARE diagnostica) for blinded laboratory analyses. After arrival the FSDs were stored at a median temperature of 21.0℃ (range: 20.0℃–21.5℃) until test evaluation, which was performed within 24 hours after shipment and conducted in a one-time measurement.
Dual-quantitative fecal immunochemical test (FIT).
Statistical analyses
Sensitivities were calculated for CRC and AA. Specificities were determined for absence of any AN. Sensitivities and specificities were computed with their corresponding Clopper-Pearson 95% confidence interval (CI) and colonoscopy results were used as reference standard.
In addition, site-specific sensitivities were determined among participants who had only proximal ANs (located proximal to or at the splenic flexure) or only distal ANs (located in the descending colon, the sigmoid colon or the rectum).
To enhance comparability of diagnostic performance between tests, sensitivities and specificities were calculated not only at the positivity thresholds recommended by the manufacturer (6.3 µg hemoglobin/g feces and 3.3 µg transferrin/g feces, respectively), but also at positivity thresholds yielding 95% specificity for both tests.
Finally, receiver operating characteristic (ROC) curves were plotted and areas under the curve (AUCs) were computed for hemoglobin, transferrin and their combination, using logistic regression models. Because of the low number of CRC patients in the screening setting and very similar sensitivities between screening and clinical CRC cases, 20 the ROC plots for CRC were constructed combining all CRC cases (16 CRC cases from the screening setting and 50 CRC cases from the clinical setting).
All statistical analyses were conducted using SAS Enterprise Guide, version 6.1 (SAS Institute Inc, Cary, NC, USA).
Results
Study population
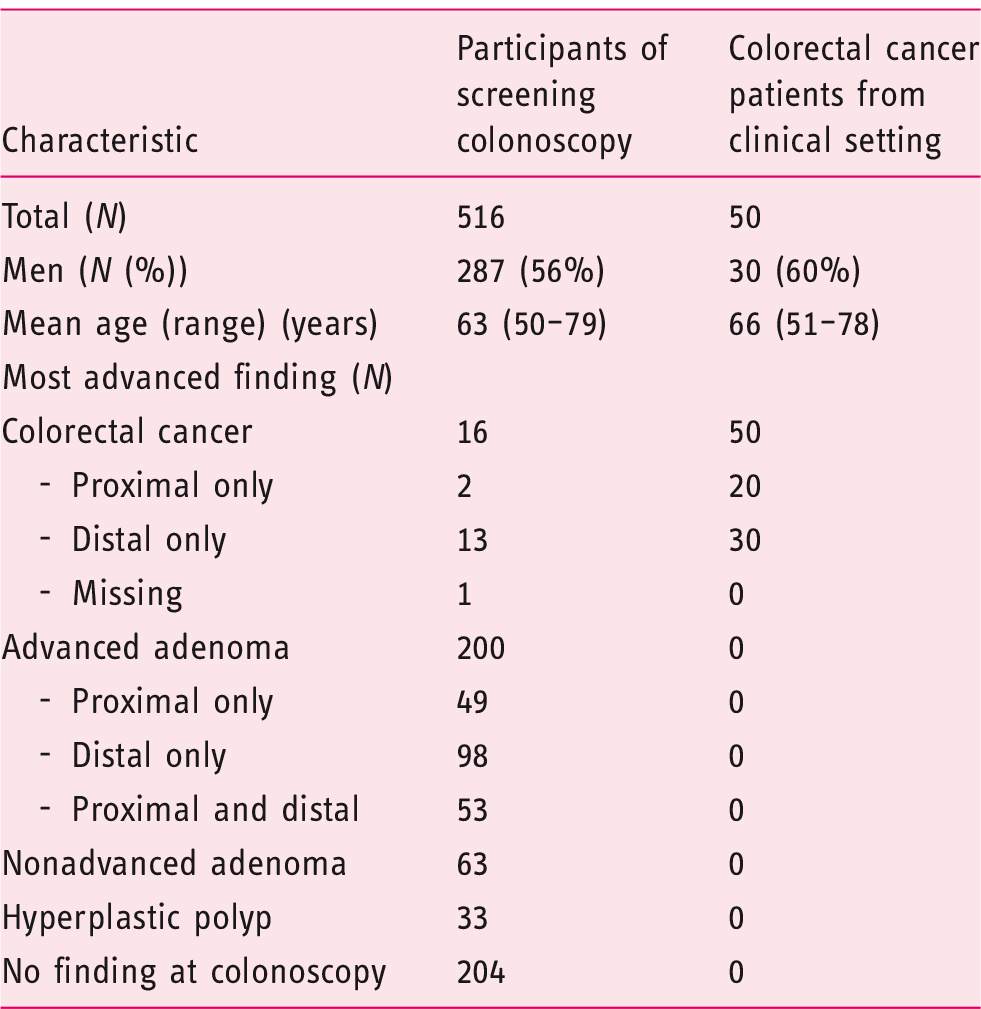
A total of 2042 participants of screening colonoscopy (BliTz study) were recruited between 2005 and 2010 and provided stool-filled containers (screening setting) (Figure 1(a)). After excluding 375 participants (exclusion criteria are shown in Figure 1(a)), 1667 eligible individuals were left. In total, 216 cases with AN (i.e. CRC or AA) and 300 randomly selected individuals without ANs were included from the screening setting.
From the DACHSplus study (clinical setting) (Figure 1(b)), a total of 184 CRC cases provided stool-filled containers. After excluding 90 participants (exclusion criteria are shown in Figure 1(b)), 94 CRC patients were eligible. In total, 50 patients with CRC from the clinical setting were included, of whom 27 were also diagnosed through a screening colonoscopy.
Study population.
Diagnostic performance
Sensitivity and specificity of hemoglobin and transferrin at preset and adjusted thresholds.
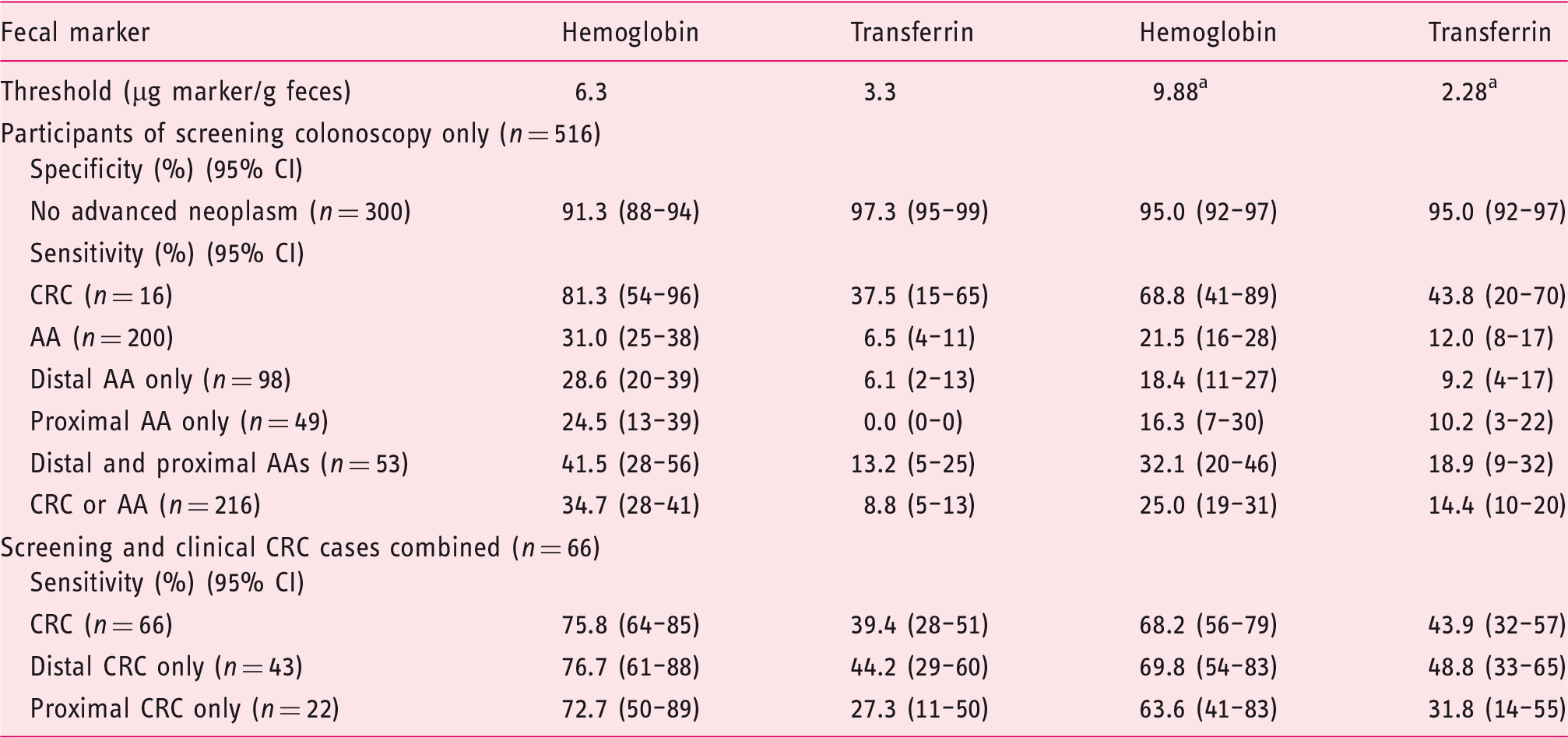
Adjusted threshold.
AA: advanced adenoma; CI: confidence interval; CRC: colorectal cancer.
At preset thresholds the sensitivities for hemoglobin were much higher than for transferrin. The sensitivities (95% CI) for all CRCs (
At adjusted thresholds, yielding the same specificity of 95.0% (95% CI, 92%–97%), sensitivity for hemoglobin was still much higher than for transferrin. The sensitivities (95% CI) for CRC and AA were 68.2% (56%–79%) and 21.5% (16%–28%), respectively, for hemoglobin and 43.9% (32%–57%) and 12.0% (8%–17%), respectively, for transferrin.
Furthermore, for hemoglobin, sensitivity was very similar for proximal and distal CRC (63.6% and 69.8%, respectively), whereas for transferrin the sensitivity for proximal CRC was substantially lower (31.8%) than for distal CRC (48.8%). The sensitivities (95% CI) for all AAs were 21.5% (16%–28%) for hemoglobin and 12.0% (8%–17%) for transferrin. The sensitivities for proximal and distal AAs only were very similar to the overall sensitivities of each marker (16% vs. 18% for hemoglobin and 10% vs. 9% for transferrin).
Figure 2 displays the ROC curves and AUCs for hemoglobin, transferrin and their combination. The AUCs for hemoglobin were consistently much higher than the AUCs for transferrin, and the combination with transferrin did not yield any further increase of the AUCs of hemoglobin. The AUCs (95% CI) for the detection of any CRC ( ROC curve for the detection of all CRC and AA, respectively, as well as for only proximal and distal ANs, respectively.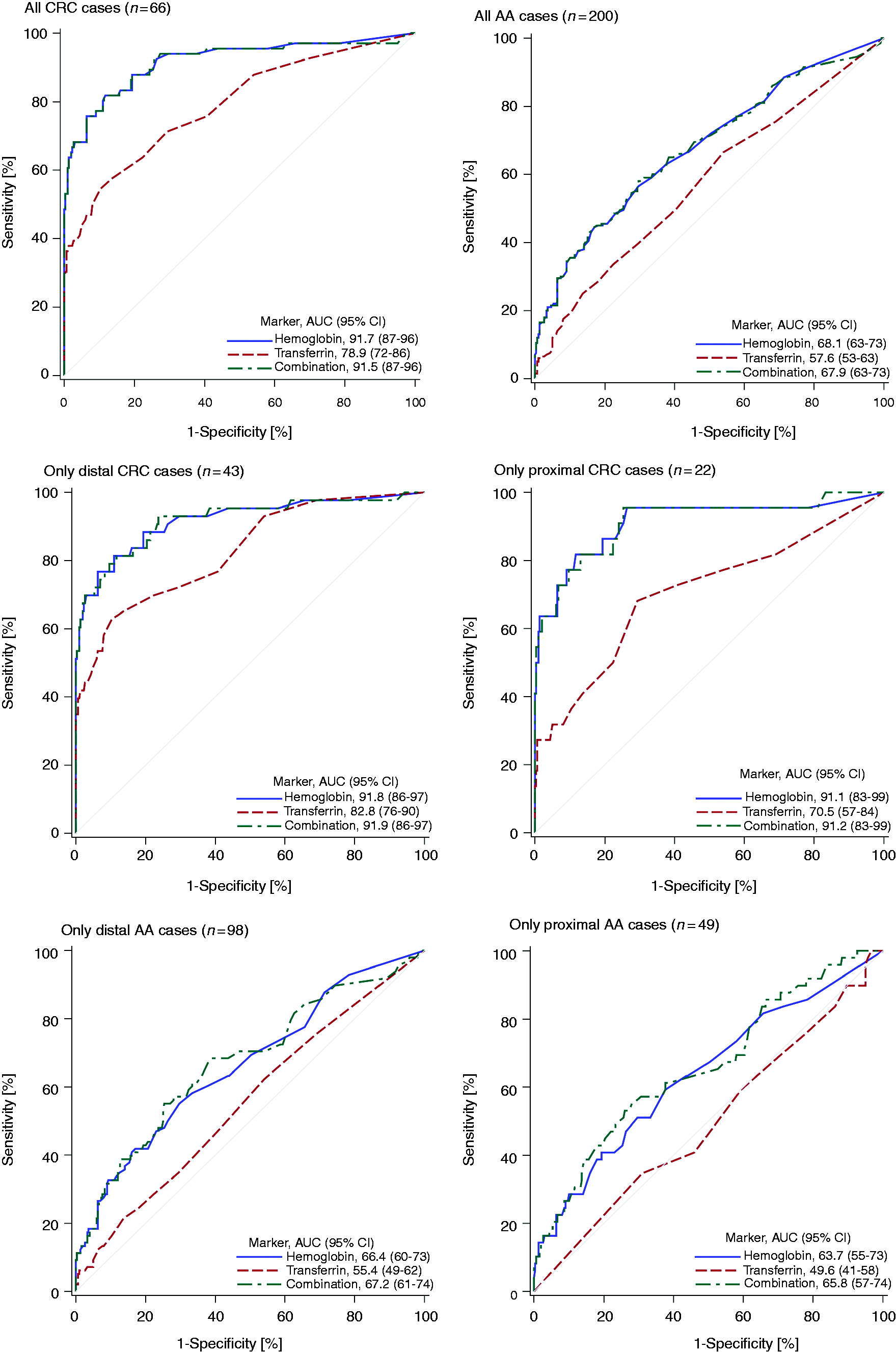
For hemoglobin, the AUCs for the detection of distal and proximal CRC were very similar (91.8% vs. 91.1%, respectively) (Figure 2, middle left and middle right). For transferrin the AUC for the detection of distal CRCs was much higher than for proximal CRCs (82.8% vs. 70.5%, respectively), but 95% CIs were overlapping. For AAs, no major site differences in ROC curves were observed for hemoglobin or for transferrin (Figure 2, bottom left and bottom right).
Discussion
We evaluated the diagnostic performance of fecal transferrin and its combination with FIT for hemoglobin in an average-risk screening population. FIT for hemoglobin had superior diagnostic performance compared to fecal transferrin, and even a combination of both markers did not improve diagnostic performance. The AUCs for CRC and AA were 92% and 68%, respectively for hemoglobin compared to 79% and 58%, respectively, for transferrin. Whereas fecal hemoglobin showed similar diagnostic performance for proximal and distal ANs, diagnostic performance of fecal transferrin was lower for proximal than for distal ANs.
Previous studies from China14–16 presented similar diagnostic performance of fecal hemoglobin and transferrin among symptomatic individuals at increased risk. In all three studies the combination of both markers led to an increase of sensitivity for CRC from 75% to 90%, 14 from 96% to 100%, 15 and from 57% to 86%, 16 respectively, but this went along with a similarly strong reduction of specificity from 88% to 71%, 14 from 72% to 61%, 15 and from 59% to 38%, 16 respectively. Because no AUCs were calculated, it was unclear whether the combination of both markers is able to improve the overall test performance, especially among average-risk screening participants.
Jin et al. 16 included an asymptomatic cohort of individuals, but only 251 participants with positive test results for either hemoglobin or transferrin were invited for an endoscopic examination. The detection rate for AA and CRC for hemoglobin, transferrin and their combination was 1.6%, 1.8% and 2.6%, respectively, but the higher detection rate for the combination went along with a higher positivity rate (15% vs. 8% and 11%) for the single tests. Because only positive-tested participants underwent colonoscopy, sensitivity and specificity could not be calculated and therefore it was essentially unclear whether a combination of both markers would improve test performance in a true screening setting. In our study among CRC screening participants at average risk, fecal hemoglobin clearly outperformed fecal transferrin as a diagnostic marker and a combination with fecal transferrin did not improve the test performance for CRC early detection.
In our study, the sensitivities for proximal compared to distal AAs were only slightly lower (24.5% vs. 28.6% at the threshold recommended by the manufacturer), and the same also applied to the AUCs (63.7% vs. 66.4%). Although de Wijkerslooth et al.
21
likewise observed FIT for hemoglobin to be similarly sensitive for the detection of proximal and distal ANs, in most of the studies evaluating site-specific diagnostic performance of FITs, the sensitivities for proximal compared to distal neoplasms were lower. 22–26 For example, in two of the largest FIT studies with more than 700 cases with ANs,22,25 significantly lower sensitivities for proximal than for distal AN were found (16% vs. 31%,
The main strength of our study is the first time evaluation of a dual-quantitative FIT in an average-risk CRC screening population, with screening colonoscopy performed independently of the FIT result in all participants. However, our study has also some limitations. First, stool samples were originally collected in 60 ml stool containers rather than with the FSD provided by the manufacturer and stored at −80℃ over several years prior to analysis. Nevertheless, the original FSD was used when collecting the fecal sample from the thawed stool, and prior homogenization of the thawed stool ruled out variation of hemoglobin and transferrin concentration within the same bowel movement. In a previous examination, we found only small differences in the diagnostic performance when the same FIT was conducted based on either frozen fecal samples or on fecal samples directly collected using the original FSD.
27
Second, despite the overall large size of the study, with targeted selection of samples from more than 1600 participants of screening colonoscopy, the number of CRC cases from the screening setting was still rather low (
Our study provides important information regarding the diagnostic performance of fecal transferrin in combination with FIT for hemoglobin. FIT for hemoglobin detects ANs in both the proximal colon and the distal colon and rectum more reliably than fecal transferrin, and a combination of both markers does not improve the diagnostic performance of FIT alone for the detection of CRC or AA. Further studies in average-risk screening populations are needed to identify markers or marker combinations that might enhance diagnostic performance of currently available screening tests.
Footnotes
Acknowledgments
The authors thank Sabine Eichenherr, Romana Kimmel and Ulrike Schlesselmann for their excellent work in laboratory preparation of stool samples. They also thank Volker Herrmann for his help in preparing the study.
Declaration of Conflict of Interest
The authors declare that there is no conflict of interest
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Written, informed consent was obtained from each participant included in the studies.
Ethics approval
Both studies have been approved by the ethics committee of the University of Heidelberg (BliTz study (178/2005): June 13, 2005 and DACHSplus study (310/2001): March 27, 2006) and by the State Chambers of Physicians of Baden-Wuerttemberg, Rhineland-Palatinate and Hesse. Both study protocols conform to the ethical guidelines of the 1975 Declaration of Helsinki (revised version from October 2000, Edinburgh) as reflected in a prior approval by each institution’s human research committee.
