Abstract
Background
The relationship between allergy and celiac disease (CD) is not clear.
Objective
The objective of this article is to investigate the association of CD and CD antibody positivity with hay fever, asthma and immunoglobulin (Ig)E sensitization in a general adult population.
Methods
A total of 2297 individuals were screened for CD antibodies and underwent allergy testing. CD antibody-positive participants were invited to undergo clinical evaluation including biopsies. Additionally, biobank blood samples from four population-based studies (6423, 973, 1718 and 1101 participants) with data on IgE sensitization to inhalant allergens were screened for CD antibodies. CD antibody-positive participants were screened for serum IgE against food allergens in three biobank studies. CD-antibody positivity was defined as IgA or IgG tissue transglutaminase ≥7 U/ml and/or IgG deamidated gliadin peptide ≥10 U/ml.
Results
The nine participants (0.4%) diagnosed with CD had significantly higher prevalence of IgE sensitization to wheat and dust mites. The prevalence of CD antibody positivity was 0.8% (18/2297), and these participants had a significantly higher prevalence of IgE sensitization to food allergens (Fx5), egg, dust mites and mugwort. In the biobank studies, the prevalence of CD antibody positivity was 0.8% to 1.2%. One study showed a positive association between CD antibody positivity and IgE sensitization for dog, horse and food allergens.
Conclusion
We found a possible association of CD and IgE sensitization to some food and inhalant allergens in the Health2006 study. In further studies, however, we could not consistently replicate these associations.
Key summary
Summarize the established knowledge on this subject:
Celiac disease is a lifelong autoimmune disease caused by an abnormal immune response in genetically susceptible individuals, triggered by ingestion of gluten. Allergy is an exaggerated specific immune response against external proteins leading to allergic inflammation. The relationship between celiac disease and allergic diseases is not clear.
What are the significant and/or new findings of this study?
Our study indicated a possible association of celiac disease and immunoglobulin E sensitization to some food and inhalant allergens. Further studies in our research biobank cohorts could not consistently replicate these associations.
Introduction
Celiac disease (CD) is a lifelong autoimmune disease affecting about 1% of the population, although many cases of CD remain undiagnosed. CD is caused by an abnormal immune response, in genetically susceptible individuals, triggered by the ingestion of gluten proteins from wheat, rye and barley.1–3 CD primarily affects the small intestine, but the clinical manifestations are broad. The treatment of CD is a lifelong gluten-free diet. A small intestinal biopsy and detection of inflammation, crypt hyperplasia and/or villus atrophy is the gold standard for the diagnosis of CD in adults. However, since individuals with CD produce disease-specific antibodies, serology is more frequently used in the diagnostic process.4,5 In contrast to the abnormal immune response in autoimmune disease in which the immune system attacks itself, allergy is an exaggerated immune response against external proteins leading to allergic inflammation. Immunoglobulin (Ig) E-mediated allergic diseases include food allergies, allergic rhinitis and allergic asthma. Allergic reactions may occur in individuals IgE sensitized to allergens, primarily proteins. The genetic or acquired predisposition to production of IgE antibodies against environmental allergens and thus risk of IgE-mediated allergic disease is often referred to as “atopy” and the diseases therefore sometimes are labeled as “atopic diseases.”
Studies have found CD to be more frequent among individuals with atopic diseases,6–9 while others, 10 but not all, 11 have found atopic diseases to be more common among individuals with CD. Ciacci et al. 12 found no significant difference between individuals with CD compared with their relatives and spouses without CD with regard to allergy prevalence, but they found the prevalence of atopic dermatitis to be three times higher in individuals with CD. Enroth and colleagues found no association between self-reported allergy and CD-related autoantibodies. 13 Pillon et al. 14 found individuals with severe food allergy to have a fivefold increased risk of CD compared with controls. Asthma has also been found to be associated with CD; individuals with CD have increased risk of asthma15,16 and CD was increased in young individuals hospitalized with asthma, 17 but the number of studies are limited.
Thus, the results are contradicting, and it is not clear whether CD is associated with increased risk of IgE-mediated allergic disease or asthma. Therefore, we aimed to investigate the association of CD and CD antibody positivity with hay fever, asthma and IgE sensitization in a general population study of Danish adults who had been screened for CD, and if positive, were examined by a specialist and underwent intestinal biopsies. Confirmation of results was sought by measurements of CD antibodies in four additional population-based studies.
Methods
The Health2006 study
This study was based on the five-year follow-up of the Health2006 study, designed as a cross-sectional population-based study. 18 A random sample living in the Southwestern part of Copenhagen aged 18–69 years were invited. All eligible participants in the Health2006 cohort were invited to a five-year follow-up examination including essentially the same study protocol19,20 with the addition of screening for CD by measurements of CD biomarkers. A total of 3405 participants were eligible for invitation, 2308 individuals (54.2% female) were reexamined (participation rate: 44.7%) and 2297 participants were screened for CD (11 were not included because of a missing blood sample or for technical reasons). The mean age at the five-year-follow-up examination was 55.7 years (range, 24–76 years). The study included structured self-administered questionnaires on general health, symptoms and diagnoses of disease, lifestyle and socioeconomic variables among other issues, physical examination and collection of blood samples.
Screening for CD
The screening for CD in the five-year follow-up of the Health2006 study has been described elsewhere.20,21 Briefly, 2297 individuals were screened for CD antibodies by the Elia™ Celikey® tissue transglutaminase (TTG) anti-IgA assay and deamidated gliadin peptide (DGP) anti-IgA and anti-IgG assays. Participants with undetectable levels of IgA-TTG were screened for IgG-TTG. The measurements were performed at Thermo Fisher Scientific, Immuno Diagnostics, Allerød, Denmark. Participants were defined as CD screen test positive and invited to a clinical examination if IgA-TTG ≥ 7.0 U/ml, IgA-DGP ≥ 10.0 U/ml and/or IgG-DGP ≥ 10.0 U/ml. (None of the participants screened for IgG-TTG had levels ≥ 7.0 U/ml.)
For this current paper diagnosed cases of CD from the Health2006 study included the eight cases of screen-detected CD diagnosed by clinical evaluation and the one participant with known CD confirmed by International Classification of Diseases, 10th revision diagnosis code in the National Patient Register. Furthermore, after evaluating the screening 21 we changed our definition of CD antibody positivity for this study to IgA-TTG ≥ 7.0 U/ml, IgG-TTG ≥ 7.0 U/ml and/or IgG-DGP ≥ 10.0 U/ml. Additionally, we conducted analyses in which CD antibody positivity was limited to IgA-TTG or IgG-TTG ≥ 10.0 U/ml.
Allergy
Serum samples were screened on a Phadia250 (ImmunoDiagnostics, Allerød, Denmark) for Phadiatop®, specific IgE to a panel of inhalant allergens, and serum-specific IgE to peanut, food mix Fx5 (a panel of food allergens), egg, soy, milk, cod, wheat, cat, horse, dog, dermatophagoides (D.) pteronyssinus, mold mix, grass, mugwort, birch and profilin birch. IgE positivity was defined as ≥0.35 kU/l.
Skin prick testing for a standard panel of aeroallergens: birch, timothy grass, mugwort, horse, cat, dog, two house dust mites (D. pteronyssinis and D. farinae), and two molds (Cladosporium herbarum and Alternaria alternata) was performed by the Soluprick SQ® test (ALK Abelló, Hørsholm, Denmark).
IgE sensitization to inhalant allergens was defined as a positive skin prick test to at least one of 10 allergens or one or more positive tests for serum-specific IgE against inhalant allergens.
Asthma and hay fever were defined as positive answers to the question “Has a doctor ever told you that you had asthma/hay fever?”
Confirmation of results in four additional studies
We aimed to confirm the association of CD antibodies and IgE sensitization against some inhalant allergens found in the Health2006 study. For this, we used our research biobank with serum samples from participants in previously performed studies. We screened four population-based studies (the Inter99 study,22,23 the 1936 cohort study, 24 the MONICA-I study 25 and the Allergy90 study26,27) with information on allergy and IgE sensitization to inhalant allergens for CD antibodies. Information about the four biobank studies and the allergy testing is included in the Supplementary material. The biobank samples were screened for CD antibodies by the Elia™ Celikey® TTG anti-IgA and DGP anti-IgG assays at Thermo Fisher Scientific. Participants with undetectable levels of IgA-TTG were screened for IgG-TTG. CD antibody positivity was defined as IgA-TTG ≥ 7.0 U/ml, IgG-TTG ≥ 7.0 U/ml and/or IgG-DGP ≥ 10.0 U/ml. Furthermore, we conducted analyses for which the CD antibody positivity only was defined as IgA-TTG or IgG-TTG ≥ 10.0 U/ml. Additionally, we screened serum samples from the CD antibody-positive participants in the 1936 cohort study, the Inter99 study and the Allergy90 study for specific IgE to a panel of food allergens (Fx5). If Fx5 positive, IgE specific to peanut, egg, milk and wheat were determined. IgE positivity was defined as positive when ≥0.35 kU/l. Prevalence of IgE positivity to food allergens in the CD antibody-positive individuals was compared with the prevalence of IgE positivity to food allergens in the CD antibody-negative individuals in the Health 2006 study.
Ethics
Informed written consent was obtained from all participants prior to participation. This study was approved by the local ethics committee (H-3-2011-081, September 6, 2011). Measurements of CD antibodies in stored blood samples were approved by the ethics committee of the Capital Region of Denmark (H-15014502, January 12, 2016). We followed the recommendations of the 1975 Declaration of Helsinki.
Statistical analyses
The association between variables was tested as appropriate: Wilcoxon test, Kruskal-Wallis tests and independent samples t test for continuous variables; and chi-squared test and Fisher exact test for categorical data. Complete case analyses were performed, thus the number of observations differs because of missing data. P values < 0.05 (two sided) were considered statistically significant. To adjust for multiple comparisons, the Bonferroni method was used. The Bonferroni adjusted significance level was 0.002 (27 outcomes). Statistical analyses were performed with software package SAS 9.4 (SAS Institute, NC, USA, USA), and the meta-analyses were performed with Stata 14 (StataCorp, College Station, TX). The study-specific estimates were combined using the inverse variance method both in fixed- and random-effects models; the results shown in Figure 1 are from the random-effects analyses. Heterogeneity across studies was assessed by the I2 test.
Meta-analyses of the different studies.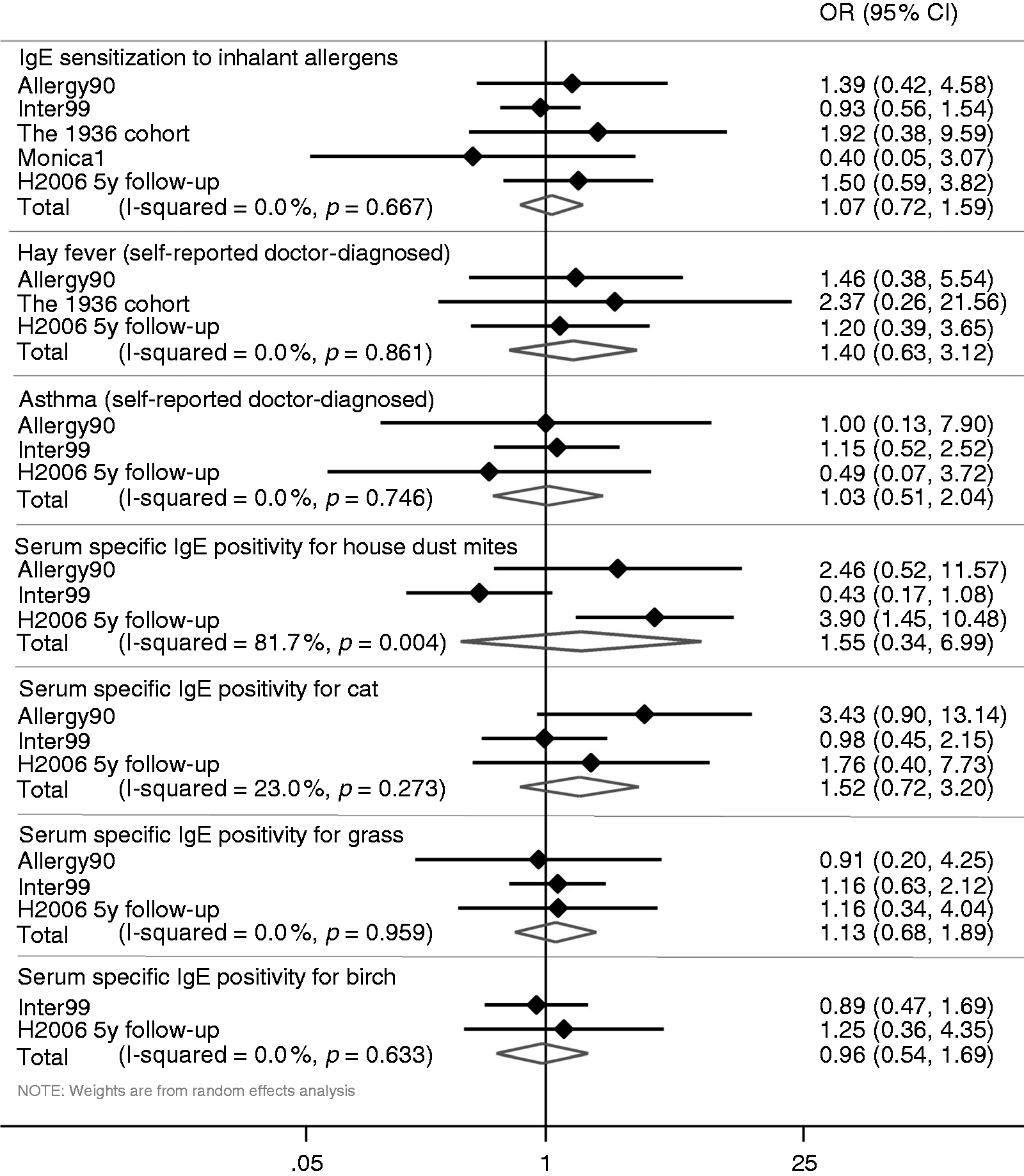
Results
General characteristics of participants stratified by CD antibody positivity.
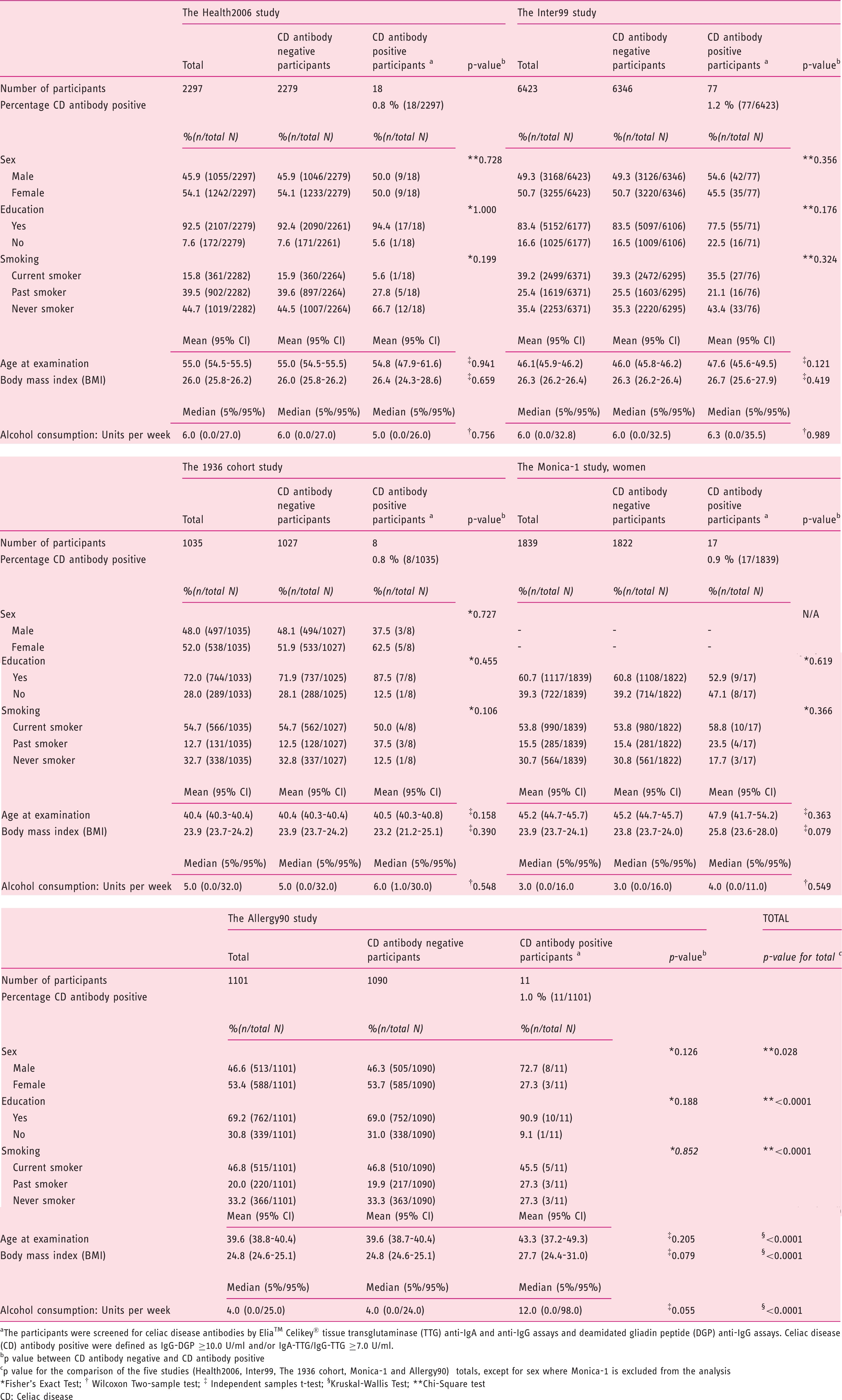
CD: celiac disease; CI: confidence interval; Ig: immunoglobulin.
Participants were screened for CD antibodies by Elia™ Celikey® tissue transglutaminase (TTG) anti-IgA and anti-IgG assays and deamidated gliadin peptide (DGP) anti-IgG assays. CD antibody positive was defined as IgG-DGP ≥ 10.0 U/ml and/or IgA-TTG/IgG-TTG ≥ 7.0 U/ml.
A p value between antibody negative and antibody positive.
Fisher exact test.
Wilcoxon two-sample test.
Independent samples t test.
Kruskal-Wallis test.
Chi-squared test.
Allergy measurements stratified by celiac disease diagnosis and antibody status a among participants in the Health2006 study.
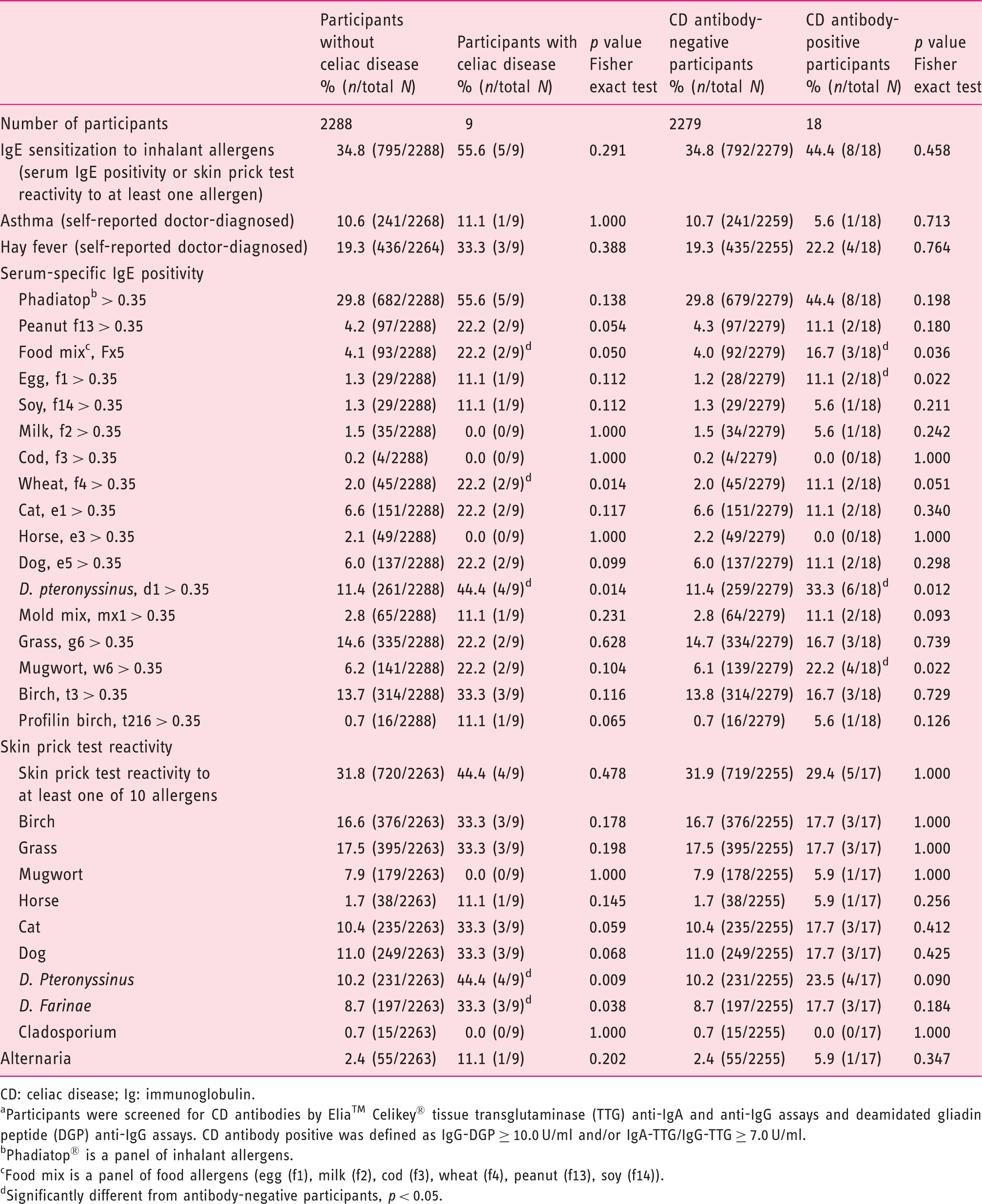
CD: celiac disease; Ig: immunoglobulin.
Participants were screened for CD antibodies by Elia™ Celikey® tissue transglutaminase (TTG) anti-IgA and anti-IgG assays and deamidated gliadin peptide (DGP) anti-IgG assays. CD antibody positive was defined as IgG-DGP ≥ 10.0 U/ml and/or IgA-TTG/IgG-TTG ≥ 7.0 U/ml.
Phadiatop® is a panel of inhalant allergens.
Food mix is a panel of food allergens (egg (f1), milk (f2), cod (f3), wheat (f4), peanut (f13), soy (f14)).
Significantly different from antibody-negative participants, p < 0.05.
Allergy measurements stratified by CD antibody status a among participants in the MONICA-1, 1936 cohort, Allergy90 and Inter99 studies.
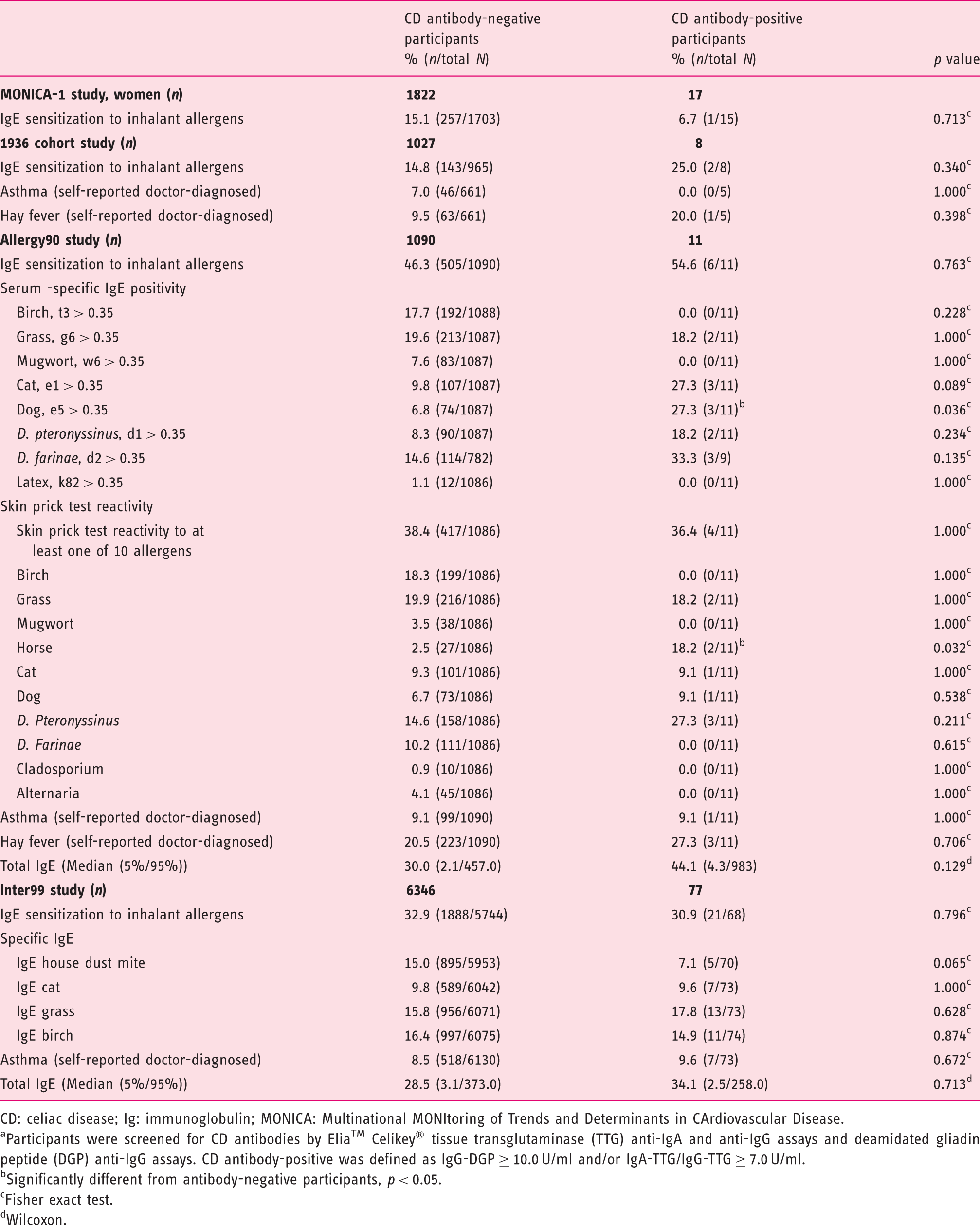
CD: celiac disease; Ig: immunoglobulin; MONICA: Multinational MONItoring of Trends and Determinants in CArdiovascular Disease.
Participants were screened for CD antibodies by Elia™ Celikey® tissue transglutaminase (TTG) anti-IgA and anti-IgG assays and deamidated gliadin peptide (DGP) anti-IgG assays. CD antibody-positive was defined as IgG-DGP ≥ 10.0 U/ml and/or IgA-TTG/IgG-TTG ≥ 7.0 U/ml.
Significantly different from antibody-negative participants, p < 0.05.
Fisher exact test.
Wilcoxon.
Allergy measurements for food allergens among CD antibody-positive participants in the 1936 cohort, Inter99 and Allergy90 studies and CD antibody-negative participants in the Health2006 study.
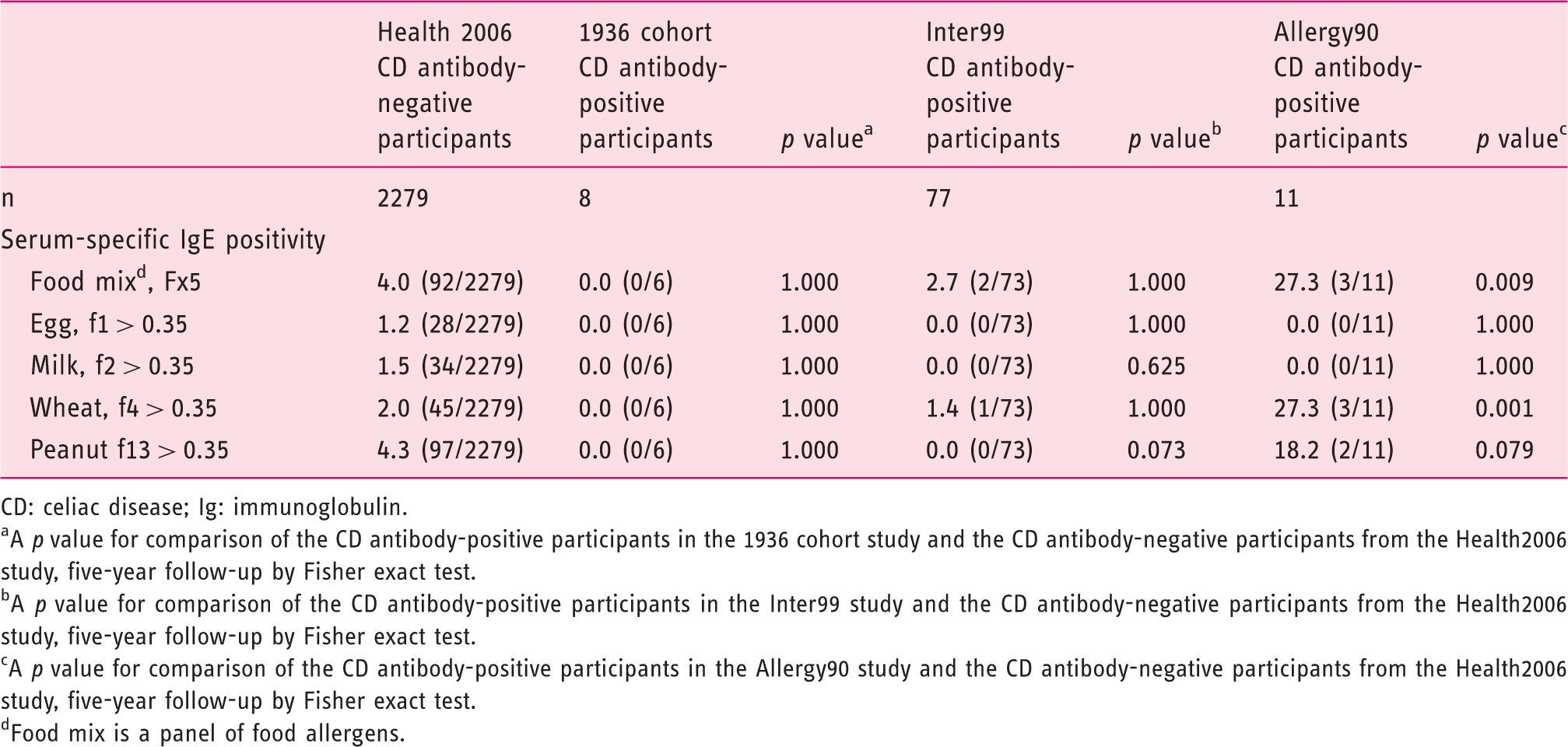
CD: celiac disease; Ig: immunoglobulin.
A p value for comparison of the CD antibody-positive participants in the 1936 cohort study and the CD antibody-negative participants from the Health2006 study, five-year follow-up by Fisher exact test.
A p value for comparison of the CD antibody-positive participants in the Inter99 study and the CD antibody-negative participants from the Health2006 study, five-year follow-up by Fisher exact test.
A p value for comparison of the CD antibody-positive participants in the Allergy90 study and the CD antibody-negative participants from the Health2006 study, five-year follow-up by Fisher exact test.
Food mix is a panel of food allergens.
When applying the Bonferroni adjusted significance level, none of the results remained statistically significant.
Figure 1 shows the results from the meta-analysis of the five studies; no significant associations were found.
Discussion
We found that individuals diagnosed with CD, or CD antibody-positive participants, had a significantly higher prevalence of IgE sensitization to some food and inhalant allergens. However, we could not replicate these associations of CD antibody positivity and IgE sensitization to inhalant allergens when screening blood samples in our research biobank from four different population-based studies for CD antibodies, although CD antibody positivity was significantly associated with a higher prevalence of IgE sensitization to dog, horse and food allergens in one study. In these five general Danish populations, the prevalence of CD antibody positivity, defined as IgA-TTG ≥ 7 U/ml, IgG-TTG ≥ 7 U/ml and/or IgG-DGP ≥ 10 U/ml., ranged between 0.8% and 1.2%.
We screened research biobank serum collected from 1976 through 2012 for CD antibodies and found no consistent increase in the prevalence of CD antibody positivity, although the prevalence steadily increased from 0.8% in 1976 to 1.2% in 2001, but in 2011–2012 (the Health2006 study) the prevalence was 0.8%. Earlier studies on the association of atopic diseases and CD have been conflicting.6–13 We found a higher frequency of IgE sensitization between individuals with CD and CD antibody-positive participants, but no significant association between overall IgE sensitization to inhalant allergens and CD or CD antibody positivity. This tendency was not, however, consistent when screening the biobank studies. Moreover, we found CD antibody-positive participants to have a significantly higher prevalence of IgE sensitization to house dust mites and mugwort and individuals with CD to have a higher prevalence of IgE sensitization against house dust mites. These associations were not confirmed in the screening of biobank serum samples. In the Allergy90 study, however, we found significantly increased serum-specific IgE positivity against dog and significantly increased skin prick test reactivity against horse. The same tendency was seen in Health2006, although not statistically significant. Our results are not consistent, but might indicate a tendency toward more allergy among individuals with CD and CD antibody-positive participants.
Pillon et al. found individuals with severe food allergy (milk, egg and wheat allergy) to have increased risk of CD. 14 In line with these findings, we found an association of IgE sensitization against food mix (Fx5), a panel of six food allergens, and CD and CD antibody positivity. Furthermore, we found significantly more serum-specific IgE positivity against egg among CD antibody-positive participants and significantly more serum-specific IgE positivity against wheat among individuals with CD. Also, CD antibody-positive participants had more specific IgE against wheat, although not statistically significant. However, the replication studies based solely on serological measurements and not clinical diagnosis of CD did not consistently support that CD antibody-positive individuals are at increased risk of specific IgE positivity to food allergens, although one study (Allergy90) found a higher prevalence of IgE positivity to food mix and wheat among CD antibody-positive individuals. Thus, the results both from Pillon et al. and two of our studies suggest that screening for CD could be considered in individuals with food allergy. A mechanism behind this possible association might be an increased intestinal permeability in individuals with CD resulting in increased flow of dietary antigens 14 that could lead to increased IgE sensitization. Other possible reasons for this association could be dysregulation of immune responses or common genetics. However, the pathogenesis of the two diseases are notably different and there is no obvious common pathway.
We could not reproduce the findings of an association of CD and asthma found in earlier studies.15–17 It is important to note, however, that in all our studies asthma was self-reported and some misclassification should be expected. Another important aspect is that the CD cases in our study were predominantly undiagnosed before the study and without substantial symptoms, probably related to low grade both of malnutrition and other possible consequences of symptomatic CD.
In the Health2006 study we screened both by using IgA-DGP and IgG-DGP, but after evaluating the screening 21 we omitted IgA-DGP from the further screenings in the additional four population-based biobank studies to limit the proportion of false-negative participants. We also included analyses for which the CD antibody positivity was limited to IgA-TTG or IgG-TTG ≥ 10 U/ml because this cutoff was found to have the highest positive predictive value, but no further significant differences were found. The prevalence of CD antibody positivity in these five study populations corresponds well to the pooled global seroprevalence of CD shown by Singh et al., while the CD prevalence in our study was lower than the pooled global prevalence of CD, 28 in line with registry studies documenting low prevalence of CD in Denmark.29,30
The population-based design is a strength of our study. However, we cannot rule out that selection bias could influence our results if presence of both undiagnosed CD and allergy biomarkers were associated with participation in the included studies, although in the Health2006 study diagnosed CD recorded in the National Patient Register was not associated with participation. 20 Another strength is the variety of information and tests on allergic diseases, including serum-specific IgE positivity, skin prick test reactions and self-reported asthma and hay fever. Including many allergy outcomes may lead to a problem of multiple testing, however. It is thus important to note that when a Bonferroni significance level was applied to adjust for multiple testing, our results did not remain statistically significant. Nevertheless, as this is an explorative study and the Bonferroni method is very conservative, we reported the results with a significance level of 0.05. Furthermore, it is a major strength that we had the possibility to test our hypothesis in the biobank studies from the same region. However, it is a limitation, as mentioned above, that the biobank studies had information only on IgE sensitization to inhalant allergens. Moreover, our study might be limited by the slight uncertainty of the diagnosis of CD in the additional biobank analyses for which clinical diagnosis and biopsies were not possible. Nevertheless, we know from our earlier study 21 that the positive predictive values of the chosen CD antibody positivity were high.
In conclusion, our study (the Health2006 study) indicated a possible association between CD and IgE sensitization to food and inhalant allergens. However, in additional analyses of other study populations we could not consistently replicate these associations between CD antibody positivity and IgE sensitization. Further research is needed to investigate the relationship between CD and allergic diseases and its possible clinical implications.
Supplemental Material
Supplemental material for The association of celiac disease and allergic disease in a general adult population
Supplemental material for The association of celiac disease and allergic disease in a general adult population by Line Lund Kårhus, Tea Skaaby, Anja Lykke Madsen, Betina Heinsbæk Thuesen, Peter Schwarz, Jüri J Rumessen and Allan Linneberg in United European Gastroenterology Journal
Footnotes
Acknowledgments
We thank all staff members at the Center for Clinical Research and Prevention (formerly Research Centre for Prevention and Health) for contributing to the data collection and health examinations. We also thank Thermo Fisher Scientific, Allerød, Denmark, Lone Søgaard and Bjarne Kristiensen for their help with the analyses and interpretation of the results, and Pia Schytte Hansen for performing the antibody measurements.
Declaration of conflicting interests
None declared.
Funding
This study was supported by the Tryg Foundation (7-11-0213), Dansk Cøliaki Forening (the Danish Celiac Disease Patient Organization), The Novo Nordisk Foundation (NNF160C0022464), Independent Research Fund Denmark and Thermo Fisher Scientific, Allerød, Denmark. Thermo Fisher Scientific performed the measurements of CD antibodies.
Informed consent
Informed written consent was obtained from all participants prior to participation.
Ethics approval
The study was approved by the local ethics committee (H-3-2011-081, September 6, 2011). Measurements of CD antibodies in stored blood samples were approved by the ethics committee of the Capital Region of Denmark (H-15014502, January 12, 2016). We followed the recommendations of the 1975 Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
