Abstract
Background
Progression of potential celiac disease (PCD) to overt celiac disease (CD) has been described in some studies from the Western Hemisphere. There are no Asian data on this aspect of CD.
Objective
We aimed to study the short-term histological course of PCD in Indian patients.
Methods
Patients with PCD were prospectively identified by screening relatives of patients with CD, the diarrheal subtype of irritable bowel syndrome (IBS-D) and patients with iron deficiency anemia (IDA). Patients with serology that was positive for immunoglobulin A antibodies against tissue transglutaminase (IgA anti-tTG) were subjected to endoscopy with duodenal biopsy. PCD was defined as a Marsh-0 to Marsh-II lesion on duodenal biopsy, along with positive IgA tTG serology. Retesting for serology and histology was done at 6-month intervals, for 12 months.
Conclusions
Our data suggested that despite the fact that nearly 80% of the patients diagnosed to have PCD continue to remain seropositive for tTG 12 months later, histological progression to Marsh-III occurred in only 7% of patients over the same time period. These observations do not justify starting a gluten-free diet in all patients with PCD, in India.
Keywords
Introduction
Celiac disease (CD) is a chronic small intestinal immune-mediated enteropathy, precipitated by exposure to dietary gluten in genetically susceptible individuals. 1 An important landmark in the diagnosis of CD was the development of reliable serological markers against endomysium, gliadin and tissue transglutaminase (tTG), which are commercially available all over the world. 2 The diagnosis of CD is based on the revised Oslo definitions, published in 2013. 1 The revised European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) guidelines underscore the importance of serological testing and suggest that duodenal biopsy may be optional in selected cases, where the serological titers are >10 times the upper limit of normal. 3
The introduction and wide availability of serological tests for the diagnosis of CD in the 1990s also led to the detection of patients with a positive serology, but with minimal or no evidence of structural injury of the small bowel. The term ‘potential celiac disease’ (PCD) was coined in 1993 by Ferguson et al., 4 who suggested that expression of T-cell mediated immunity to gliadin in the gut occurs across a spectrum of histological and functional abnormalities, wherein the minimal lesion would appear as a virtually normal biopsy with a high count of intra-epithelial lymphocytes (IELs). The single most important risk factor for CD is having a first-degree relative with already diagnosed celiac disease, particularly a sibling. Evidence for a familial risk in CD has accumulated from many sources, including biopsy and serological studies in families with known CD. 5 The terms latent CD and potential CD have often been used interchangeably in the past; however, the recent Oslo guidelines discourage the use of the term latent CD. 1
The natural history of PCD is of interest mainly because of two key issues: first, to know what proportion of patients with PCD evolve into overt CD; and second, to know the time period over which this evolution occurs. Pediatric literature from Europe suggests that between 31–62.5% of children with PCD will progress to overt CD with villous atrophy, over a period of 1–3 years.6,7 Published literature on the natural history of PCD in adults is scarce. Biagi et al. 8 recently report that there is maintenance of normal duodenal mucosal histology for many years, as well as spontaneous symptom resolution despite a gluten-containing diet (GCD), in a subset of adult patients with PCD; however, a randomized controlled trial from Finland suggests there is an unequivocal benefit of a gluten-free diet (GFD) in adult patients with PCD. 9 In the last decade, CD has been increasingly recognized in the predominant wheat-eating population of northern India. The prevalence of the disease in northern India is no different from the figure of 1% that is reported from Europe.10,11 We therefore undertook this study to determine the proportion of patients with PCD whom progress to overt CD, over a short-term period of 12 months.
Materials and methods
This prospective cohort study was conducted in the Department of Gastroenterology, Govind Ballabh Pant Hospital, New Delhi, from April 2010 to October 2013. Patients diagnosed with PCD on the basis of a positive serology for immunoglobulin A antibodies (IgA) against tTG, along with a duodenal biopsy showing Marsh-0 to Marsh-II changes, formed the study group. 12 We identified these patients by screening first-degree relatives of index cases of CD, patients with a diarrheal variety of irritable bowel syndrome (IBS-D) or those suffering from unexplained iron deficiency anemia (IDA). Patients with Marsh-III changes on duodenal biopsy with a positive serology were put on a GFD, and were excluded from this study.
A detailed history was obtained from all patients who fulfilled the inclusion criteria, followed by a clinical examination. Blood samples were taken from all patients, for determination of blood chemistry and hematological studies. Anemia was arbitrarily defined as hemoglobin <12 gm/dl in males and <11 gm/dl in females. Serological tests for CD were done using enzyme-linked immunosorbent assay (ELISA) kits (AESKULISA, Germany) for IgA tTG. We considered a value >18 IU/ml a positive test for IgA anti-tTG, as per the manufacturers’ guidelines. We obtained the duodenal biopsy from the second part of the duodenum (6–8 pieces), to send for histopathological examination. The slides were stained with hematoxylin and eosin (H&E), and were reported independently by two trained histopathologists, following the modified Marsh classification.13,14
We repeated the serological marker and duodenal histology tests at 6 and 12 months, in those diagnosed to have PCD. Patients developing Marsh-III changes during the course of the study were put on a GFD and not subjected to further testing. Informed consent was obtained from each patient, prior to inclusion in the study. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki’s 6th revision from 2008, as reflected in a priori approval by the institutional ethics committee. The patients were followed up at regular intervals, for 1 year on out patient department basis.
Statistical methods
Continuous variables were expressed as the mean and range. Categorical variables were reported as percentages. The kappa score for inter-observer agreement was calculated. The SPSS software version 19.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis.
Results
Demographic and clinical data of patients with PCD

BMI: Body mass index; CD: celiac disease; IBS: irritable bowel syndrome; PCD: potential celiac disease
A past history of diarrhea was present in 22 study participants (38.5%). Mean body mass index (BMI) of the study population was 21.5 kg/m2 (range: 12.8–32.8 kg/m2). The mean value of hemoglobin was 11.65 gm/dl (range: 5.7–16.2 gm/dl). Nine patients (6 female) were diagnosed to be anemic; the anemia was microcytic hypochromic, in all the patients. Upper gastrointestinal (GI) endoscopy was essentially normal in 38 patients. Duodenal biopsy showed normal villous pattern in 28 patients (Marsh-0), normal villous pattern with increased IELs in 27 patients (Marsh-I) and normal villous pattern with crypt hyperplasia with increased IELs in two patients (Marsh-II). The mean IgA anti-tTG value was 58.6 (22–124) U/ml. All the patients were followed up for 1 year. None of the study patients were put on a GFD. None of the patients had any clinical deterioration, during the course of the follow-up period. None of the patients had any features suggestive of autoimmune diseases, during the course of the study.
Reassessment at 6 months
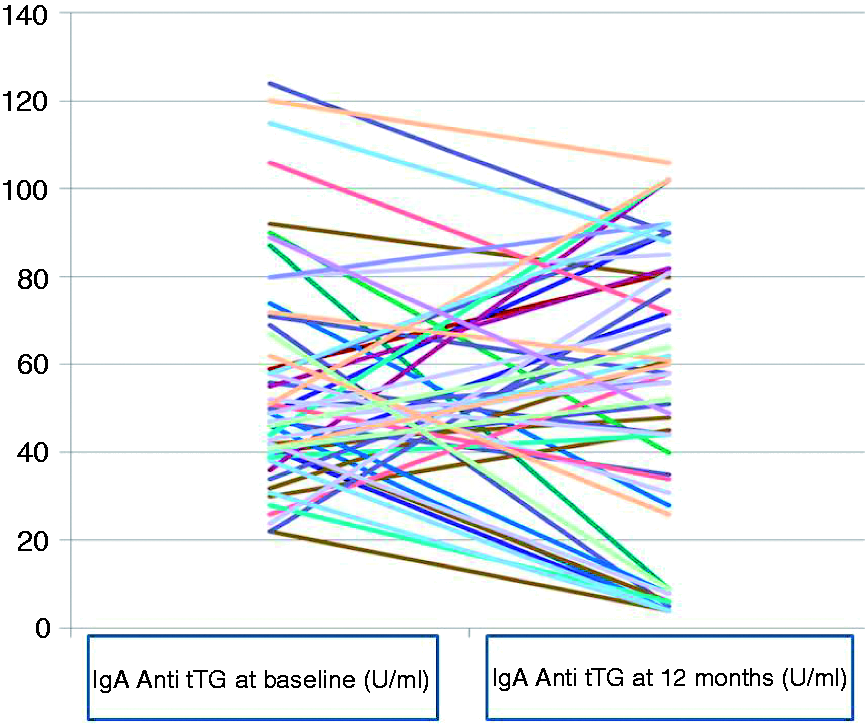
Of the 57 patients who were positive for IgA anti-tTG antibody, 45 continued to remain positive, but 12 became negative.
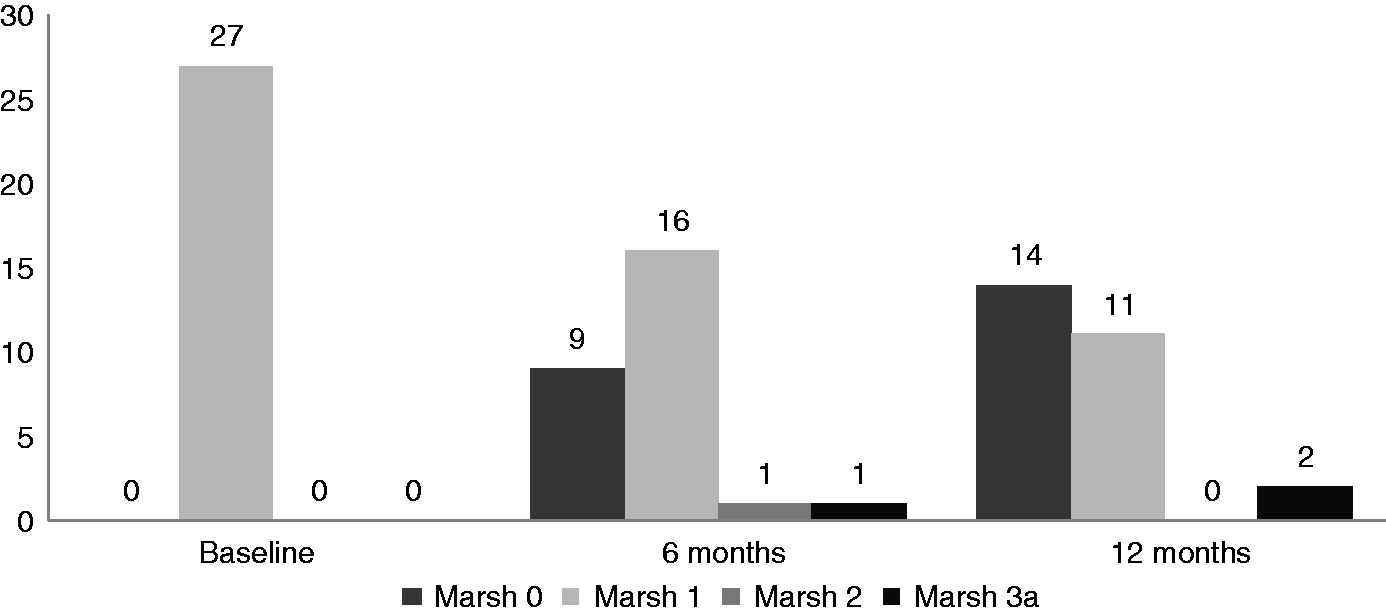
Histological evaluation at the 6-month follow up revealed that 22 of the 28 (78.5%) with Marsh-0 changes showed no histological progression, but five patients had progressed to Marsh-I and one patient had progressed to Marsh-II.
In 16 of the 27 (59.2%) patients with Marsh-I changes at the initial assessment, there was no histological change; whereas nine patients had a regression to Marsh-0. Progression of the histological lesion to Marsh-II and Marsh-IIIa was seen in one patient each.
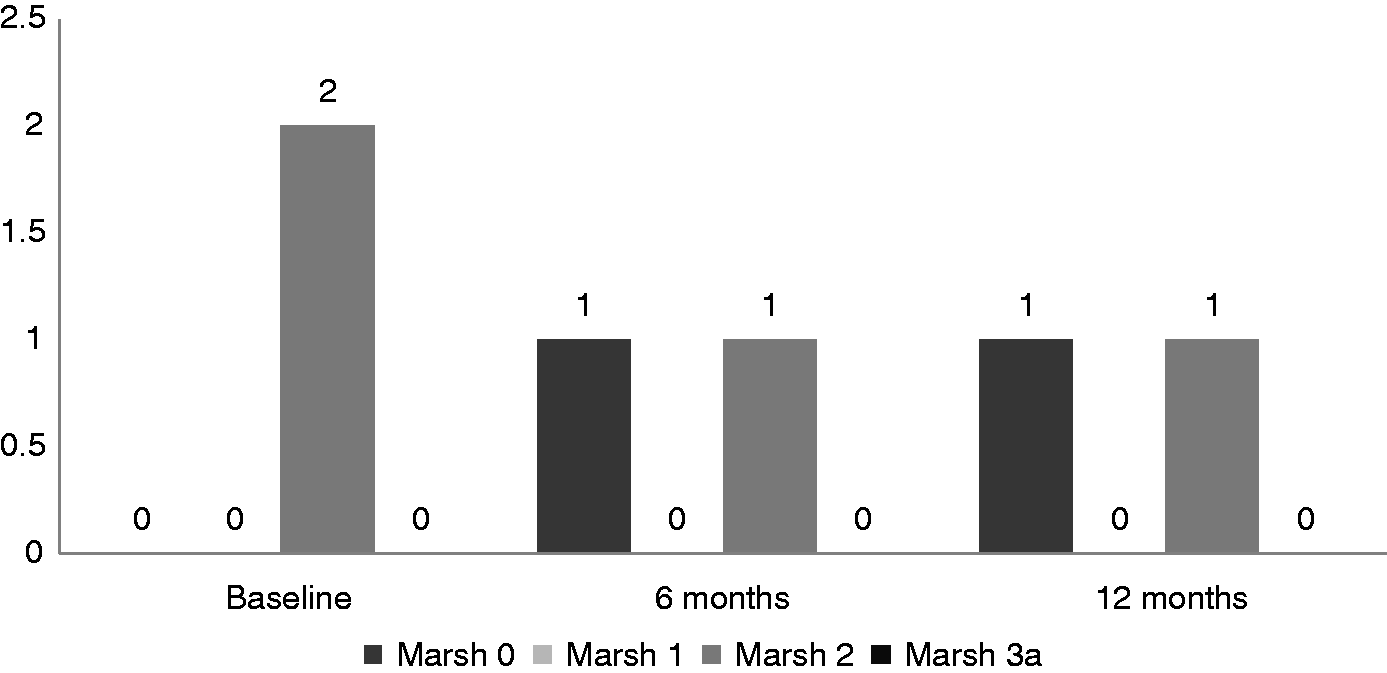
Of the two patients with Marsh-II changes at initial assessment, one remained at the status quo 6 months later and one showed regression to Marsh-0.
These findings are summarized in Figures 1–3.
Bar diagram showing follow-up histology at 6 and 12 months, in the 28 patients who were Marsh-0 at baseline. Bar diagram showing follow-up histology at 6 and 12 months, in the 27 patients who were Marsh-I at baseline. Bar diagram showing follow-up histology at 6 and 12 months, in the two patients who were Marsh-II at baseline.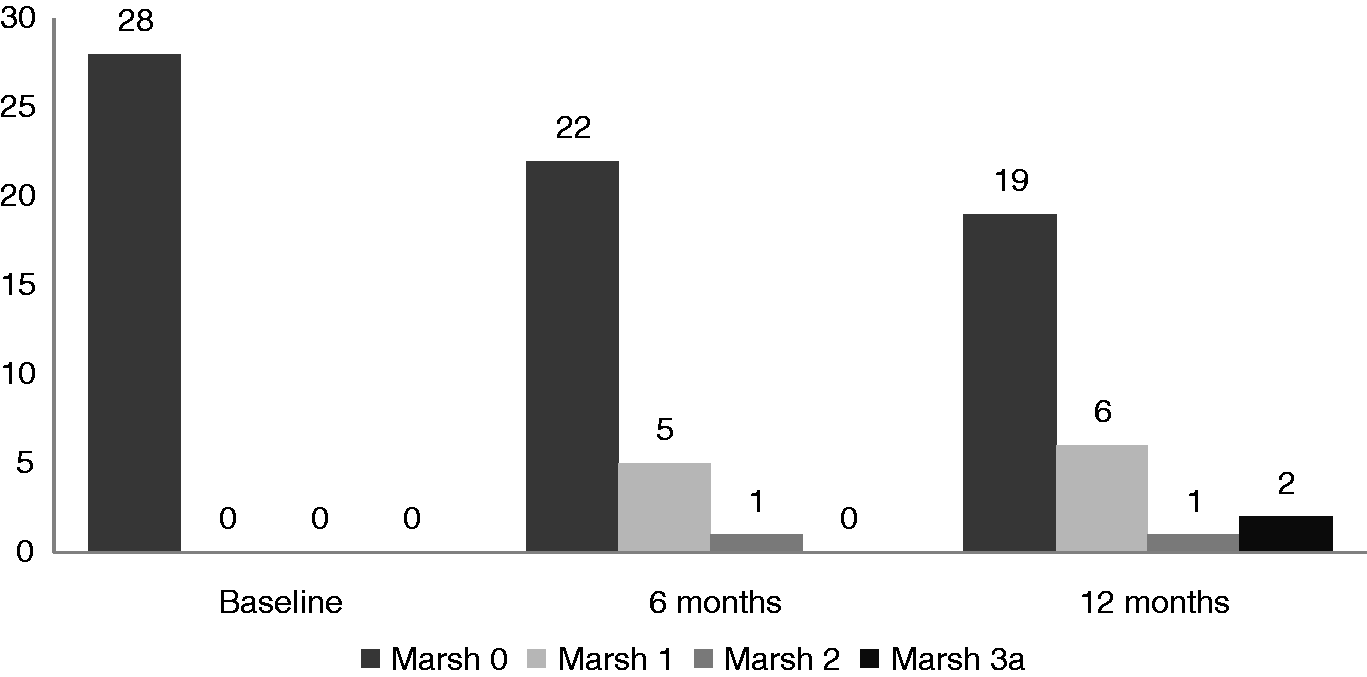
Reassessment at 12 months
Follow-up serology at 12 months, in 57 study patients

IgA: immunoglobulin Type A; Neg: negative; Pos: positive; tTG: tissue transglutaminase
Follow-up serology at 12 months, in the 57 study patients.
The histological findings at the 12-month follow-up revealed that of the 28 patients classified as Marsh-0 at baseline, 19 remained as such; whereas six of these patients progressed to Marsh-I, one patient progressed to Marsh-II and two patients progressed to Marsh-IIIa.
Of the 27 patients who were initially classified as Marsh-I, 14 patients became Marsh 0; while 11 patients remained as Marsh-I and two patients became Marsh-IIIa.
Of the two patients who were initially Marsh-II, one remained as such at follow up, whereas one showed regression to Marsh-0.
These findings are summarized in Figures 1–3. The kappa score for inter-observer agreement was 0.743, implying there was substantial agreement; thus in summary, progression to Marsh-III was documented in only four (7%) of our 57 study patients with PCD, over a period of 12 months.
Discussion
The primary aim of this study was to evaluate the short-term natural history of PCD in terms of progression to overt CD, as documented by worsening duodenal histology. In this study from a tertiary referral center in northern India, 57 adult patients with a mean age of 28.7 years were diagnosed as having PCD and prospectively evaluated over a period of 12 months. The commonest symptom at presentation was intermittent diarrhea, in 22 patients (38.5%). Anemia was documented in nine patients (15.7%), upon investigation. We compared the paired duodenal biopsies of all patients diagnosed with PCD at the time of the initial presentation, with the subsequent biopsy taken 12 months later.
Of the 28 patients with a normal villous pattern (Marsh-0) at baseline, 19 remained at the same histology grade (Marsh-0), whereas progression to Marsh-I, -II and -IIIa was seen in six, one and two patients, respectively; over a period of 12 months.
Of the 27 patients with Marsh-I histology at baseline, 11 continued to be in the same category 12 months later; whereas 14 of them showed regression of the histology to Marsh-0. Progression of the histological grade to Marsh-IIIa was documented in only two patients. Of the two patients who were initially Marsh-II, one showed no change in the subsequent biopsy, whereas regression to Marsh-0 was noted in the other patient.
In summary, the duodenal histology was non-progressive in 46 of the 57 patients (80%), when evaluated 12 months after the initial biopsy. Conversely, these data suggested a histological worsening, from Marsh-0 to Marsh-II to overt CD (Marsh-IIIa), in only four patients (7%) over a period of 12 months.
A similar trend of non-progression was also seen with regard to the serological data. Of the 57 patients who were positive for anti-tTG IgA antibodies initially, 45 retained positivity; whereas 12 patients became seronegative by the end of 12 months. Variation in the serological tests at baseline versus follow up could be due to an initial false positivity, or more likely due to fluctuation in the immunological status of the subjects over the course of time. They could also be attributed to the phenomena of transient celiac auto-immunity, as described by Mahadev et al. 15 Overall, 12 patients (21%) lost these antibodies during the course of the study, over a 12-month period. These observations are not very different from those of Tosco et al., 6 who observed that IgA antibodies to tTG disappeared in 15% of subjects and fluctuated in 33% of them, over a follow-up period of 3 years.
Existing data on the natural history of PCD is sparse and is limited to a few studies from the West6–9; data from South Asia is non-existent, despite the fact that the prevalence of CD in some parts of Asia10,11,16 is at a par with the figures from the West. Most of the data on the natural history of PCD is restricted to children6,7 and the natural history of the disease in the adult population is largely unknown. Existing data from the West suggests that PCD would progress to overt CD in nearly one-third of pediatric patients, over a relatively short period of 3 years. 6 Kurppa et al. 7 show an even more alarming rate of histological progression from Marsh-I to Marsh-III, in 62.5% children over a 1-year period; however, the natural history the disease elicits in children seems to be bi-directional with reference to serological tests, as nearly 15% become persistently seronegative. 6 These data suggested that patients with PCD need to be followed up regularly, with both serology and histopathology, to determine their future course.
The basic question then arises as to what should be done in the subgroup of patients diagnosed as PCD after the initial workup? Unfortunately, the existing data does not provide a unanimous opinion for this vexing problem. Finnish investigators, on the basis of a randomized controlled trial, recommend that all symptomatic patients with mild enteropathy may benefit from a GFD, as all PCD patients would eventually progress to develop villous atrophy. 9 On the contrary, this opinion is not shared by Tosco et al., 6 as nearly 15% of their patients became seronegative when followed for up to 2 years. The same group has recently reported that at the 3-year follow up, no histological progression was seen in 86% of the cases. Extended follow-up at 9 years has shown non-progression in two-thirds of the patients. 17
Two issues need to be considered, regarding the implications of our study: First, the ever-increasing awareness of the disease in the predominantly wheat-eating areas across northern India, coupled with the widespread use of serological tests for the diagnosis of CD, suggests that the volume of patients diagnosed with PCD is going to increase significantly, in the future. Second: In this scenario, it is mandatory to give unequivocal guidelines to practitioners, regarding the management of these PCD patients. With a population >1 billion and a prevalence rate of 1%, it is obvious that the number of patients diagnosed with PCD in India in the near future will be sizeable. Since histological progression did not occur in the vast majority of patients, even after 9 years, there is little justification in starting a GFD in all these patients at the outset. 17
Our observations add further weight to a ‘wait and watch’ policy, as only a minority (<10%) of patients with PCD would eventually progress to overt CD. The only marker that could help in the early identification of the patients at a higher risk to develop intestinal damage seems to be the presence of anti-TG2 IgA intestinal deposits, at the time of the initial biopsy. Intestinal deposits of TG2 are reported to be present at the mucosal level before the appearance of serum anti-TG2 antibodies, so their detection has been proposed to be the most sensitive and specific test for predicting the diagnosis of CD. 6
The limitations of our study are manifold. Besides having a small sample size, the follow-up period in our study is only 12 months. Most other studies pertaining to this issue have been of >2 years duration. Additionally, we did not perform tissue testing for TG2 antibodies, as tests are not available in India. Other limitations include the non-quantification of the exact amount of gluten intake, over the follow-up period. Despite these limitations, this is the first study on PCD from Asia having a predominantly adult population as study subjects.
In conclusion, our short-term follow-up study suggested that despite nearly 80% of the patients with PCD continuing to remain seropositive for IgA anti-tTG at 12 months, histological progression to Marsh-III occurred in only 7% of the subjects, over the same time period. These observations do not justify starting a GFD in all patients with PCD in India. As the natural history of the disease may be variable in different regions of the world, larger studies are needed to accurately describe the natural history of PCD, before universally-applicable recommendations can be made.
Footnotes
Acknowledgements
The authors are grateful to Peter HR Green for his valuable suggestions and guidance, during the preparation of this manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
