Abstract
Background
Human leukocyte antigen (HLA) DQ2 and DQ8 are important risk factors for some autoimmune diseases such as celiac disease (CD), but their possible role in other diseases and health conditions is not fully explored.
Objectives
The objective of this article is to examine the distribution of HLA DQ2 and HLA DQ8 in an adult general population, and their association with health indicators (diseases, symptoms and biomarkers).
Methods
In this cross-sectional, population-based study, 2293 individuals were screened for HLA DQ2 and DQ8; CD-associated alleles (DQA*0201*03*05/DQB*02*0301/0304*0302/0305) and DQB1*02 homozygosity were determined for screen-positive participants. The National Patient Registry provided diagnosis information.
Results
A total of 47.7% (1093/2293) individuals were positive for DQ2 and/or DQ8: 31.2% (716/2293) only DQ2, 11.9% (273/2293) only DQ8, 4.1% (93/2293) both DQ2 and DQ8. Among nine individuals diagnosed with CD, 89.9% (8/9) had DQ2.5
Conclusion
In this general Danish population, 47.7% were HLA DQ2/DQ8 positive and thus potentially at risk for CD. All individuals with CD were DQ2/DQ8 positive; the majority DQ2.5. Surprisingly, DQ2/DQ8-positivity was associated with lower IBS prevalence.
Key summary
Summarize the established knowledge on this subject:
Celiac disease is a lifelong autoimmune disease caused by a harmful immune response to gluten proteins in genetically susceptible individuals. Human leukocyte antigen (HLA) DQ2 and HLA DQ8 are important risk factors for some autoimmune diseases such as celiac disease; however, their possible role in other diseases and health conditions is not fully explored. What are the significant and/or new findings of this study?
Our study showed that 47.7% of individuals were HLA DQ2/DQ8 positive and thus potentially at risk for celiac disease in this general Danish population. We found that all individuals with celiac disease were HLA DQ2/DQ8 positive; the majority DQ2.5. Furthermore, HLA DQ2/DQ8 positivity was associated with lower prevalence of irritable bowel syndrome, but not with other diagnoses/diseases.
Introduction
Celiac disease (CD) is a lifelong autoimmune disease caused by a harmful immune response to gluten proteins. Ingestion of gluten-containing grains, such as wheat, rye and barley, results in inflammation of the small intestine and villous atrophy in genetically susceptible individuals.1,2 Like other autoimmune diseases, CD has a strong human leukocyte antigen (HLA) association. 2 The physiological role of HLA is to present antigenic peptides to T lymphocytes. HLA class II genes are encoded in the major histocompatibility complex on the short arm of chromosome 6 and include the principal factors for genetic susceptibility for CD. 3 An essential step in CD pathogenesis is the HLA DQ-mediated presentation of gluten peptides to CD4+ T lymphocytes: Deamidated gluten peptides (deaminated by the enzyme transglutaminase 2) bind to HLA-DQ molecules and are presented to gluten-specific CD4+ T cells stimulating the immune response leading to CD.2,4 However, CD is a multifactorial and polygenic disorder, thus HLA is necessary but not sufficient for the development of CD. 2 Approximately 30%–40% of Western populations carry HLA DQ2 or DQ8, while only 1% is affected by CD. 3
About 90% of individuals with CD carry HLA-DQ2.5. HLA-DQ2.5 can be expressed in
Although HLA DQ2 and DQ8 are known to be associated with autoimmune diseases such as CD,2,9–11 knowledge is limited about the association of HLA DQ2 and DQ8 with other diseases and health indicators.
The distribution of HLA DQ2 and DQ8 in a general Danish population has not previously been reported, although the distribution of HLA-DR in Danes was studied in 1981.12–14 We have conducted a screening for CD in a general Danish population. 15 In a further analysis of this cohort we aimed to map the distribution of HLA DQ2 and HLA DQ8 haplotypes and examine their association with CD and other diseases, symptoms and biomarkers.
Material and methods
Study population
This study was based on the five-year follow-up of the Health2006 cohort and designed as a cross-sectional, population-based study. A detailed description of the Health2006 cohort profile has been published elsewhere. 16 The participants invited to the Health2006 cohort were drawn as a random sample from the background population aged 18–69 years, living in 11 municipalities in the Southwestern part of the greater Copenhagen area. All eligible participants in the Health2006 cohort were invited to a five-year follow-up examination including essentially the same study protocol 15 with the addition of screening for HLA DQ2/DQ8 known to be associated with CD, and screening for CD by measurements of CD-related antibodies: immunoglobulin (Ig)A against tissue transglutaminase (TTG), IgA and IgG against deamidated gliadin peptide, and for those with IgA deficiency: IgG TTG. The screening for CD has been described elsewhere.15,17 A total of 3405 participants were eligible for invitation and 2308 individuals (54.2% female) were re-examined. The mean age at the five-year-follow-up examination was 55.7 years (range: 24–76 years). The study included: structured self-administered questionnaires on general health, symptoms and diagnoses of disease, lifestyle, socioeconomic variables among other issues; physical examination and collection of blood samples.
Screening for HLA DQ2 and HLA DQ8
A total of 2293 individuals (15 not included because of missing blood sample or for technical reasons) were screened for HLA DQ2/DQ8 by the Celiac Gene Screen test, and alleles associated with CD were determined for 1175 HLA DQ2/DQ8-positive participants with the Celiac Gene Alleles test (BioDiagene, Palermo, Italy, distributed by Thermo Fisher Scientific, Allerød, Denmark) identifying by a sequence-specific primer-polymerase chain reaction (SSP-PCR) the HLA alleles DQA1*0201, *03, *05 and DQB1*02, *0301/0304, *0302/0305, as well as DQB1*02 homozygosity.
Covariates
Allergy
Serum samples were screened for specific immunoglobulin E (sIgE) to a panel of inhalant allergens using Phadiatop® (ImmunoDiagnostics, Allerød, Denmark) and a panel of food allergens by using food mix Fx5. Participants were considered Phadiotop positive and Fx5 positive when ≥0.35 kU/l.
Skin prick test reactivity against a panel of 10 common inhalant allergens was performed and atopy was defined as a positive reaction (≥3 mm) to at least one of 10 allergens. Groups of allergens (pollens, animals, or house dust mites) were also considered. Methods for allergy testing have been described elsewhere. 18
Irritable bowel syndrome (IBS)
Definition of IBS was based on a symptom cluster of frequent self-reported gastrointestinal symptoms during the past 12 months: “Subjects stating that they
Biomarkers
At follow-up, fasting blood samples were analyzed for glucose and glycated hemoglobin (HBA1c), and hemoglobin and anti-cyclic citrullinated peptide (anti-CCP) were measured. In addition, at baseline serum levels of 25-hydroxyvitamin D3 (25-OH-D3), anti-thyroperoxidase (anti-TPO), parathyroid hormone (PTH), thyroid-stimulating hormone (TSH) and free thyroxine (FT4) were measured.
Registry-based diagnoses
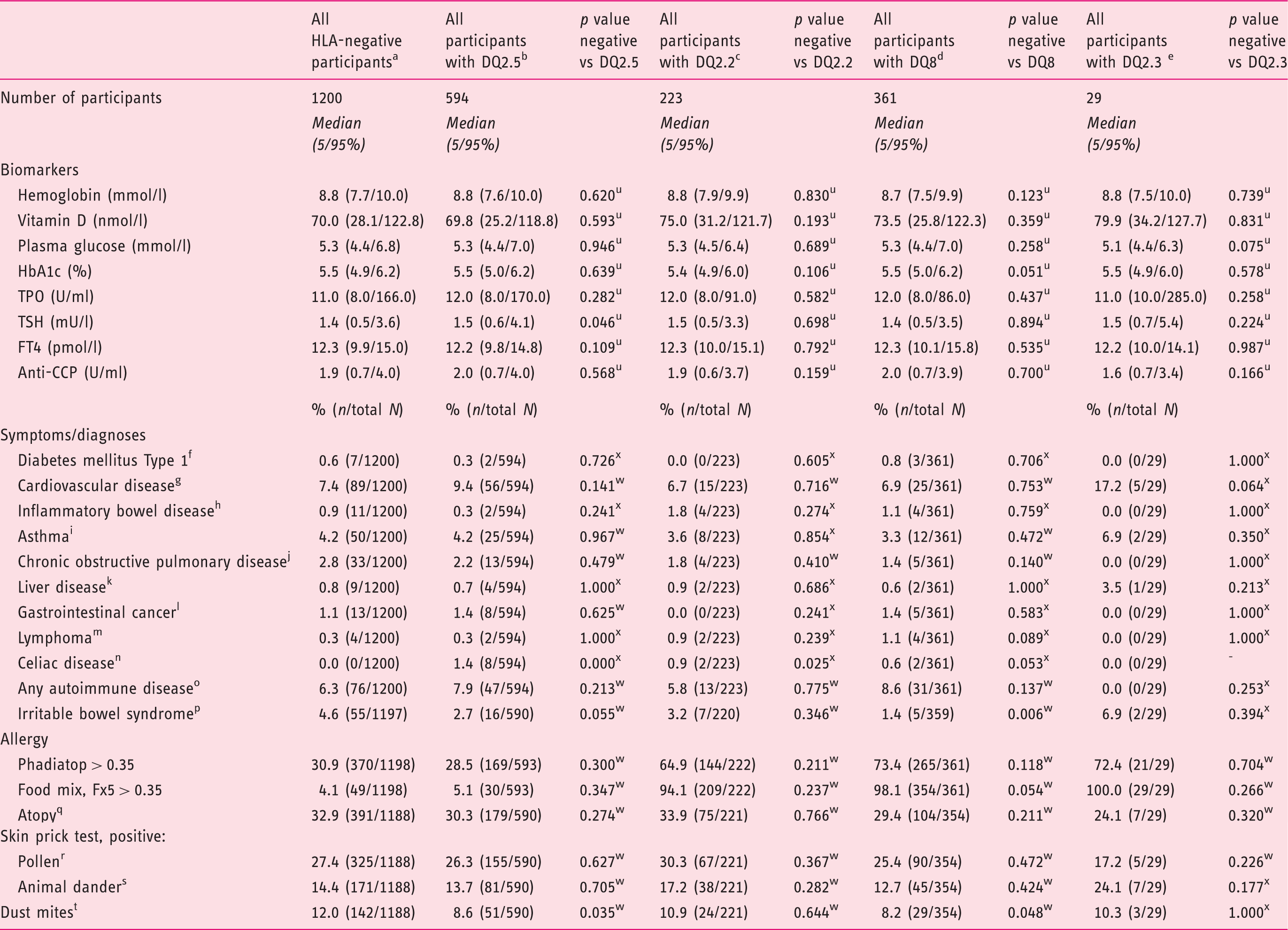
Results of biochemical analyses, symptoms and diagnoses stratified by HLA DQ2 and HLA DQ8 status among participants in the Health2006 five-year follow-up study.
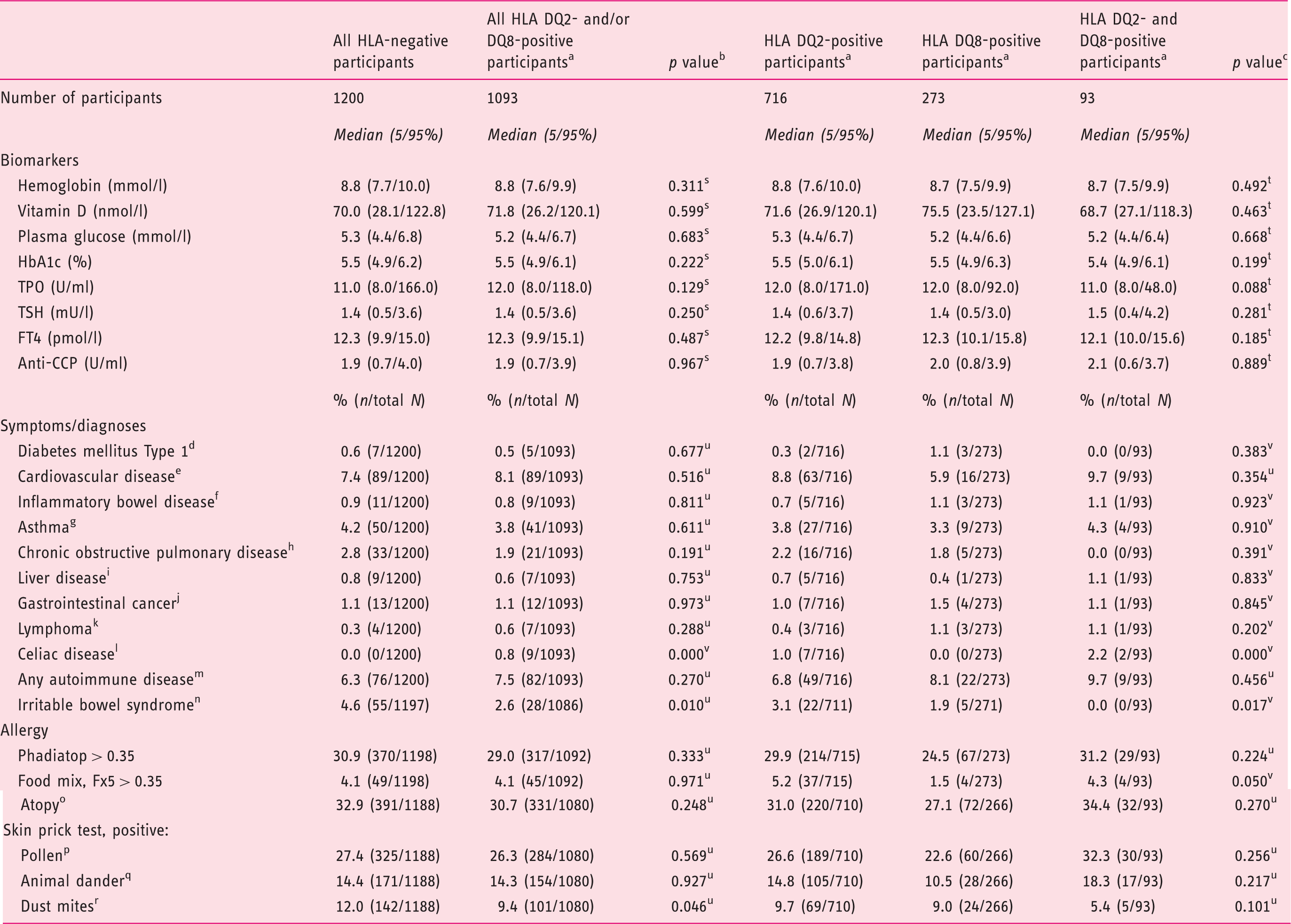
The participants were screened for HLA DQ8 and HLA DQ2 with the Celiac Gene Screen test, and the alleles associated with CD were identified by Celiac Gene Alleles test (BioDiagene, Palermo, Italy, distributed by Thermo Fisher Scientific, Allerød, Denmark).
Diabetes mellitus Type 1: ICD-8: 249, ICD-10: E10.
Including: ischemic heart disease (ICD-8: 410–414, ICD-10: I20–I25) and stroke (ICD-8: 431–434, 436, ICD-10: I60–I69).
Including: Crohn’s disease (ICD-8: 563.01, ICD-10: K50) and ulcerative colitis (ICD-8: 563.19, ICD-10: K51).
Asthma: ICD-8: 493, ICD-10: J45, J46.
Chronic obstructive pulmonary disease: ICD-8: 491–492 and ICD-10: J42–J44.
Liver disease: ICD-8: 570–573 and ICD-10: K70–K77.
Gastrointestinal cancer: ICD-8: 140–154 and ICD-10: C00–C18, C20–C21.
Lymphoma: ICD-8: 200-202 and 275.59 and ICD-10: C81–86, C88 and C915.
Celiac disease: ICD-8: 269.00 and ICD-10: K90.0.
aIncluding thyrotoxicosis(ICD-8: 242.00, ICD-10: E05.0), type 1 diabetes (ICD-8: 249, ICD-10: E10), multiple sclerosis (ICD-8: 340, ICD-10: G35), iridocyclitis (ICD-8: 364, ICD-10: H20), Crohn’s disease (ICD-8: 563.01, ICD-10: K50), ulcerative colitis (ICD-8: 563.19, ICD-10: K51), psoriasis vulgaris (ICD-8: 696.09–10, 696.19, ICD-10: L40), seropositive rheumatoid arthritis (ICD-8: 712.19, 712.39, 712.59, ICD-10: M05–M06), polymyalgia rheumatica (ICD-8: 446.30–31, 446.39, ICD-10: M31.5–6, M35.3), pernicious anemia (ICD-8: 281.0, ICD-10: D51.0), autoimmune hemolytic anemia (ICD-8: 283.90–91, ICD-10: D59.1), idiopathic thrombocytopenic purpura (ICD-8: 446.49, ICD-10: D69.3), autoimmune thyroiditis (ICD-8: 245.03, ICD-10: E06.3), primary adrenocortical insufficiency (ICD-8: 255.1, ICD-10: E27.1), Guillain-Barré syndrome (ICD-8: 354, ICD-10: G61.0), autoimmune hepatitis (ICD-8: 571.93, ICD-10: K75.4), primary biliary cirrhosis (ICD-8: 571.90, ICD-10: K74.3), celiac disease (ICD-8: 269.00, ICD-10: K90.0), pemphigus (ICD-8: 694, ICD-10: L10), pemphigoid (ICD-8: 694.05, ICD-10: L12), alopecia areata (ICD-8: 704.00, ICD-10: L63), vitiligo (ICD-8: 709.01, ICD-10: L80), juvenile arthritis (ICD-8: 712.09, ICD-10: M08), Wegener’s granulomatosis (ICD-8: 446.29, ICD-10: M31.3), dermatopolymyositis (ICD-8: 716, ICD-10: M33), myasthenia gravis (ICD-8: 733.09, ICD-10: G70.0), systemic sclerosis (ICD-8: 734.0, ICD-10: M34), systemic lupus erythematosus (ICD-8: 734.19, ICD-10: M32), Sjögren’s syndrome (ICD-8: 734.90, ICD-10: M35.0), and nkylosing spondylitis (ICD-8: 712.49, ICD-10: M45).
Irritable bowel syndrome is defined as a cluster of frequent self-reported gastrointestinal symptoms during the past 12 months: “Subjects stating that they often experience both abdominal pain and distension and additionally, either borborygmi or altering stool consistency, or both.”
Atopy defined as a positive reaction to at least one of 10 allergens: birch, grass (
Pollen, at least one positive of birch, grass (
Animal dander, at least one positive for horse, cat or dog.
Dust mites, at least one positive for one of two house dust mites:
Wilcoxon test.
Kruskal–Wallis test.
Chi-square test.
Fisher’s exact test.
Anti-CCP: anti-cyclic citrullinated peptide; CD: celiac disease; FT4: free thyroxine; HbA1c: hemoglobin A1c; HLA: human leukocyte antigen; ICD-8: International Statistical Classification of Diseases and Related Health Problems, eighth revision; ICD-10: International Statistical Classification of Diseases and Related Health Problems, 10th revision; TPO: thyroperoxidase; TSH: thyroid-stimulating hormone.
Statistical analysis
To investigate the association between variables, different tests were used: Wilcoxon test, Kruskal–Wallis tests and independent samples
Ethics
Informed written consent was obtained from all participants prior to participation. This study was approved by the Ethics Committee of the Capital Region of Denmark (H-3-2011-081, September 6, 2011), and we followed the recommendations of the 1975 Declaration of Helsinki.
Results
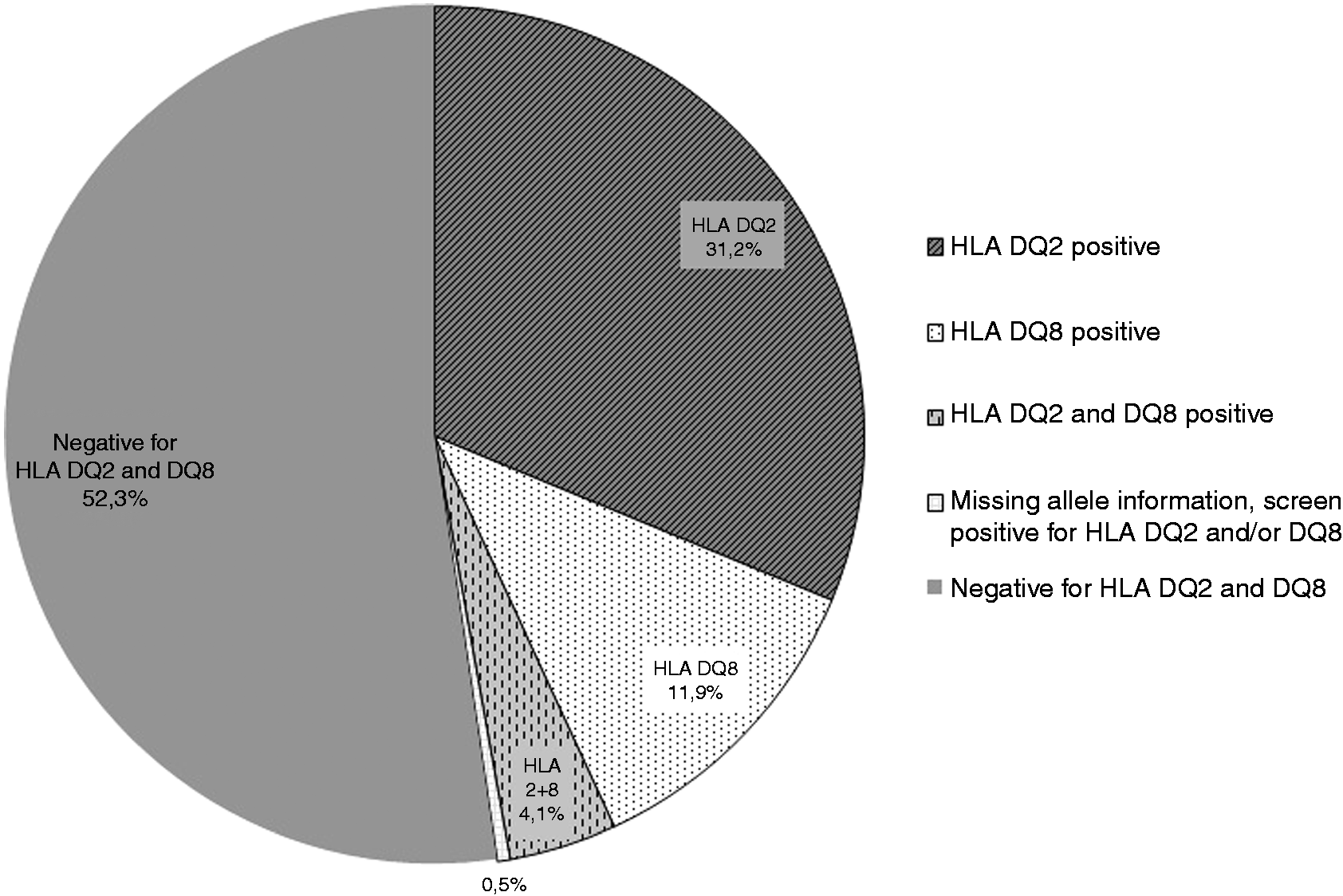
Figure 1 illustrates the flow of participants. A total of 2293 individuals were screened for HLA DQ2 and HLA DQ8 and 1093 (48%) were positive. Among the HLA DQ2- and HLA DQ8-positive participants, 1082 (99%) were allele classified. We found that 31% were DQ2 positive, 12% were DQ8 positive and 4% were both DQ2 and DQ8 positive (Figure 2).
Overview of study and flow of participants in the cohort. Prevalence of celiac disease predisposition in a Danish general population.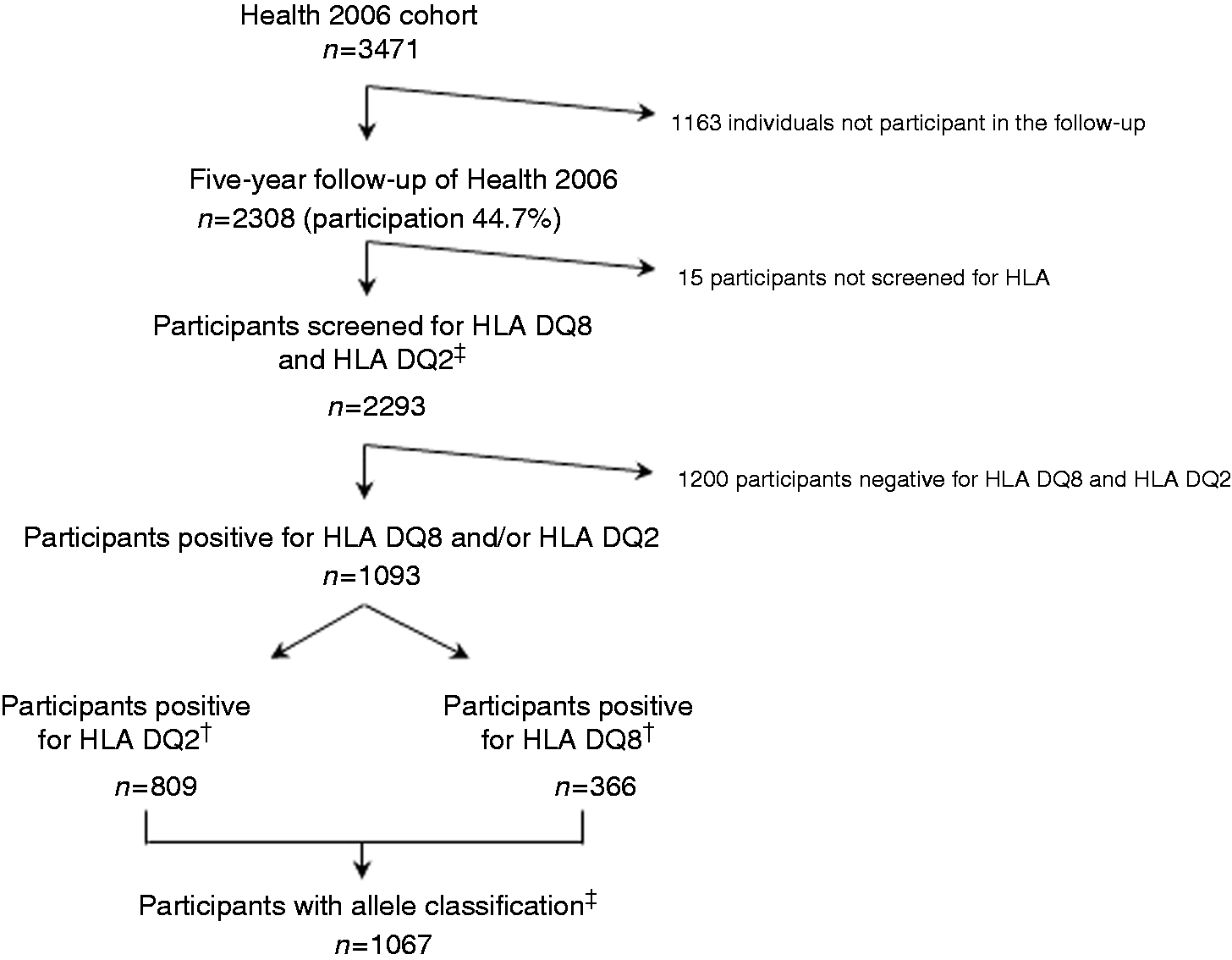
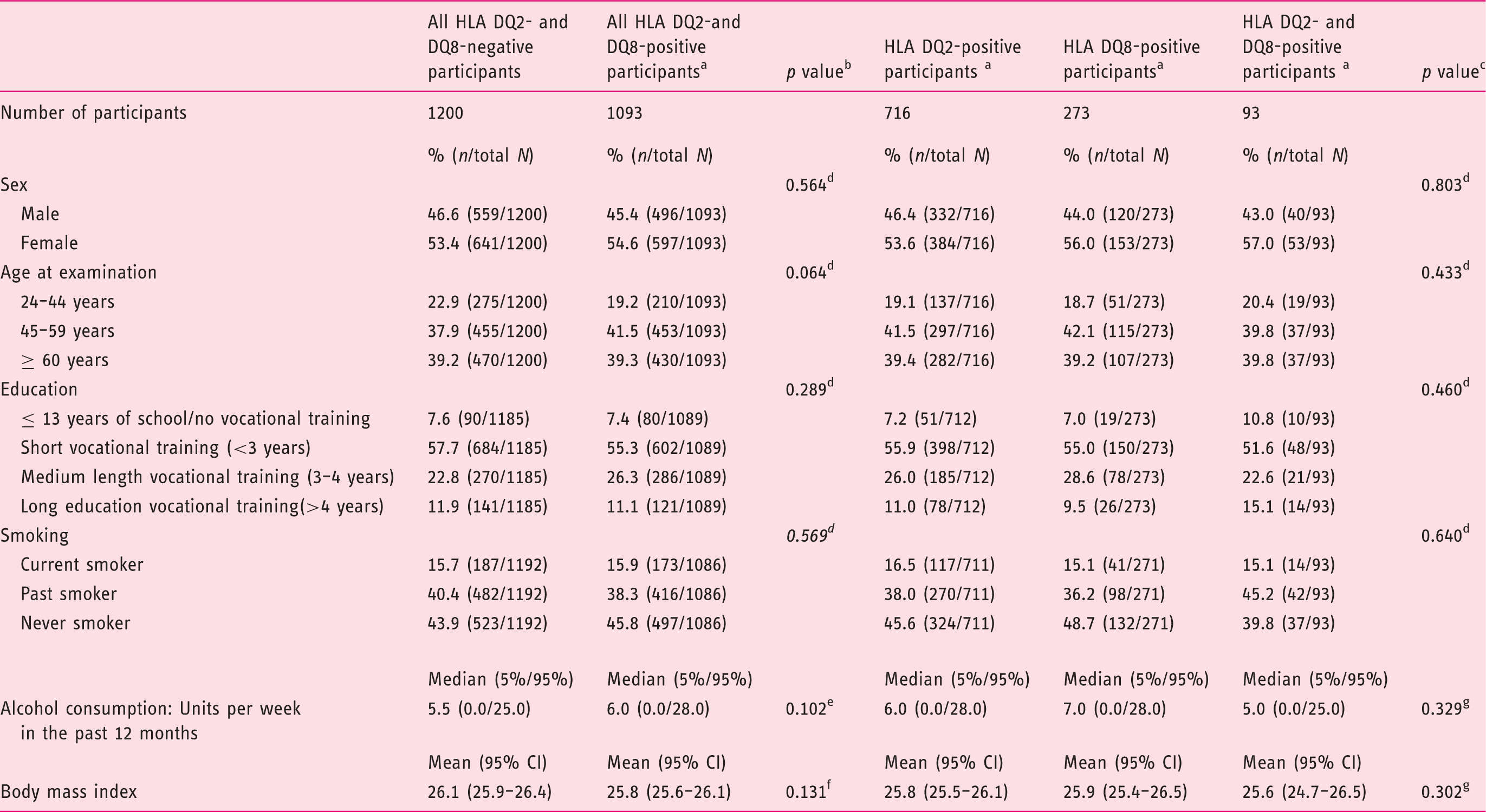
Characteristics of the participants positive and negative for HLA DQ2 and HLA DQ8.
The participants were screened for HLA DQ8 and HLA DQ2 with the Celiac Gene Screen test, and the alleles associated with CD were identified by the Celiac Gene Alleles test (BioDiagene, Palermo, Italy, distributed by Thermo Fisher Scientific, Allerød, Denmark).
Chi-square test.
Wilcoxon two-sample test.
Independent samples
Kruskal–Wallis test.
CD: celiac disease; HLA: human leukocyte antigen.
Results of biochemical analyses, symptoms and diagnoses stratified by HLA haplotypes DQ2.5, DQ2.2, DQ2.3 and DQ8 among participants in the Health2006 five-year follow-up.
The participants were screened for HLA DQ8 and HLA DQ2 with the Celiac Gene Screen test, and the alleles associated with CD were identified by Celiac Gene Alleles test (BioDiagene, Palermo, Italy, distributed by Thermo Fisher Scientific, Allerød, Denmark).
DQ2.5cis: DQB1*02, DQA1*05 and DQ2.5trans: encoded by DQA1*05-DQB1*03:01 and DQA1*02:01-DQB1*02.
DQ2.2: DQB1*02, DQA1*0201.
DQ8: DQA1*03, DQB1*0302/0305.
DQ2.3: DQB1*02, DQA1*03.
Diabetes mellitus Type 1: ICD-8: 249, ICD-10: E10.
Including: ischemic heart disease (ICD-8: 410–414, ICD-10: I20–I25) and stroke (ICD-8: 431–434, 436, ICD-10: I60–I69).
Including: Crohn’s disease (ICD-8: 563.01, ICD-10: K50) and ulcerative colitis (ICD-8: 563.19, ICD-10: K51).
Asthma: ICD-8: 493, ICD-10: J45, J46.
Chronic obstructive pulmonary disease: ICD-8: 491–492 and ICD-10: J42–J44.
Liver disease: ICD-8: 570–573 and ICD-10: K70–K77.
Gastrointestinal cancer: ICD-8: 140–154 and ICD-10: C00–C18, C20–C21.
Lymphoma: ICD-8: 200–202 and 275.59 and ICD-10: C81–86, C88 and C915.
Celiac disease, ICD-8: 269.00 and ICD-10: K90.0.
aIncluding thyrotoxicosis (ICD-8: 242.00, ICD-10: E05.0), Type 1 diabetes (ICD-8: 249, ICD-10: E10), multiple sclerosis (ICD-8: 340, ICD-10: G35), iridocyclitis (ICD-8: 364, ICD-10: H20), Crohn’s disease (ICD-8: 563.01, ICD-10: K50), ulcerative colitis (ICD-8: 563.19, ICD-10: K51), psoriasis vulgaris (ICD-8: 696.09–10, 696.19, ICD-10: L40), seropositive rheumatoid arthritis (ICD-8: 712.19, 712.39, 712.59, ICD-10: M05–M06), polymyalgia rheumatica (ICD-8: 446.30–31, 446.39, ICD-10: M31.5–6, M35.3), pernicious anemia (ICD-8: 281.0, ICD-10: D51.0), autoimmune hemolytic anemia (ICD-8: 283.90–91, ICD-10: D59.1), idiopathic thrombocytopenic purpura (ICD-8: 446.49, ICD-10: D69.3), autoimmune thyroiditis (ICD-8: 245.03, ICD-10: E06.3), primary adrenocortical insufficiency (ICD-8: 255.1, ICD-10: E27.1), Guillain-Barré syndrome (ICD-8: 354, ICD-10: G61.0), autoimmune hepatitis (ICD-8: 571.93, ICD-10: K75.4), primary biliary cirrhosis (ICD-8: 571.90, ICD-10: K74.3), celiac disease (ICD-8: 269.00, ICD-10: K90.0), pemphigus (ICD-8: 694, ICD-10: L10), pemphigoid (ICD-8: 694.05, ICD-10: L12), alopecia areata (ICD-8: 704.00, ICD-10: L63), vitiligo (ICD-8: 709.01, ICD-10: L80), juvenile arthritis (ICD-8: 712.09, ICD-10: M08), Wegener’s granulomatosis (ICD-8: 446.29, ICD-10: M31.3), dermatopolymyositis (ICD-8: 716, ICD-10: M33), myasthenia gravis (ICD-8: 733.09, ICD-10: G70.0), systemic sclerosis (ICD-8: 734.0, ICD-10: M34), systemic lupus erythematosus (ICD-8: 734.19, ICD-10: M32), Sjögren’s syndrome (ICD-8: 734.90, ICD-10: M35.0), and nkylosing spondylitis (ICD-8: 712.49, ICD-10: M45).
Irritable bowel syndrome is defined as a cluster of frequent self-reported gastrointestinal symptoms during the past 12 months: “Subjects stating that they often experience both abdominal pain and distension and additionally, either borborygmi or altering stool consistency, or both.”
Atopy defined as a positive reaction to at least one of 10 allergens: birch, grass (
Pollen, at least one positive of birch, grass (
Animal dander, at least one positive for horse, cat or dog.
Dust mites, at least one positive for one of two house dust mites:
Wilcoxon test.
Chi-square test.
Fisher’s exact test (
Anti-CCP: anti-cyclic citrullinated peptide; CD: celiac disease; FT4: free thyroxine; HbA1c: hemoglobin A1c; HLA: human leukocyte antigen; ICD-8: International Statistical Classification of Diseases and Related Health Problems, eighth revision; ICD-10: International Statistical Classification of Diseases and Related Health Problems, 10th revision; TPO: thyroperoxidase; TSH: thyroid-stimulating hormone.
We also compared the different groups on allergy parameters, IgE tests and skin prick test. When comparing the HLA DQ2/DQ8-positive with the HLA-negative participants, we found that the negative had a significantly higher number of positive skin prick tests for dust mites; however, there were no other significant differences with respect to allergy (Table 1). The same results were found when comparing the HLA-negative participants with all HLA DQ2.5-positive and all HLA DQ8-positive separately, but the haplotypes DQ2.2 and DQ2.3 did not have the same low number of positive skin prick tests for dust mites (Table 3). However, when comparing the four groups (described above), we found no significant differences among dust mite allergy, although there were significant differences among the groups when screened for serum-specific IgE to food allergens by food mix (Fx5) (Table 1). This difference was not found in the analysis where haplotypes were compared separately (Table 3).
We found no significant associations with respect to different diagnosis groups from the National Patient Registry, apart from a significantly higher prevalence of CD among HLA DQ2- and HLA DQ8-positive individuals as expected (Table 1).
Looking at IBS, we found significant differences between HLA DQ2/DQ8-negative and -positive, as well as the four groups (described above), showing that HLA DQ2/DQ8-positive participants had a significantly lower frequency of IBS (Table 1). This difference did not remain significant in the separate analyses of HLA DQ2.5-, DQ2.2- and DQ2.3-positive participants (Table 3).
All analyses were also conducted separately for individuals without CD, but the exclusion of CD did not change the results (data not shown).
HLA DQ2 and HLA DQ8 haplotype distribution in a general Danish population.
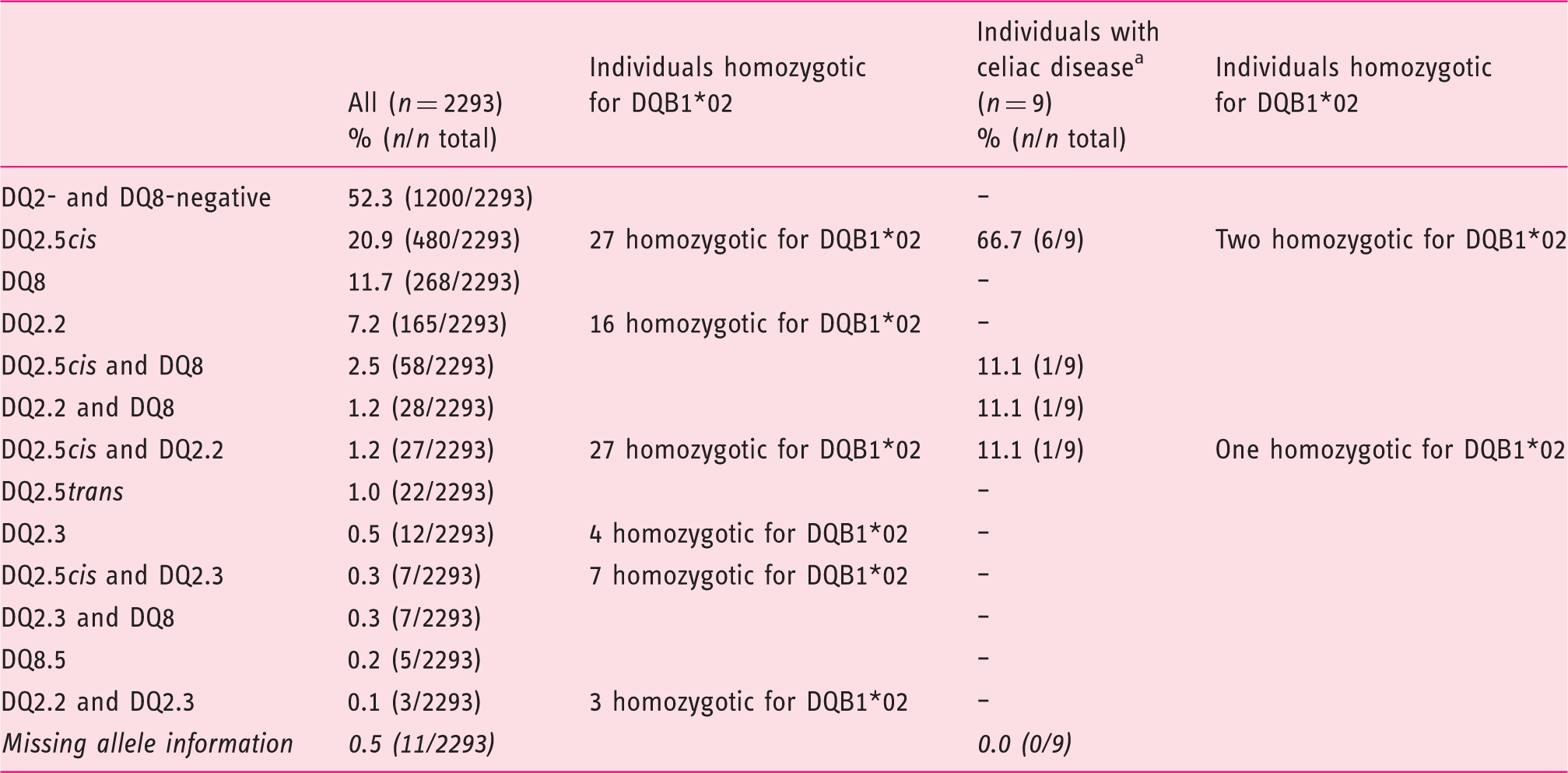
Celiac disease cases: eight found by screening (published earlier 12 ) and one with known celiac disease from the National Patient Registry.
HLA: human leukocyte antigen.
Allele distribution in a general Danish population.

Discussion
In this general Danish population, 47.7% were positive for either HLA DQ2 or DQ8 or both. All individuals with CD were HLA DQ2 and/or HLA DQ8 positive, the majority HLA DQ2.5 being positive. We compared the different HLA DQ2 and HLA DQ8 groups with respect to biomarkers, diagnoses, symptoms and allergy test results and found that HLA DQ2.5 was associated with higher TSH levels, while HLA DQ2/DQ8-positive participants had significantly lower frequencies of IBS. There was no statistically significant association between HLA DQ2/DQ8 and diagnoses other than CD.
Surprisingly, we found that this general Danish population had a higher prevalence of HLA DQ2/DQ8 than reported in other studies,3,22–24 although the prevalence in Denmark of CD has been reported to be lower than other European countries even though it is increasing.
25
The lower prevalence of CD in Denmark may be due to differences in exposure to environmental risk factors or underdiagnosis of CD. Our study extends the results from other studies since DQ2.5 (both
Our findings suggest that measuring total HLA DQ2/8 risk alleles as compared with only DQ2.5
As it is known that CD and diabetes Type 1 share links to HLA,9,10,32 we had expected an association between Type 1 diabetes and HLA DQ2/DQ8, but we found no significant difference among the groups with respect to diabetes. This might be due to a small proportion of individuals with diabetes Type 1 in the cohort, caused by selection for the study population and low prevalence of diabetes Type 1, as well as the fact that susceptible HLA antigens for diabetes Type 1 are more numerous than for CD, giving less power. We found no association between HLA DQ2/DQ8 positivity and autoimmune diseases,9–11 which might be due to a lack of power; however, twin studies have shown that CD is more highly genetically determined than other autoimmune diseases. 12 Furthermore, we found an association between TSH and DQ2.5 that has not been previously known, and not found for the other thyroid markers (TPO, FT4). A reason could be that TSH is a more sensitive marker for hypothyroidism, as well as the fact that TSH is a quantitative parameter measured in all participants, giving more power to detect differences.
In line with a study from Italy, 24 we found that the proportion of IBS was lower among HLA DQ2/8-positive individuals. However, the Italian study also found that IBD and liver diseases were more prevalent among HLADQ2/8-positive individuals, which we could not confirm in this present study. Other studies have found IBS symptoms to be more prevalent in patients with CD33,34; however, none of the nine individuals with CD in this present study had IBS according to our IBS definition.
We found a significant difference between HLA DQ2/8-positive and -negative individuals for dust mite allergy. HLA has earlier been associated with allergy, playing a role in antigen/allergen presentation35,36 and IgE dysregulation 37 ; however, the association is not clear and different alleles and allergies have been tested. Few studies have associated HLA DQ2/DQ8 with allergy. One found an association between DQ8 and hevein-specific IgE immune response in individuals with latex allergy, 38 while others found DQ2 to be associated with olive pollen 39 and the allele DQB1*0301 to be associated with grass pollen allergy. 36 Thus, the association of HLA DQ2/8 with allergy remains unclear.
The population-based design and the screening of all participants both for CD antibodies and for HLA DQ2/DQ8 are major strengths of our study. However, the relatively low prevalence of CD limits the possibilities of comparing participants with and without CD. Our study is limited to the HLAs known to be associated with CD and we were therefore not able to describe other HLA alleles, and since the test is an SSP-PCR for specific alleles, we cannot be sure about the typing with respect to homozygosity and heterozygosity on the alleles other than DQB1*02. Selection bias is not likely as we have no reason to believe that HLA genotype is associated with participation in the study. The linkage to the National Patient Registry is a strength that gives us the possibility of accurate information about diagnoses. However, there is a risk of misclassification in the registries that can influence the frequencies of diseases. There is also a risk of misclassification of IBS that was based on the self-reported questionnaire for IBS symptoms, and we had no information on duration and severity. Also, the differences between the IBS definition used and the Rome criteria could lead to misclassification of IBS and inconsistent results between studies.
In conclusion, 48% of individuals were positive for either HLA DQ2 or HLA DQ8 or both in this general Danish population and thus potentially at risk for CD. All individuals with CD were HLA DQ2 and/or DQ8 positive, and the majority HLA DQ2.5. We found that HLA DQ2.5 was associated with higher TSH levels, while HLA DQ2- and/or HLA DQ8-positive participants had a significantly lower prevalence of IBS. HLA DQ2/DQ8 was strongly associated with CD, but no other registry-based diagnoses.
Supplemental Material
Supplementary table -Supplemental material for The distribution of HLA DQ2 and DQ8 haplotypes and their association with health indicators in a general Danish population
Supplemental material, Supplementary table for The distribution of HLA DQ2 and DQ8 haplotypes and their association with health indicators in a general Danish population by Line Lund Kårhus, Betina H Thuesen, Tea Skaaby, Jüri J Rumessen and Allan Linneberg in United European Gastroenterology Journal
Footnotes
Acknowledgements
We would like to thank all staff members at the Research Centre for Prevention and Health for contributing to the data collection and health examinations. We would also like to thank Thermo Fisher Scientific, Allerød, Denmark; Lone Søgaard and Bjarne Kristiensen for their help with the HLA analyses and interpretation of the results; and Pia Schytte Hansen for performing the HLA screening.
Declaration of conflicting interests
None declared.
Funding
This work was supported by the Tryg Foundation (7-11-0213), Dansk Cøliaki Forening (the Danish Celiac Disease Patient Organization), and Thermo Fisher Scientific, Allerød, Denmark. Thermo Fisher Scientific performed the HLA screening.
Informed consent
Informed written consent was obtained from all participants prior to participation.
Ethics approval
This study was approved by the Ethics Committee of the Capital Region of Denmark (H-3-2011-081, September 6, 2011), and we followed the recommendations of the 1975 Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
