Abstract
Background
Although a 1-day low-fibre diet before colonoscopy is currently recommended, some endoscopists prescribe a 3-day diet.
Objective
The objective of this study was to compare the influence of a 3-day versus a 1-day low-fibre diet on bowel preparation quality, patient tolerability and adherence.
Methods
Outpatients scheduled for total colonoscopy were randomized in two groups, 3-day versus 1-day low-fibre diet, performing a 4-litre polyethylene glycol split-dose. The primary outcome was a reduction of inappropriate preparations in the 3-day low-fibre diet arm from 15% to 5% (bowel preparation was assessed by the Boston Bowel Preparation Scale). Secondary outcomes were adherence to, difficulty to perform, difficulty to obtain and willingness to repeat the diet. Intention-to-treat (ITT) and per-protocol (PP) analyses were conducted for the primary outcome.
Results
A total of 412 patients were randomized (206 per group). Bowel preparation quality was similar between groups. On ITT analysis (
Conclusion
A 3-day low-fibre diet does not bring benefit to the bowel preparation quality and is harder to perform than a 1-day diet.
Key Summary
Established knowledge on this subject
A 1-day low-fibre diet is the gold standard prior to colonoscopy. Some endoscopists recommend a 3-day low-fibre diet, although no randomized studies support that prescription.
What are the significant and/or new findings of this study?
A 3-day low-fibre diet did not bring advantages over the recommended 1-day diet in bowel preparation results, especially if performing split-dose. Longer diet times increase the complexity and difficulty of the prescription and may limit bowel preparation results.
Introduction
Colonoscopy is essential for the diagnosis and treatment of lower gastrointestinal tract disorders and is generally safe and accurate. Several quality indicators are described, such as bowel cleanliness, with recent guidelines suggesting the achievement of good preparation in at least 90% of all colonoscopies.1,2 Improving bowel preparation is a leading concern with several international guidelines in this area.3,4 Recent improvements in bowel preparation results have been made with the introduction of split-dose strategies, achieving a good preparation in 85% compared with 63% of those taking the non-split-dose, independently from the laxative. 5 Also the study of patient-related risk factors for inadequate bowel preparation contribute to a better preparation result, opening the possibility to design tailored strategies to improve outcomes.6–8
The influence of diet in bowel cleansing has also been studied with two recent meta-analyses summarizing the evidence in this area, recommending a low-fibre diet (LFD) as the standard diet for bowel preparation prior to colonoscopy, after several studies proving its non-inferiority against clear-liquid diets, with improved patient tolerability and no major differences in side-effects.9,10 An LFD is not properly defined, but some authors agree in a <10 g/day fibre intake.11,12
The European Society of Gastrointestinal Endoscopy recommends an LFD on the day preceding the examination. In addition, no recommendations are made regarding the use of an LFD for more than 24 hours before the procedure, although the experts recognize a group of endoscopists routinely prescribing the recommended diet for a 3-day period prior to the colonoscopy; however, at the time only one study is published comparing both dietary prescriptions.3,13
Therefore, the purpose of this study was to test the influence of a 3-day versus 1-day LFD in bowel preparation results and the effect on patient tolerability and adherence, in a general population of outpatients undergoing colonoscopy.
Methods
Study design
The design is a prospective, randomized, single-blind trial conducted at the endoscopy unit of the Gastroenterology Department of the Portuguese Oncology Institute of Coimbra, a tertiary referral hospital in the Central region of Portugal, for the diagnosis and treatment of malignant and pre-malignant conditions, during 2017 and 2018. The unit performs approximately 2000 colonoscopies per year, mainly in morning sessions.
The study protocol was approved by the local ethics committee (TI 07/16) and registered at ClinicalTrials.gov (NCT02955901).
Patients
All patients scheduled for total colonoscopy in the morning slots were invited to participate. Exclusion criteria were as follows: inpatients, colonoscopies under sedation, urgent procedures, colonoscopies not intended to reach the caecum, patient with previous colectomy, patient refusal to participate, and absolute contraindication to colonoscopy.
Process of enrolment
All patients were invited to participate at the outpatient facility. Upon acceptance, the doctor explained the study protocol, obtained both colonoscopy and study informed consent and recorded demographics, medical history and risk factors for inadequate bowel preparation. On that same visit the patient was randomized and received information regarding the diet scheme to perform. All patients were prescribed with the same split-dose regime with a 4-litre (L) polyethylene glycol (PEG) formulation. Patients were instructed to take a 3-L PEG preparation on the late afternoon of the day prior to the examination and a 1-L PEG preparation in the morning of the day of the examination, ending 4 hours before the scheduled examination time.
Randomization process and groups description
Simple randomization in a 1:1 sequence was obtained by a computer-generated table. Allocation concealment was done by sealed, numbered, opaque envelopes. Patients were not blinded to the intervention and could be randomized in one of two groups: D3 group, performing a 3-day LFD for all meals until dinner of the day before colonoscopy plus a split-dose 3 + 1L PEG; or D1 group, assigned a 1-day LFD for all meals until dinner of the day before colonoscopy with the same laxative. Diet suggestions were given in the outpatient clinic visit and are part of the in-house bowel preparation leaflet in use at the unit and previously revised by the Nutrition Department (online supplementary material – Appendix 1 s).
Colonoscopy procedures and data collection
All colonoscopies were performed in the morning sessions and were conducted by five board-certified gastroenterologists with more than 10 years of practice and two supervised fellows in training. Assessment of the bowel preparation was made according to the Boston Bowel Preparation Scale (BBPS), with a total BBPS <6 or a BBPS <2 in any segment being defined as inappropriate. All physicians were trained in the use of the BBPS by online training available at http://cori.org/bbps/, 14 and have been using the BBPS for at least 3 years, prior to the beginning of the study.
Before entering the endoscopy suite, the patient was interviewed by the department nurse collecting information regarding the adhesion to the prescribed diet and bowel preparation and instructed not to reveal to the gastroenterologist team the diet assigned. Complaints and side-effects were also registered. Patient self-assessment of the difficulty to perform and to buy the diet prescribed, as well the willingness to repeat the scheme was evaluated by a visual analogue scale (1–10; 1 representing the most favourable outcome and 10 the most unfavourable).
Variables collected
Patient-related variables
Patient variables collected were demographics, indication for colonoscopy, previous experience with split-dose and history of previous inadequate bowel preparation. Risk factors for inadequate bowel preparation were collected: age >65 years, body mass index >30 kg/m2, Parkinson’s disease, previous stroke, constipation (<3 weekly bowel movements), previous abdominal surgery, diabetes, opioid or tricyclic antidepressant intake, low educational level (basic education or inferior), and third-party dependency (Eastern Cooperative Oncology Group performance status ≥2). 15 Compliance with diet, preparation and split-dose timing were also collected, as well as any side-effects and complaints.
Colonoscopy-related variables
Colonoscopy variables collected were BBPS (overall and segmental); number, location and morphological classification of lesions according to the Paris classification; caecal intubation rate and reasons for not achieving it; withdrawal time for colonoscopies without any diagnostic or therapeutic procedures; and colonoscopy complications.
Statistical analysis and sample size calculation
When designing the study an internal audit pointed out a 15% rate of inadequate bowel preparations in our unit (unpublished data). We intended to achieve a reduction from 15% to 5% of inappropriate preparation result admitting that a 3-day LFD would be superior to 1-day LFD. Assuming a normal distribution, a power of 90% and a type I error of 0.05, the calculated sample size for each group was 188; allowing a 10% dropout rate, the sample size is 206 per group (412 patients overall). The two groups were compared using the chi-squared statistic for categorical and Student’s
Study outcomes
Primary outcome
This was to achieve a reduction from 15% to 5% of inappropriate preparation in the intervention arm; BBPS was used as the grading system, with inappropriate bowel preparation defined as total BBPS <6 or BBPS <2 in any segment.
Secondary outcome
This was to evaluate the patient’s compliance with diet, preparation and split-dose timing. A 10-point subjective visual analogue scale was used to evaluate the difficulty on performing and obtaining the recommended diet and the willingness to repeat the entire protocol. Adverse events were assessed asking the patients if they suffered from any of the following: fatigue, hunger, significant alteration in daily activities, no complaints; an open field was included for reporting other complaints.
Results
Out of 796 patients assessed for eligibility, a total of 412 patients were randomized, resulting in 412 patients for ITT analysis (206 in each group) (Figure 1). Both baseline characteristics and risk factors for inadequate bowel preparation result were equivalent between groups (Table 1). Twelve patients presented with deviations to the protocol: 4 did not attend colonoscopy, 3 had incomplete colonoscopies (due to patient intolerance), 3 were not compliant with the prescribed diet and 2 bought a different laxative than the one prescribed (Figure 1). In ITT analysis all these patients were assigned as having the most unfavourable result. Therefore, PP analysis for primary outcome had 400 patients, 199 in D3 group and 201 in D1 group (Figure 1).
Study flowchart. Characteristics of the study population and risk factors for inadequate bowel preparation (intention-to-treat analysis, Defined as <3 spontaneous bowel movements per week. Defined as an Eastern Cooperative Oncology Group performance status >2. Non-colic abdominal surgeries. Defined as basic or lower education. IQR: interquartile range.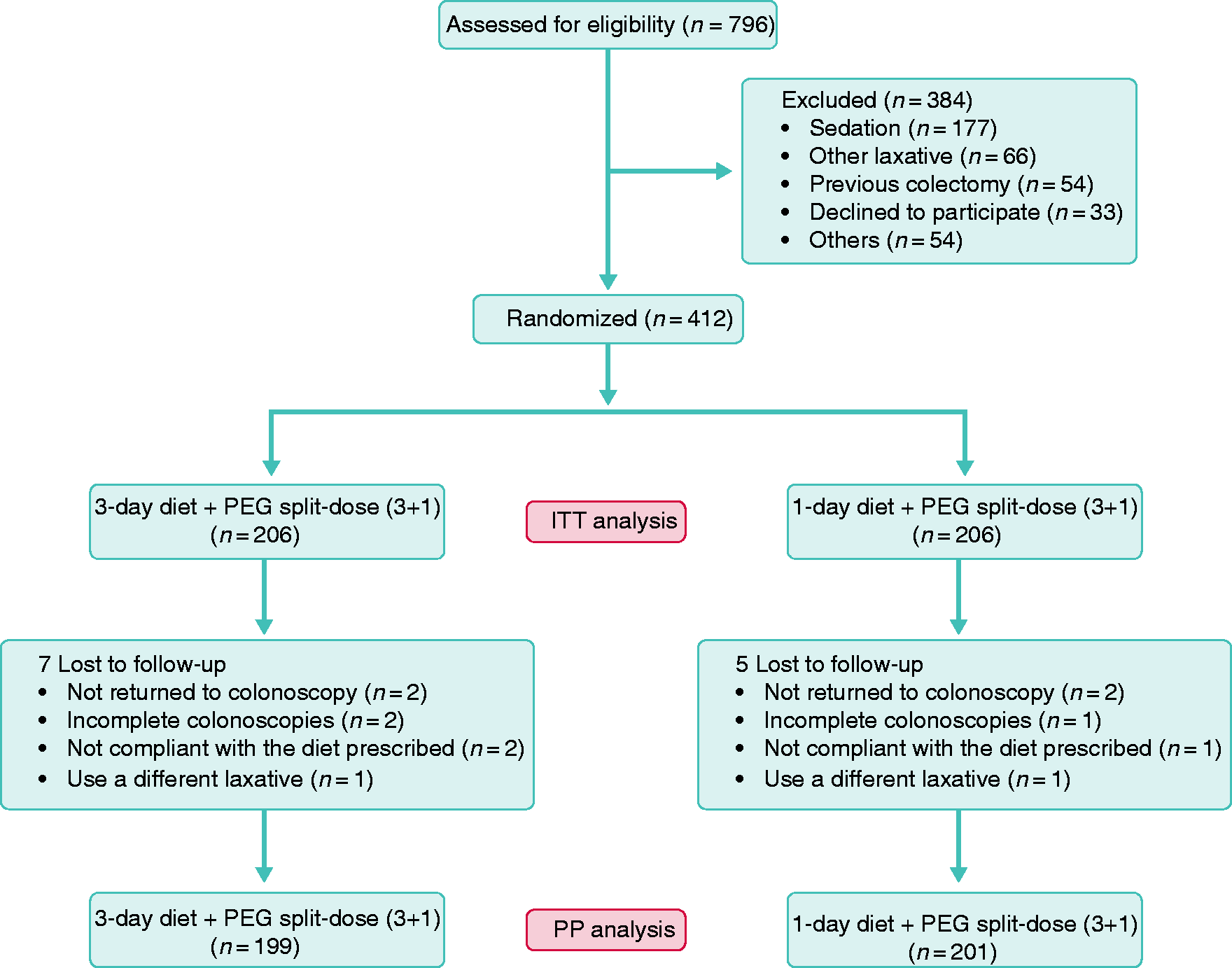
Primary outcomes
Comparison of intestinal preparation result between groups.
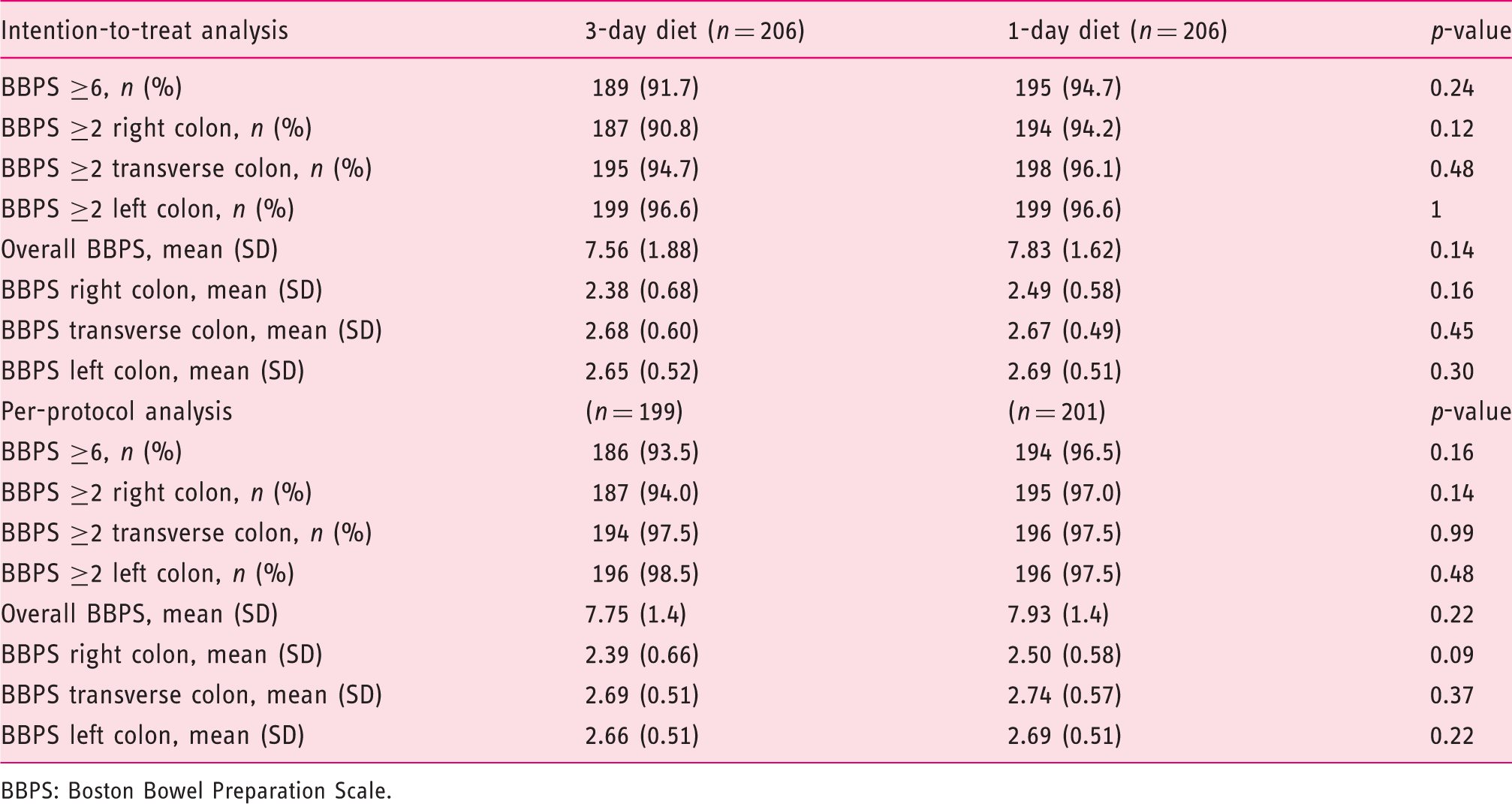
BBPS: Boston Bowel Preparation Scale.
Risk factors for inadequate bowel preparation
Risk factors for inadequate preparation (intention-to-treat analysis,
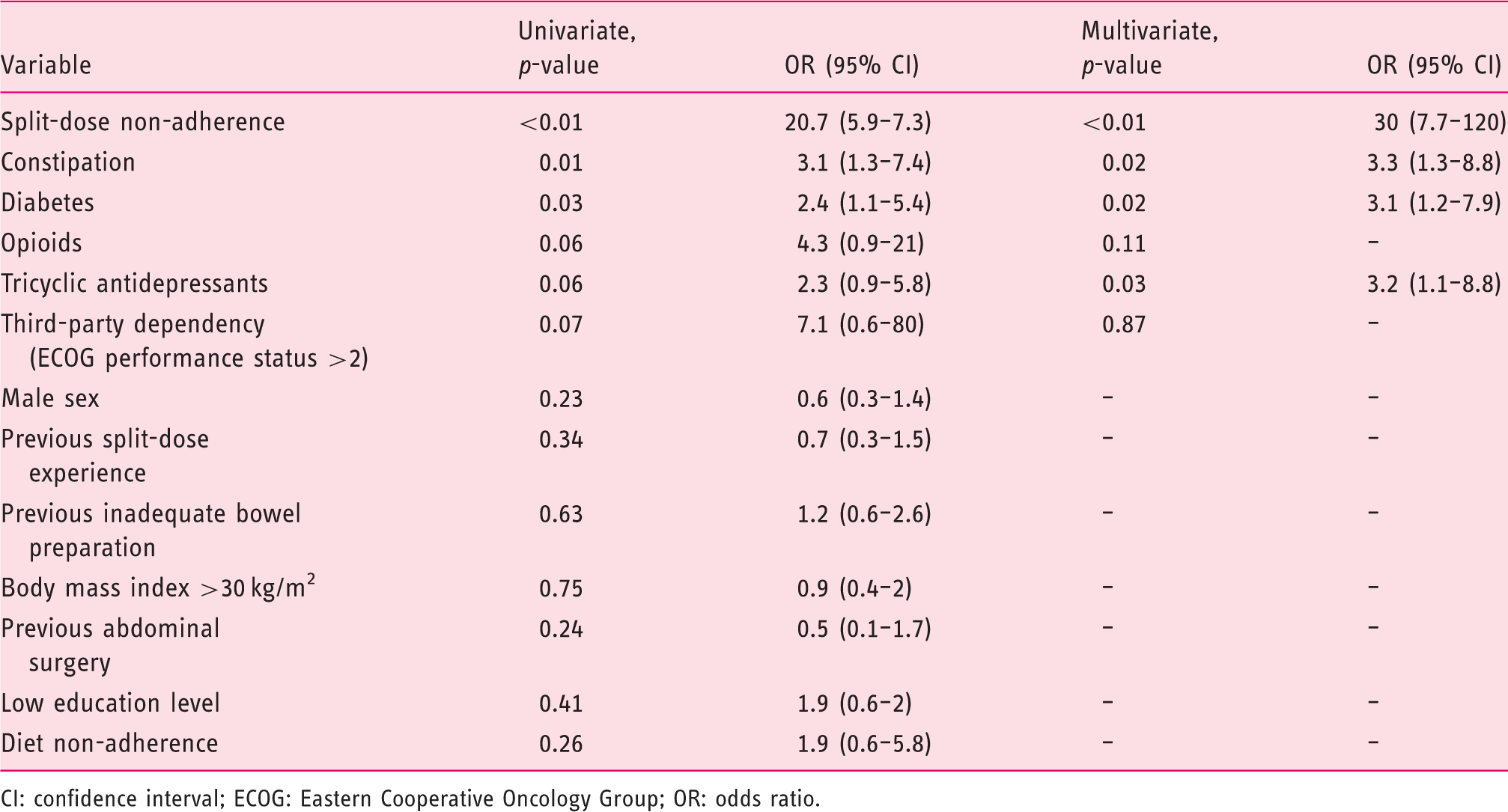
CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; OR: odds ratio.
Colonoscopy findings
Colonoscopy findings (intention-to-treat analysis,
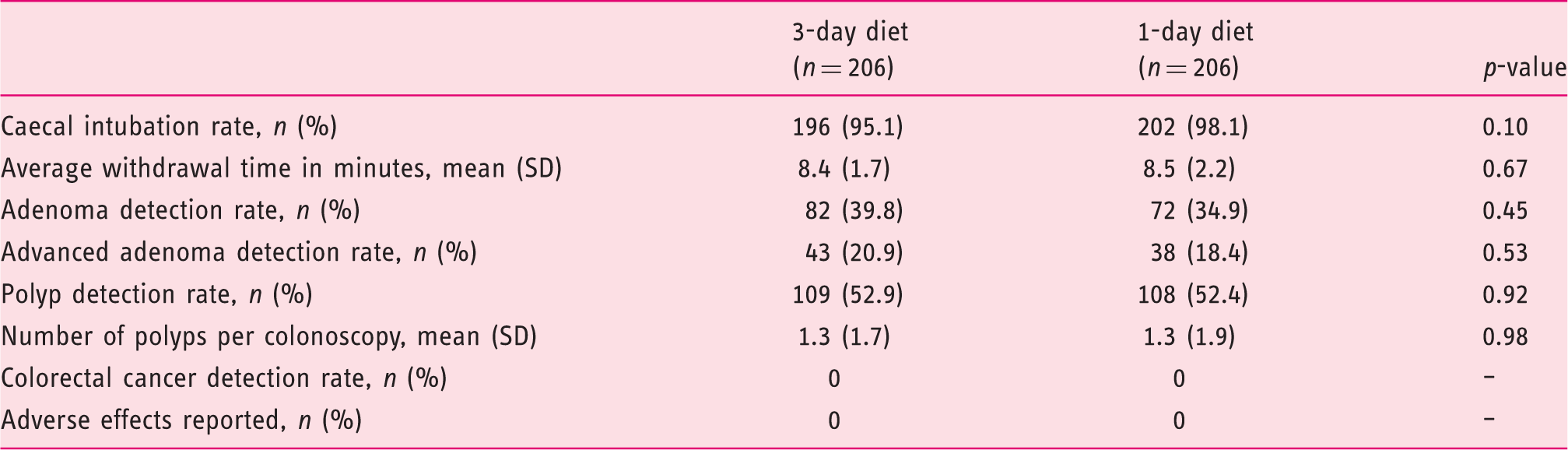
Secondary outcomes
Patient-related outcomes
Adverse events related to the prescribed preparation and diet (intention-to-treat analysis,
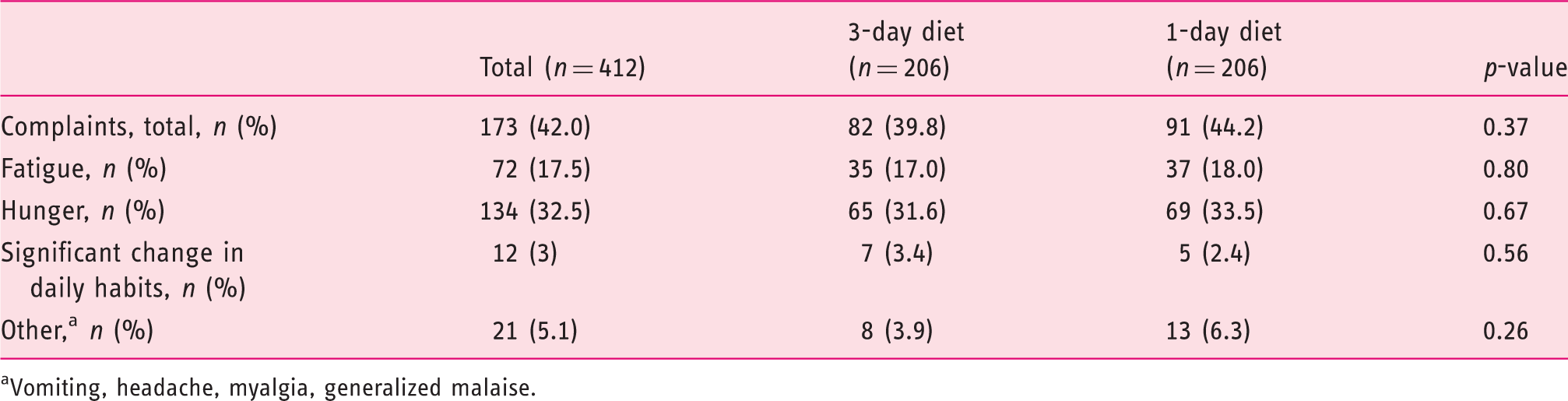
Vomiting, headache, myalgia, generalized malaise.
Diet-related outcomes (intention-to-treat analysis,
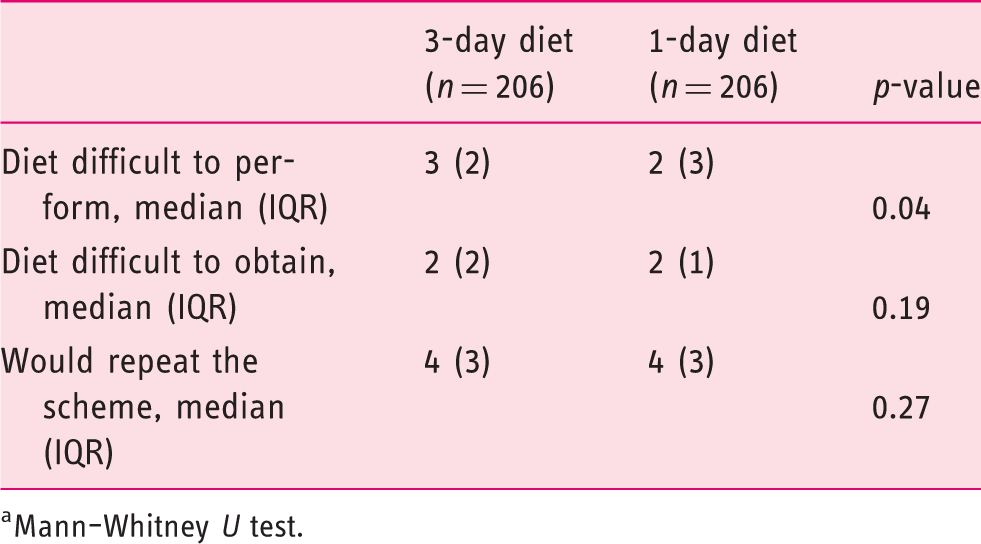
Mann–Whitney
Discussion
High standard colonoscopy will only be achieved with optimal bowel preparation and split-dose is fundamental to achieve it; however, diet prescription can have an important role. Current guidelines suggest a 1-day LFD prior to colonoscopy with moderate evidence, but there is no substantial data for extending the timing of diet. 3
The current randomized, controlled trial showed that a bowel preparation protocol based on a 3-day LFD associated with a standard 4L PEG split-dose preparation was not superior to 1-day LFD diet in the same conditions (91.7% versus 94.7%). Therefore, prescribing more than one day of an LFD in the general population in an outpatient clinic setting does not seem to be necessary. Furthermore, and although no differences were found between groups on the difficulty to obtain the diet and in the willingness to repeat, a significantly higher difficulty was verified when evaluating the difficulty to follow the 3-day diet, favouring the current recommendation. To our knowledge, this is the second randomized trial on this subject with a recent published work obtaining the same non-superiority results favouring the 1-day LFD, obtaining similar BBPS results in both groups (82.7% versus 85.6%). The same study also found a tendency towards the 1-day LFD when evaluating satisfaction and difficulty following the protocol, assuming this as the most favourable in those particular aspects, although with no statistical significance. 13 Also a recent observational study found that only the diet performed in the day prior to the colonoscopy had significant impact with the BBPS scores, when evaluating the diet in the 3 days before the colonoscopy, stating that multi-day dietary instructions can be confusing to patients and not adding significant value to the bowel preparation result. 16 A recent meta-analysis found no significant difference in the bowel preparation results with the use of an LFD or a clear-liquid–based diet in the day before colonoscopy; however, the tolerance and the willingness to repeat were significantly higher in the LFD cohorts. 10
At the current point we do not have an explanation for the failure of our intervention; however, both groups improved BBPS scores, within the recommended standards. 1 Dietary fibre influences colonic transit, for example insoluble fibre promotes a faster colonic transit by mechanical and neuroendocrine mechanisms.12,17 We can hypothesize that reducing this stimulus leads to a decrease in colon transit, contributing to justify the failure of our intervention. Also, the overall improvement of BBPS scores might be explained by the use of split-dose, since only around 30% of patients had split-dose previous experience, associated with the personalized intervention in the outpatient clinic at randomization, with influence as previously documented. 18 Therefore simplification of bowel preparation schemes appears to be decisive in the tolerability of the prescription, and possibly have influence in the results.
Poor bowel preparation is related to several factors studied in two recent meta-analyses19,20 suggesting that the influences of certain health conditions such as diabetes or the intake of tricyclic antidepressants have the higher burden causing inadequate bowel preparations, corroborating our results.
The strengths of our study are its design, a controlled, randomized trial addressing the issue of comparing the duration of an LFD prior to colonoscopy. It is easily reproducible by other groups in similar settings. The prescribed diet was reviewed by a nutrition specialist before the beginning of the study. The reproducibility and accuracy of the primary outcome is secured by the previous experience of the endoscopists on the use of BBPS, complemented with online training.
On the other hand, the single-centre design and our specific type of hospital can be limiting factors on reproducibility. Our cohort is homogenous; however, since we serve a very specific population, we currently do not follow patients with inflammatory bowel disease or cirrhosis, and therefore we cannot generalize our conclusions to those specific populations. Finally, our results are based in a standard 4L PEG solution and we cannot generalize our conclusion to other laxatives, although the similar protocol was recently published where a low-volume 2L PEG plus ascorbic acid was used with similar results. 13
Concluding, the recommendation of a 3-day LFD does not bring added value to the bowel preparation quality especially if performing split-dose. Increasing the time of diet can also increase the difficulty of the overall preparation, possibly undermining the result.
Supplemental Material
UEG883176 Supplemental Material - Supplemental material for A 3-day low-fibre diet does not improve colonoscopy preparation results compared to a 1-day diet: A randomized, single-blind, controlled trial
Supplemental material, UEG883176 Supplemental Material for A 3-day low-fibre diet does not improve colonoscopy preparation results compared to a 1-day diet: A randomized, single-blind, controlled trial by Filipe Taveira, Miguel Areia, Luís Elvas, Susana Alves, Daniel Brito, Sandra Saraiva and Ana T Cadime in United European Gastroenterology Journal
Footnotes
Author contribution
FT was responsible for conception and design of the study, analysis, interpretation of data, drafting the paper, and final approval of the version to be published. MA was responsible for conception and design of the study, analysis, interpretation of data, revising the draft for important intellectual content, and approval of the version to be published. LE, SA, DB, SS and ATC were responsible for acquisition of data, reviewing the draft for important intellectual content, and approval of the version to be published.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study protocol was in accordance with the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the local ethics committee.
Funding
This study was supported by a grant from the Portuguese Digestive Endoscopy Society (1/2016).
Informed consent
Written informed consent was obtained from all patients.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
