Abstract
Background
There are few large prospective cohort studies evaluating predictors of outcomes in acute pancreatitis.
Objectives
The purpose of this study was to determine the role of age and co-morbid disease in predicting major outcomes in acute pancreatitis.
Methods
Data points were collected according to a predefined electronic data collection form. Acute pancreatitis and its complications were defined according to the revised Atlanta classification. Univariable and multivariable analyses were conducted using Cox proportional hazard regression and multiple logistic regression.
Results
From June 2013–February 2015, 1655 adult patients were recruited from 23 centres across Spain. Co-morbid disease, obesity, open surgical necrosectomy within 30 days, and pancreatic necrosis were independently associated with both 30-day mortality and persistent organ failure (
Conclusion
Comorbidity and obesity are important determinates of mortality and persistent organ failure in acute pancreatitis, but in the absence of organ failure they do not appear to independently contribute to morbidity. This has important implications for severity classification and predictive models of severity in acute pancreatitis.
Introduction
Approximately 70% of patients with acute pancreatitis (AP) have mild disease and are discharged within a week, and only a minority of patients suffer from mortality during admission or develop significant morbidity requiring a longer hospital stay. 1
The key determinants of morbidity and mortality in AP are organ failure (OF) and infected pancreatic necrosis (IPN). While the revised Atlanta classification (RAC) of 2012 defined severe AP as persistent organ failure (POF), the determinant-based classification (DBC) defined the presence of either POF or IPN as severe AP.2,3 Despite an exacerbation of comorbid disease being defined as moderately severe AP in the RAC, there is a paucity of data evaluating the impact of comorbid disease on severity in AP.4,5 There have been a large number of studies evaluating a host of various clinical markers as predictors of severity in AP.6–9 However, the majority of these studies have either been in a single centre, had a small sample size or were derived from large administrative databases, thus suffering from problems with generalisability and reproducibility. In addition, few studies have incorporated comorbid disease into their clinical models when evaluating determinates of severity.
The primary aims of this study were two-fold. Firstly, to evaluate the influence of age, elevated body mass index (BMI), comorbid disease and a number of known predictors of severity on mortality and OF. Secondly, to evaluate the impact of elevated BMI and comorbid disease on both length of stay (LOS) and other markers of morbidity in AP.
Methods
The Atlantis project, a prospective cohort study, was created under the auspices of the Spanish Association of Pancreatology (AESPANC) and the Spanish Association of Gastroenterology (AEG), to ascertain the role of age, obesity and comorbidity on the course of AP as well as to validate and compare the determinants of severity and the severity classifications. The present study is focused on the first aim. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in approval from the institutional review board at the principal investigator's centre, Comité ético de investigación clínica del Hospital General Universitario de Alicante on 29 June 2014. The study was also approved by the relevant local ethical committee at each centre. Consent by the patient or their healthcare advocate was required for enrolment in the study.
Patients
Adult (≥18 years) patients with AP were prospectively enrolled at 23 Spanish centres (20 tertiary referral centres and three community centres) from June 2013–February 2015. AP was defined in accordance with the RAC. 2 Patients with documented chronic pancreatitis were excluded. Each centre had one to two gastroenterologists or surgeons responsible for recruiting the patients and collecting data, and a radiologist, blinded to the clinical course and outcomes, responsible for the description of local complications on computerised tomography (CT) imaging. CT imaging was not pursued in those patients with mild AP (self-limited abdominal pain, absence of clinical and/or laboratory markers of systemic inflammatory response, and successful oral refeeding). We assumed that these patients did not have local complications.
Definitions
Comorbidity was defined according to the Charlson Co-morbidity Index (CCI), age was not included in the score. 10
BMI, OF, transient organ failure (TOF), POF, inpatient mortality and LOS are defined in Supplementary Material Methods.
The radiologists at each centre recorded the presence of parenchymal and extrapancreatic necrosis based on the RAC definitions. IPN was defined as either parenchymal and/or extrapancreatic seen on CT with a positive culture obtained from the (peri) pancreatic bed. 11
Interventions were defined as the need for open surgical necrosectomy, transmural endoscopic drainage and/or necrosectomy of (peri) pancreatic collections or radiologically placed percutaneous drain during hospitilization.
Nutritional support was defined as the need for nasoenteric (NE) supplementary feeding or the need for total parenteral nutrition (TPN).
Statistical analysis
Continuous and categorical data were compared between groups using standard parametric and non-parametric tests. Time to event analysis was conducted using the Kaplan-Meier method with log rank testing. Censoring was performed at discharge from hospital. Multivariable Cox proportional hazard regression analyses were performed to evaluate the factors associated with 30-day inpatient mortality and POF. OF was not included in the model for mortality as OF is a known intermediary variable for mortality in AP, thus its inclusion in a mortality models result in an over-adjustment bias.
12
A description of how continuous variables were modelled and how missing data was handled is provided in the supplementary material methods statistical analysis. Multivariable logistic regression analysis was performed to determine the factors associated with a prolonged length of admission (top 25th centile). The results are presented as estimated hazard ratio (HR) or odds ratio (OR) with respective 95% confidence interval (CI) and
Results
Demographics of acute pancreatitis cohort.
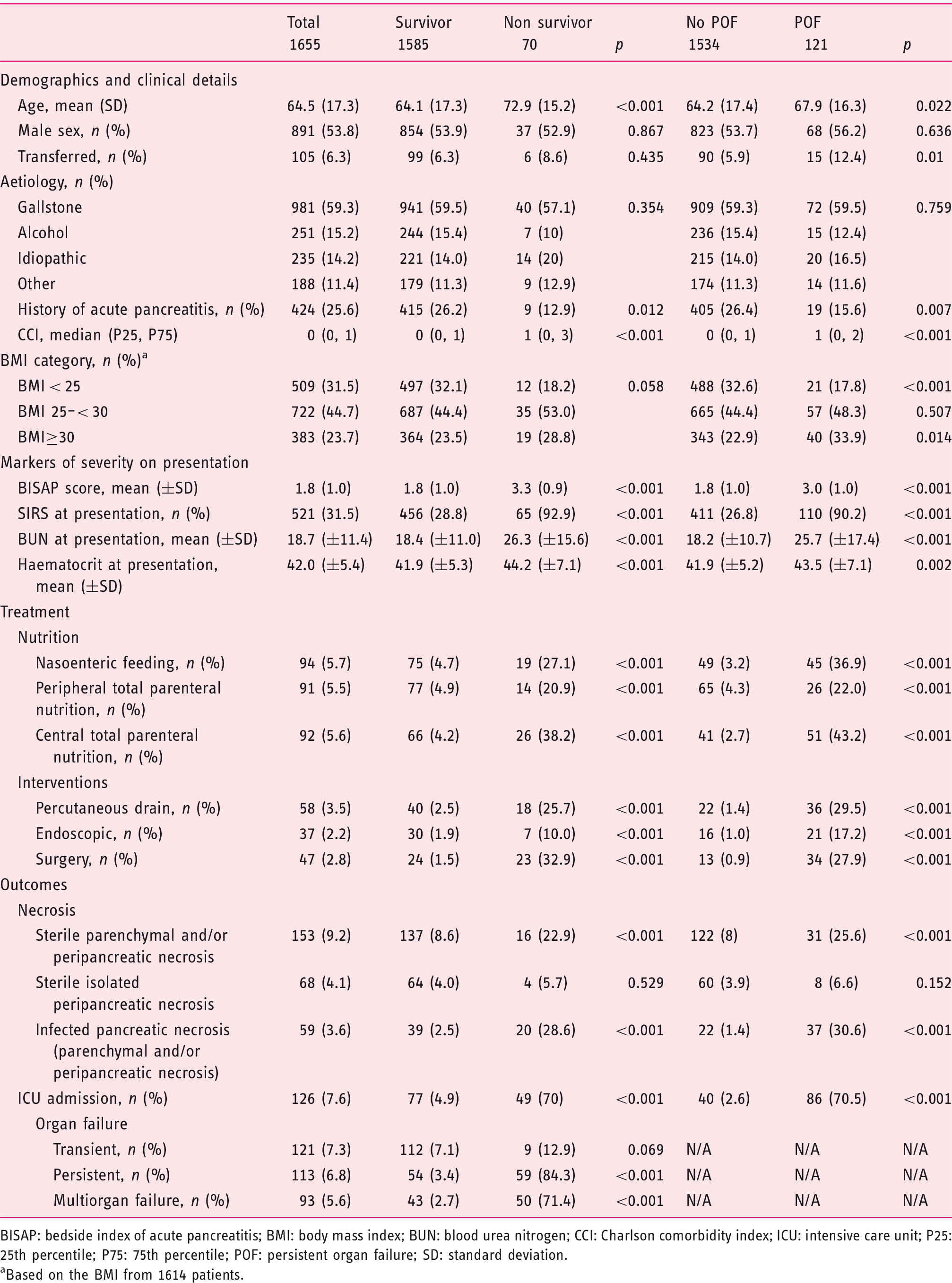
BISAP: bedside index of acute pancreatitis; BMI: body mass index; BUN: blood urea nitrogen; CCI: Charlson comorbidity index; ICU: intensive care unit; P25: 25th percentile; P75: 75th percentile; POF: persistent organ failure; SD: standard deviation.
Based on the BMI from 1614 patients.
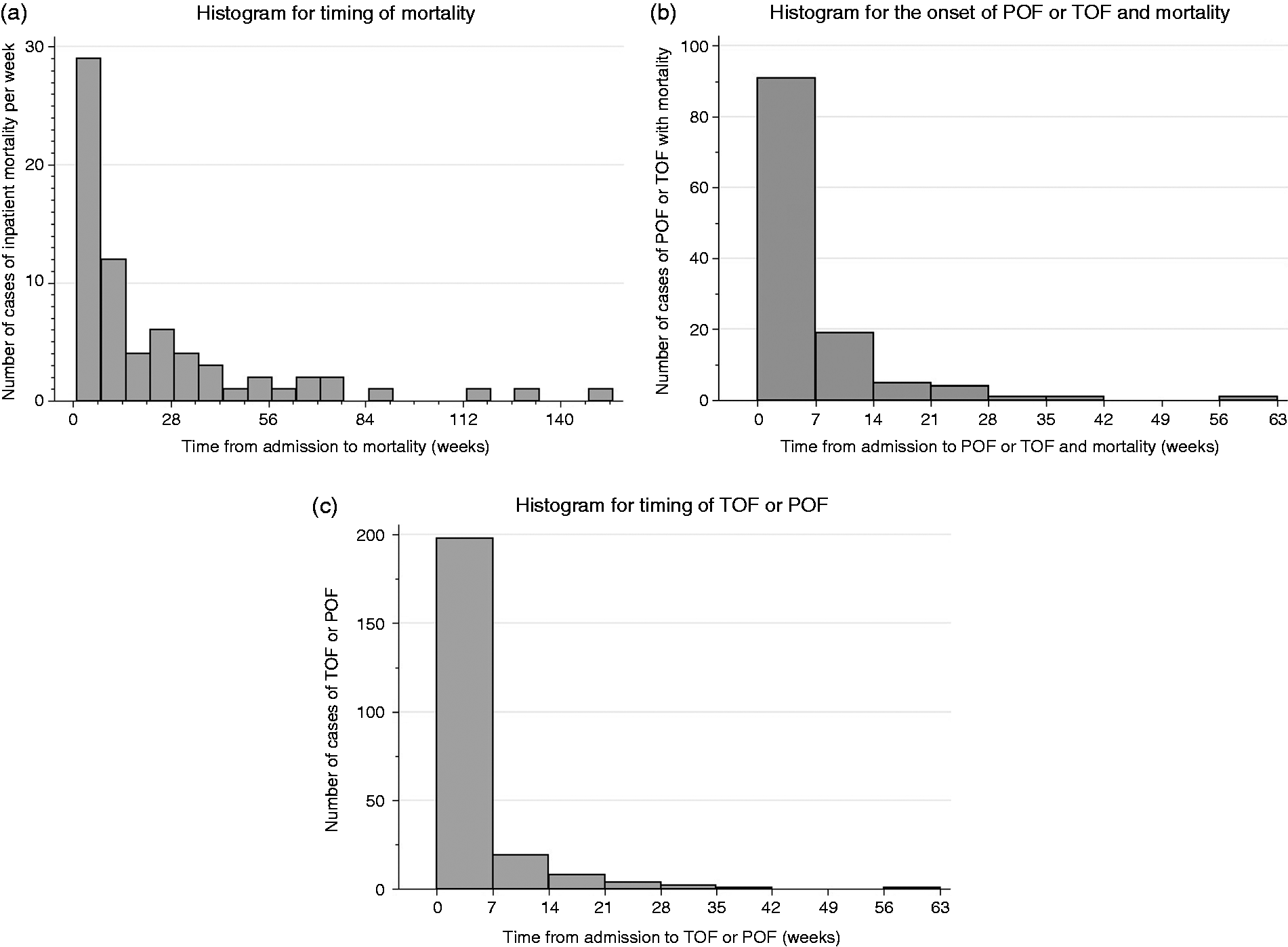
(a) Histogram for timing of mortality in weeks; (b) histogram for the onset of persistent organ failure (POF); (c) histogram for the onset of transient organ failure (TOF) or POF.
Of the 70 inpatient deaths, 59 patients had POF, nine had TOF and two died without OF. The cause of mortality in the nine patients who died of TOF is as follows: two patients died of sudden death, two died due to septic shock, one patient has an acute aspiration event, one patient had a stroke, one patient had an acute exacerbation of chronic obstructive lung disease and the cause of death was not reported in two patients. The two patients who did not have OF died of an acute aspiration event with subsequent cardiac arrest and the sudden onset of a fatal cardiac arrhythmia, respectively. These patients were 83 and 91 years old on admission, respectively.
30-Day inpatient mortality
Supplementary Material Figure 1 presents the Kaplan-Meier curves for 30-day inpatient mortality. Advanced age, higher CCI and BMI categories, parenchymal necrosis and open surgical necrosectomy within 30 days of presentation were all associated with increased risk of 30-day inpatient mortality on unadjusted and multivariable cox regression analysis (Figure 2 and Supplementary Material Table 1). Recurrent acute pancreatitis (RAP) was associated with a reduced risk of mortality on unadjusted ( Forest plot of the multivariable Cox model for the development of 30-day inpatient mortality. The Cox model has been adjusted for each listed variable in addition to acute recurrent pancreatitis, parenchymal necrosis, peripancreatic necrosis, infected pancreatic necrosis and early surgery within 30 days of presentation. BMI: body mass index; CCI: Charlson Comorbidity Index.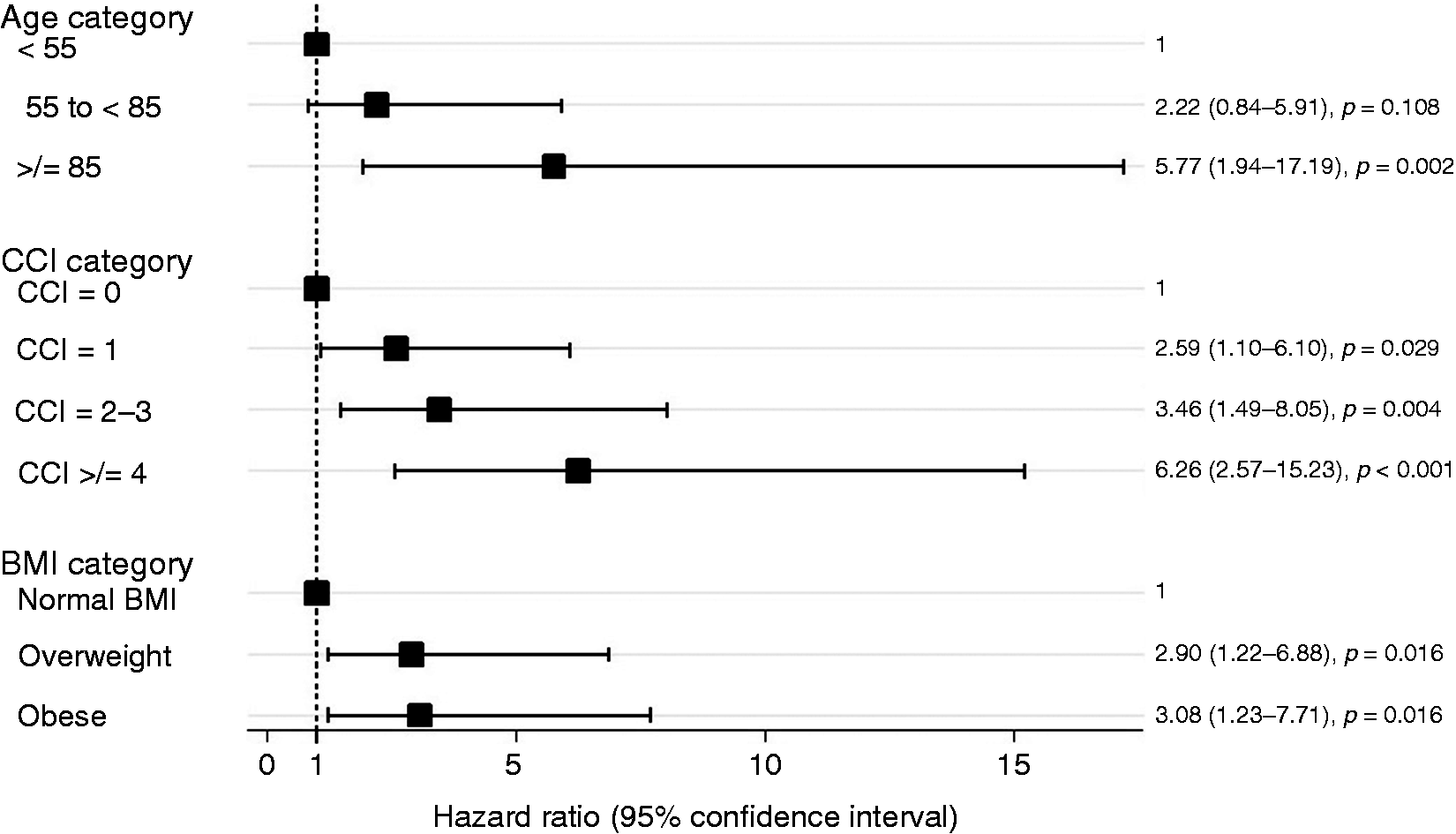
When the same multivariable Cox model was applied to total inpatient mortality, the model fit was superior for 30-day inpatient mortality as compared to total inpatient mortality (AIC: 592.65 vs 723.77). In addition, there was a violation of the proportional hazards assumption for the total inpatient mortality model (global test based on Schoenfeld residuals,
POF
A total of 121 patients developed POF (including TOF in non-survivors) during admission. Supplementary Material Figure 2 presents the Kaplan-Meier curves for the probability of developing POF. Higher CCI categories, obesity, parenchymal necrosis, isolated peripancreatic necrosis, IPN and open surgical necrosectomy within 30 days of presentation were all associated with increased risk of developing POF in unadjusted and multivariable cox regression models (Figure 3 and Supplementary Material Table 2). Being overweight was associated with an increased risk of POF on unadjusted but not multivariable Cox regression analysis. Advanced age was not associated with developing of POF on unadjusted or multivariable Cox regression analysis. Alcohol aetiology was not associated with developing POF on unadjusted Cox regression analysis ( Forest plot of the multivariable Cox model for the development of persistent organ failure. The Cox model has been adjusted for each listed variable in addition to acute recurrent pancreatitis, parenchymal necrosis, peripancreatic necrosis, infected pancreatic necrosis and early surgery within 30 days of presentation. BMI: body mass index; CCI: Charlson Comorbidity Index.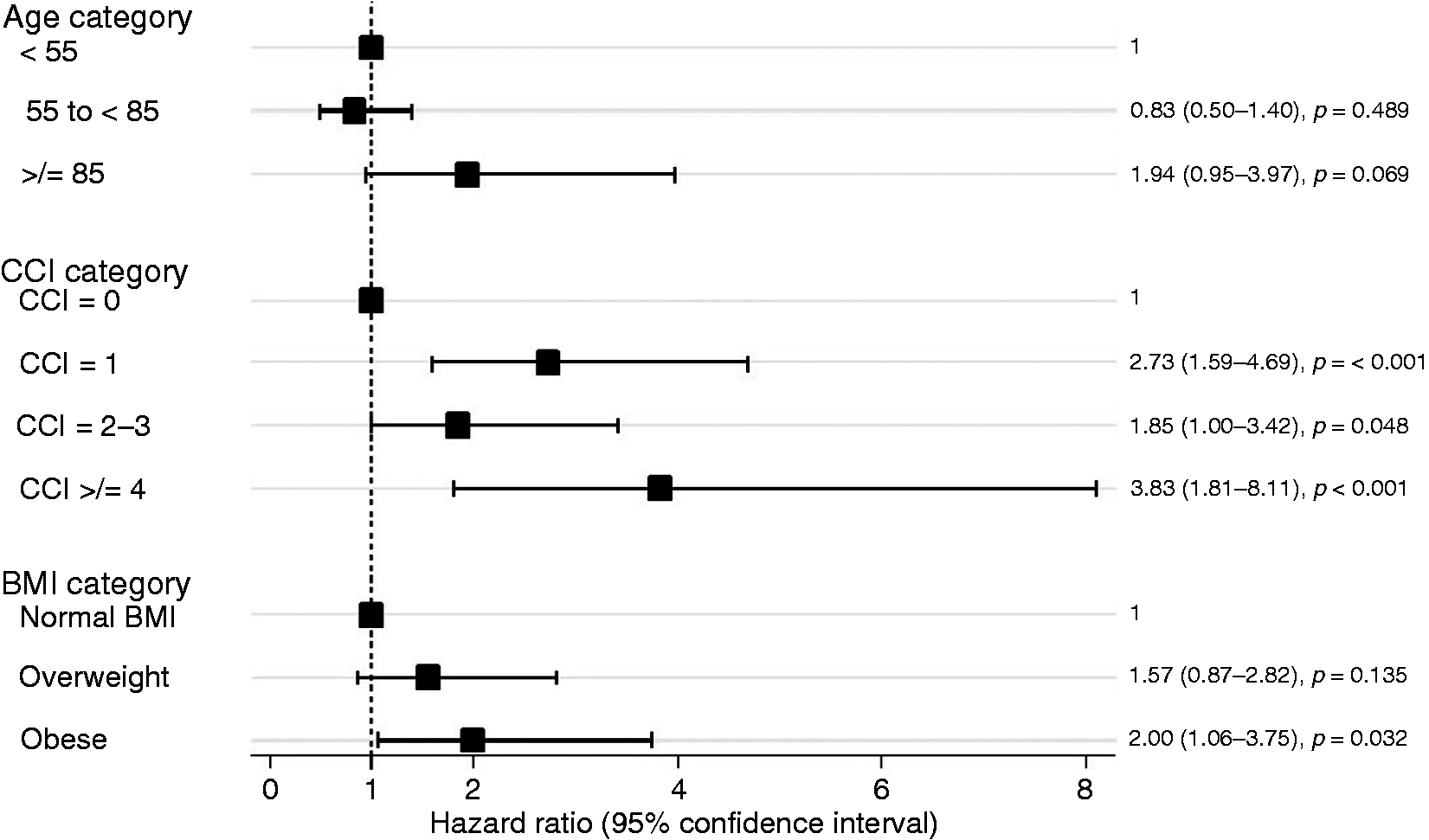
Length of stay
The median (P25, P75) LOS for all patients was 7 (5, 12) days. LOS of ≥ 13 days was chosen as the cutoff point to mark a prolonged LOS as it represented the top 25th centile for LOS. Prolonged LOS was evaluated with a single and multivariable logistic regression analysis (Figure 4 and Supplementary Material Table 3). The presence of parenchymal necrosis, isolated peripancreatic necrosis, IPN, TOF, POF and the need for endoscopic or surgical intervention and percutaneous drains were associated with a prolonged LOS on both single and multivariable logistic regression analysis ( Forest plot of the multivariable logistic regression model for increased length of stay (top 25%). Adjusted for the presence of parenchymal necrosis, peripancreatic necrosis, transient organ failure (TOF), persistent organ failure (POF), percutaneous drains, endoscopic therapy, surgical necrostomy and mortality. BMI: body mass index; CCI: Charlson Comorbidity Index; OF: organ failure; OR: odds ratio.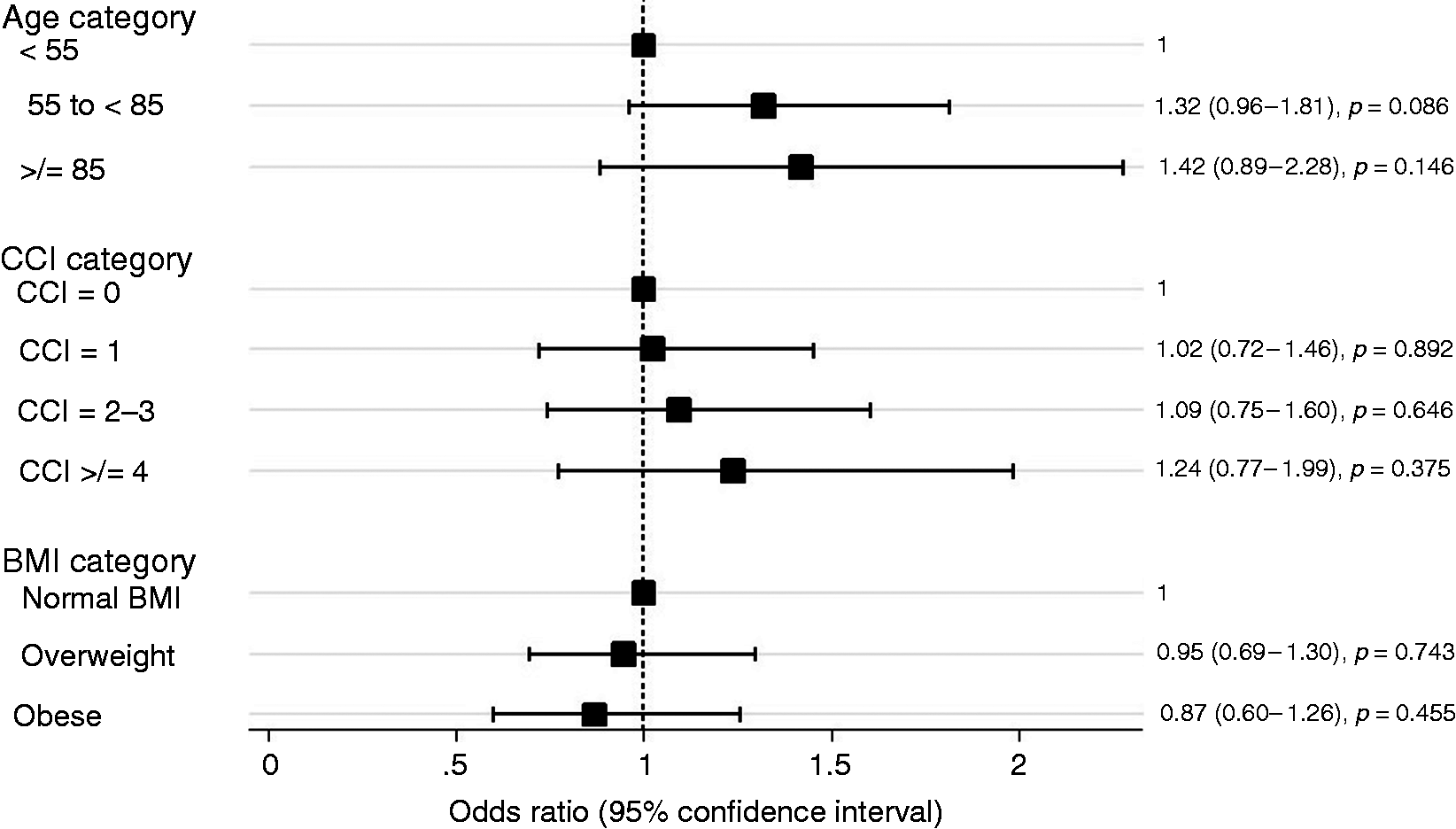
A sensitivity analysis was performed to evaluating LOS in the cohort of survivors who did not develop TOF, POF or mortality (
Need for intervention and presence of pancreatic necrosis
BMI categories, CCI scores and age were not independently associated with the need for intervention or nutritional support on multivariable logistic regression adjusted for each predictor variable in addition to TOF and POF (
A sensitivity analysis was performed to evaluating interventions in the cohort of survivors who did not develop TOF or POF (
Discussion
The present study has many important findings. Firstly, comorbid disease, elevated BMI, open surgical necrosectomy within the first 30 days of presentation and pancreatic necrosis are independently associated with both 30-day inpatient mortality and POF, while only the extreme of age (>85 years old) was associated with 30-day inpatient mortality but not POF. Secondly, after adjusting for OF and other markers of morbidity, comorbid disease and elevated BMI were not independently associated with a prolonged LOS, a marker of morbidity.
Comorbidity has been demonstrated to be a strong independent risk factor for mortality and morbidity across both a host of individual medical diagnosis and in unselected patients requiring acute hospital admission.13–15 While many tools exist, the CCI is the most extensively utilised clinical tool for assessing the impact of comorbid disease on clinical outcomes. 16 Considering the importance of comorbidity in predicting outcomes for other diseases, there is an overall paucity of literature regarding the role of comorbidity in AP. Apart from the limited recording of comorbid disease in the Acute Physiology and Chronic Health Evaluation II (APACHE II) score, comorbid disease is not incorporated into clinical predictive models of severity in AP. 17 In a study of an administrative database, the individual comorbidities contained within the CCI were found to be associated with mortality and the development of multiorgan failure (MOF) within the first two weeks of admission with AP. 4 This study was limited by it retrospective nature, its reliance on an administrative data base with ICD 9 codes and the lack of utilization of survival analysis. Comorbidity was demonstrated to be a strong independent predictor of 30-day inpatient mortality and POF in the present study with increasing levels of comorbidity being associated with a progressively higher risk of 30-day inpatient mortality and POF.
Obesity is a known validated predictor of mortality, OF and local and systemic complications in patients with AP. 9 Elevated levels of intrapancreatic fat are associated with increased BMI.18,19 In addition elevated BMI is associated with elevated visceral fat which surrounds the pancreas. Pancreatic lipase is thought to induce lipolysis in adipocytes resulting in increased unsaturated fatty acids which in turn drives the inflammatory cascade. 20 Obesity is known to be associated with an excess of medical comorbidities. 21 In the present study being overweight or obese was independently associated with an increased risk of POF and being obese was independently associated with 30-day inpatient mortality even after adjusting for other medical comorbidities. This is in keeping with the intrinsic mechanism of lipolysis associated with elevated BMI.
Older age has been studied extensively as a marker of severity and mortality in AP and is incorporated in the APACHE II score, Ranson score, the bedside index of acute pancreatitis (BISAP) score and the Japanese severity score (JSS) as a marker of severity, with an age range of>45 up to>70 years old being associated with adverse clinical outcomes in these scoring systems.17,22 Contrary to previously published studies, after adjusting for comorbid disease in the present study, only the very extreme of age (>85 years old) was associated with 30-day inpatient mortality and POF. The likely explanation for this is that advancing age is associated with a higher degree of comorbid diseases, consequently, after adjusting for comorbid disease the relationship between older age and adverse clinical while remaining significant but is no longer as strong. 23 A numerical age does not necessarily reflect a person's underlying health status, a concept embodied by the theory of biological vs chronological age. 24
The International Association of Pancreatology and American Pancreatic Association evidence-based guidelines describe using host risk factors, such as age, co-morbidity and BMI as part of a three-pronged approach in predicting outcomes of AP. The results described within the present study support the use of co-morbidity and BMI for the risk stratification of patients with AP. In order for these host factors to be adequately incorporated into our management of AP, predictive tools that adequately incorporating these variables will need further development. 25
Delaying open surgical necrosectomy has been shown to be associated with a reduction in mortality. 26 This has led to the adoption of a step-up approach where a gradual step up from more minimally invasive interventions to an open surgical necrosectomy is performed if necessary. This has been shown to be associated with a reduction in new onset MOF in a randomised controlled trial. 27 Recent mortality data from an international cohort of nearly 2000 patients supports the use of minimally invasive surgical or endoscopic drainage/debridement procedures over open surgical necrosectomy. 28 The higher risk of 30-day inpatient mortality in patients who have open surgical necrosectomy within 30 days of presentation in the current study further highlights that patients should not undergo early surgical necrosectomy as the standard of care in 2017.
Both isolated peripancreatic necrosis and parenchymal necrosis (±peripancreatic necrosis) are known predictors of morbidity, POF and mortality in AP, the results of the present study validate these finding.2,29 Interestingly, IPN was not associated with 30-day inpatient mortality but was associated with overall hospital mortality, supporting the published literature that deaths due to IPN occur later in the disease. 30 This is an important yet challenging factor to consider when developing clinical predictive models of mortality in AP since assessing 30-day mortality is standard for most diseases, and using total mortality during the entire length of admission to incorporate late mortality events associated with IPN may result in variables violating the assumptions made in survival analysis, as was the case in the present study.
Importantly comorbidity and elevated BMI were not associated with increased LOS or other markers of morbidity on adjusted analysis, incorporating OF. Consequently, co-morbid disease and elevated BMI can be seen to increase hospital morbidity by increasing the risk of OF, but in the absence of OF, co-morbid disease does not increase morbidity (Figure 5). Inclusion of ‘exacerbation of co-morbid disease’ in the moderate severity of the RAC is potentially redundant, as exacerbation of co-morbid disease resulting in increased morbidity is accounted for by the development of OF which is presently a key component of the RAC. Finally, only the extreme of age was associated with increased LOS. It is possible that increased LOS in this older population may be a surrogate for living alone and needing to be at a more advanced stage in their rehabilitation or have a social support package in place prior to discharge. Data from the UK reports that 50% of the UK population older than 75 years lives alone, while 26% of 65–74 year olds and 16% of 45–64 year olds live alone.
31
From a practical viewpoint, patients older than 85 years are generally not included in clinical trials.
Relationship between elevated body mass index (BMI), co morbidity, persistent organ failure (POF) and morbidity.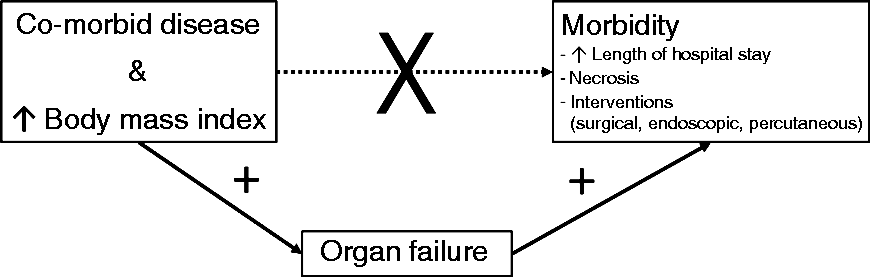
The main strength of the present study is that it is a large multicentre, prospective study that included tertiary and non-tertiary centres with predefined definitions and data collection sheet. Secondly, survival analysis was utilised for the primary analysis, POF and 30-day mortality. In comparison to standard logistic regression which has been used in most epidemiological studies of AP, this type of analysis is more powerful, allows for censoring and is commonly used in epidemiological studies of major disease outcomes and 30-day mortality.32,33 Finally, as opposed to using previously published cut off points for age and CCI, these variables were individually modelled in order to achieve the best fit of these variables for this data set. There are a number of limitations in the present study. Imaging data was evaluated by a local radiologist and not a centralised radiologist. Evaluating for the present of peripancreatic and parenchymal necrosis is known to be associated with a high inter-reader variability. 34 However, the results for necrosis are broadly in keeping with the published literature arguing against significant flaws in this data. Lastly, supportive measures such as fluid resuscitation which have been associated with major clinical outcomes were not accounted for. While this may have impacted the results, it does allow for the broad generalisability of the studies current findings.
In conclusion, comorbid diseases, including BMI are significant determinates of POF and mortality in AP and their inclusion in clinical models and predictive scores evaluating major clinical outcomes in AP is important. After accounting for OF, comorbid disease, including BMI is not associated with a prolonged LOS or other markers of morbidity, thus inclusion of ‘exacerbation of co-morbid disease’ in the moderate severity of the RAC may be redundant.
Summary of the established knowledge on this subject
Obesity is associated with organ failure in clinical studies of acute pancreatitis and has been shown to be associated with mortality in a meta-analysis. There is a limited description of the role of co-morbid disease in the development of morbidity, organ failure and mortality in acute pancreatitis.
What are the significant and/or new findings of this study?
Comorbid disease is independently associated with both organ failure and morality in acute pancreatitis. Co-morbid disease and obesity are not independently associated with markers of morbidity, including: length of hospital stay, development of necrosis (pancreatic, peripancreatic or infected) and interventions (surgery, endoscopy or percutaneous drains). Age is not independently associated with persistent organ failure and age is only associated with mortality in the very elderly (>85 years old).
Supplemental Material
Supplemental material for Influence of age, body mass index and comorbidity on major outcomes in acute pancreatitis, a prospective nation-wide multicentre study
Supplemental material for Influence of age, body mass index and comorbidity on major outcomes in acute pancreatitis, a prospective nation-wide multicentre study by Robert A Moran, Guillermo García-Rayado, Daniel de la Iglesia-García, Emma Martínez-Moneo, Esther Fort-Martorell, Eugenia Lauret-Braña, Mar Concepción-Martín, Fabio Ausania, Carlos Prieto-Martínez, Miguel González-de-Cabo, Noé Quesada-Vázquez, M Asunción Marcaide-Ruiz-de-Apodaca, José A Pajares-Díaz, Francia C Díaz, José L de-Benito, Jennifer Hinojosa-Guadix, Pilar Marqués-García, Jaume Boadas, Eduardo Bajador-Andreu, Oswaldo Moreno, Federico Argüelles-Arias, Gregorio Martín-Benítez, Carla Tafur-Sánchez, Jesús Leal-Téllez, Beatriz Romero-Mosquera, Ruben Hernaez, Georgios I Papachristou, Vikesh K Singh and Enrique de-Madaria in United European Gastroenterology Journal
Footnotes
Acknowledgements
For reviewing the manuscript prior to submission, the authors wish to thank Dhiraj Yadav, Division of Gastroenterology, University of Pittsburgh Medical Center, Pittsburgh, USA.
The participating investigators were:
Ana M Argüelles‐Arias, Federico Bertoletti, María P Bodenlle‐Bello, Juan I Cervera, Ángel Ferrández, Roberto García‐Figueiras, Cristina Gil‐García‐Ollauri, Silvia Guzmán‐Suárez, Ricardo Ituarte‐Uriarte, Nelly Lareu‐Balza, José Lariño‐Noia, Patricia Latorre, María M Menso, Alicia Mesa‐Álvarez, Elida Oblitas, María Rodríguez‐Peláez, José I Rodríguez‐Prada, Carmen L Rosales‐Alexander, María J Soria‐de‐la‐Cruz, Jordi Soriano, María A Udaondo‐Cascante, Federico Bolado, Héctor J Canaval‐Zuleta, Carlos Marra‐López, Ana I Hernando‐Alonso, Adolfo del‐Val‐Antoñana, Robin Rivera‐Irigoin, Francisco J Grau‐García, Lluís Oms, Judith Millastre‐Bocos, Isabel Pascual‐Moreno, David Martínez‐Ares, Juan A Rodríguez‐Oballe, Antonio López‐Serrano, María L Ruiz‐Rebollo, Alejandro Viejo‐Almanzor, Belén González‐de‐la‐Higuera, Aitor Orive‐Calzada, Ignacio Gómez‐Anta, José Pamies‐Guilabert, Fátima Fernández‐Gutiérrez‐del‐Álamo, Isabel Iranzo‐González‐Cruz, Mónica E Pérez‐Muñante, María D Esteba, Ana Pardillos‐Tomé and Pedro Zapater.
Author contributions were as follows:
Planning the study, conducting the study, collecting and interpreting data and drafting the manuscript: Enrique de‐Madaria. Statistical analysis, collecting and/or interpreting data and drafting the manuscript: Robert A Moran and Ruben Hernaez. Conducting the study, collecting and interpreting data and drafting the manuscript: Guillermo García‐Rayado; Daniel de la Iglesia‐García; Emma Martínez‐Moneo; Esther Fort‐Martorell; Eugenia Lauret‐Braña; Mar Concepción‐Martín; Fabio Ausania; Carlos Prieto‐Martínez; Miguel González‐de‐Cabo; Noé Quesada‐Vázquez; Asunción M Marcaide‐Ruiz‐de‐Apodaca; José A Pajares‐Díaz; Francia C Díaz; José L de‐Benito; Jennifer Hinojosa‐Guadix; Pilar Marqués‐García; Jaume Boadas; Eduardo Bajador‐Andreu; Oswaldo Moreno; Federico Argüelles‐Arias; Gregorio Martín‐Benítez; Carla Tafur‐Sánchez; Jesús Leal‐Téllez and Beatriz Romero‐Mosquera. Interpreting data and drafting the manuscript: Georgios I Papachristou and Vikesh K Singh.
Declaration of conflicting interests
Vikesh Singh is a consultant to Abbvie, Akcea and Ariel Precision Medicine. Enrique de‐Madaria is consultant to Mylan. All other authors have no disclosures.
Ethics approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in approval from the institutional review board at the principal investigator's centre, Comitéé tico de investigación clínica del Hospital General Universitario de Alicante on 29 June 2014. The study was also approved by the relevant local ethical committee at each centre.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not‐for‐profit sectors.
Informed consent
Informed consent by the patient or their healthcare advocate was acquired for all patients enrolled in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
