Abstract
Background
Anxious patients and those with poor tolerance to previous esophagogastroduodenoscopy (EGD) usually have poor tolerance for EGD.
Aims
To investigate the effect of meperidine on these patients during EGD.
Methods
A total of 110 patients undergoing diagnostic EGD were randomized to receive either meperidine (n = 55) or placebo (n = 55) before EGD. The primary outcome was patient discomfort scores during esophageal intubation.
Results
Patients in the meperidine group reported less discomfort during esophageal intubation (median score of 5.0 and interquartile range (IQR) 1.5–7.0) compared with the control (median score of 6.0, IQR 5.0–8.5, P = .003). Patients in the meperidine group had better tolerance during the procedure (median score of 2 (IQR 1.0–3.0) versus 3 (IQR 1.0–4.0), P = .048), and the endoscopists reported higher overall satisfaction scores (median score of 9 (IQR 7.0–9.0) versus 8 (IQR 7.0–9.0), P = .043). There was significantly less increase in heart rate and blood pressure during the procedure in the meperidine group than in the placebo group (23 bpm (IQR 9–32) versus 30 bpm (IQR 18–52); P = .006); (7 mmHg (IQR 1–14) versus 18 mmHg (IQR 2–30); P = .008). Connect-the-numbers test showed comparable results before and after EGD between the two groups.
Conclusion
For patients expected to have poor tolerance during EGD, meperidine reduced discomfort, decreased cardiovascular distress, and improved endoscopist satisfaction, without adverse effects on their psychomotor function after the procedure.
Key summary
Minimal sedation with a single use of meperidine is a useful adjunct to improve patient tolerance and reduce discomfort during esophagogastroduodenoscopy (EGD). The use of meperidine was beneficial for patients who are expected to have poor tolerance to EGD by reducing the discomforts during esophageal intubation. Patients receiving meperidine achieved better tolerance and higher overall satisfaction scores as rated by the endoscopists. Meperidine reduced cardiovascular distress during EGD. Adverse effects, including psychomotor impairment, were compatible between meperidine and placebo.
Introduction
Esophagogastroduodenoscopy (EGD) is an important diagnostic tool for upper gastrointestinal disorders. Although sedation for EGD is widely utilized in the USA, 1 its use varies in Europe and Asia.2–5 From the perspective of the patients, sedation for EGD has the benefit of improving satisfaction, eliminating anxiety and discomfort, and increasing the willingness to repeat the procedures.6–8 For the endoscopists, sedation can improve the success rate and quality of examination for diagnostic EGD, as well as enhance the treatment outcomes of therapeutic EGD. 8 Sedation, however, contributes to a significant proportion of morbidity and mortality associated with EGD.5,7,9 Besides, sedation increases the cost and the time needed for preprocedure preparation, the procedure per se, and post-procedure recovery, not to mention the indirect costs in loss of work time in patients and their family caregivers.5,7,9
Unsedated EGD, albeit without the drawbacks of sedation and well tolerated by a modest proportion (61%) of patients, 10 is considered uncomfortable by many patients. Factors associated with discomfort during unsedated EGD include the presence of gag reflex, young age, high level of anxiety, female, and poor tolerance to prior examinations.10–12 An alternative method that is effective for those patients expected to experience discomfort during EGD but without resorting to full sedation is desirable.Minimal sedation with a single use of meperidine has been reported to be a useful adjunct to EGD in improving patient tolerance and reducing discomfort. 4 Its adverse effects were also comparable to that in the placebo group except more self-limited dizziness was observed. 4 However, the previous study was targeted at the general population, a significant proportion of which would have good tolerance to EGD with or without meperidine anyway. The real challenge to meperidine lies in those who are expected to have poor tolerance during EGD. Furthermore, the psychomotor impairment by meperidine after EGD had not been assessed in the previous study.The aim of this double-blind, randomized, controlled trial was to investigate the use of meperidine as a single sedative agent for patients who were expected to have poor tolerance to EGD, including patients with high anxiety levels and/or poor tolerance to prior unsedated EGD. Our hypothesis is that meperidine could reduce the discomforts associated with esophageal intubation for these patients.
Materials and methods
This double-blind, randomized, controlled trial was conducted at Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Taiwan, from September 2013 to March 2017. Patients undergoing unsedated diagnostic EGD performed by the participating endoscopists were considered for enrollment. Patients who had undergone previous EGD were asked about the experience of their last EGD. Only patients rating their previous experience very uncomfortable were invited to join this study. For patients undergoing EGD for the first time, anxiety level was assessed, and those with high anxiety levels (anxiety score ≥5; 0 = not anxious at all; 10 = extremely anxious) were enrolled. Patients were excluded if they had any of the following conditions: a therapeutic EGD, sedated EGD, bidirectional endoscopy, contraindication to hyoscine N-butylbromide, allergy to meperidine, American Society of Anesthesiology physical status classification Class 3 or higher, renal failure, decompensated cirrhosis, age less than 18 years or more than 65 years, being pregnant, or refusal to provide written informed consent. The EGDs were performed by three board-certified endoscopists with experience of more than 5000 to 10,000 diagnostic EGDs. All participants signed written informed consents. The study was approved by the Institutional Review Board of Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation on 20 March 2013 (IRB B10201018). The trial was registered in ClinicalTrials.gov# NCT01948921. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution’s human research committee.
A total of 110 patients were allocated into two groups in a one-to-one ratio based on a random number list generated by a computer. The randomization codes were contained in prearranged opaque envelopes, which were opened immediately before insertion of the scope to reveal either meperidine or placebo by a research assistant. To blind the patients, endoscopists, and the study assistant, all subjects received a single unlabeled intramuscular (IM) injection 5–10 min before the procedure. Patients in the meperidine group received 25 mg of meperidine (National Bureau of Controlled Drugs, Department of Health, Taipei, Taiwan) and 20 mg of hyoscine N-butylbromide (Boehringer Ingelheim, Taipei, Taiwan) in the injection, whereas patients in the placebo group received 20 mg of hyoscine N-butylbromide only. The medications were prepared and administered by an endoscopy nurse who was not involved in the study. All patients then ingested 15 ml of dimethylpolysiloxane (Yungshin Pharmaceutical, Taichung, Taiwan) and received 10 puffs of 10% lidocaine spray (Xylocaine 10% Spray, AstraZeneca, Södertalje, Sweden) (1 puff = 10 mg of lidocaine) delivered to the soft palate, posterior part of the tongue, and pharynx.
The procedures were performed by using a standard video esophagogastroduodenoscope (EG-590; Fujinon, Tokyo, Japan; GIF-Q-260; Olympus Optical Co., Ltd, Tokyo, Japan). All procedures started with the patient in the left lateral decubitus position. During the examination, electrocardiogram, blood pressure, and oxygen saturation of the patient were monitored.
The primary outcome was the patient’s self-reported discomfort scores during esophageal intubation, assessed with a 0–10 (0 = none; 10 = unbearable) visual analog scale. The secondary outcomes were discomfort score during the procedure and overall satisfaction with the procedure (0 = not satisfied at all; 10 = very satisfied) as evaluated by the patients; patient tolerance to the procedure, ease of intubation of the endoscope, and satisfaction with the procedure as evaluated by the endoscopists; and number of coughing and retching episodes during the procedure counted by a blinded study assistant.
Demographic data (age, sex, height, and weight), indications for EGD, history of drinking or smoking, and anxiety level were obtained before EGD. After the procedure, patients assessed their discomfort induced by the lidocaine spray, during esophageal intubation, and during the procedure (0 = none; 10 = unbearable); overall satisfaction with the procedure (0 = not satisfied at all; 10 = very satisfied); and their willingness to receive EGD in the same way if needed in the future (yes or no). All patients were asked whether they had suffered from dizziness, nausea and/or vomiting, and sore throat after the procedure.
At the end of the procedure, the endoscopists were asked to respond to a questionnaire to rate whether the procedure was completed, the difficulty of intubation (0 = very easy; 10 = very difficult), patient tolerance to the procedure (0 = very well; 10 = extremely poor), and overall satisfaction with the procedure. One score was given to each of the four areas (esophagus, stomach, duodenum to 2nd portion, and retroflex for proximal stomach) if adequately examined and necessary biopsies performed (0 = not complete study in all four areas; or 4 = complete study in all four areas). The endoscopic findings, esophageal intubation time (the time from insertion of the endoscope into the patient’s mouth to passage through the cricopharyngeus), total procedure time, and the time patients spent in bed after EGD were also recorded.
To elevate the psychomotor function after the procedure, the number connection test A (NCT-A) was used. 13 The NCT-A is a test of visual-spatial orientation and psychomotor speed. Each patient was shown an A4-sized piece of paper with 25 consecutively numbered circles (1–25) randomly arranged on the paper. The task was to connect the numbers in ascending order as fast as possible. The test result was the time needed, including error correction time, to complete the task. The test was administered before the EGD and was repeated 10 min after the procedure with another sheet of test paper with a different arrangement of the numbers. If a patient could not complete the task within 2 min, the test would be repeated 10 min later.
Statistical analysis
The sample size calculation was based on the data from our previous study, 4 which showed that the difference in the patient discomfort score during esophageal intubation was 1.6 with a standard deviation (SD) of 2.9 between the two groups. Power calculations estimated that 55 patients in each arm would ensure an 80% power of detecting the difference (5% significance level, two-sided test). Statistical analysis was performed using SPSS version 19.0 software (SPSS Inc., Chicago, Illinois, USA). Normally and non-normally distributed data were represented by using mean (SD) and median (interquartile range; IQR), respectively. The Chi-squared test, with Yates’ correction for continuity or Fisher’s exact test, was used for the comparison of categorical data, as appropriate. The Mann–Whitney U test was used to compare differences in continuous variables between groups. A P value < .05 was considered significant.
Results
Baseline characteristics of enrolled patients
A flow diagram of enrollment and intervention allocation is shown in Figure 1. Of the 2031 patients considered for enrollment, 1921 patients were excluded. A total of 110 patients undergoing diagnostic EGD were enrolled in this study. The mean age of the patients was 48.6 y (SD = 14.2 y); the majority (87.3%) of the patients were female. The baseline characteristics of both groups are shown in Table 1. The two groups were similar in terms of sex, age, height, smoking status, use of alcohol, enrollment criteria, and indication for endoscopy. The mean body weight in the meperidine group was significantly higher than the placebo group (60.9 kg versus 55.9 kg; P = .023). The distribution of the indications was also comparable, with dyspepsia being the most common one (63.6%).
Flow chart of enrollment and intervention allocation. Baseline characteristics of patients undergoing esophagogastroduodenoscopy. EGD: esophagogastroduodenoscopy; GERD: gastroesophageal reflux disease; SD: standard deviation. P values for continuous variables were obtained by t-test. P values for categorical variables were obtained by Fisher’s exact test.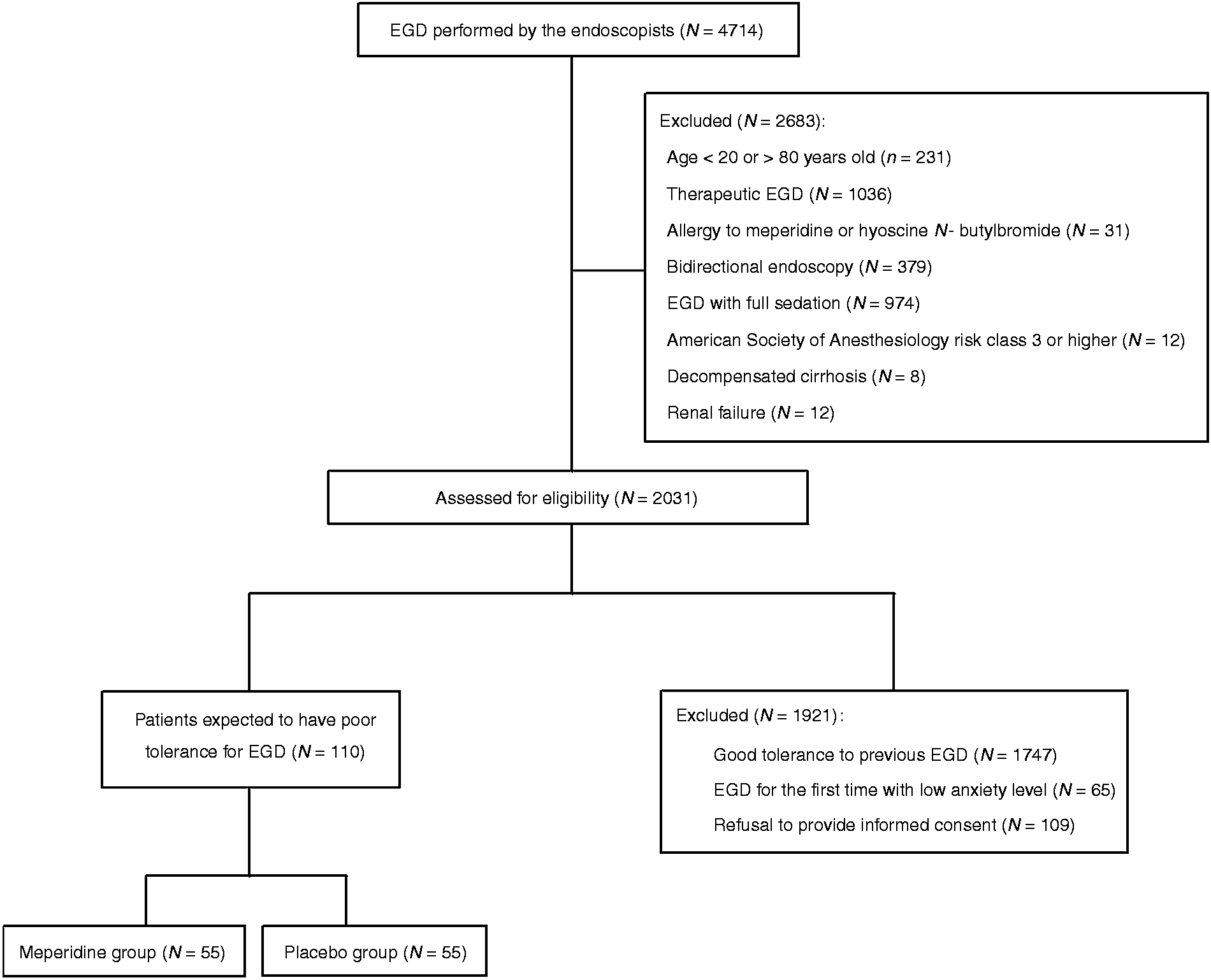
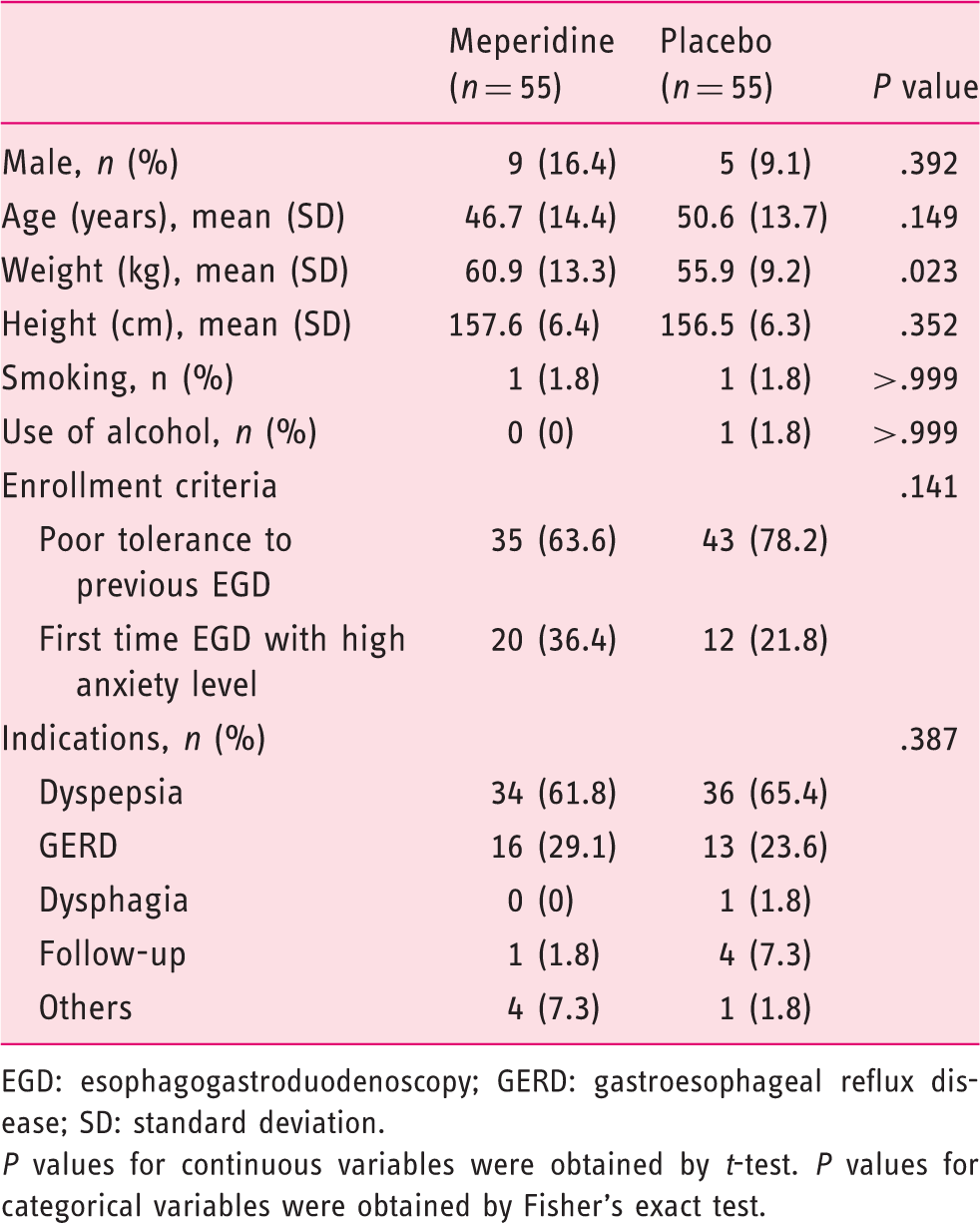
Patient evaluation scores
Patient evaluation scores for esophagogastroduodenoscopy.

EGD: esophagogastroduodenoscopy.
Data are expressed as median (interquartile range) unless otherwise specified.
P values for continuous variables were obtained by non-parametric Mann–Whitney U test. P values for categorical variables were obtained by Fisher’s exact test.
0 = not anxious at all; 10 = extremely anxious.
0 = none; 10 = unbearable.
0 = not satisfied at all; 10 = very satisfied.
Endoscopists’ evaluations and number of retching and coughing episodes
Endoscopists’ evaluations and number of retching and coughing episodes encountered during esophagogastroduodenoscopy.
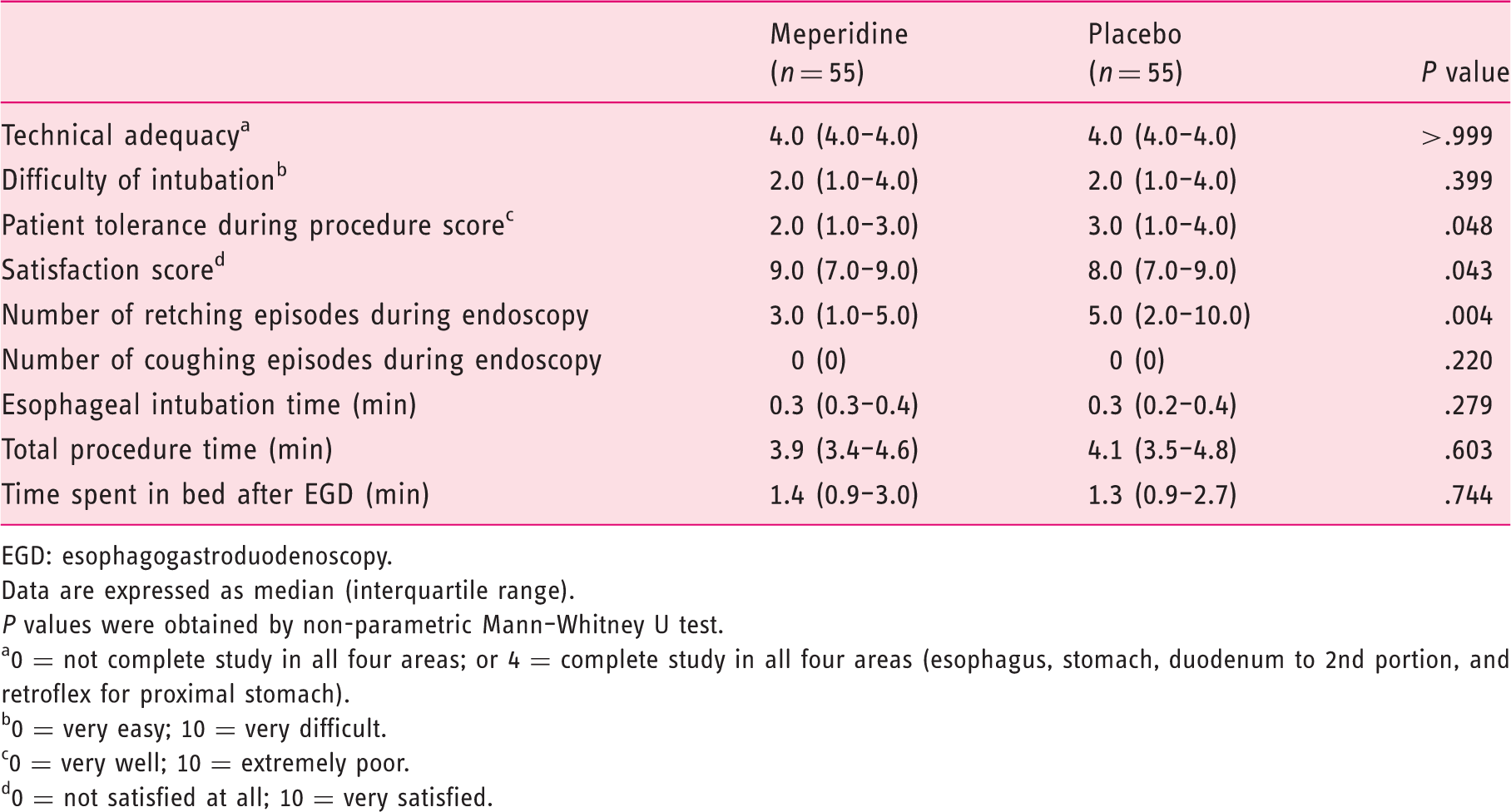
EGD: esophagogastroduodenoscopy.
Data are expressed as median (interquartile range).
P values were obtained by non-parametric Mann–Whitney U test.
0 = not complete study in all four areas; or 4 = complete study in all four areas (esophagus, stomach, duodenum to 2nd portion, and retroflex for proximal stomach).
0 = very easy; 10 = very difficult.
0 = very well; 10 = extremely poor.
0 = not satisfied at all; 10 = very satisfied.
Psychomotor function, hemodynamic changes, and adverse effects
Vital signs changes and adverse effects.
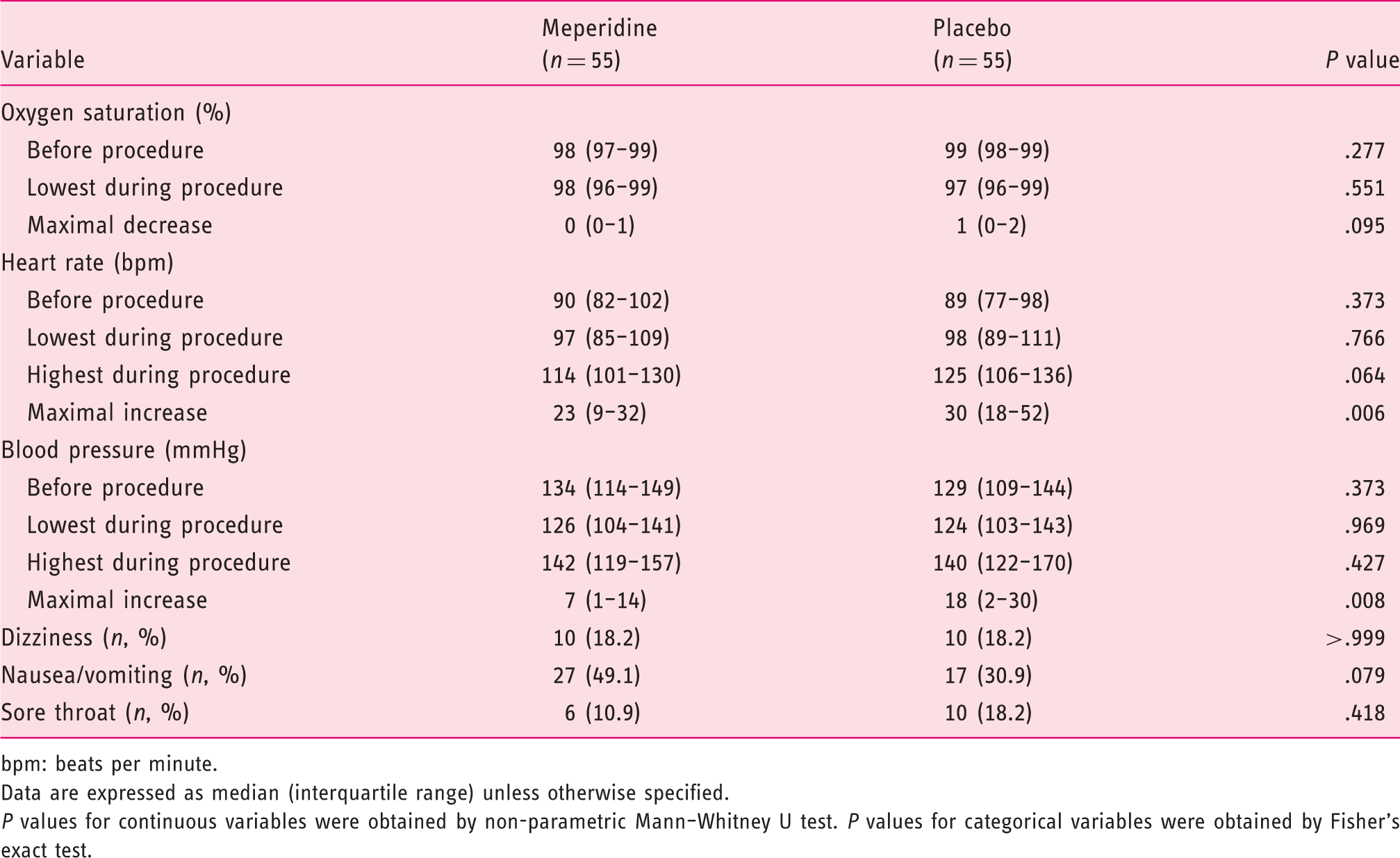
bpm: beats per minute.
Data are expressed as median (interquartile range) unless otherwise specified.
P values for continuous variables were obtained by non-parametric Mann–Whitney U test. P values for categorical variables were obtained by Fisher’s exact test.
Discussion
In this double-blind, randomized, controlled study, we found that the IM injection of 25 mg of meperidine was useful in patients expected to poorly tolerate EGD by reducing discomfort during esophageal intubation, boosting endoscopists satisfaction through better patient tolerance, and decreasing cardiovascular stress. This type of patient generally suffers the most during unsedated EGD, and therefore is the most important target population for sedation, and usually full sedation. The current study offers an alternative method of sedation for these patients.
Meperidine is the most frequently administered opiate in sedated EGD. 1 It is usually used in combination with a benzodiazepine in the USA. 1 Dies et al. found that patients receiving meperidine, as compared with ketorolac, experienced less gagging and tachycardia, and required less midazolam during EGD. 14 Diab et al. randomized 120 patients to receive either 50 mg of meperidine or 1 mg of midazolam, adding incremental doses of midazolam at the discretion of the endoscopist. 15 Those receiving meperidine needed less midazolam and showed better tolerance than those receiving only midazolam. 15 Laluna et al. compared midazolam alone with meperidine plus midazolam for sedating patients undergoing EGD. 16 The endoscopists rated better tolerance for patients in the combination group, which also entailed a lower dose of midazolam. In short, adding meperidine to midazolam improves patient tolerance and reduces the dosage of midazolam.
There are sparse reports on the use of opiate as a single sedative agent for EGD. Ishido et al. showed that intravenous (IV) fentanyl, a short acting opiate, could increase the tolerance of patients during EGD, as reflected by less coughing and retching. 17 Another report by Stephens et al. showed that fentanyl was even better than diazepam at enhancing the ease of procedure, as evaluated by the endoscopists. 18 Our group had conducted a randomized, controlled trial comparing meperidine with placebo, with results showing that patients receiving meperidine had better tolerance, as reported by both the patients and the endoscopist. There were also fewer retching episodes in the meperidine group as reported by a third independent observer. 4 In contrast, Wang and Lin reported that meperidine was not superior to placebo for sedation during EGD; however, their study was not a randomized, controlled trial, and no detailed information was provided. 19 The present study focusing on the population with expected poor tolerance to EGD further verifies the benefit and safety of meperidine as a single agent for EGD sedation.
A rapid IV injection of meperidine may result in respiratory depression, hypotension, circulatory disorder, cardiac arrest, etc. Thus the use of IV route is advised to be restricted to settings in which a narcotic antagonist and facilities for assisted or controlled respiration are immediately available. 8 IM administration of meperidine carries a lower risk of cardiopulmonary adverse effect and does not require IV cannulation. Therefore we chose IM administration of meperidine in our study. The analgesia of IM meperidine begins 5–10 min after injection and its peak plasma concentrations are measured at 15–60 min after IM injection.20,21
The primary outcome of the current study was patient discomfort scores during esophageal intubation, which was reported to be the most important contributing factor to a poor overall tolerance during EGD. 22 We found that meperidine significantly decreased the discomfort during esophageal intubation as compared to placebo (Table 2). Meperidine has the effect of suppressing the pharyngeal reflex. Our observation is compatible with a recent report by Yamasaki et al. evaluating the best sedation method for pharyngeal observation using transoral endoscopy by comparing meperidine with midazolam and placebo. 23 By suppressing the gag reflex without impairing the ability to vocalize, the meperidine group achieved the highest observation score. The meperidine group also had lower discomfort scores than that of the placebo group during pharyngeal observation. The suppressing effect of gag reflex by meperidine was also supported by our observation that significantly fewer episodes of retching were noted in the meperidine group.
In the current study, the endoscopists were more satisfied with the EGD when meperidine, instead of placebo, was injected at the start of the procedure, probably because the patients tolerated the procedure better with meperidine. On the other hand, the overall satisfaction of the patients with the procedure did not differ between the two groups. Although less discomfort during the procedure was associated with higher patient satisfaction, other factors might also play a part, such as personal manner and technical skills of the endoscopists, personal manner of the nurses, physical environment, and more time to discuss the procedure with the endoscopists. 24
Our results showed that a single injection of low dose (25 mg) meperidine did not adversely affect the psychomotor function of the patients after EGD, as shown by the NCT-A test. A previous cumulative-dosing study showed that opioids such as meperidine produced dose-related increases in the impairment of psychomotor function, whereas no detrimental effect was noted at a low dose (such as meperidine 17.5 mg/70 kg). 25 The psychomotor effects of meperidine, when present, were short-lived; they peaked at 5 min after drug injection and recovered, sometimes to baseline levels, within 55 min. 25 In line with the absence of psychomotor disturbance, the patients in the current study were fully alert during the procedure without requiring monitoring or assistance from an anesthesiologist. Most patients receiving meperidine could leave the examination table almost immediately, entailing minimal post-procedure monitoring. They were discharged without an escort but were instructed not to drive for at least 2 h after the procedure. 21 However, there is no official recommendation about the use of meperidine and driving.
Due to the subjective nature of any assessment of patient tolerance and satisfaction, alternative methods for evaluating safety and tolerability would be preferable. Assessments of heart rate and blood pressure are both objective methods of evaluating tolerability of endoscopic procedures. Adachi et al. reported that cardiac stress increased during unsedated EGD as indicated by a 66% increase in rate-pressure product. 26 In the current study, the maximal increase in both heart rate and blood pressure were attenuated in the meperidine group as compared with the placebo group, reflecting a decrease in cardiac stress. The desaturation of arterial oxygen was minimal and there was no difference in arterial oxygen saturation between the meperidine group and the control group.
Several limitations of this study should be mentioned. First, as the study was conducted at a single center, the results need to be validated by other endoscopists in different clinical settings. Second, a relatively low dose of meperidine (25 mg) was injected intramuscularly in this study, and whether a different dose or a different administered route would be more effective will require further investigations. Third, as the Asian sample population of the current trial had a lower mean body weight (mean 58.4 kg; SD 11.6 kg), the findings might not be transferable to the Western population especially with higher body weight, for which a higher or weight-based dose of meperidine might be needed. Fourth, although meperidine could significantly reduce the discomfort score during esophageal intubation, the median score of 5 was still high. Future studies to further reduce the discomfort by using a more potent opiate or in combination with other modalities are warranted.
Conclusions
For patients expected to have poor tolerance during EGD, meperidine reduced their discomfort, decreased cardiovascular distress, and improved endoscopist satisfaction, without adverse effects on their psychomotor function after the procedure.
Footnotes
Author contributions
YHH and CWT defined the research theme, designed methods, carried out the study, interpreted the results, and wrote the paper. MK analyzed the data and revised the paper. KCT helped to collect the cases. All authors approved the final version of the paper.
Declaration of conflicting interests
The authors declare no conflict of interest.
Funding
This work was supported by a research fund from Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation.
Ethics approval
The study was approved by the Institutional Review Board of Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation on 20 March 2013 (IRB B10201018).
Informed consent
Written informed consent was obtained from each patient included in the study.
