Abstract
Background and aim
We present a systematic review on the performance of currently available methods for serological diagnosis of celiac disease (CD) and the role of human leukocyte antigen (HLA) typing.
Objective
A literature survey was conducted using PubMed, MeSH database, Web of Science as well as manual searches.
Results
Tissue transglutaminase antibodies (tTG) (IgA) (tested in nine studies) show sensitivities and specificities in the range of 0.76–0.968 and 0.909–0.98, and deamidated gliadin peptide (DGP) (IgA and IgG) (tested in eight studies) show sensitivities and specificities in the range of 0.69–0.984 and 0.903–1. Endomysial antibodies (EMA) (tested in five studies) show sensitivities and specificities in the range of 0.61–0.937 and 0.98–1, respectively. Combination assays (tested in three studies) using DGP + tTG and DGP (IgA + IgG) show sensitivities and specificities in the range of 0.87–1 and 0.8–1, respectively. HLA DQ2/DQ8 may be necessary for the development of CD—HLA DQ2 in particular. A possible close correlation may also exist between CD and HLA-G.
Conclusion
DGP and tTG for serological testing for CD show equivalent diagnostic performance. More studies with, in particular, DGP alone and in combination with tTG are necessary before a firm recommendation can be made. HLA typing to exclude CD may still be controversial. It still seems premature to diagnose celiac disease in adults based on serology alone.
Introduction
Celiac disease (CD) is an autoimmune, gluten-sensitive enteropathy where intake of foods containing gluten, a protein found in grain species such as wheat, barley and rye, to cause mucosal damage in the small intestine, leading to malabsorption. 1
Ninety percent of all patients with celiac disease carry HLA DQ2 and probably all of the remaining 10% carry HLA DQ8. However, these haplotypes are carried by about 30% of the unaffected population as well. 2 The prevalence of CD is estimated to be around 1% in the general population. 3 It is assumed that HLA DQ2/DQ8 represents less than 50% of the genetic explanation of CD’s etiology,4,5 which means that there must be other genetic factors playing a role in disease development. It is still controversial whether HLA DQ2 or DQ8 is necessary for the development of CD.
The area of serological testing for CD has developed greatly in recent years. The most commonly used antibodies are antigliadin antibodies (AGA), endomysial antibodies (EMA) and tissue transglutaminase antibodies (tTG).6,7 The AGA test was developed in the early 1980s, and more recently developed assays may have higher sensitivities and specificities than AGA, which is considered obsolete.8–13 The recently introduced deamidated gliadin peptide antibody (DGP) has shown promising performance as compared to EMA and tTG test results.6–9,13–15 Data and experience concerning the usage of DGP are still preliminary, however.
EMA was developed in the mid-1980s after AGA and has generally shown high specificity and sensitivity. However, drawbacks of this assay are the resource-demanding use of immunofluorescence and the operator-dependent nature of the test results, possibly diminishing the objectivity of the test.8,9,16 The tTG assay, which uses enzyme-linked immunosorbent assay (ELISA), was developed in the late 1990s and has shown sensitivities and specificities close to EMA. Besides, tTG immunoglobulin (Ig) G may be usable in diagnosing CD in individuals with IgA deficiency.3,13 The current literature on diagnosis of CD reflects controversies among clinicians with respect to preferred screening tests, screening algorithms and the role played by HLA typing in the diagnosis of CD. Are we approaching a scenario in which CD can be diagnosed or excluded in adults without performing a small intestinal biopsy? We will address these questions in the present systematic overview of recent papers on serological testing for CD in adults.
Material and methods
Search strategy in MeSH database (limits: 2006–2013)
Initial search:
The same method was used in the following searches:
Search strategy on PubMed (limits: 2006–2012)
Initial search:
Summary of the examined studies
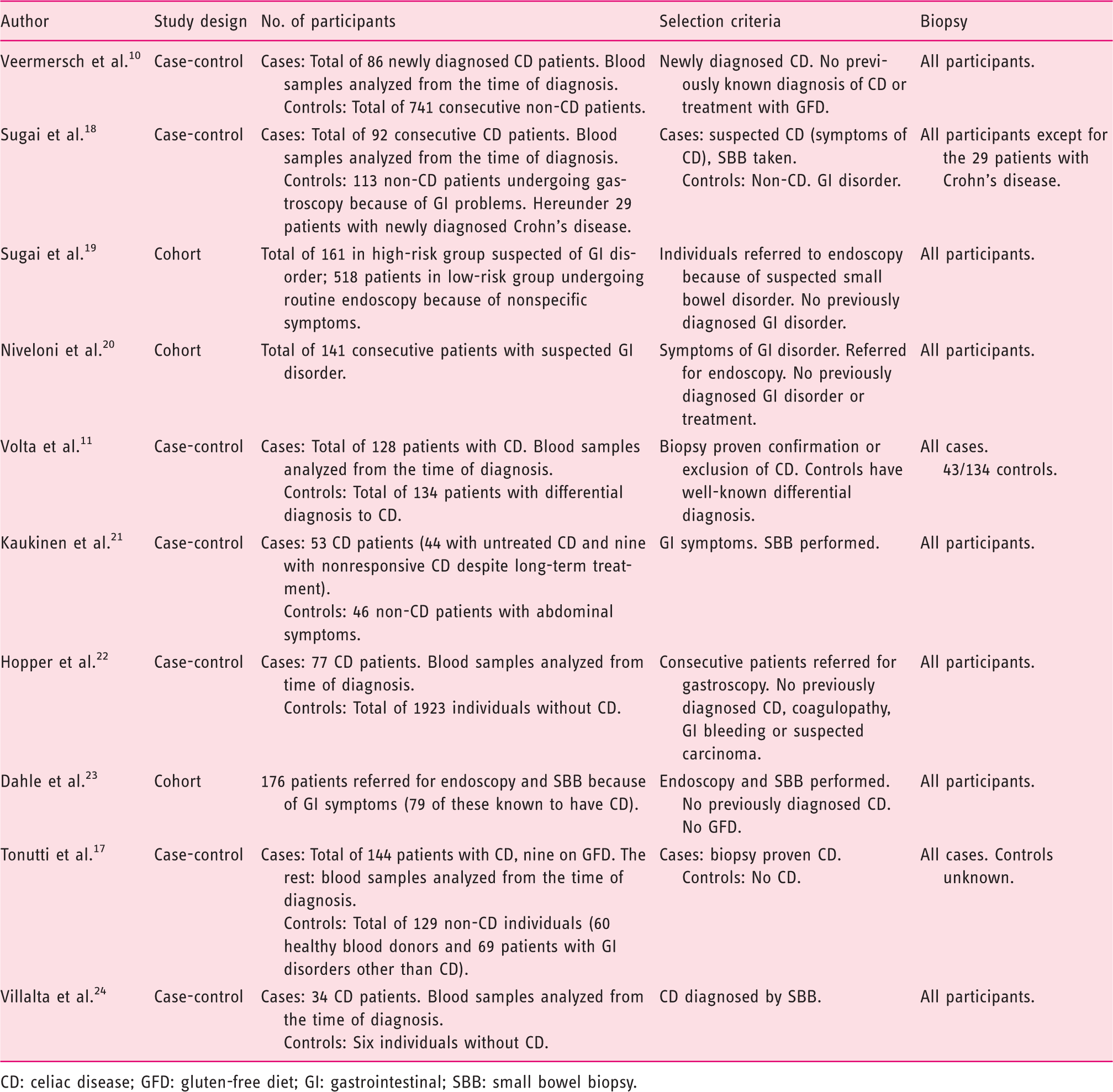
CD: celiac disease; GFD: gluten-free diet; GI: gastrointestinal; SBB: small bowel biopsy.
Median (range) for assays tested in different studies
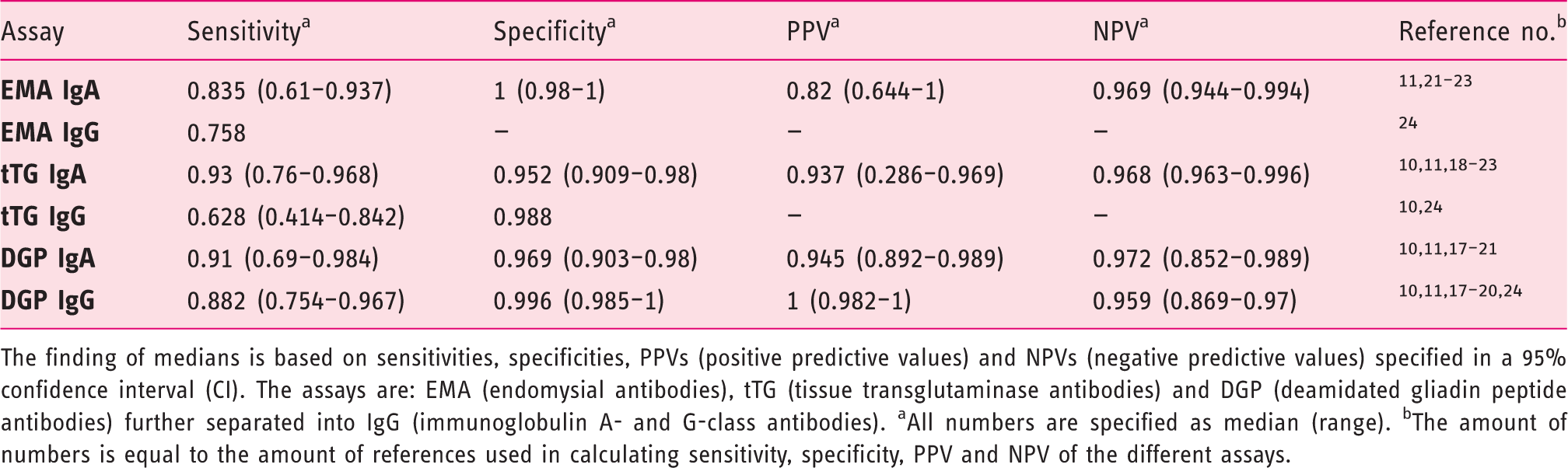
The finding of medians is based on sensitivities, specificities, PPVs (positive predictive values) and NPVs (negative predictive values) specified in a 95% confidence interval (CI). The assays are: EMA (endomysial antibodies), tTG (tissue transglutaminase antibodies) and DGP (deamidated gliadin peptide antibodies) further separated into IgG (immunoglobulin A- and G-class antibodies). aAll numbers are specified as median (range). bThe amount of numbers is equal to the amount of references used in calculating sensitivity, specificity, PPV and NPV of the different assays.
All studies used met the requirement that all diagnoses of CD were finally confirmed by a biopsy and not by serologic testing alone, as biopsy is considered necessary by many for diagnosing CD.2,8,15,16 Studies that, before testing, had taken IgA deficiency into account as being a possible condition among the participants (by measuring total IgA) were preferred to avoid this being a source of error in the final results, as IgA deficiency is common in CD patients. 24
Results
EMA
Five studies (three case-control and two cohort studies) studied EMA assays: Four studied EMA IgA tests11,21–23 and one studied a EMA IgG test. 24
EMA IgA
The median
EMA IgG
The study by Villalta et al.
24
tested the performance of an EMA IgG assay. The only value reported was the
(See Table 1 for further information about the studies.)
tTG
Nine different studies (four case-control and five cohort studies) studied tTG assays: Eight studied tTG IgA10,11,18–23 and two tTG IgG.10,24
tTG IgA
The median
tTG IgG
Only two values of
(See Table 1 for further information about the studies.)
DGP
Eight different studies (five case-control and three cohort studies) studied DGP assays: Seven studied DGP IgA tests10,11,17–21 and seven studied DGP IgG tests.10,11,17–20,24
DGP IgA
The median
DGP IgG
Here the median
(See Table 1 for further information about the studies.)
Combination assays (DGP and tTG)
Median (range) for combination assays tested in different studies
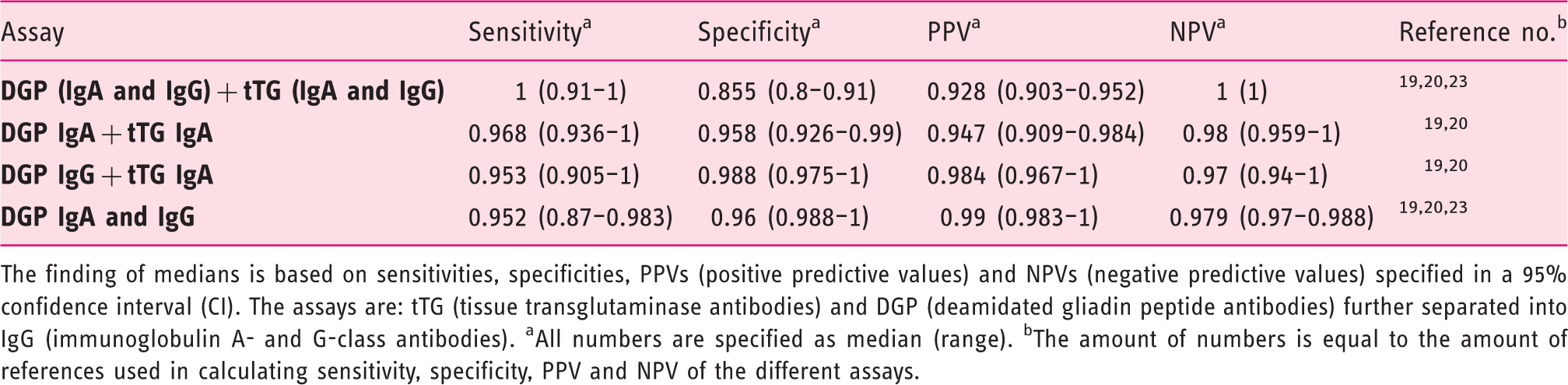
The finding of medians is based on sensitivities, specificities, PPVs (positive predictive values) and NPVs (negative predictive values) specified in a 95% confidence interval (CI). The assays are: tTG (tissue transglutaminase antibodies) and DGP (deamidated gliadin peptide antibodies) further separated into IgG (immunoglobulin A- and G-class antibodies). aAll numbers are specified as median (range). bThe amount of numbers is equal to the amount of references used in calculating sensitivity, specificity, PPV and NPV of the different assays.
Prevalences in cohort-studies
In the three cohort studies by Sugai et al., 19 Niveloni et al. 20 and Dahle et al. 23 included in this work, all patients were referred for gastroscopy because of suspected gastrointestinal (GI)/malabsorption disorder. All patients underwent duodenal biopsy.19,20,23 The prevalence of CD in the three studies extends from 39.1% to 44.9%, which is very high in consecutive patient materials, however. PPVs and NPVs are reported in two of the studies,19,20 and here all PPVs and NPVs are higher than those of the case-control studies, except for the NPVs of tTG IgA and the PPVs of DGP IgG, which are comparable with the case-control results. The same cut-off of 20 U/ml was used in testing all assays19,20,23 except for AGA, for which the cut-off was 25 U/ml. Dahle et al. 23 used the cut-off of 5 U/ml for the tTG IgA assay.
HLA-DQ
HLA DQ2/DQ8 typing is typically used to exclude CD in patients when misdiagnosis is suspected or in patients on a gluten-free diet for whom the diagnosis is unclear and gluten-challenge is not possible.9,14–16,26,27 Some find HLA DQ2 or DQ8 negativity to be sufficient to rule out a diagnosis of CD6,13,13,26,27 as the tissue types are considered necessary for the development of CD.28–30
In a study by Karell et al.,
31
61/1008 CD patients were found to be missing HLA DQ2 and DQ8 hetero-dimers. Of these 61 patients, 57 patients expressed
Torres et al. 28 investigated whether HLA G (coding for major histocompatibility complex (MHC) class I molecules) is associated with CD. All 24 CD patients studied expressed HLA G while none of the nine non-CD individuals did the same. In addition, raised levels of serum HLA G in patients with other diagnosis, e.g. Down syndrome or autoimmune thyroid disease, were observed while there was found low or negative values in CD patients on a gluten-free diet for more than five years. Fabris et al. 29 confirmed the results of Torres et al. Trynka et al. recently found 13 new CD risk loci, bringing the number of known loci (including HLA) to 40. 36
Discussion
IgA tests
EMA has a low sensitivity: (0.835, (0.61–0.937)), compared to earlier studies that rate EMA as the best test (comparable with tTG).6,26,33 Dahle et al. 23 explain the low sensitivity of EMA IgA in their study by the relatively old age of all participants included (half of them are 50 years or above) as old age has been connected to less true-positive EMA results compared to studies that include younger adults. Despite the generally low sensitivity, EMA IgA has the highest specificity of all assays together with a high NPV, and therefore it is usable to identify CD, e.g. in persons in a low-risk group. tTG IgA has the highest sensitivity of all (single) assays and a high specificity overruled only by DGP IgA, which also has the second highest sensitivity. This indicates that the assay is suitable for identifying persons with CD and can be used for screening purposes. The high values match the results from other studies that also find tTG IgA to be the most successful test together with EMA.3,6,8,26,34,37
IgG tests
There is insufficient data about EMA IgG and tTG IgG in the selected studies to make firm conclusions. Earlier studies have shown both low sensitivities and low specificities of the two assays.8,33 Kaukinen et al. 21 show an impaired performance of AGA assays when non-CD patients have symptoms compatible with CD. In their study, 54% of the non-CD control group was tested positive for AGA.
DGP IgG shows superior performance compared to DGP IgA and other IgG assays. It has a relatively high sensitivity, a high specificity and the highest PPV of all (single) assays. DGP IgG could be especially effective in persons with IgA deficiency (concluded by others as well8,10,20,24) and it could therefore be used in routine testing as a supplement to a tTG IgA test.
All combination assays have higher sensitivities than each single assay alone (Table 3). Almost all combinations have higher specificities exceeded only by DGP IgG (0.996 (0.985–1)) and EMA IgA (1 (0.98–1)). All the PPVs of the combinations are higher than those of AGA and EMA IgA (single assays) while all the NPVs are higher than those of the single assays alone.
When comparing the different study designs, cohort studies are preferable over case-control studies if the prevalence rates of CD in the cohort are realistic (Table 2). Cohort studies use consecutively chosen patients with symptoms compatible with CD and provide reliable predictive values if prevalence rates are relevant. None of the patients involved in the included cohort studies were previously diagnosed with or excluded from having CD (except for Dahle et al. 23 (see Table 1)) and thereby expectation bias is avoided, which can be an issue in case-control studies if they are not properly blinded. Cases are specifically chosen and have all been diagnosed with CD, while the control group often consists of healthy individuals without symptoms or other patient groups. Biopsy is omitted (or not reported) in some control groups11,17 and therefore the presence of CD cannot be excluded with certainty.
Comparison of the three cohort studies included in this work by Sugai et al., 19 Niveloni et al. 20 and Dahle et al. 23 show prevalences of CD extending from 39.1% to 44.9%. This is a very high prevalence of CD in consecutive materials even in highly specialized practice, although reservations due to overlapping patient material must be taken into account for Sugai et al. 19 and Niveloni et al. 20 The PPVs and NPVs reported in these two studies19,20 show that all PPVs and NPVs are higher in these study populations than those of the case-control studies, except for the NPVs of tTG IgA and the PPVs of DGP IgG, which are comparable with the case-control results. The cut-off values of the assays are the same in the three cohort studies, which justifies comparison of the study results.
For many years it has been known that there is a strong association between the tissue types HLA DQ8 and DQ2 and the risk of developing CD.5,6 Karell et al.
31
confirm this, as 57 patients lacking the HLA DQ2 dimer express
In conclusion, tTG IgA and DGP IgG show the best performance from a clinical diagnostic standpoint. It seems as preferable as combining two or more assays (especially DGP (IgA and IgG) + tTG (IgA and IgG) or DGP (IgA and IgG)) as this shows both very high sensitivities and specificities (Table 3) compared to the single assays as concluded by others.7,9,14,16 However, data derive from three studies only, and additional prospective studies as well as cost-benefit analyses are necessary before final recommendations can be made. In addition, more cohort studies should be conducted using DGP assays (both alone and combined with tTG) to confirm or refute the usefulness of these tests compared to other assays (tTG or EMA alone). Based on the above studies in adults, it is still premature to recommend a diagnostic strategy for CD in adults that omits duodenal biopsy. Such a strategy seems feasible in subgroups of children with CD. Here intestinal biopsies can be avoided in one-third or more of seropositive individuals in the presence of high CD-specific antibody titers, gluten-dependent symptoms and the presence of HLA DQ2 and/or HLA DQ8. 13 Further cohort studies in adults, also taking predictive values of antibody titers into consideration, are anticipated, preferably with designs allowing evaluation of diagnostic strategies that aim to omit intestinal biopsies.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
