Abstract
Objectives
Altered immune homeostasis and involvement of T cells has been reported in chronic pancreatitis (CP). We evaluated the role of Bach2 (BTB and CNC homology basic leucine zipper transcription factor 2), a key regulator of immune homeostasis in the chronicity of CP.
Methods
Expression of Bach2 and T-cell transcription factors, enumeration of BACH2+/CD4+ T-lymphocytes were performed by qRT-PCR and flow cytometry respectively. Bach2silenced human CD4+ T-lymphocytes were exposed to CP tissue extract to assess T-cell lineage commitment. Aryl hydrocarbon receptor (Ahr) and Deubiquitinase enzyme A (DUBA/OTUD5gene) were evaluated as markers of persistent Th17 cell differentiation. Bach2 gene (exons) was sequenced to identify risk variants and functionally validated.
Results
Decrease in Bach2 (p < 0.0001) and increase (p < 0.001) in TBX21, RORC, Ahr, PRDM1, IL23R mRNA were noted in pancreatic tissues, while BACH2+/CD4+ T-lymphocytes were decreased (p < 0.01) in circulation and tissues. Exposure of Bach2 silenced CD4+ T-lymphocytes to CP tissue extract showed increased Ahr, decreased OTUD5, and enhanced Th17 cell differentiation. Sequencing of Bach2 gene revealed association of novel variant (rs9111 in 5′-UTR) with advanced disease and luciferase assay confirmed its role in Bach2 repression.
Conclusion
Bach2 repression mediates Th17 cell induced inflammation and rs9111-TT in individuals with primary genetic susceptibility to CP is associated with clinical features of advanced disease.
Introduction
Although, recurrent episodes of acute pancreatitis (AP) and persistent inflammation are known to progress to chronic pancreatitis (CP), the natural course of transition and progression from AP to CP is not completely elucidated. 1 While earlier studies reported altered immune homeostasis, recent investigations demonstrated an increase in Th1 and Th17 cells both in circulation and pancreatic tissues of CP patients.2,3 Differentiation of naïve T cell to Th1 cell lineage depends on the transcription factor TBX21 and RORC specifies Th17 cell lineage.4,5 Developmental flexibility of Th17 cells and differentiation to Th1 cell like phenotype have been reported in the pathogenesis of inflammatory diseases of gastrointestinal system such as colitis, celiac, and Crohn’s.6–9 Although several studies have suggested a role for the cellular adaptive immune system in the pathogenesis of CP both in humans and rodent models, the nature of specific transcriptional programs initiating niche specific local T-cell lineage commitment and polarization leading to advanced form of the disease has not been studied.
Bach2, a BTB and CNC homology basic leucine zipper transcription factor 2, was recently identified as a key regulator of T-cell mediated immune homeostasis.10,11 The locus encoding Bach2 is recognized to have a prominent T-cell super enhancer architecture involving cytokines and cytokine receptors, wherein genes critical for T-cell differentiation are repressed by BACH2. 12 It is also shown to balance immunity and tolerance, 13 regulate CD8(+) T-cell differentiation, 14 as well as promote tumor immunosuppression. 15 In addition, polymorphisms within the Bach2 locus in humans were found to be associated with susceptibility to inflammatory and auto immune diseases.16–20 We conducted this study to assess the clinical relevance of Bach2 associated inflammatory T-cell phenotypes and their role in persistent inflammation in CP. We sequenced the entire exonic region of Bach2 gene in CP patients to identify polymorphisms, with plausible association to the disease. Our results demonstrate that Bach2 is repressed in CP with concomitant polarization of T cells towards Th17 axis. In addition, a novel single-nucleotide polymorphism (SNP) (rs9111) in the 5′-UTR region was found to be associated with repression of BACH2 and advanced disease.
Patients and methods
Patients
Patients with documented CP were recruited prospectively between 2013 and 2015 (Figure 1) and were confirmed to have definite CP as per M-ANNHEIM criteria (recurrent pancreatitis, calcifications, ductal lesions, and typical pancreatic histology). CP patients (n = 35) undergoing surgery for pain management were screened and idiopathic CP patients (n = 28) without alcohol consumption and smoking were included as part of the discovery cohort for evaluating expression of T-cell transcription factors (tissues) and genotyping (blood). Pancreatic tissues obtained from patients undergoing resections for pancreatic adenoma and malignancies other than pancreatic without history of CP (n = 22) formed the control group. Patients with pancreatic malignancies, endocrine tumors, acute on CP, and autoimmune problems were excluded. CP was confirmed either with EUS (Rosemont criteria), MRCP (presence of ductal changes), ERCP (Cambridge criteria), CECT (presence of pancreatic atrophy and/or ductal dilatation and/or calculi/calcification), or trans-abdominal USG (presence of pancreatic atrophy and/or ductal dilatation and/or calculi/calcification). Development of diabetes subsequent to the onset of CP was considered to be pancreatogenic diabetes (type 3 C) with fasting glucose levels of more than 100 mg/dL.
21
Blood samples were obtained from healthy controls (n = 198) and CP patients (idiopathic-302, alcoholic-50) for validation. Ductal dilatation, main pancreatic duct size, side branch involvement, calcifications, strictures, obstruction in pancreatic duct were the criteria used to classify advanced CP as per Cambridge classification.
22
Whole blood and tissues were used for enumeration of BACH2+/CD4+ T-lymphocytes, DNA and RNA isolation. The study was approved by ethical review board of Asian Institute of Gastroenterology (December 2013).Written informed consent was obtained from all the participating individuals and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
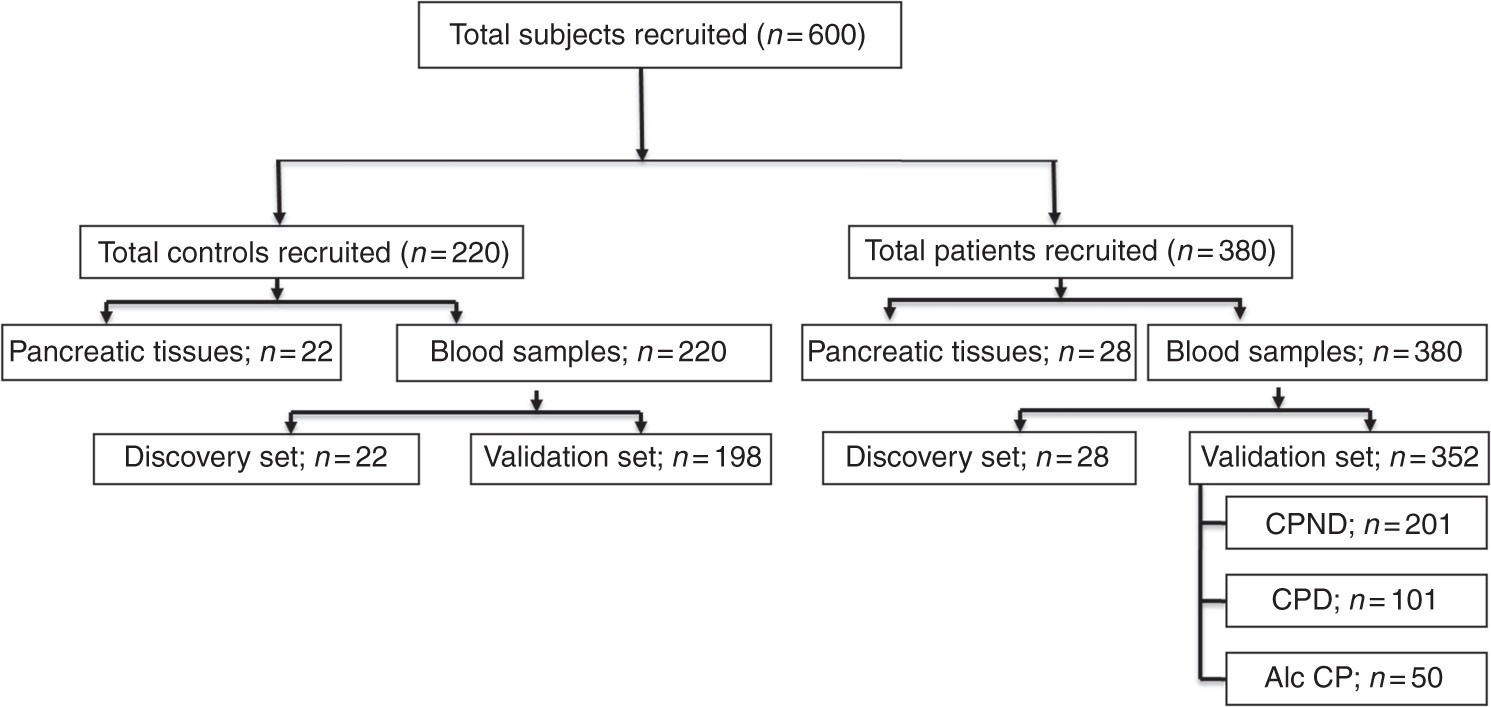
Methods
Expression of Bach2 (mRNA and protein) and T-lymphocyte transcription factors were evaluated (primers – see Supplementary Table 1 online) and BACH2+/CD4+ T-lymphocytes were enumerated both in pancreatic tissue and circulation. T-lymphocyte differentiation pattern was studied after exposing the Bach2 silenced lymphocytes to CP tissue extract and Th17 inducing conditions. Bach2 exons were sequenced to identify association of variants with severity of the disease. Characteristics of advanced form of CP were statistically evaluated between the genotypes of Bach2. Detailed methodology is given online in the supplementary methods section.
Results
Clinical characteristics of CP patients.

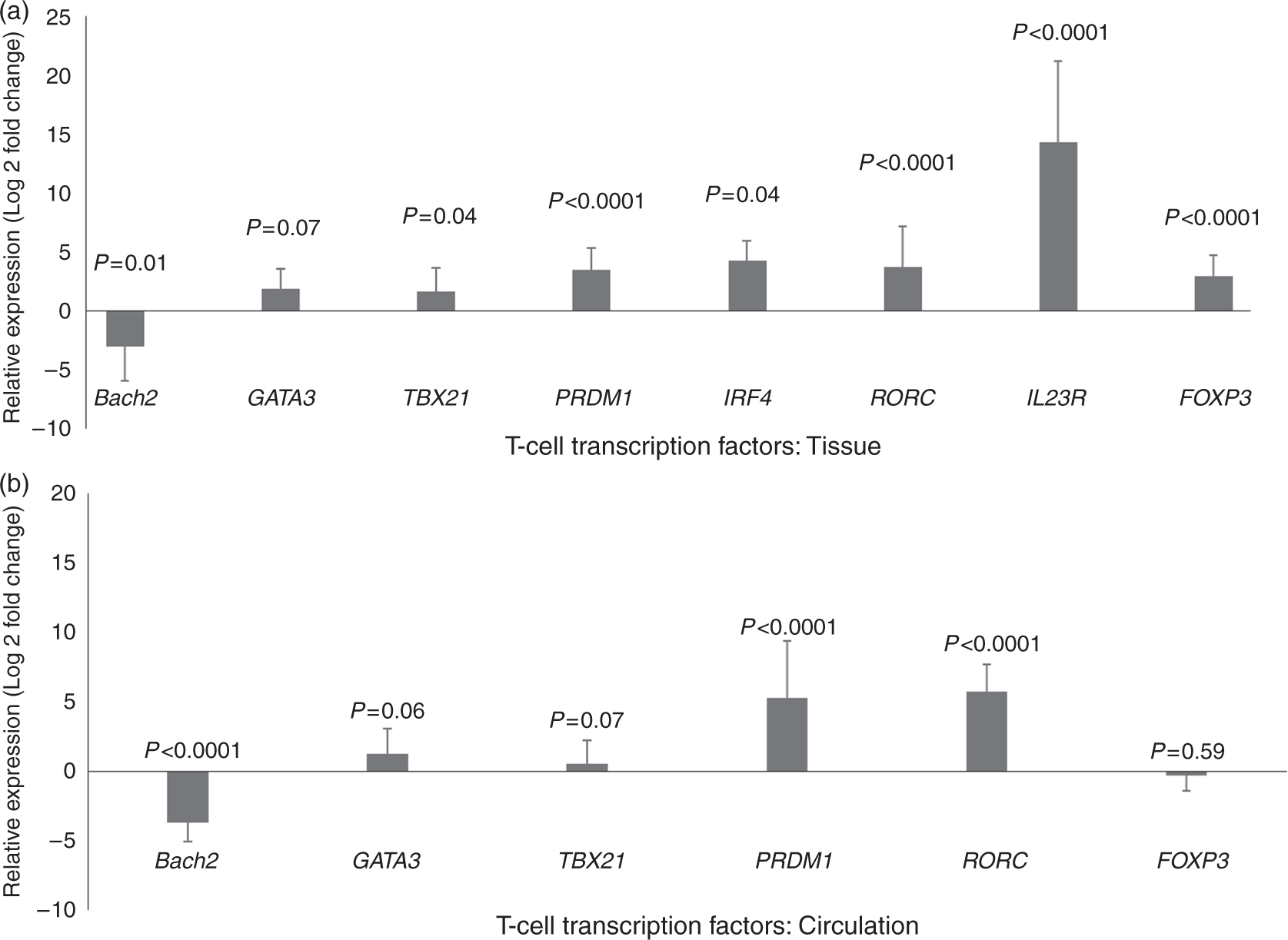
mRNA of Bach2 and effector T-lymphocyte transcription factors are differentially expressed in CP
Bach2 was decreased in CD4+ T-lymphocytes (−3.0 ± 2.93 fold, p = 0.01) isolated from pancreatic tissue, while TBX21 (1.64 ± 2.00 fold, p < 0.04) and RORC (3.74 ± 3.43 fold, p < 0.0001) levels were increased in CP as compared to controls. A significant increase was also noted in IL23 receptor gene expression (IL23R) in pancreatic tissue of CP patients (14.36 ± 6.88 fold, p < 0.0001). PRDM1 and IRF4 required for T-helper cell polarization and FoxP3 required for regulatory T-lymphocyte differentiation were also increased (3.5 ± 1.86, 4.3 ± 1.69 and 2.94 ± 1.80 fold, respectively; p < 0.0001, Figure 2). A similar decrease in Bach2 expression was observed in circulating T-lymphocytes (−3.68 ± 1.35 fold, p < 0.0001) of CP patients along with increased expression of RORC and PRDM1 (5.75 ± 1.89 fold and 5.28 ± 4.11 fold, respectively; p < 0.0001, Figure 2).
Bach2+/CD4+ T-lymphocytes are decreased in CP
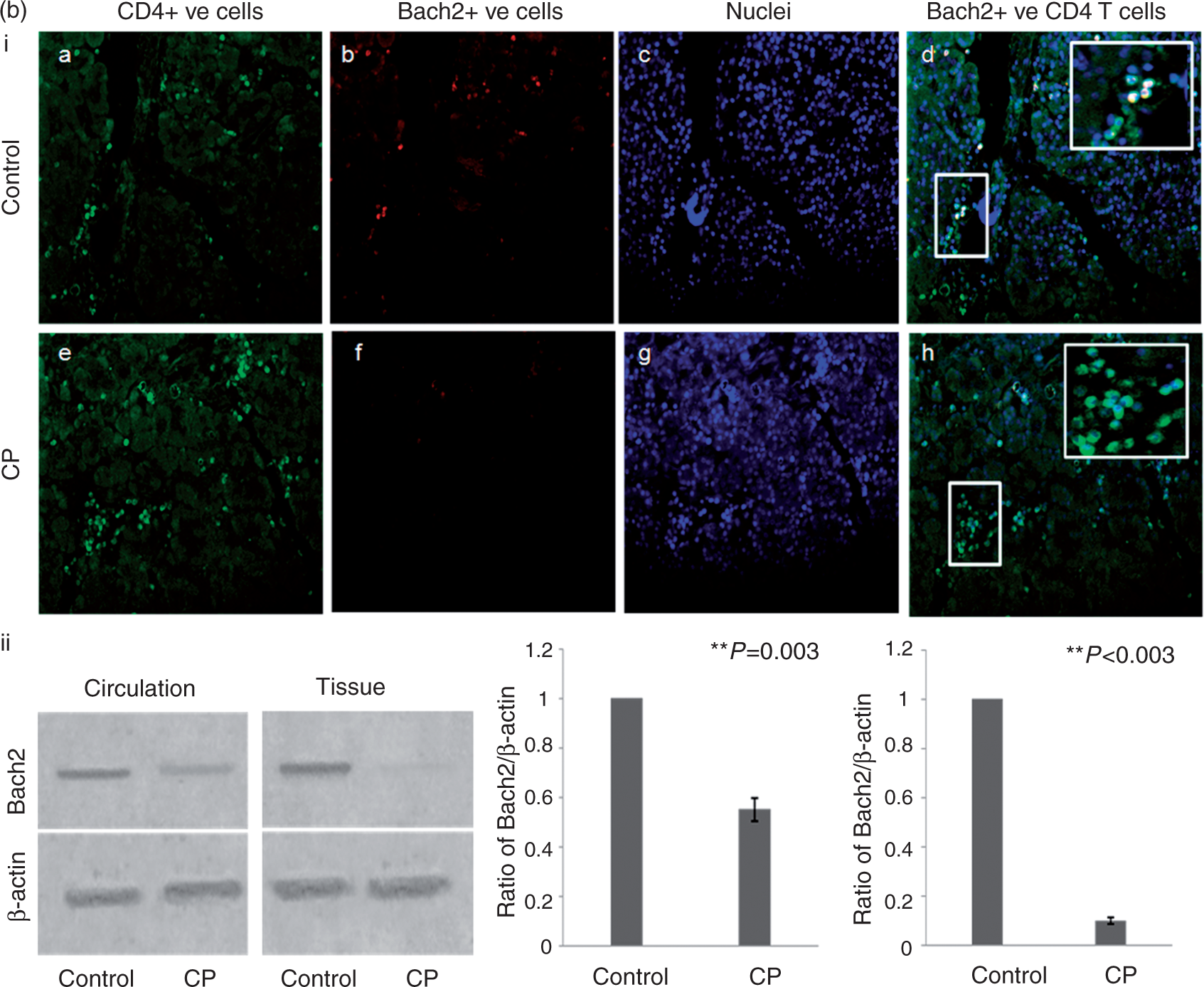
While the total number of circulating CD4+ T-lymphocytes increased in CP patients (control: 18.7 ± 3.66; CP: 29.81 ± 6.88; p < 0.028), the number of Bach2+/CD4+ T-lymphocytes decreased [control: 24.67 ± 5.92; CP: 4.27 ± 2.04; p < 0.004; Figure 3(a)]. A similar increase in CD4+ T-lymphocytes (control: 63 ± 21/hpf; CP: 110 ± 27/hpf) and decrease in Bach2+/CD4+ T-lymphocytes (control: 53 ± 26/hpf; CP: 25 ± 12/hpf) was observed upon immunohistological and immunofluorescent staining of tissue sections obtained from CP patients (see Supplementary Figure 1). Western blot revealed a decrease in BACH2 protein expression [p < 0.05; Figure 3(b)].
(a) (b) (i) Immunofluorescent staining of pancreatic tissues for CD4+ (green), Bach2 (red) depicts Bach2+/CD4+ T cells (double positive cells seen in yellow) in controls and CP patients. (ii) Representative western blots of Bach2 protein in circulation and pancreatic tissue in controls and CP patients.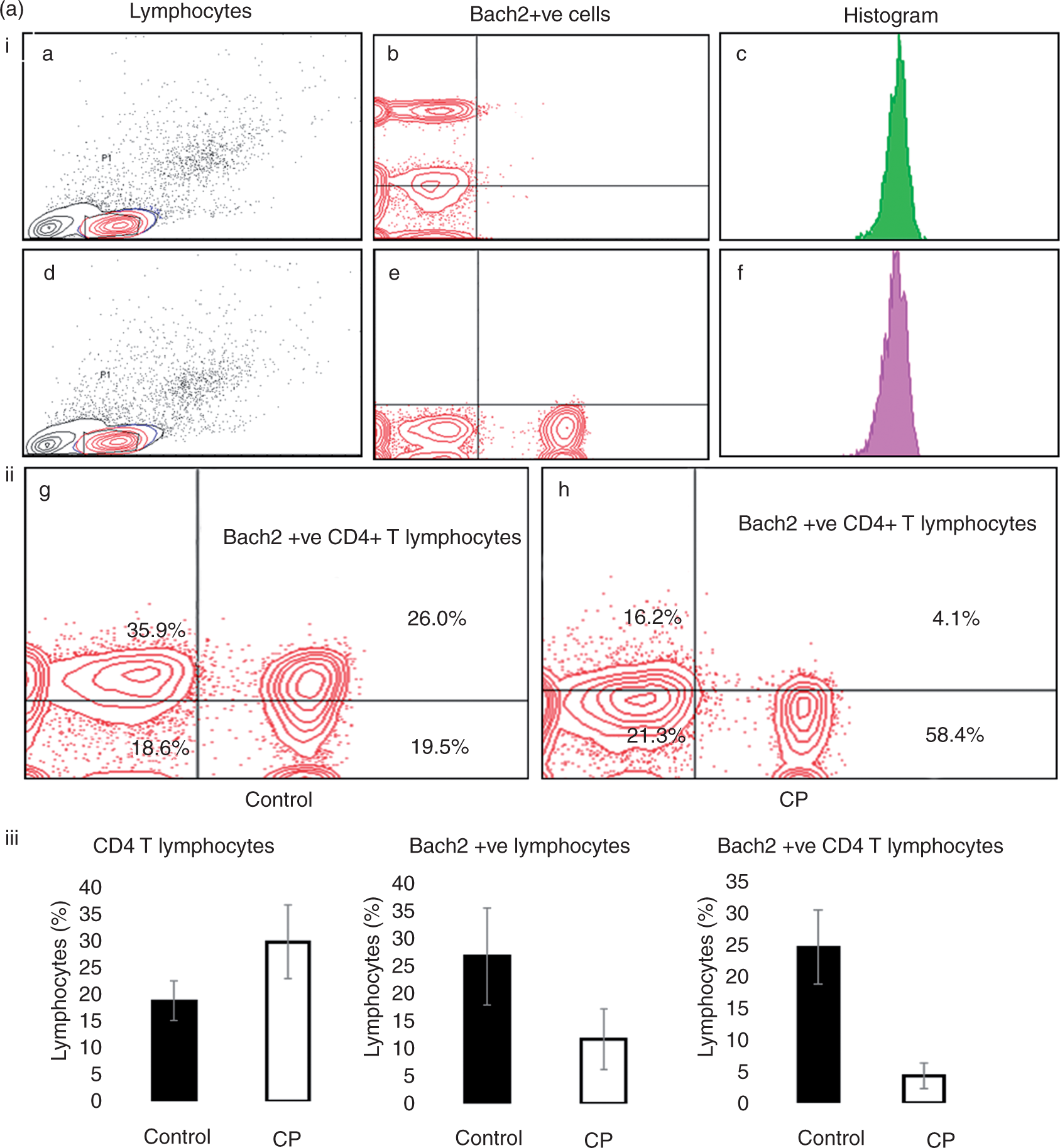
ShRNA mediated Bach2 silencing and exposure to CP tissue extract/Th17 inducing conditions contribute to effector T-lymphocyte differentiation in vitro
Bach2 silenced cells showed increased effector T-lymphocyte differentiation (Th1, Th2, and Th17 cells) in almost equal proportions (31.0 ± 3.0, 21.1 ± 1.7, and 20.1 ± 1.0), while a propensity towards Th1 and Th17 cells was observed on exposure of Bach2 silenced cells to both CP tissue extract (45.0 ± 4.2, 42.6 ± 2.9, and 38.9 ± 2.6) and Th17 inducing conditions (59.9 ± 5.4, 56.6 ± 4.0, and 49.6 ± 3.6; Figure 4) in comparison to controls (25.0 ± 2.3, 16.3 ± 1.4, and 11.4 ± 1.2; Figure 4).
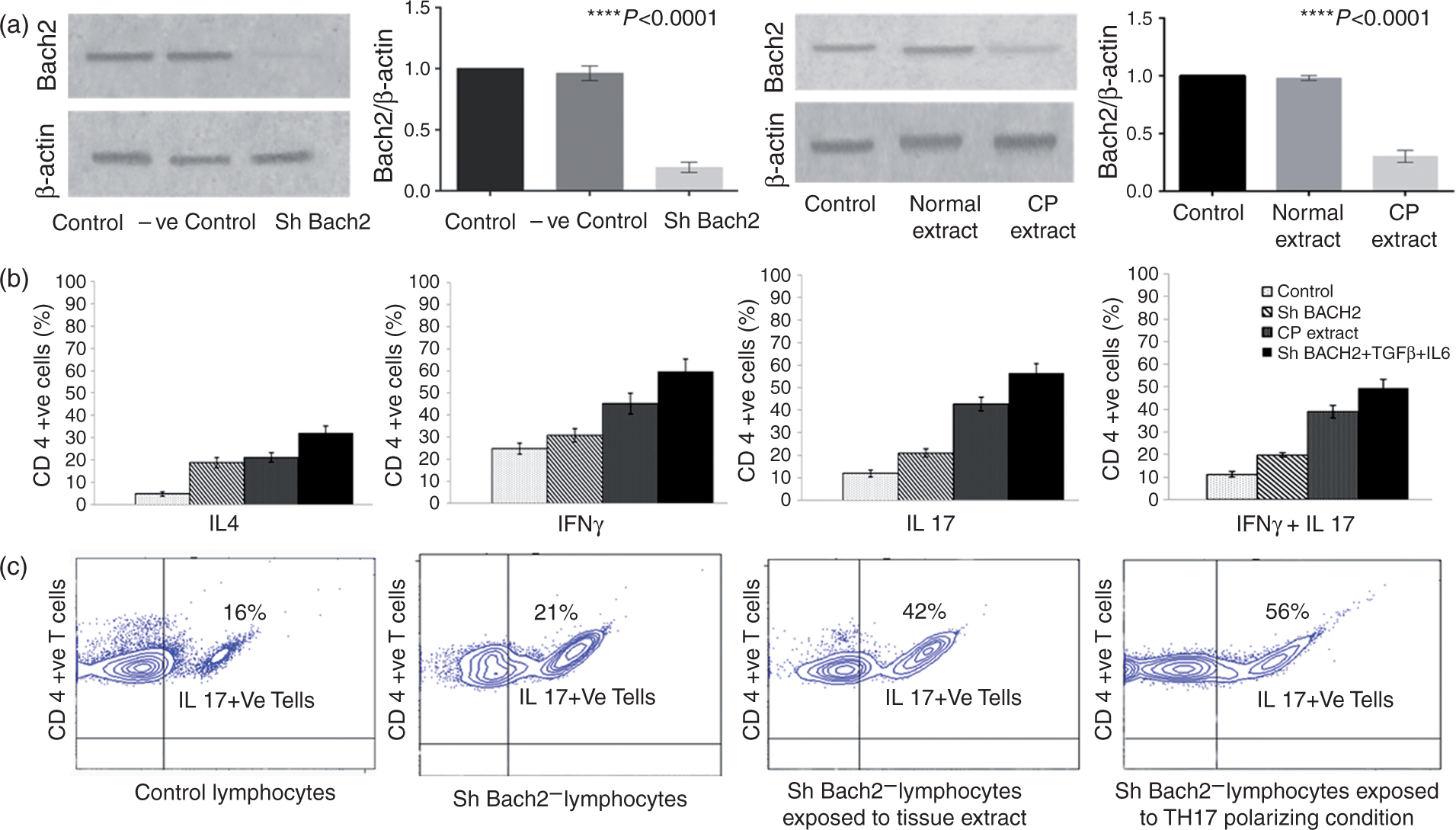
Increased aryl hydrocarbon receptor ascertain Th17 cell differentiation in CP
The aryl hydrocarbon receptor (Ahr) required for Th17 cell differentiation was upregulated (3.29 ± 0.99 fold, p < 0.01), confirming the differentiation of naïve T-lymphocytes to Th17 cells under inflammatory conditions in CP. DUBA (a cell intrinsic suppressor of IL17 production; OTUD5 gene) required to ubiquitylate RORC for its clearance was seen to be marginally increased (2.86 ± 1.20 fold, p < 0.05) in CP lymphocytes. However, exposure of CD4+ T-lymphocytes to CP tissue extract showed increase in Ahr and decrease in DUBA expression (Supplementary Figure 2).
Bach2 exon sequencing reveals SNPs in 5′-UTR in CP
Sequencing Bach2 exons (primers – see Supplementary Table 2) identified association of two intronic (rs45483891, rs182382) and two exonic SNPs with CP in the discovery set (Supplementary Table 3). The two exonic SNPs were located in exon 2 which is part of 5′-UTR. One SNP was novel (rs9111) with the major allele “T” being significantly associated with CP (p = 0.04, odds 4.8). The “T” allele of the other exonic SNP (rs45553631) was also associated (p = 0.05, odds 3.88).
Allelic frequency in the validation set
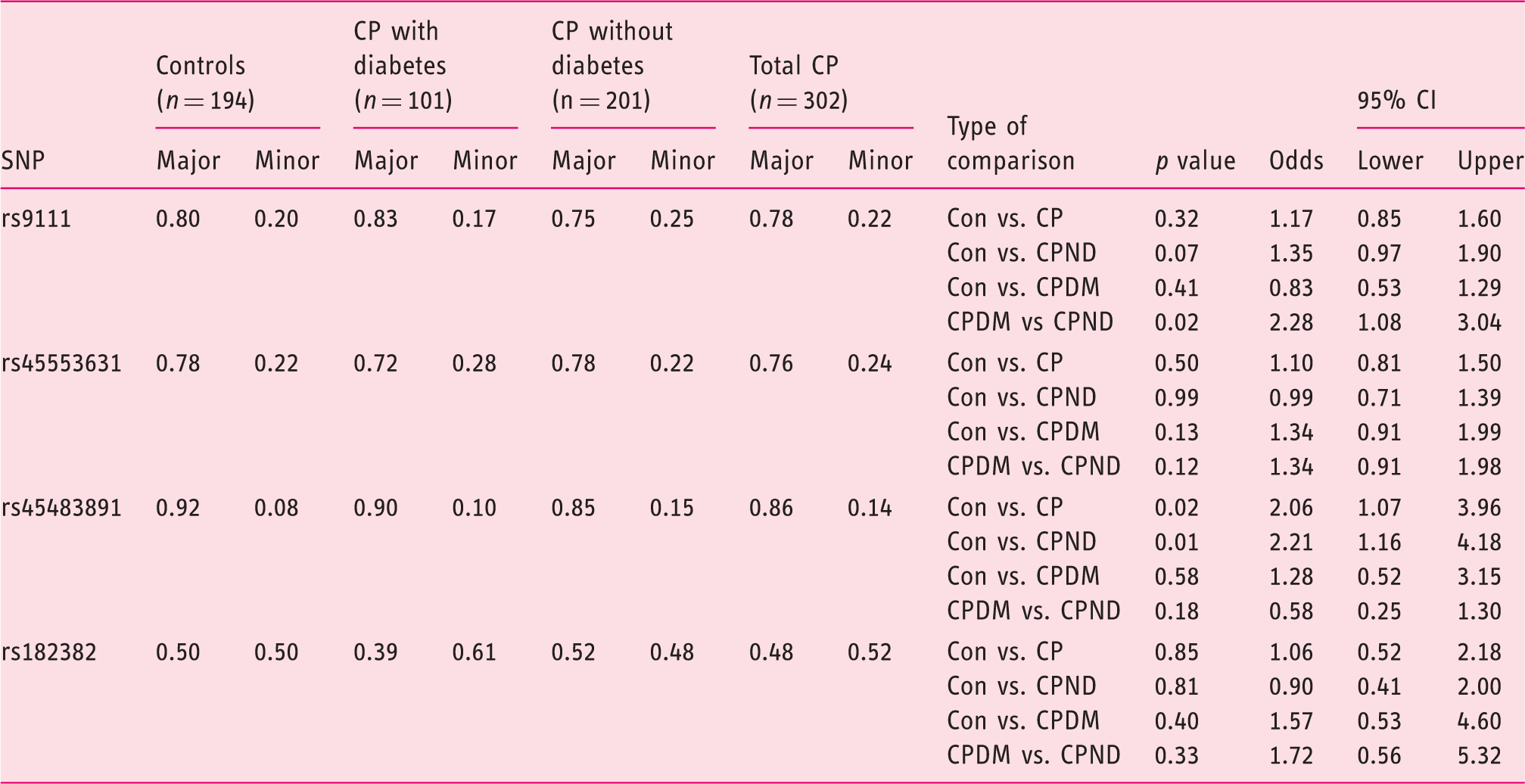
Con – control; CP – chronic pancreatitis; CPND – chronic pancreatitis without diabetes; CPDM – chronic pancreatitis with diabetes.
Luciferase assay confirms the functional role of rs9111
Both rs9111 and rs45553631 were included in the luciferase experiments as they are close (4 bases apart) to the intron-exon boundary. C allele at both the positions was considered as “wild type”, T-rs9111; C-rs45553631 as “single mutant” and T-rs9111; T-rs45553631 as “double mutant”. In comparison to wild type, a significant increase in Bach2 expression was noted (by 20% and 40%, respectively) with single and double mutants [Figure 5(a)]. However, upon exposure of the constructs to CP tissue extract, 70% and 81% decrease in Bach2 expression was observed as compared to the wild type [p = 0.04; Figure 5(b)].
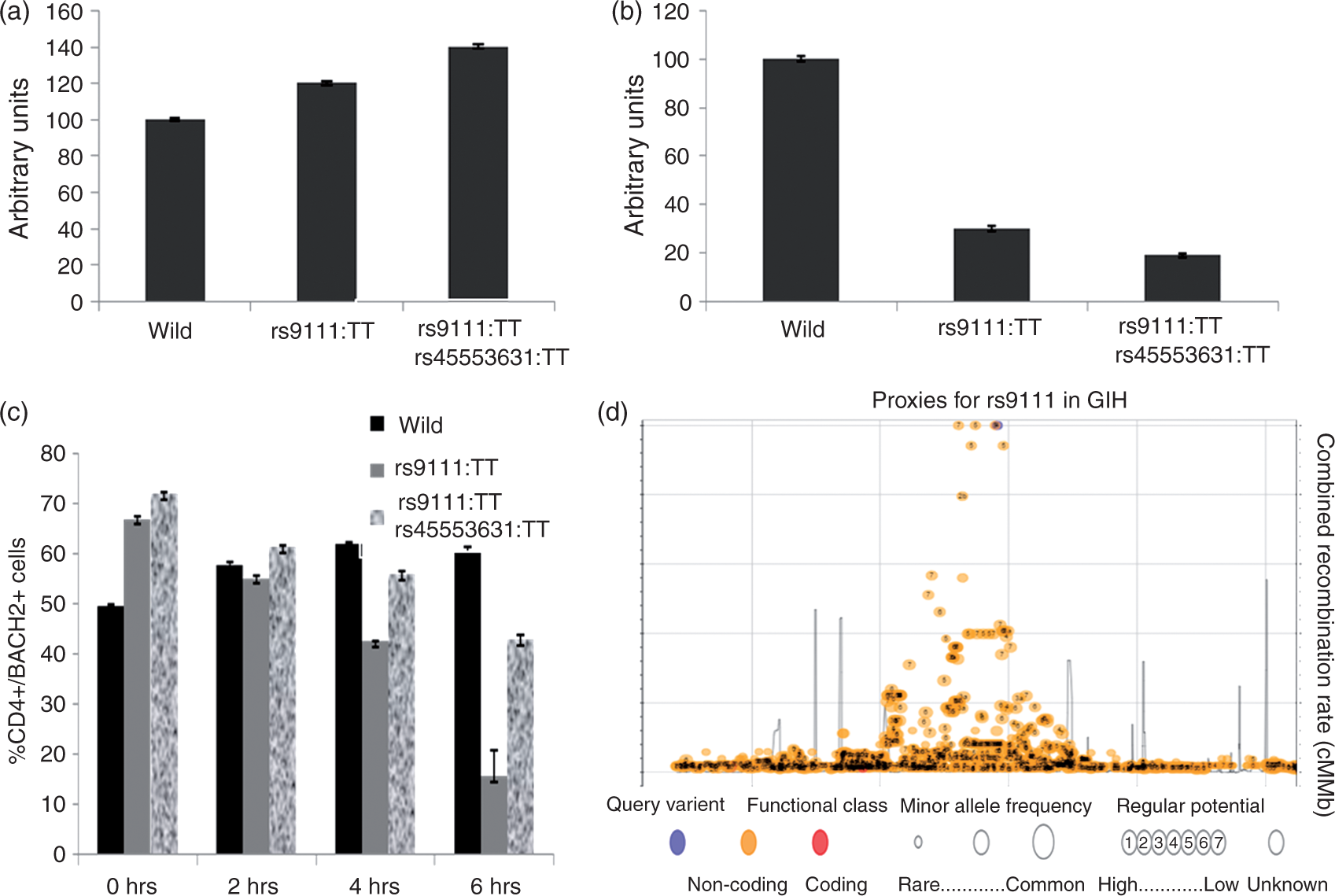
Exposure of CD4+ T-lymphocytes with variants in rs9111 to CP tissue extract revealed genotype based differences in BACH2+/CD4+ T-lymphocytes
CD4+ T-lymphocytes from individuals with various genotypes were exposed to CP tissue extract to assess genotype based differences in Bach2+/CD4+ T-lymphocytes. On exposure, the percentage Bach2+/CD4+ T-lymphocytes at base line was lower in wild type (rs9111; C; rs45553631; C 49.4%) as compared to single (rs9111; T 66.8%) and double mutants (rs9111; T and rs45553631; T 71.8%). While a gradual increase was observed in the number of cells from 0, 2, 4, and 6 h in wild type (49.4%, 57.7%, 61.8%, and 60.2%), a gradual decrease was observed in both single (66.8%, 54.9%, 42.3%, 15.4%) and double mutants (71.8%, 61.2%, 55.7%, and 42.7%) [Figure 5(c)]. Linkage and proxy analysis with GIH (Gujrati-Indian from Houston) data for rs9111 revealed a cluster with rs9111 being the only variant with regulatory potential [Figure 5(d)].
Association of rs9111-TT genotype with advanced disease
rs9111-TT was found to be associated (p = 0.002) with higher MPD size (6.22 ± 3.04 in TT vs. 3.98 ± 2.34 in CT and CC) and higher number of CP patients with ductal dilatation (61% in TT vs. 33% in TC and CC; p = 0.001), pancreatic atrophy (15% in TT vs. 3.2% in TC and CC; p = 0.003), and ductal obstruction (31% in TT vs. 19% in TC and CC; p = 0.01). The phi coefficient between genotype and MPD size is 0.35 (p = 0.004).
Discussion
In this study, we demonstrate repression of Bach2, known to regulate immune homeostasis leading to polarization of T-helper cells towards Th17 cell axis and higher inflammation in CP. Additionally, we identified a novel SNP in the 5′-UTR region that is found to be associated with its repression and advanced disease.
Differential expression of T-lymphocyte transcription factor mRNAs (TBX21, GATA3, RORC, PRDM1, IRF4, and FOXP3) in CP patients (tissue and circulation) point to altered immune homeostasis. A significant increase in mRNA levels of TBX21 and RORC in pancreatic tissue suggests differentiation of T cells towards Th1 and Th17 locally in CP (Figure 2). We and others have earlier reported an increase in circulating and intra pancreatic Th1 and Th17 cells in CP patients.2,3 In contrast to observations made with tissue, no significant alterations in TBX21 expression were seen in circulation indicating locally induced differentiation of T-lymphocytes in CP. Interestingly, we noticed a highly significant increase in transcription factor RORC mRNA levels in pancreatic tissue as well as in circulation, suggesting both local and systemic differentiation of Th17 cells. Since it is recognized that naïve T-lymphocyte differentiation into effector cells/Tregs is regulated by Bach2, 10 significant decreases observed in the expression of Bach2 both at mRNA and protein levels point to its role in altered immune homeostasis in CP.
Noticeable increase in mRNA levels of other T-lymphocyte transcription factors such as PRDM1 and IRF4 normally stifled by Bach2 to facilitate the differentiation of Tregs also indicates imbalance in CP. Surprisingly, mRNA of FOXP3 increased in CP tissues, possibly due to elevated levels of PRDM1 in Bach2 repressed state, 25 and it is shown that CP is associated with disease specific Treg responses. 26 Roychoudari et al. recently showed that cellular differentiation was diverted towards Th2 lineage in the lymph nodes of Bach2 knockout mice. 10 Our study demonstrated that under Bach2 repressed condition in pancreas, CD4+ T-cell differentiation was triggered towards IL17 secreting Th17 cellular axis. Recently, overexpression of IL17 was shown to induce multiple tissue inflammation including pancreatitis.27,28 It is of importance to note that despite increase in CD4+ T-lymphocytes in CP, Bach2+/CD4+ T-lymphocytes were reduced. These altered transcriptional programs towards Th17 cellular axis indicate higher inflammatory conditions under Bach2 repressed state in human CP. However, it would be interesting to study the contribution of T cells in chemically induced CP in animal models. Th17 cells are recognized to have key pro-inflammatory roles in autoimmune diseases, inflammatory bowel disease (IBD), and graft versus host disease.29–32 In our study, up regulation of IL23R mRNA in pancreatic tissue of CP patients clearly suggest the involvement of pathogenic Th17 cells. Thus, elevated expression of RORC and IL-23R in the same tissue imply the ascendancy of pathogenic Th17 cells over Th2 lineage in CP. Absence of detectable IL23R in circulation further suggests the influence of altered micro environment at tissue level that influence naïve Th17 cells to attain pathogenic phenotype.
It is to be noted that ShRNA mediated silencing of Bach2 in human CD4+ T-lymphocytes resulted in differentiation of almost equal proportions of Th1, Th2, and Th17 cells as reported earlier in mice. It is interesting to note that Bach2 expression was also repressed when normal CD4+ T-lymphocytes were exposed to CP tissue extract and T-lymphocyte differentiation was more towards Th1 and Th17 akin to the higher expression levels of T-lymphocyte transcription factor mRNAs such as TBX21, RORC seen in CP (Figure 4).
Much greater T-lymphocyte differentiation towards Th17 was noted when Bach2 silenced lymphocytes were exposed to Th17 inducing conditions indicating that TGFβ and IL6 have an added effect on differentiation of T-lymphocyte into Th17 cells in CP (Figure 4). It is well established that CP tissue contains elevated levels of cytokines, 33 including TGFβ and IL6 which promote differentiation of Th17 cells in Bach2 repressed state in CP. It is also known that Th17 cells transdifferentiate into regulatory T-lymphocyte, a hallmark of resolution of inflammation, is largely dependent on canonical TGF beta signaling and Ahr status. 34 The observed increased expression of Ahr which favors Th17 cell differentiation also affirm T-cell lineage commitment towards Th17 under inflammatory conditions in CP. Rutz et al. showed that the deubiquitylating enzyme DUBA is a negative regulator of IL17A production in T-lymphocyte and mice with DUBA-deficient T-lymphocyte developed exacerbated inflammation in the small intestine. Although, DUBA showed marginal increase in CD4+ T-lymphocytes obtained from CP patients, its decrease upon exposure of normal CD4+ T-lymphocytes to CP tissue extract is a sign of persistent accumulation of RORγt. This can trigger persistent local differentiation of Th17 cells and higher inflammation. 35 These results established that Bach2 repression resulting in altered immune cell transcriptional programs, increased TGFβ and IL6 might be playing a crucial role in higher inflammation and severity of CP.
Since Bach2 expression was found to be repressed both in circulation and in pancreatic tissue, identifying the likely contribution of SNPs to the down regulation of Bach2 gene was assessed. The intronic SNP (rs45483891) was associated with a two-fold higher risk for CP and the exonic SNP (rs9111) was significantly associated with diabetic phenotype in CP (considered as higher inflammation/advanced disease).It is interesting to note that majority of the Bach2 variants reported to be associated with various human autoimmune and inflammatory diseases are in the intronic region. To our knowledge, this is the first report demonstrating association of exonic SNP (rs9111).Further, the exonic variant is part of low haplotype diversity, essentially forming an LD block with SNPs that are associated with other autoimmune diseases like Graves’s, autoimmune thyroid disease, celiac, Type 1 diabetes, and Crohn’s disease (http://analysistools.nci.nih.gov/LDlink/?tab=Idhap). While all these variants are in the introns, rs9111 that is part of 5′-UTR could be the functionally relevant SNP that is associated with these phenotypes. The association of rs9111-TT with higher inflammation (phenotype manifested as diabetes) could be due to its contributory role in down regulating Bach2 gene expression. It has been shown that Bach2 inhibition also promotes cytokine induced β cell apoptosis. 36
Significant increase observed in fluorescence units in single (20%) and double mutant (30%) in comparison to wild type, upon functional evaluation of rs9111 employing luciferase assay, indicates increased Bach2 expression in mutants. However, gradual decrease in Bach2+/CD4+ T-lymphocytes upon exposure of CD4+ T-lymphocytes with wild and mutant genotypes to CP tissue extract indicate the role of Bach2 variant in downregulating its expression under inflammatory conditions.
In addition, patients with primary susceptibility to CP (variants in PRSS1-2 and CTRC genes) and rs9111-TT genotype were found to have advanced disease. Constant pancreatic injury arising as a result of primary susceptibility in these patients and repressed Bach2 state arising due to rs9111-TT in Bach2 gene may lead to higher inflammation and advanced disease. Our results in CP corroborate with a recent study by Lee et al published in Nature genetics demonstrating independent nature of genetic susceptibility in the initiation and progression of Crohn’s disease. 37
Association of rs9111-TT genotype with ductal dilatations, MPD size and ductal obstructions indicate a role for this SNP in higher inflammation and advanced disease in these patients. Further, functional validation from this study demonstrates the role of rs9111 in transcriptional repression of Bach2 under inflammatory conditions. The extent of repression is influenced by the genotype and the inflammatory environment as demonstrated by exposing the T-lymphocytes with different genotypes to CP tissue extract. This repression of Bach2 along with Th17 polarizing conditions prevailing in CP (increased TGFβ and IL6) might elicit activation signals locally leading to enhanced differentiation of CD4+ T-lymphocytes towards pathogenic Th17 cells resulting in higher inflammation ensuing severe form of fibrosis. As a result, ductal distortions and obstructions may develop leading to advanced form of the disease.
In conclusion, our study demonstrates that transcriptional repression of Bach2 gene in CD4+ T-lymphocytes of CP patients can be a key event that drives polarization of pathogenic Th17 cells. Further, presence of variant in 5′-UTR of Bach2 gene along with primary susceptibility to CP can aggravate the disease towards a more advanced/severe form. Our results also point to the therapeutic potential of modulating Bach2 expression to reduce persistent inflammation in CP.
Footnotes
Acknowledgments
The authors thank Dr RM Mukherjee, Asian Healthcare Foundation for participating in the discussions on the topic. We thank Dr HVV Murthy, Asian Healthcare Foundation for his help with statistical analyses. The authors are grateful to Prof. Dayananda, Dr Sunil from University of Hyderabad, and Prof. Pratibha Nallari, Department of Genetics, Osmania University for the help rendered in cloning Bach2 gene and Prof. C Subramanyam for editing the manuscript.
Declaration of conflicting interests
The authors report no conflicts of interest.
Ethics approval
The study was reviewed and approved by Institutional Ethics committee of Asian Institute of Gastroenterology (December 2013)
Funding
This work was supported by intramural funds from Asian Healthcare Foundation.
Informed consent
Written informed consent was obtained from all the participating individuals and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
