Abstract
Background
Bile leak after liver transplantation (LT) is commonly treated with endoscopic retrograde cholangiopancreatography (ERCP); however, there are limited data regarding the optimal treatment strategy.
Objective
We aimed to examine the role of ERCP in LT recipients with bile leaks at two large institutions.
Methods
We reviewed all ERCPs performed in LT recipients with bile leak and duct-to-duct biliary anastomosis at two high-volume transplant centers.
Results
Eighty patients were included. Forty-seven (59%) patients underwent ERCP with plastic stent placement (with or without sphincterotomy) and 33 patients (41%) underwent sphincterotomy alone. Complete resolution was obtained in 94% of the stent group vs. 58% of the sphincterotomy group (
Conclusion
ERCP with plastic stent placement is highly successful and more effective than sphincterotomy alone for post-LT bile leak treatment. These results indicate that ERCP and plastic stent placement should be considered the standard of care for the treatment of bile leaks in LT.
Keywords
Introduction
Biliary adverse events constitute an important cause of morbidity and mortality in patients after liver transplantation (LT). Among the different types of biliary adverse events after LT, bile leak is the second most frequent with an incidence of 2% to 25%.1–7 Bile leaks are classified as anastomotic, T-tube-related, cystic duct-related or, in the case of living donor liver transplants (LDLT), cut-surface-related. 6 Common risk factors for the development of bile leaks are related to surgical technique, hepatic artery thrombosis, LT from donors after cardiac death, ABO mismatch, prolonged warm and cold ischemia times, and T-tube use. 8 The standard of care for the treatment of bile leaks after LT is not well established. Patients with a peri-T-tube leak can sometimes be managed conservatively by keeping the T-tube drainage open for a prolonged period of time. 9 Endoscopic retrograde cholangiopancreatography (ERCP) is also very effective and high-resolution rates are near 90%.1–4,6–8 A variety of ERCP techniques have been described for managing leaks, including: nasobiliary drainage,10–13 sphincterotomy alone,14–17 plastic biliary stent placement with or without out sphincterotomy,3,4,6,7,18–24 and placement of fully covered self-expandable metallic stents (FCSEMS).25–27 Regardless of method used, the suggested mechanism of healing relates to the equalization of pressures in the bile duct and duodenum, which allows antegrade bile flow into the duodenum.8,10 Nevertheless, the optimal endoscopic approach has not yet been established and to date there are very few studies that compare the different endoscopic treatment options. In addition, there are no data that define factors predictive of endoscopic success in patients with post-LT bile leaks. The aim of this analysis was to evaluate data from two large institutions to determine the role of ERCP in the treatment of bile leaks following LT.
Materials and methods
This study was approved by the Ethical and Institutional Review Board at the Hospital Clinic of Barcelona (Barcelona, Spain) and Mayo Clinic (Rochester, MN, USA). The protocol, Evaluation of Endoscopic Therapy of Bile Leaks after Liver Transplantation, was approved at both institutions in May and August 2015. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institutions’ human research committee. Informed consent of the procedures was obtained from all patients included in the study. LT recipients with clinical or radiologic suspicion of a bile leak referred for ERCP were included. We reviewed all ERCPs in patients following LT at Mayo Clinic from July 2000 to June 2013 and at Hospital Clinic from January 2003 through January 2015. Mayo Clinic performs between 100 and 120 LTs and approximately 2000 ERCPs yearly. Hospital Clinic performs between 80 and 90 LTs and approximately 450 ERCPs yearly. Data were collected and entered after each case. The electronic medical records and endoscopy database of both institutions were accessed to abstract demographic, clinical, surgical, and endoscopic data. All procedures were performed with sedation, with levels from moderate to general anesthesia. All patients received intravenous broad-spectrum prophylactic antibiotics. Administration of pharmacologic agents and pancreatic stent placement for prevention of post-ERCP pancreatitis were not routinely performed.
ERCP was performed using standard techniques. After cannulation, cholangiography was performed to confirm the leak. If necessary, an occlusion cholangiogram using a balloon catheter was performed to determine the leak site. After confirmation of the bile leak, endoscopic intervention (complete sphincterotomy or stent with or without complete sphincterotomy) was performed at the proceduralist’s discretion. Biliary stents were placed across the site of the bile leak, when technically feasible; in some cases patients were treated with biliary sphincterotomy alone.
Definitions
Bile leaks were diagnosed based on the following criteria: (1) clinical symptoms (i.e. abdominal pain, ascites, fever and/or jaundice); (2) the presence of a new fluid collection consistent with a leak on cross-sectional imaging; (3) increasing or persistent bilious output from an intra-abdominal drain; (4) extravasation of contrast seen on T-tube cholangiography. Intrahepatic leaks were defined as extravasation of contrast due to bile duct rupture and extravasation of bile into the hepatic parenchyma. Patients with a peri-T-tube leak were referred for an ERCP only if the condition persisted after leaving the T-tube to open drainage. We only included patients with a suspected bile leak confirmed by ERCP.
We defined resolution of the bile leak as the cessation of symptoms and drain output, and/or the absence of contrast extravasation on ERCP performed eight weeks after the initial ERCP, with a maximum of two more ERCP treatments. In contrast to postcholecystectomy leaks, where the stent is usually removed in four weeks, in post-LT leaks it is preferred the stent be left in place for approximately eight weeks because of potential problems with delayed healing that may occur because of immunosuppression. 6 If initial resolution was not achieved after the placement of one biliary stent, a posterior ERCP was performed using a larger diameter biliary stent or by placement of an additional biliary stent. FCSEMS were not placed in any patient. Those patients with persistent leaks were referred for surgery or percutaneous transhepatic biliary drainage (PTBD) and were categorized as failures. Definitions of individual adverse events and their severity after ERCP (i.e. pancreatitis, cholangitis, hemorrhage, perforation) were defined by criteria as established by Cotton et al.28,29 Mild events were considered when hospitalization was prolonged by two to three days, moderate by 4–10 days, and severe by more than 10 days.
Statistical analysis
Patient demographics, clinical and procedure data and outcomes were analyzed. χ2 or Fisher exact test were used for categorical variables and the Student
Results
Patient baseline characteristics.
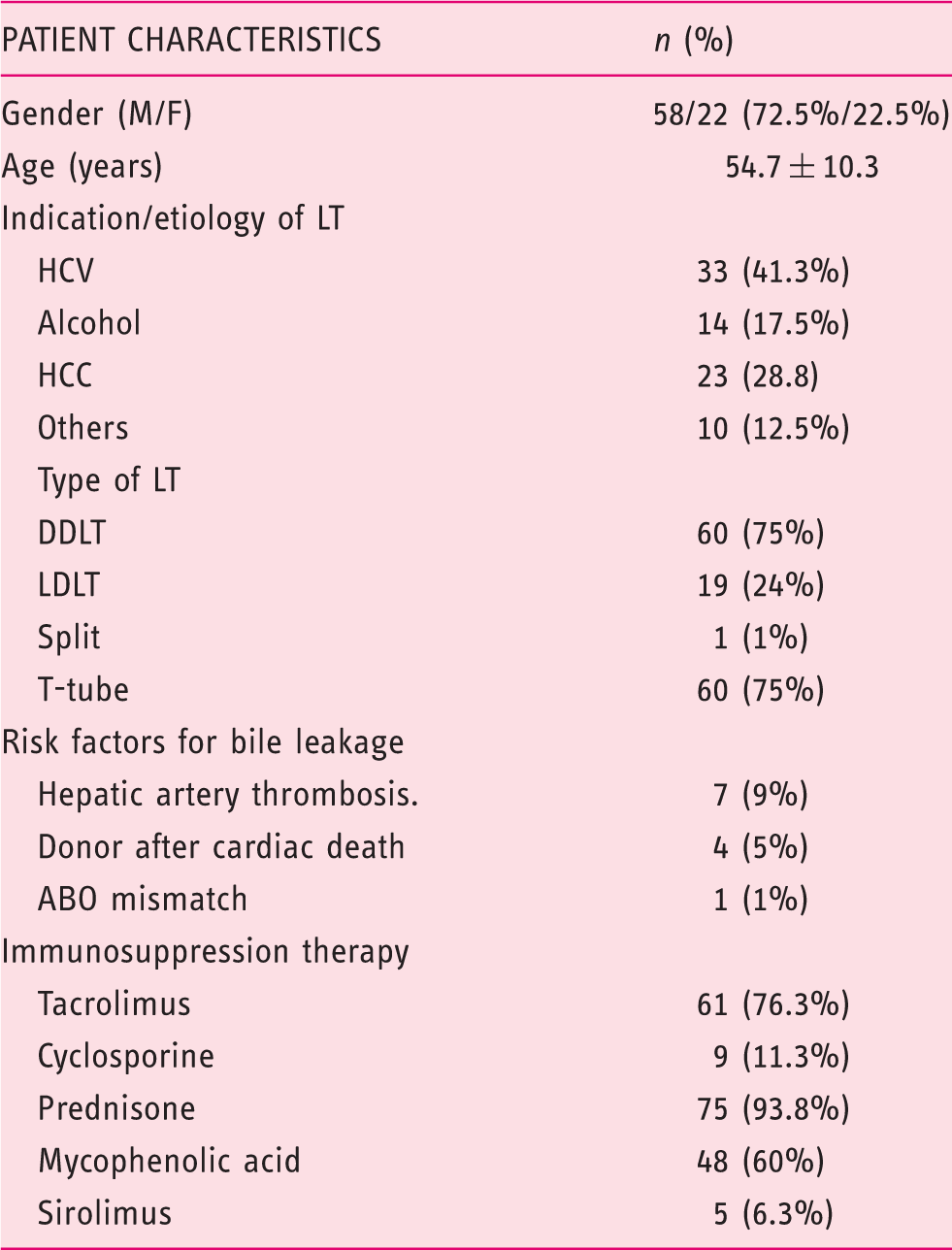
M: male; F: female; LT: liver transplant; HCV: hepatitis C virus; HCC: hepatocellular carcinoma; DDLT: deceased donor liver transplant; LDLT: living donor liver transplant.
Endoscopic findings.
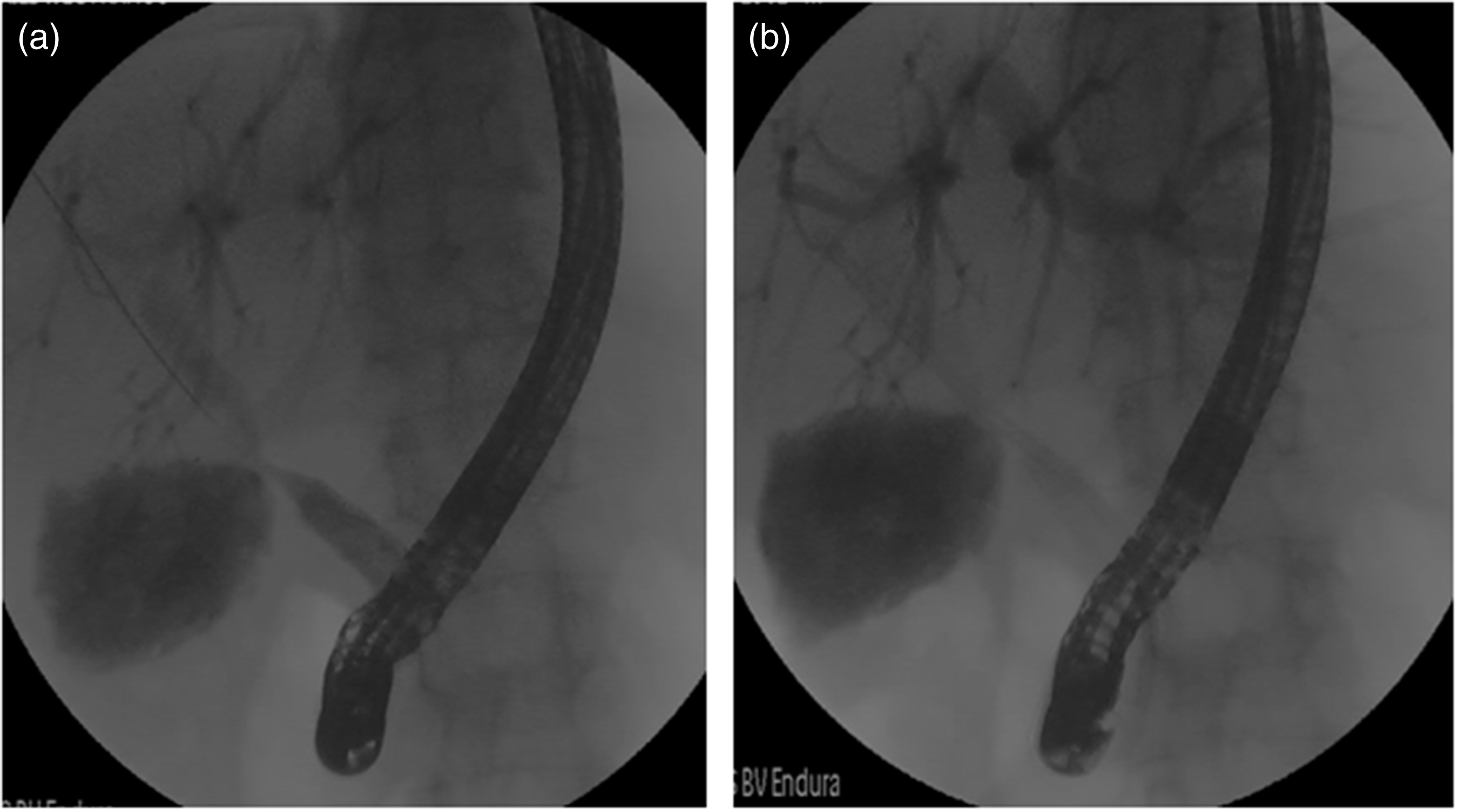
Characteristic findings of a bile leak with an anastomotic stricture. (a) Bile leak and anastomotic stricture. (b) Stent placement across the bile leak site.
Factors associated with bile leak resolution.
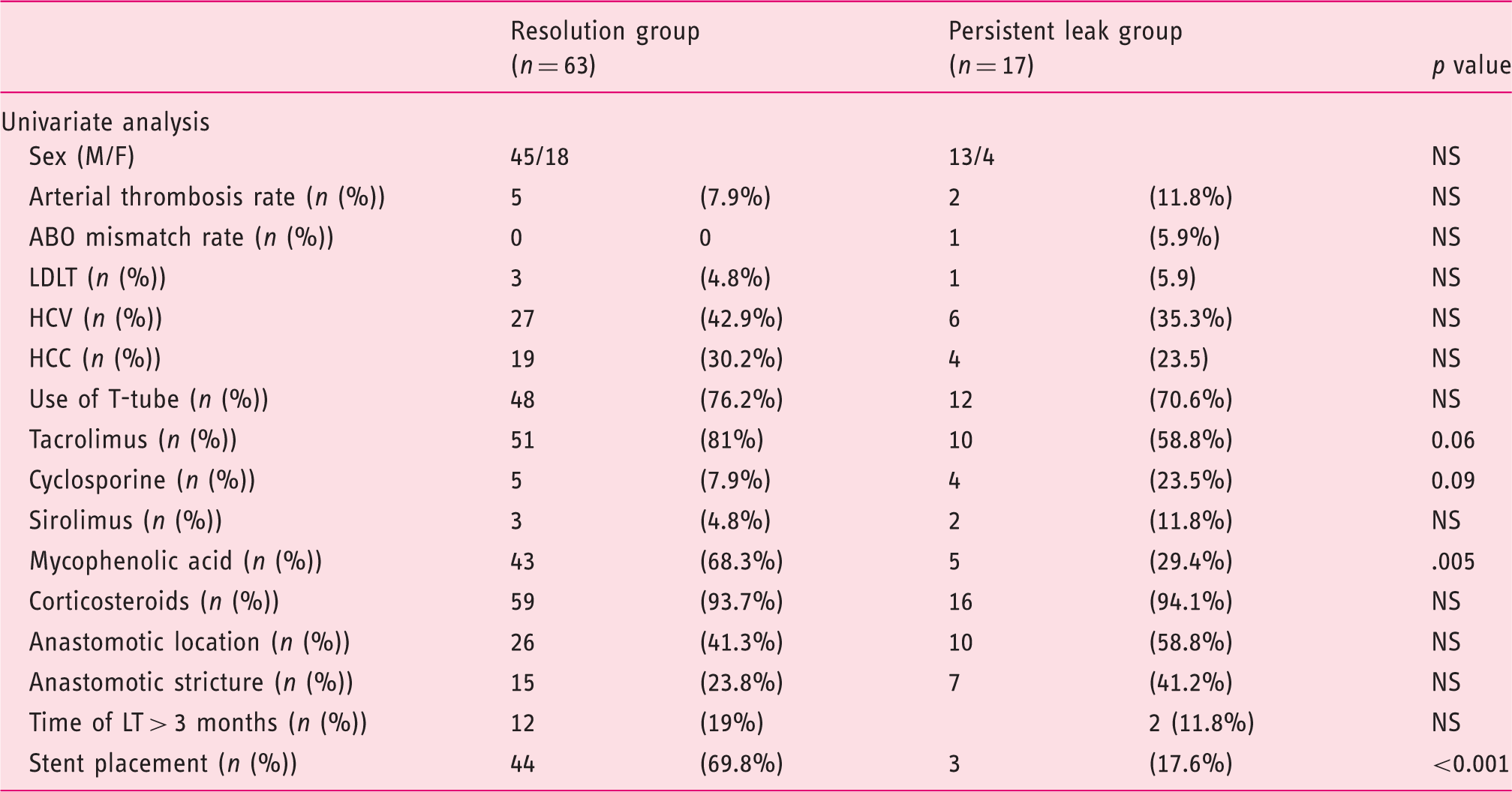
M: male; F: female; LDLT: living donor liver transplant; HCV: hepatitis C virus; HCC: hepatocellular carcinoma; LT: liver transplant.
Multivariate logistic regression model assessing factors associated with resolution of bile leaks after liver transplantation.
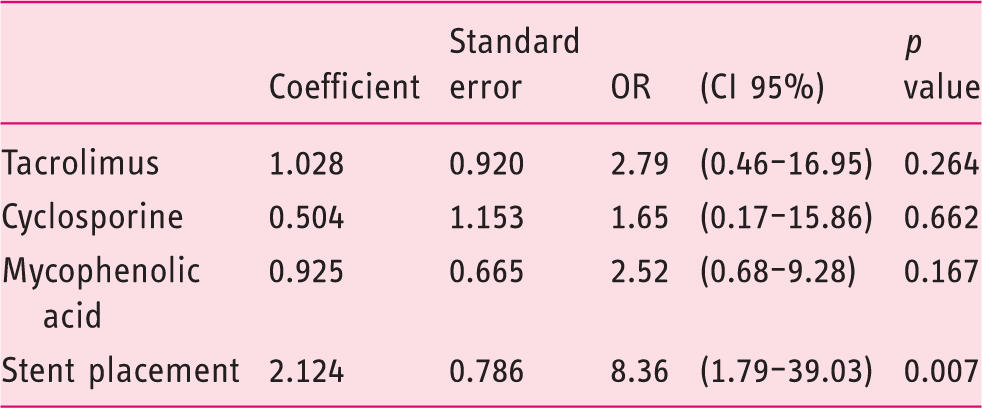
OR: odds ratio; CI: confidence interval.
ERCP and patient-related outcomes.
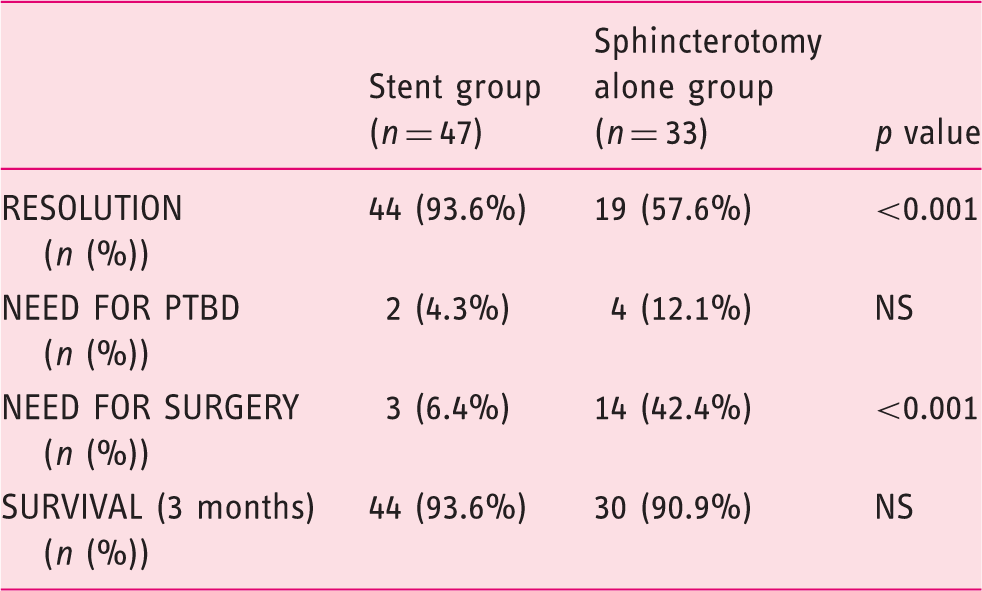
ERCP: endoscopic retrograde cholangiopancreatography; PTBD: percutaneous transhepatic biliary drainage.
Failure of ERCP therapy occurred in 14 (42.4%) patients in the sphincterotomy group and three patients in the stent group (6.4%). In these cases, two patients underwent PTBD, two patients underwent PTBD plus surgery and 11 underwent surgery without prior PTBD. In two patients no further therapy was performed. Eight (10%) patients developed post-ERCP adverse events: mild acute pancreatitis (
Discussion
We sought to analyze ERCP-guided treatment approaches for patients with bile leaks after LT. The results of this analysis from two large academic medical centers indicate that ERCP therapy with placement of a biliary plastic stent (with or without biliary sphincterotomy) is highly successful and significantly more effective than biliary sphincterotomy alone.
Bile leaks are the second most common biliary adverse event following LT and constitute significant morbidity for LT recipients. In addition, bile leaks are considered an independent risk factor for the development of early or late anastomotic biliary strictures and thus require a prompt, safe and highly effective therapy.8,30–32 In this analysis 75% of patients had T-tube placement. T-tubes have been routinely placed as a prophylactic measure for anastomotic stricture development. However, the results of several comparative studies, systematic reviews and meta-analyses suggest no major differences in the incidence of biliary complications, and the current trend has favored more the abandonment of the use of T-tubes after LT in most centers.
33
Most bile leaks can be resolved non-operatively with early intervention. The most widely accepted treatment approach in patients with a duct-to-duct biliary anastomosis is early ERCP-guided endoscopic therapy.1–4,6–8 ERCP-guided endoscopic therapy of bile leaks can be performed with a combination of biliary sphincterotomy and plastic stent placement or with sphincterotomy alone. Some authors propose the use of biliary sphincterotomy alone as it is easy to perform and patients do not require a subsequent ERCP for stent removal; however, most of the available data supporting this practice stem from patients with bile leaks following cholecystectomy.14,16,34,35 In the case of bile leaks after LT, available data regarding the use of biliary sphincterotomy alone for bile leaks are scant and limited to large series of patients treated for an array of biliary adverse events after LT.14–17 The success rate of this approach in LT is poorly understood as no randomized controlled trials have directly compared this strategy to sphincterotomy plus biliary plastic stent placement. We believe plastic stent placement for bile leaks after LT has the advantage of preferentially diverting bile flow to the duodenum through the elimination of the transpapillary pressure gradient, and perhaps could be the reason why stent placement was responsible for better outcomes when compared to sphincterotomy alone. In this analysis, a center effect was clear as most of the cases with sphincterotomy alone were performed at Hospital Clinic. The resolution rate at Hospital Clinic was 69% vs. 91% at Mayo Clinic (
Several studies have reported outcomes of endoscopic therapy for bile leaks after LT, but to our knowledge this is the only study that has analyzed the effectiveness of biliary sphincterotomy alone vs. plastic stent placement with or without sphincterotomy in two high-volume centers. Among the factors associated with the resolution of the bile leak after LT, multivariate analysis identified ERCP with stent placement as the only factor predictive of bile leak resolution (odds ratio, 8.36; 95% confidence interval, 1.79–39.03;
Though controversial, some authors recommend different endoscopic techniques based on the size of the bile leak, suggesting that small leaks may be managed with sphincterotomy alone.14,24 In contrast, our findings suggest that placement of a biliary stent with or without sphincterotomy for the treatment of bile leaks results in a high rate of resolution compared to sphincterotomy alone regardless of the bile leak location. Aside from these two approaches (sphincterotomy alone and sphincterotomy with plastic stent placement), some authors also advocate the use of FCSEMS for patients with bile leaks (mainly post-cholecystectomy leaks) given the high resolution rates (93%–95%) in early studies.26,27,38,39 In our clinical experience FCSEMS are useful in some LT recipients with large or refractory bile leaks, though FCSEMS were not placed during the present study. While FCSEMS may be effective for bile leaks, some stents carry a risk of new stricture formation in LT recipients, thus more information is needed in these patients. 25
A number of studies indicate that the location of the bile leak is a key factor predicting ERCP-directed treatment success. Pfau et al. 4 described a significantly lower success rate of endoscopic treatment of anastomotic leaks (42.9%) compared with T-tube-related leaks (95.2%). In a recent study by Tewani et al. 21 that included a variety of post-surgical bile leaks (i.e. post-cholecystectomy, hepatobiliary surgery, LT), post-cholecystectomy bile leak was identified as a predictive factor for ERCP therapy success compared with leaks after hepatobiliary surgery. Interestingly, in a subgroup analysis 90% of post-LT bile leaks were successfully treated after a single ERCP with stent placement. 21 Finally, one group proposed classifying post-cholecystectomy leaks into low-grade and high-grade. Leaks visualized at the intrahepatic biliary branches were considered low grade, while those seen below the intrahepatics were considered high grade. Low-grade leaks managed with sphincterotomy alone had a high success and high-grade leaks managed with stents and sphincterotomy also had very high success. 24 Our study focuses only on LT recipients and we found that the majority of bile leaks occurred at the anastomotic site (45%) and peri-T-tube site (34%); however, leak site was not a predictor of endoscopic success.
There are inherent limitations in our study. Although this analysis was carried out at two high-volume centers, it is a retrospective analysis including a relatively low number of patients. The study is a descriptive analysis of the ERCP practices at two centers and not a true comparative study of two endoscopic approaches. Also, we were unable to record the bile duct mismatch and size of the bile leaks. Finally, the management strategy for patients in which the first ERCP failed to resolve the bile leak was at the discretion of each proceduralist.
In summary, our study indicates that ERCP with biliary plastic stent placement with or without sphincterotomy is a highly effective therapy for bile leaks after LT and should be considered the standard of care in this clinical situation.
Footnotes
Acknowledgments
Author contributions are as follows: O Sendino: acquired, analyzed and interpreted the results, performed the statistical analysis and drafted and revised the manuscript. A Fernández-Simon: acquired, analyzed and interpreted the results, performed the statistical analysis and drafted and revised the manuscript. R Law: acquired, analyzed and interpreted the results, performed the statistical analysis and drafted and revised the manuscript. B Abu Dayyeh: acquired, analyzed and interpreted the results; performed the statistical analysis and drafted and revised the manuscript. M Leise: acquired, analyzed and interpreted the results; performed the statistical analysis and drafted and revised the manuscript. K Chavez-Rivera: acquired, analyzed and interpreted the results and revised the manuscript. H Cordova: interpreted the results and revised the manuscript. J Colmenero: interpreted the results and revised the manuscript. G Crespo: interpreted the results and revised the manuscript. C Rodriguez de Miguel: acquired, analyzed and interpreted the results. C Fondevila: interpreted the results and revised the manuscript. J Llach: interpreted the results and revised the manuscript. M Navasa: interpreted the results and revised the manuscript. TH Baron: acquired, analyzed and interpreted the results; conceived the study and participated in its design; drafted and revised the manuscript. A Cárdenas: acquired, analyzed and interpreted the results; conceived the study and participated in its design; performed the statistical analysis and drafted and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
None declared.
Funding
Part of the research reported in this article was funded by the Institut d’Investigacions Biomèdiques August Pi-Sunyer (IDIBAPS) and Ciber de Enfermedades Hepáticas y Digestivas (CIBERHED), Barcelona, Spain.
Ethics approval
This study was approved by the Ethical and Institutional Review Board at the Hospital Clinic of Barcelona (Barcelona, Spain) and Mayo Clinic (Rochester, MN, USA).
Informed consent
Informed consent was obtained from all patients included in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
