Abstract
The objective of this study was to evaluate the risk factors and survival outcomes of biliary complications (BCs) after living donor liver transplantation (LDLT) based on our single-center experience. From 2007 to 2010, 112 adult patients were assessed. Forty-nine patients (43.8%) experienced at least one episode of BCs, including biliary stricture and bile leak, occurring in 37.5% and 16.1% of the patients, respectively. Multivariate analysis indicated that hepatic artery thrombosis (relative risk (RR), 5.692; 95% CI, 2.132 to 15.201; p < 0.001), a hepatic duct diameter of less than 3 mm (RR, 2.523; 95% CI, 1.295 to 4.914; p = 0.005), ductoplasty (RR, 2.175; 95% CI, 1.134 to 4.174; p = 0.018), and cytomegalovirus infection (RR, 4.452; 95% CI, 1.868 to 10.613; p = 0.001) were independent risk factors for the development of BCs. However, these factors and BCs showed no prominent impact on the overall survival (OS) and graft survival (GS). In addition, the patients who developed vascular complications demonstrated poor outcomes in terms of OS (five-year, 56.3% vs. 78.1%; p = 0.017), GS (five-year, 56.3% vs. 77.1%; p = 0.023), and BC-free survival (five-year, 25.0% vs. 63.5%; p = 0.007) compared with patients without vascular complications. In conclusion, BCs remain a common problem after LDLT, especially for patients using duct-to-duct anastomosis. Hepatic artery thrombosis, a short duct diameter, ductoplasty, and cytomegalovirus infection lead to an increased incidence of BCs. The occurrence of BCs manifested no significant influence on the long-term survival outcomes. However, our findings await verification through large-scale randomized studies regarding the risk factors for the development of BCs and their impact on the prognosis.
Keywords
Introduction
Liver transplantation has been established for decades; however, despite advanced surgical techniques, its application is limited owing to a shortage of available organs. 1 The deficiency of donor livers has led to the development of living donor liver transplantation (LDLT), which exploits the regenerative abilities of the liver.2,3 However, because of several shortcomings, such as a loss of partial liver function from the hepatic resection in donors and a high incidence of biliary complications (BCs), further evaluation of the outcomes of LDLT is still necessary.4,5
It is widely known that biliary stricture (BS) and bile leak (BL) are the most frequent types of BCs. A recent study from the United States indicated that BC was the most common type of complication after LDLT with a rate of 53.7%, of which anastomotic stricture accounted for 41.5%. 6 Another study from Russia found that 40/147 patients (27.2%) developed BCs, including 29 cases of BL (72.5%) and 11 (27.5%) cases of BS. 7 Likewise, the reported morbidity rate of BC is quite diverse from different centers. The current consideration on the incidence of BCs is primarily associated with the blood supply to the hepatic duct, length of the duct, tension at the anastomosis, reconstruction methods, and technical issues.8,9 However, despite the abundant advances in various aspects, the incidence of BCs has not been remarkably reduced.
Herein, we conducted the present study to establish integrated risk factors for the development of BCs. In addition, we reviewed all of the clinical characteristics and postoperative complications and analyzed their impact on the cumulative overall survival (OS), graft survival (GS), and BC-free survival (BCFS) after adult-to-adult right-lobe LDLT.
Materials and methods
Study patients
From January 2007 to December 2010, 116 adult patients underwent LDLT at the Department of Liver Surgery, Ren Ji Hospital (Shanghai, China). Four patients were excluded from the analysis because of possible extrahepatic metastasis before the operation. Finally, 112 patients with various hepatic diseases were formally enrolled in the present study, including 52 patients with malignant hepatic tumors and 60 patients with benign end-stage liver diseases. Of the malignant hepatic tumors, hepatocellular carcinoma accounted for 49 cases, and cholangiocarcinoma accounted for three cases. Of the benign end-stage liver diseases, hepatitis B virus (HBV)-associated liver cirrhosis accounted for 34 cases, fulminant liver failure accounted for 21 cases, primary biliary cirrhosis accounted for four cases, alcoholic liver cirrhosis accounted for two cases, and Wilson’s disease accounted for one case. The patients were diagnosed by comprehensively observing the following factors: clinical symptoms, imaging studies, and postoperative pathology. The patients were divided into two groups according to the transplant stage to assess our learning curve. The first half of patients who underwent LDLT from January 2007 to October 2007 was assigned to the early cases, and the latter half of patients who underwent LDLT from November 2007 to December 2010 was enrolled into the later cases.
Preoperative assessment
The following steps were carried out before the surgical operation. (1) All of the data, including the age, height, weight, past medical history, and relationship between the donor and recipient were collected, and the donor underwent a preliminary ethical and psychological evaluation by specialists. (2) Further laboratory studies and routine imaging studies were completed if needed. (3) The anatomy of the vessels of the liver and the biliary tract was confirmed, and the dimensions of the liver were calculated by liver spiral computed tomography angiography (SCTA) and magnetic resonance cholangiopancreatography (MRCP). (4) Discussions of the examination findings of the donors and recipients were carried out within the department. For patients with special conditions, the relevant departments were consulted. If the discussions were successful, the examination findings, the results of the discussion, a confirmation of the relationship, and other documents were submitted to the ethics committee of the hospital for a decision.
Surgical procedures
For the donors, intraoperative real-time cholangiography was accomplished to confirm the biliary anatomy. The graft resection was performed using cut-ultrasound aspiration. The middle hepatic vein was included when the graft was inadequate for the recipient to provide sufficient functional liver, for which nine cases were carried out. After isolation from the abdominal cavity, the graft was perfused with 4℃ Celsior solution. The recipient operation was started when the hepatic hilar structure of the donor was exposed. The reconstruction of the branches (segment V and VIII) was performed with 6-0 Prolene when the tributary had a diameter greater than 5 mm, and the other short veins were occluded with the use of clips or were ligatured. Duct-to-duct anastomosis was the only type of biliary reconstruction except for one patient with sclerosing cholangitis who received hepaticojejunostomy. Doppler ultrasonography was carried out during the operation to test the blood flow. Right-lobe LDLT was the only type of surgery performed.
Data collection and follow-up
The prospectively collected LDLT database was used in the description of the preoperative, intraoperative, and postoperative data. A comprehensive evaluation of the clinical manifestations, serological examinations, and imaging studies was carried out for the confirmation of BCs. For the patients with clinical manifestations, the imaging studies were immediately followed for a definite confirmation, and the patients with asymptomatic or non-specific symptoms with biochemical abnormalities, including serum alkaline phosphatase, bilirubin, and gamma-glutamyl transferase, were placed into a high index of suspicion, and imaging studies were carried out immediately to make a definite diagnosis. Routine surveillance of fluid drainage was performed to detect BL, and the samples were collected for a definite confirmation when abnormal accumulation was detected. Cytomegalovirus (CMV) infection was defined as active CMV infection (disease), including de novo infection and reactivation of latent CMV. Routine serologic antibody testing (immunoglobulin (Ig)M and IgG) was the first step to estimate CMV infection. The definite diagnosis was made after the confirmation of CMV DNA and/or pp65 antigenemia and after treatment with intravenous ganciclovir injection; both clinical and subclinical infections were involved. Within one week after the operation, routine ultrasonography was performed daily. Routine blood parameters, liver function, and the blood concentration of immunosuppressants were monitored within two weeks after the operation. A liver-enhanced CT scan was performed within one month after discharge. Serological examinations were performed once a week within one month after discharge, once every two weeks during the second month, and once a month after the second month. Serological examinations, chest X-ray, abdominal ultrasound, and upper abdomen contrast-enhanced CT were performed in the clinic at each visit once every six months from the second year and annually from the third year. The patients who did not visit the clinic were followed up through telephone inquiry. The endpoints of this study were OS, GS, and BCFS. OS was calculated according to the time of LDLT to the death or loss of contact. BCFS was defined as the period between LDLT to the date of a high index of suspicion in patients who eventually developed BCs. The follow-up investigation was performed in a range of 1 to 94 months, and 60 months was the minimum follow-up period for the survivals.
Statistical analysis
SPSS for Windows version 19.0 software (SPSS Inc, Chicago, IL, USA) was used for statistical analysis. The number of cases and percentages were presented as categorical variables. The median (minimum–maximum) was used in the description of central tendency and dispersion. The survival curves were produced using the Kaplan-Meier method. Univariate analysis was used in identifying prognostic factors, and the statistically significant variables were used in the multivariate analysis to confirm independent factors. The risk factors were evaluated by relative risk (RR) and the 95% confidence interval (CI). A p value <0.05 represented statistical significance.
Results
Patient characteristics
Baseline characteristics of the recipients.
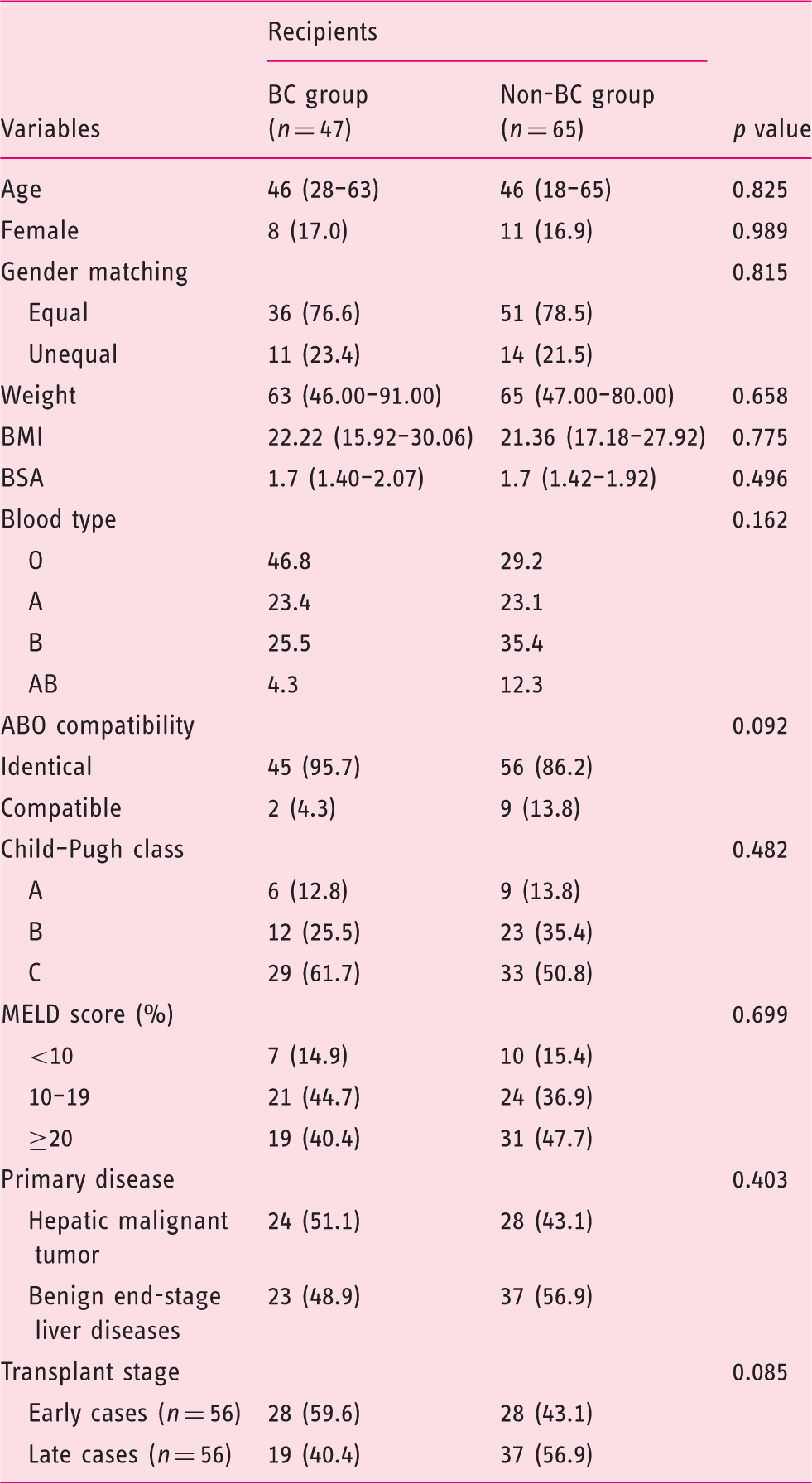
BC: biliary complications; BMI: body mass index; BSA: body surface area; MELD: Model for End-Stage Liver Disease.
Baseline characteristics of the donors.
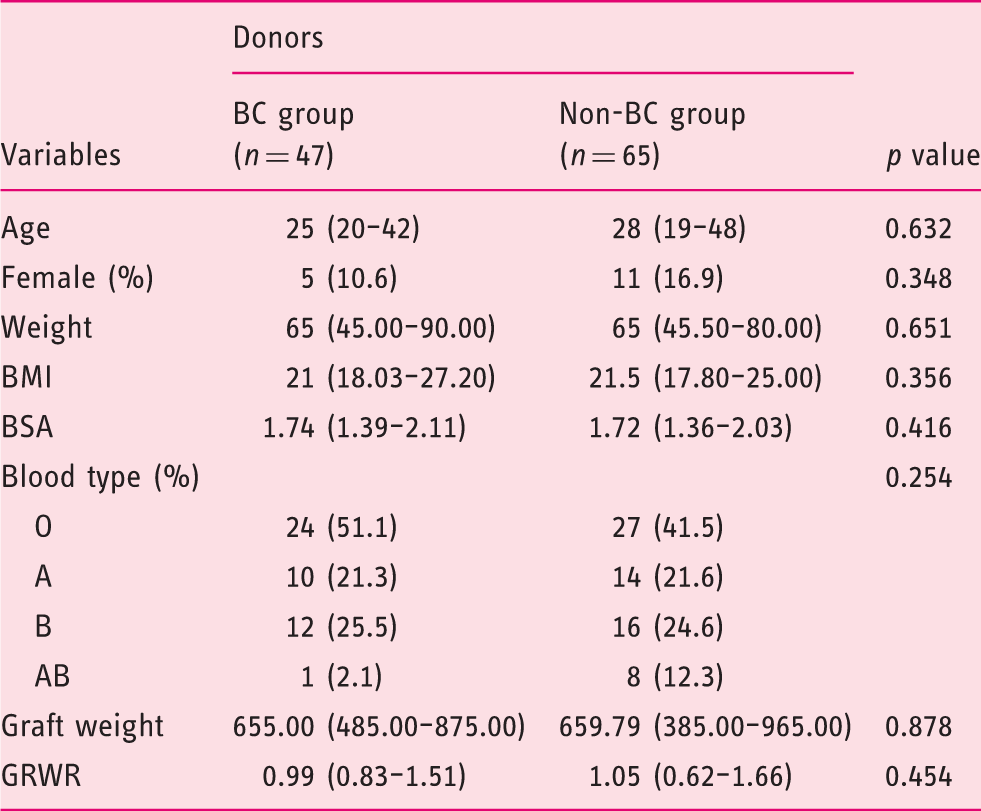
BC: biliary complication; BMI: body mass index; BSA: body surface area; GRWR: graft-to-recipient weight ratio.
Incidence of postoperative complications and their impact on OS and GS
Incidence of complications.
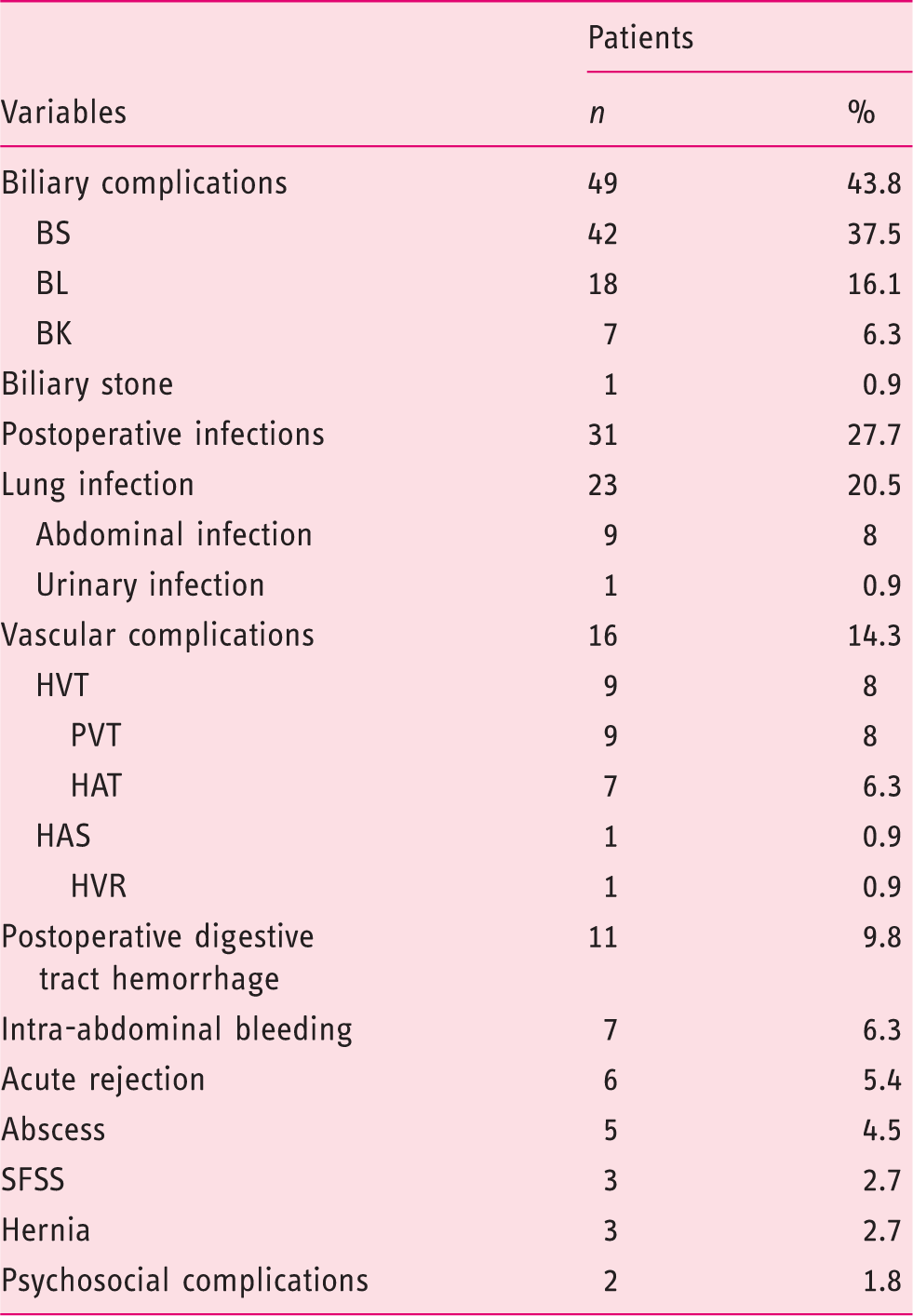
BS: biliary stricture; BL: bile leak; BK: bile duct kinking; CMV: cytomegalovirus; HVT: hepatic vein thrombosis; PVT: portal vein thrombosis; HAT: hepatic artery thrombosis; HAS: hepatic artery stricture; HVR: hepatic vein rupture; SFSS: small-for-size syndrome.
Univariate analysis of all the postoperative complications was performed to identify the influential factors. Six variables were found to significantly affect the survival outcomes, and those were used in the multivariate analysis. The results revealed that small-for-size syndrome (SFSS) (RR, 14.040; 95% CI, 3.090 to 63.788; p = 0.001), abdomen infection (RR, 3.112; 95% CI, 1.068 to 9.070; p = 0.038), digestive tract hemorrhage (RR, 3.772; 95% CI, 1.463 to 9.723; p = 0.006) and abscess (RR, 3.930; 95% CI, 1.059 to 14.580; p = 0.041) were the independent risk factors for the cumulative OS.
In addition, the patients were stratified according to the presence of VCs. Our data revealed that the presence of VCs negatively affected both the OS and GS (Figure 1(a)). The one-year and five-year OS rates of the patients with and without VCs were 68.8% vs. 86.5% and 56.3% vs. 78.1%, respectively. A relatively high RR of 3.887 was found with a 95% CI of 1.281 to 11.800 (p = 0.017). An analog was detected in the GS (RR, 3.550; 95% CI, 1.194 to 10.560; p = 0.023) (Figure 1(b)). When further stratified into the hepatic artery complications, portal vein complications, and hepatic vein complications, none of these subgroups revealed significance separately.
Impact of vascular complications on the overall, graft, and biliary complications-free survival. VC: vascular complications; CI: confidence interval.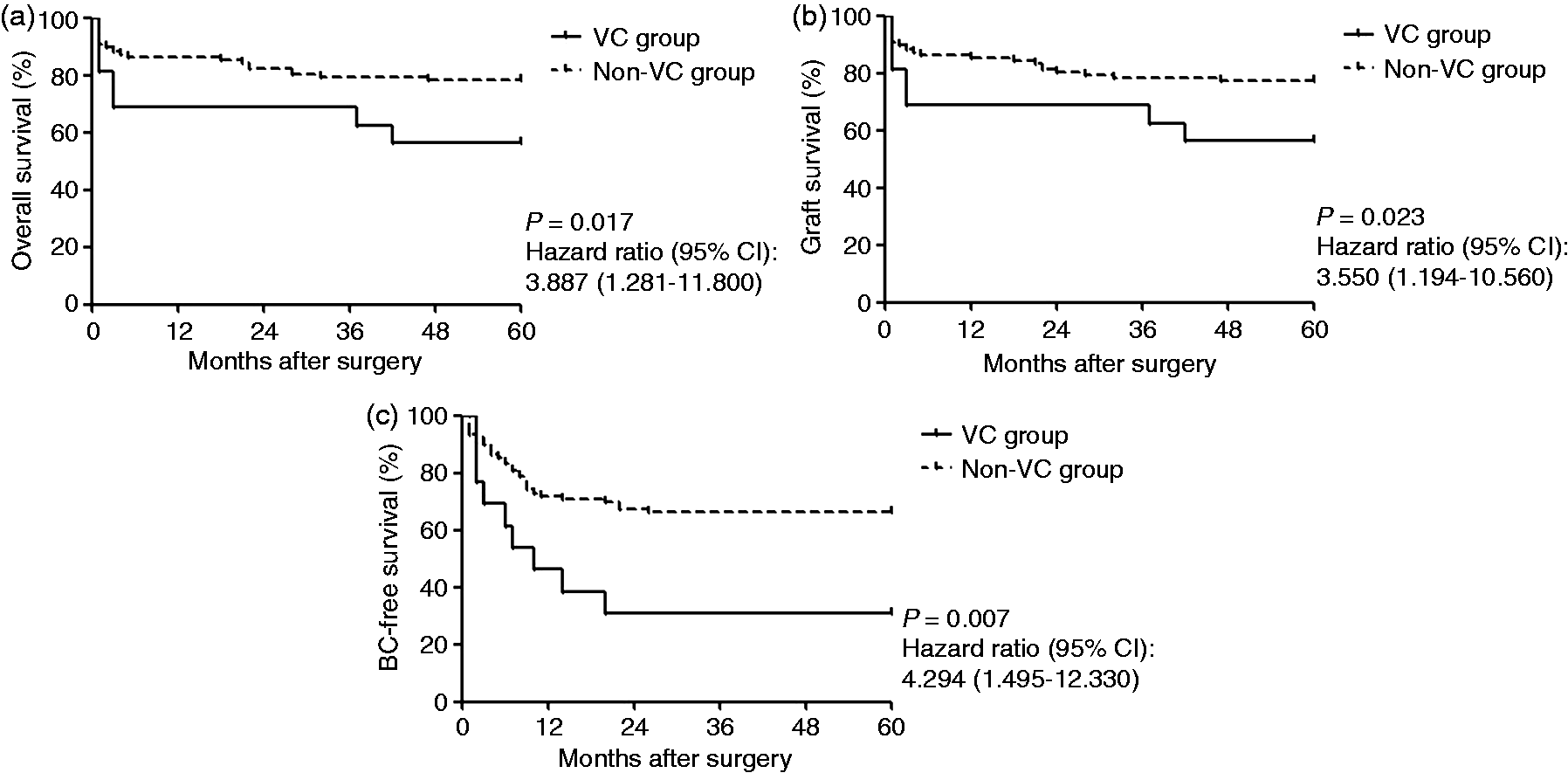
Risk factors for the development of BCs
Univariate and multivariate analyses of complications that significantly affected overall survival.
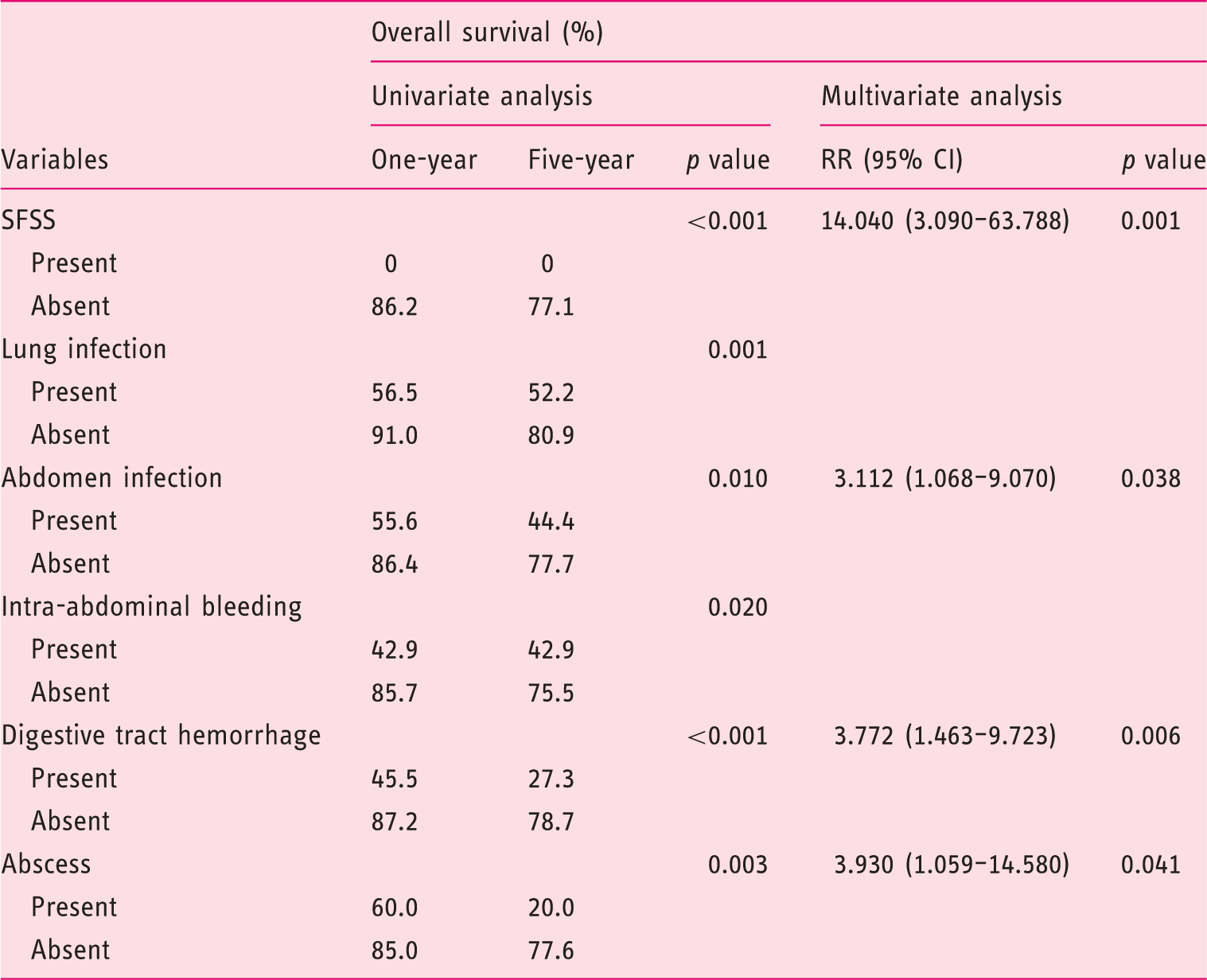
OS: overall survival; GS: graft survival; SFSS: small-for-size syndrome; RR: relative risk; CI: confidence interval.
Independent risk factors for the incidence of biliary complications.
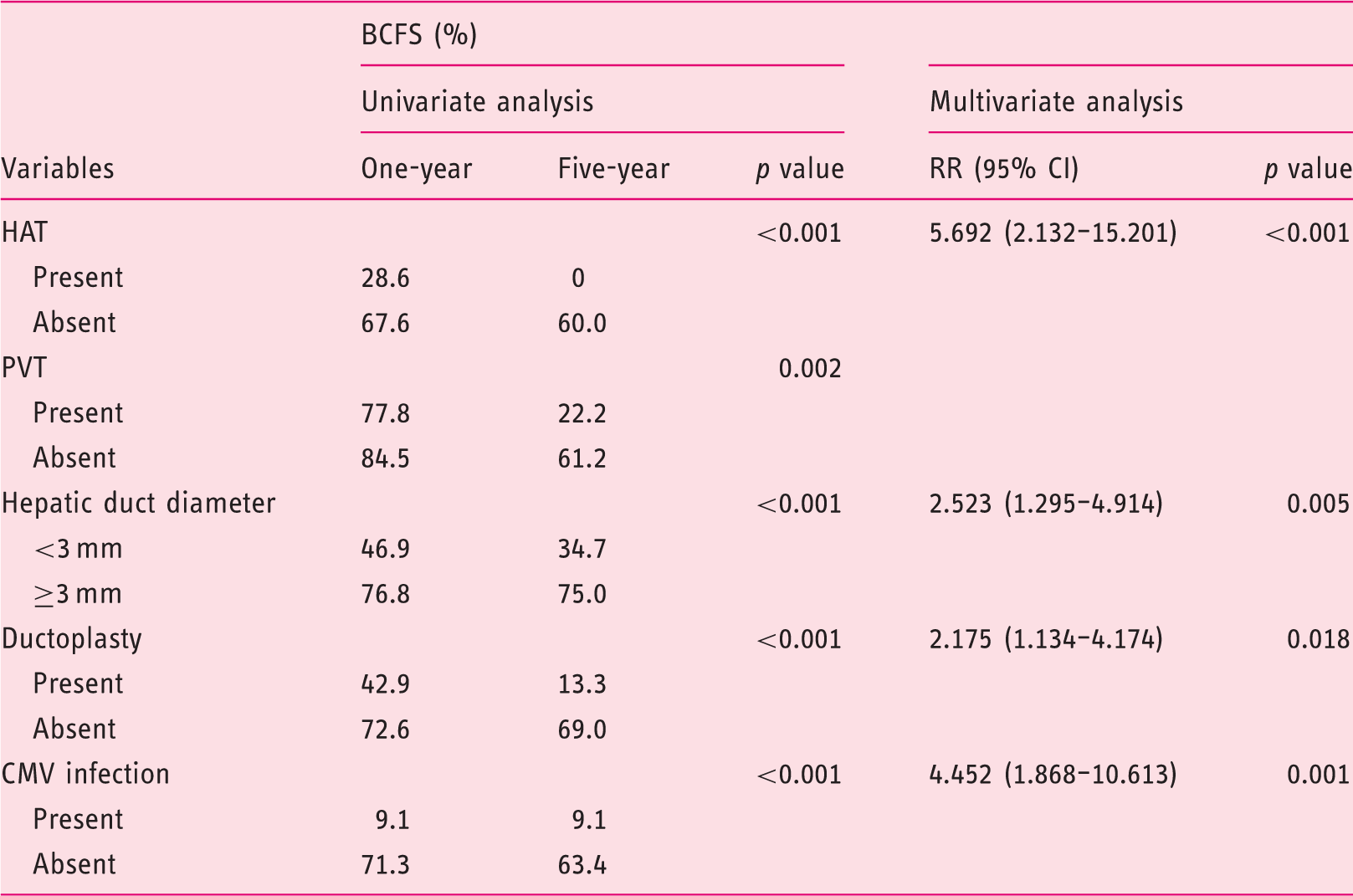
HAT: hepatic artery thrombosis; PVT: portal vein thrombosis; CMV: cytomegalovirus; RR: relative risk; CI: confidence interval.
Impact of BCs on the OS and GS
There was no significant difference between the patients with and without BCs; the one-year and five-year OS of the patients with and without BCs were 91.8% vs. 77.8% and 79.6% vs. 71.4%, respectively (Figure 2(a)). The GS also revealed no difference between the BC and non-BC groups (Figure 1b, p = 0.412). When the BC group was stratified into three subgroups (BL, BS, and BK) to confirm whether there is a difference between survival outcomes of the three major BCs, the five-year OS of the BL, BS, and BK groups were 72.2%, 81.0%, and 85.7%, respectively, demonstrating no significant differences (Figure 2(c), p = 0.616). The log-rank test for trend analysis also showed no significance. The GS outcomes manifested similar results to the OS with five-year GS of 66.7%, 81.0%, and 85.7%, respectively (Figure 2(d), p = 0.374).
Impact of biliary complications on the overall and graft survival of the patients. BC: biliary complications; CI: confidence interval.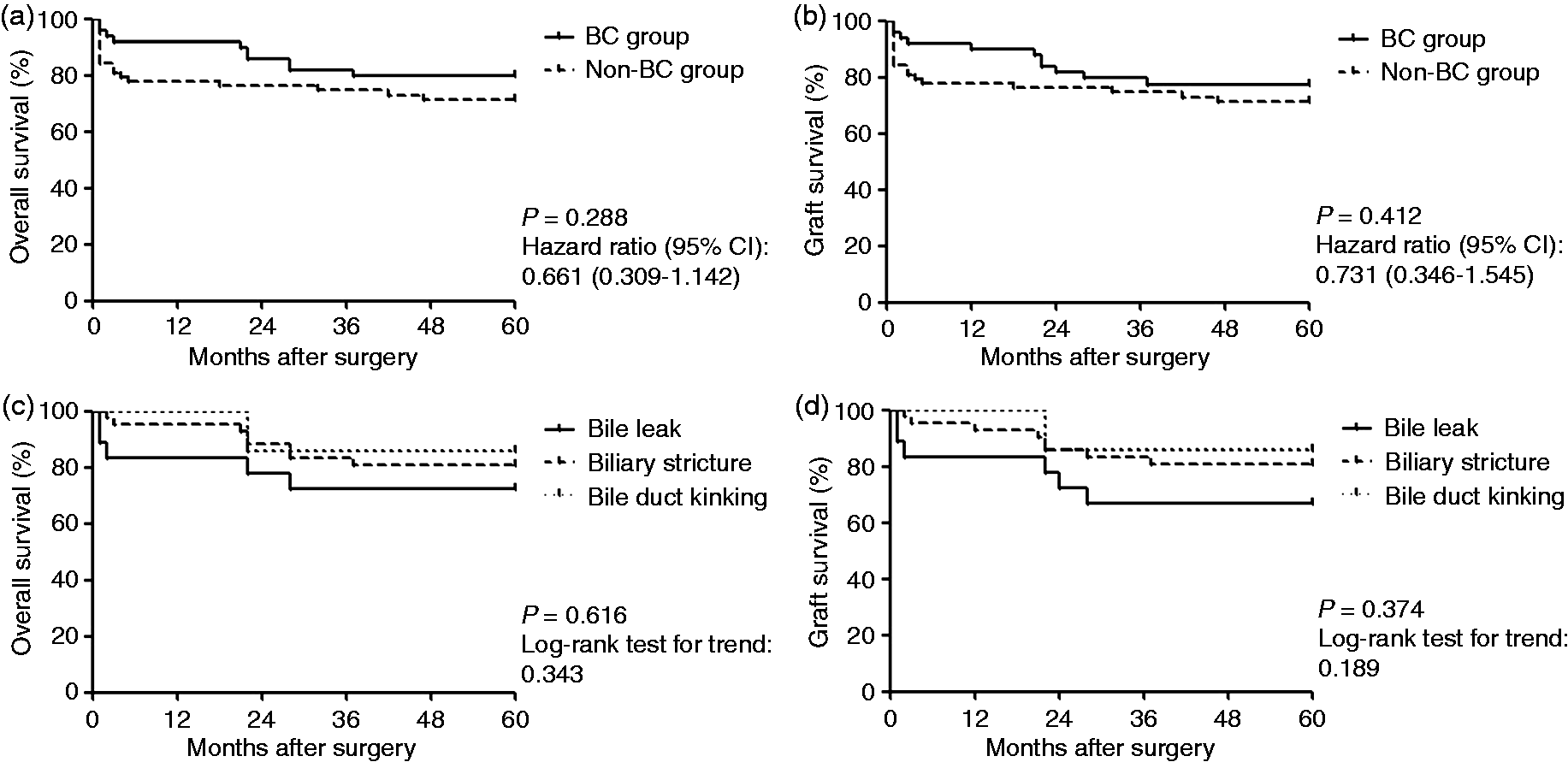
Discussion
To date, tremendous efforts have been made to determine the risk factors for the development of BCs, such as the biliary anatomy of donors and surgical techniques.12,13 Our report adds another series of risk factors for BCs and confirms that the occurrence of BCs is related to a moderate order of severity with endoscopic treatment. In the present study, the incidence of BCs was affected by the presence of HAT, hepatic duct diameter, ductoplasty, and CMV infection. In terms of distinct complications, the patients were separately evaluated according to the incidence of BCs and VCs. The BC group achieved comparatively acceptable prognosis, despite the poor outcomes in the VC group that were also correlated with the decrease in BCFS.
Our results intensively support that surgical techniques are involved in the incidence of BCs in terms of experience and anastomotic methods. Similar to other surgical operations, a learning curve effect is obvious in LDLT.14,15 A resent publication by Kim et al. 16 demonstrated that LDLT performed by young surgeons was found to be a risk factor for the development of BL, suggesting that a senior surgeon has to monitor biliary reconstruction until the young surgeon successfully conducts at least 20 of the procedures. As described in our previous study, the incidence of BCs decreased as our center accumulated surgical experience. 17 Even the experienced surgeon who accomplished more than 20 cases showed a slight difference as the surgeon obtained more experience. Thus, we suggest that at least one specialized biliary reconstruction surgeon is necessary for each transplant center. Furthermore, another important surgical technique to decrease the incidence of BCs is maintaining the blood supply to the bile ducts in donor surgery, a procedure that already has been proven to be a key step in terms of preventing the development of BCs. 18 On the other hand, a retrospective study of 74 adult patients who underwent LDLT from South Korea indicated that hepaticojejunostomy was correlated with a lower incidence of BCs (11.1%) than biliary stent-associated duct-to-duct anastomosis (48.5%) and duct-to-duct anastomosis alone (33.3%), and they concluded that hepaticojejunostomy is associated with improved long-term survival outcomes. 19 By contrast, a study from Asan hospital reported that, although duct-to-duct anastomosis is associated with more frequent BCs after LDLT, it was significantly better in achieving favorable overall outcomes. 20 Beyond doubt, duct-to-duct anastomosis has several advantages, including a shorter duration of operation and ease of the endoscopic assessment in the case of complication.21,22 Nonetheless, a previous meta-analysis of 16 trials from our center demonstrated no definite evidence of a preponderance of either of the two major types of biliary reconstruction. 23 In the present study, all of the patients underwent duct-to-duct anastomosis, except for one patient with sclerosing cholangitis who received hepaticojejunostomy, indicating that duct-to-duct anastomosis might produce a relatively high incidence of BCs (43.8%).
VCs, including thrombosis and stenosis of the hepatic artery, hepatic vein, and portal vein, are one of the major concerns after LDLT in terms of severity and frequency. 24 The incidence of VCs is quite diverse across different centers; it ranges from 5.6% to 21.6%.25–27 Gad et al. 27 indicated that the presence of VCs severely influenced the OS, of which the patients with VCs merely achieved a 36.1% of survival (13/36), whereas the patients without VCs obtained a significantly better OS rate (79/131, 60.3%; p = 0.010). Another study specializing in PVT reported that PVT is a comparative predictor of unfavorable prognosis after LDLT, for which the five-year survival rates were 60.6% for patients with severe PVT, 69.3% for patients with mild PVT, and 80.4% for patients without PVT. 28 Although they performed eversion thrombectomy with a modified technique, which they insisted was feasible in most cases of PVT, their survival outcomes showed a slight difference (p = 0.059). In the present study, when divided into the composite VC group, the presence of VCs significantly affected the OS, GS, and BCFS, indicating that the development of VCs is a major risk factor in the overall prognosis after LDLT. However, no such independent prognostic factor was identified when analyzed separately; only HAT demonstrated an independent association with the development of BCs, leading us to suggest that all these factors (PVT, HVT, HVR, and HAS) might independently affect the survival outcomes in large-scaled studies.
Infections are a common cause of mortality in LDLT, of which the incidence ranges from 13.3% to 80%, and 39 of our patients (34.8%) were involved. 29 The most frequent type of infection was lung infection with an incidence rate of 20.5%, followed by CMV infection (9.8%) and abdomen infection (8.0%). Among them, only abdomen infection significantly affected the OS and GS, and CMV infection influenced the incidence of BCs. In addition, abdomen infection was further identified to be independently associated with the incidence of BCs when subgroup analysis of patients without HAT was performed. Another important issue is infection by CMV, which is a genus of viruses in the family Herpesviridae that conceals itself within the body over a long period of time and promotes the constitutive expression of genes under its control, thereby causing ulceration and mucosal hemorrhage in the gastrointestinal tract. 30 CMV infection has been considered as a possible cause or trigger for the development of BCs after transplantation.31–33 A recent prospective single-center study of 210 biliary atresia infants by Zani et al. 34 demonstrated that CMV infection promotes biliary atresia, and the patients with CMV infection revealed infant native liver survival (p < 0.001) and increased OS (p = 0.002), further indicating that this group of patients showed a reduced response to Kasai portoenterostomy. Wadhawan et al. 35 indicated that CMV reactivation and disease were strongly correlated with acute cellular rejection after LDLT, but CMV reactivation and disease showed no impact on OS. In short, CMV infection might be an etiological factor for the development of BCs without affecting OS.
This report has several underlying limitations. First, the limited number of patients enrolled in our study who underwent LDLT at our center may be a weakness of the present retrospective investigation. Second, the present study mainly focused on the incidence of BCs; the detailed treatments were not systematically described. Third, most of the patients in our study were associated with HBV, while primary sclerosing cholangitis and hepatitis C virus infection are the most common etiological factors for patients with end-stage liver diseases in Japan and Western countries. Therefore, our results call for confirmation in Japanese or Western populations.
In conclusion, HAT, a hepatic duct diameter<3 mm, ductoplasty, and CMV infection are the independent risk factors for the development of BCs. Unlike SFSS, abdomen infection, digestive tract hemorrhage, and abscess, the presence of BCs showed no impact on the OS and GS. In addition, VCs revealed a significant association with poor OS, GS, and BCFS, but they showed no significant impact when analyzed separately. Multi-institutional large-scale studies including two major biliary reconstruction methods are required to more comprehensively conclude the risk factors for the development and prognostic impact of BCs.
Footnotes
Acknowledgments
This report was a retrospective study that was designed and overseen by a guiding committee that included academic investigators and employees of the Department of Liver Surgery, Ren Ji Hospital. This report was conducted in accordance with the principles of Ren Ji Hospital guidelines and the Declaration of Helsinki and was approved by Shanghai Jiaotong University School of Medicine, Renji Hospital Ethics Committee. All of the authors participated in the study design and had access to the data, which were analyzed by one author (Seogsong Jeong). The manuscript was written by the first author (Seogsong Jeong) and was drafted and revised by all of the authors. Jianjun Zhang, Lei Xia, Ying Tong, Yi Luo, Xin Wang, and Qiang Xia were the specialized surgeons who performed the surgical operations on the patients involved in this study, and they edited the surgical procedures in the manuscript.
Declaration of conflicting interests
None declared.
Funding
This work was supported by the Key Joint Research Program of Shanghai Health Bureau (grant number 2013ZYJB0001) and Technological Innovation and Action Project of the Shanghai Science and Technology Commission (grant number 15411950401).
