Abstract
Background
Inhibition of the renin–angiotensin system (RAS) was associated with longer survival in patients with different solid malignancies.
Objective
The objective of this study was to investigate the effect of RAS inhibitor (RASi) treatment (angiotensin-converting enzyme inhibitors or angiotensin-II-receptor blockers) on survival of patients with hepatocellular carcinoma (HCC).
Methods
Patients diagnosed with HCC and Child-Pugh A between 1992 and 2013 who received sorafenib, experimental therapy, or best supportive care were eligible for the Vienna cohort. The Mainz cohort included patients with HCC and Child-Pugh A who received sorafenib treatment between 2007 and 2016. The association between RASi and overall survival (OS) was evaluated in univariate and multivariate analyses.
Results
In the Vienna cohort, 43 of 156 patients received RASi for hypertension. RASi treatment was associated with longer OS (11.9 vs. 6.8 months (mo); p = 0.014) and remained a significant prognostic factor upon multivariate analysis (HR = 0.6; 95% CI 0.4–0.9; p = 0.011). In subgroup analysis, patients treated with sorafenib plus RASi had better median OS (19.5 mo) compared to those treated with either sorafenib (10.9 mo) or RASi (9.7 mo) alone (p = 0.043). The beneficial effect of RASi on survival was confirmed in the Mainz cohort (n = 76).
Conclusion
RAS inhibition is associated with longer survival in HCC patients with Child-Pugh class A.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the second most common cause of cancer-related mortality globally. 1 HCC usually develops in patients with underlying liver cirrhosis, predominantly caused by chronic viral hepatitis or alcohol consumption,2–4 and nonalcoholic steatohepatitis (NASH) as rapidly growing cause in Western countries. 5 Treatment of HCC depends on tumor stage, liver function, and general condition of the patient. 3 Potential curative treatment options are reserved for patients with small tumors and well-preserved liver function. 3 As patients who develop HCC are usually asymptomatic, about 50% of all patients are diagnosed at advanced tumor stages, where only systemic medical treatment is available. 3 The oral multikinase inhibitor sorafenib, a pan-vascular endothelial growth factor (VEGF) receptor tyrosine kinase inhibitor (TKI), remains the only efficacious treatment in first-line, and provides a modest prolongation of median overall survival (OS) (2.8 months). 6 A closely related TKI drug (regorafenib) recently showed increased OS in second-line (after sorafenib failure). 7 None of the other agents tested in the first-line or second-line setting in advanced HCC showed a survival benefit versus sorafenib or placebo. 8 Thus, there is still an urgent need for further improvements in the treatment of this deadly disease.
Angiotensin-converting enzyme inhibitors (ACEi) and angiotensin receptor blockers (ARB) are widely prescribed antihypertensive drugs, and may also have beneficial effects in cancer patients. ACEi/ARB use was associated with reduced risk of breast cancer recurrence, 9 increased tumor response in rectal cancer, 10 and longer OS in patients with pancreatic, renal cell (RCC), lung, and brain cancer.11–16 However, it was recently reported that the use of renin–angiotensin system inhibitors (RASi) at baseline was not a significant independent prognostic factor for improved survival in a pooled analysis of metastatic RCC patients treated with pazopanib or sunitinib. 17
There is some evidence that these drugs could have beneficial effects in patients with HCC.18–21
Here, we assessed the association between RASi and survival in a cohort of 156 patients with HCC and Child-Pugh class A treated with sorafenib or experimental/supportive therapy, and confirmed the results in an independent cohort of 76 Child-Pugh A patients treated with sorafenib.
Materials and methods
Patients
Vienna cohort
Patients ≥ 18 years diagnosed with HCC at the Division of Gastroenterology & Hepatology, Medical University of Vienna between December 1992 and January 2013 who received sorafenib or – especially before the sorafenib era – experimental therapy (not standard according to current guidelines, 3 e.g. rapamycin, octreotide, thalidomide, etc.) or best supportive care (BSC) as first-line therapy were eligible for this study. Patients treated with surgery or local ablative treatment (i.e. percutaneous ethanol injection, radiofrequency ablation, transarterial chemoembolization (TACE)) were excluded from this analysis. Treatment decision was based on an interdisciplinary approach involving hepatologists, interventional radiologists, and surgeons. According to current recommendations for HCC trials, 22 and due to the potential severe side effects (hypotension, renal insufficiency/failure) of RAS inhibitors in advanced cirrhosis, 23 representing a potential source of bias, only patients with Child-Pugh class A were included into this study. We then identified HCC patients that received ACEi or ARB for treatment of arterial hypertension and investigated the effect of RASi on survival. The retrospective analysis of these data was approved by the Ethics Committee of the Medical University of Vienna.
Mainz cohort
Patients ≥ 18 years with HCC who received sorafenib treatment at the University Medical Center, Johannes Gutenberg University Mainz between January 2007 and April 2016 were eligible. These patients were screened for ACEi and ARB therapy for arterial hypertension. Similar to the Vienna cohort, only patients with Child-Pugh class A were included and treatment decision was based on an interdisciplinary consensus. The retrospective analysis of the data was approved by the local Ethics Committee.
Data collection
Data were collected retrospectively and entered into a database in both cohorts. The date of HCC diagnosis (Vienna cohort) and the date of sorafenib start (Mainz cohort) were considered the baseline of this study.
Statistical analysis
Baseline characteristics were summarized using descriptive statistics. A chi-square test or Fisher’s exact test were used to compare nominal data. A t-test was used to compare metric data. The median duration of RASi treatment was calculated from the date of RASi start or date of HCC diagnosis (if RASi was started before diagnosis) until the date of discontinuation or last follow-up visit where RASi intake was confirmed. In patients whose last follow-up visit took place within one month prior to death, we assumed that RASi were taken until death. OS was defined as time from date of diagnosis (date of biopsy if available or diagnostic imaging; Vienna cohort)/sorafenib start (Mainz cohort) until date of death or last contact. Survival curves were calculated using the Kaplan–Meier method and compared by log rank test (univariate analysis). Variables that reached a p-value of < 0.1 were entered into a multivariate analysis. The multivariate analysis was performed using Cox proportional hazard regression. Statistical tests were two-sided and a p-value < 0.05 was considered significant. All statistical analyses were performed using SPSS version 17.0 (SPSS Inc., Chicago, IL).
Results
Patient characteristics
Of 1050 patients diagnosed with HCC at the Division of Gastroenterology & Hepatology, Medical University of Vienna between December 1992 and January 2013, 447 subjects received sorafenib, experimental therapy, or BSC. Of these, 156 individuals had Child-Pugh class A and were therefore eligible (Figure 1). Forty-three (28%) of these patients already received RASi treatment (ACEi, n = 40; ARB, n = 3) at the time of HCC diagnosis. Patients who received RASi were older (mean age, 69 vs. 64 years) and included more sorafenib-treated subjects (33% vs. 17%) compared to those who were not treated with RASi. All other baseline characteristics were equally distributed and are shown in Table 1. Median follow-up time was 96.6 months and 147 (94%) patients died during follow-up. Median duration of RASi treatment was 12.7 months (95% confidence interval (CI) 2.7–22.6 months).
Flow chart of patient selection. Patient characteristics (Vienna cohort). ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; ECOG PS, Eastern Cooperation Oncology Group performance status; RAS, renin–angiotensin system. ACEi, n = 40; ARB, n = 3. Missing, n = 1 for RAS inhibitor ‘No’ group.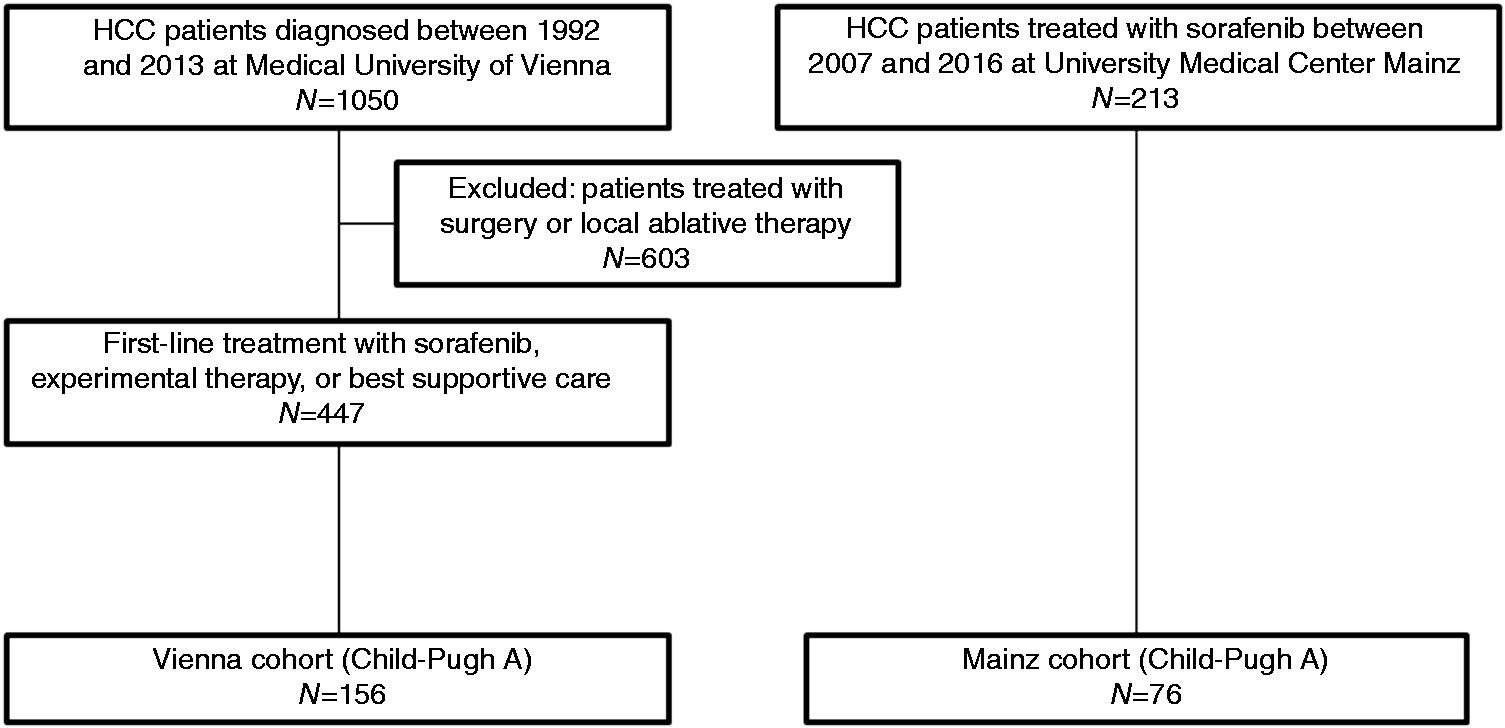
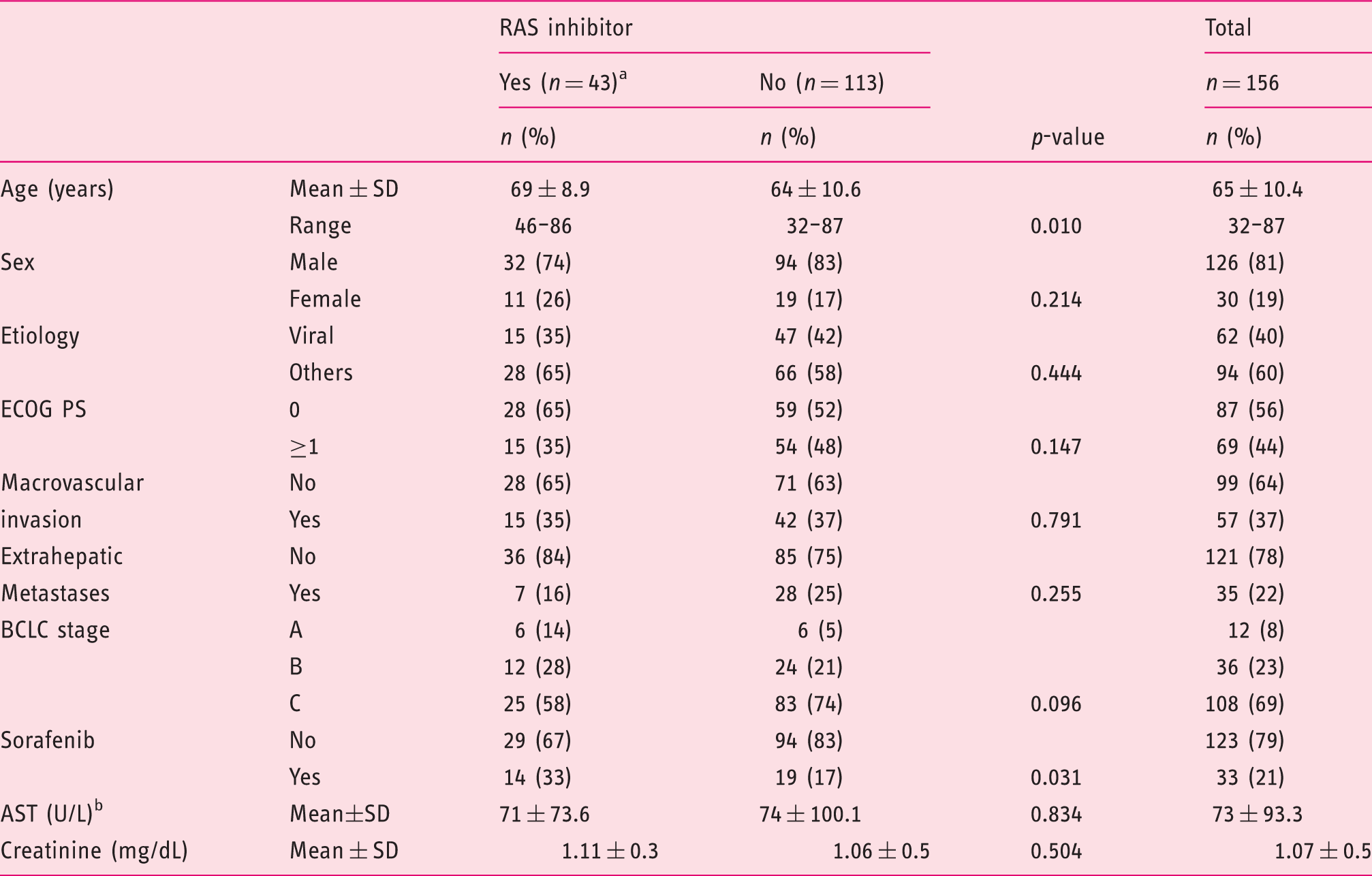
Patient characteristics (Mainz cohort).

ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; ECOG PS, Eastern Cooperation Oncology Group performance status; RAS, renin–angiotensin system.
ACEi, n = 27; ARB, n = 10; both, n = 1.
Missing, n = 2 for RAS inhibitor ‘Yes’ group and n = 1 for RAS inhibitor ‘No’ group.
Missing, n = 1 for RAS inhibitor ‘No’ group.
Missing, n = 4 for RAS inhibitor ‘No’ group.
Univariate and multivariate analyses of prognostic factors
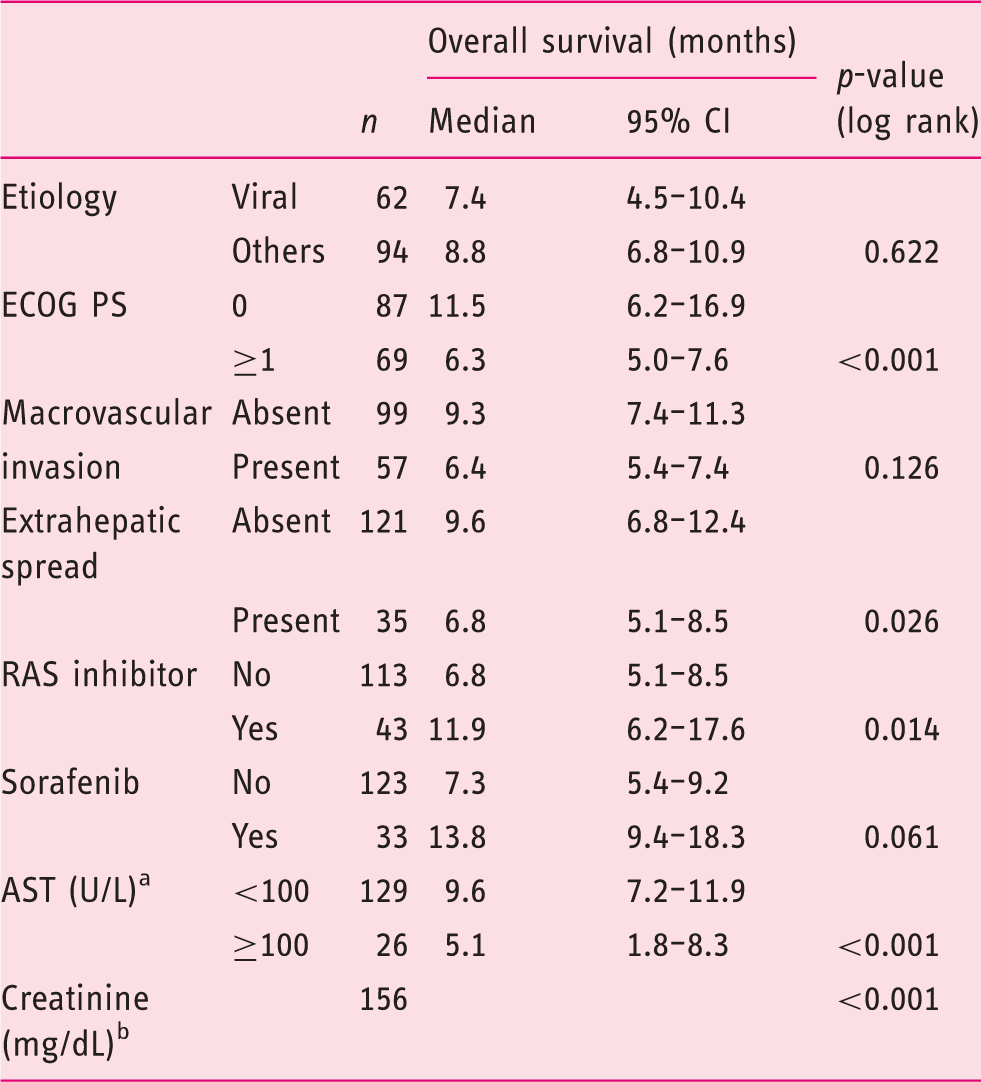
Median OS of the Vienna cohort (n = 156) was 8.3 months (95% CI 6.3–10.2 months). The 1-, 3-, and 5-year survival rates were 36%, 11%, and 4%, respectively.
Treatment with RASi was significantly associated with OS (median OS, 11.9 vs. 6.8 months; 95% CI 6.2–17.6 vs. 5.1–8.5 months; p = 0.014) (Table 3; Figure 2(a)). Eastern Cooperative Oncology Group (ECOG) performance status (PS), extrahepatic spread, AST, and serum creatinine were also significantly associated with OS (p < 0.05 for all variables; Table 3) and therefore included into multivariate analysis. Finally, sorafenib treatment achieved a p-value of < 0.1 and thus was also included (Table 3).
Kaplan–Meier survival curves. Survival according to renin–angiotensin system inhibitor (RASi) treatment in the Vienna (a) and Mainz cohort (b) and according to treatment with sorafenib (SOR) alone, neither SOR nor RASi, SOR+RASi, and RASi alone in the Vienna cohort (c). Univariate analysis of prognostic factors (Vienna cohort). AST, aspartate aminotransferase; ECOG PS, Eastern Cooperation Oncology Group performance status; RAS, renin–angiotensin system. Missing, n = 1. Continuous variable.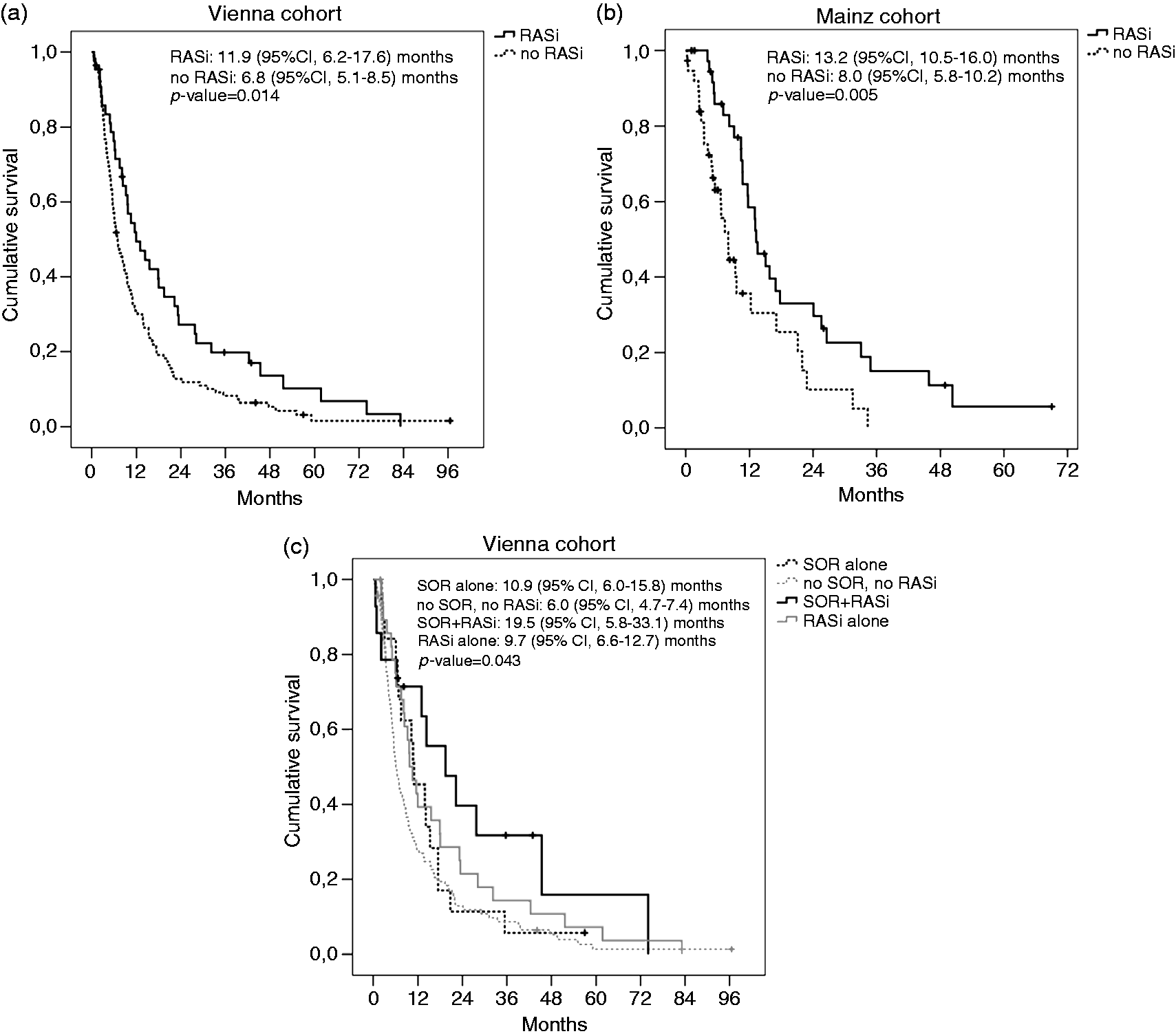
Multivariate analysis of prognostic factors (Vienna cohort).
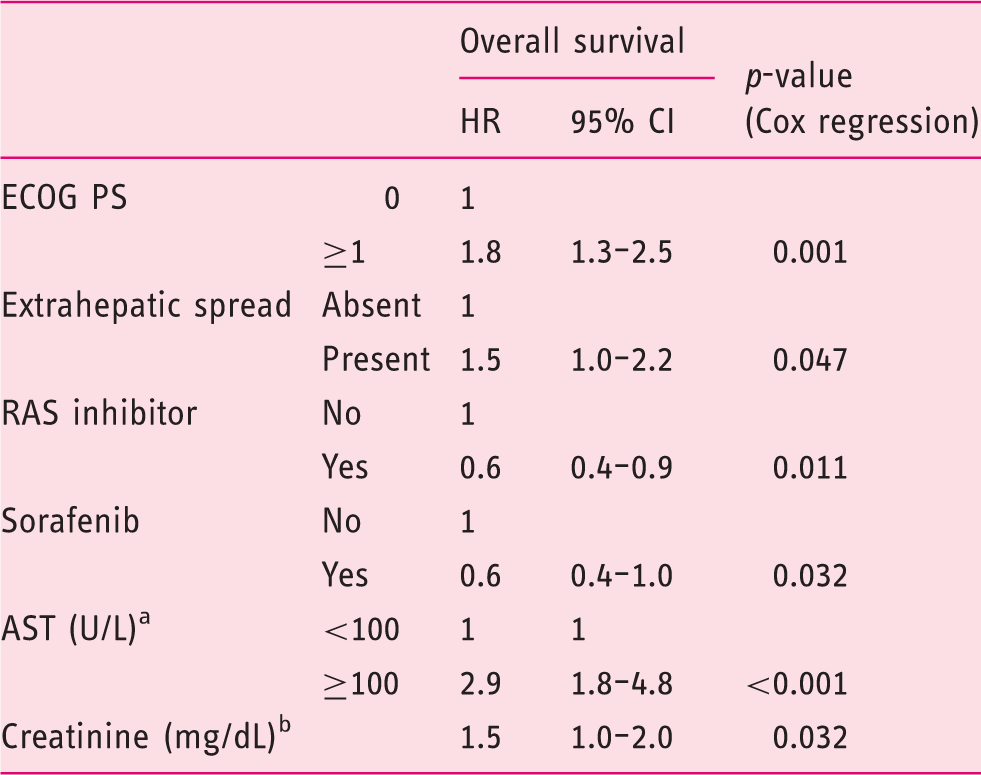
AST, aspartate aminotransferase; ECOG PS, Eastern Cooperation Oncology Group performance status; RAS, renin–angiotensin system.
Missing, n = 1.
Continuous variable.
Multivariate analysis of prognostic factors (Mainz cohort).
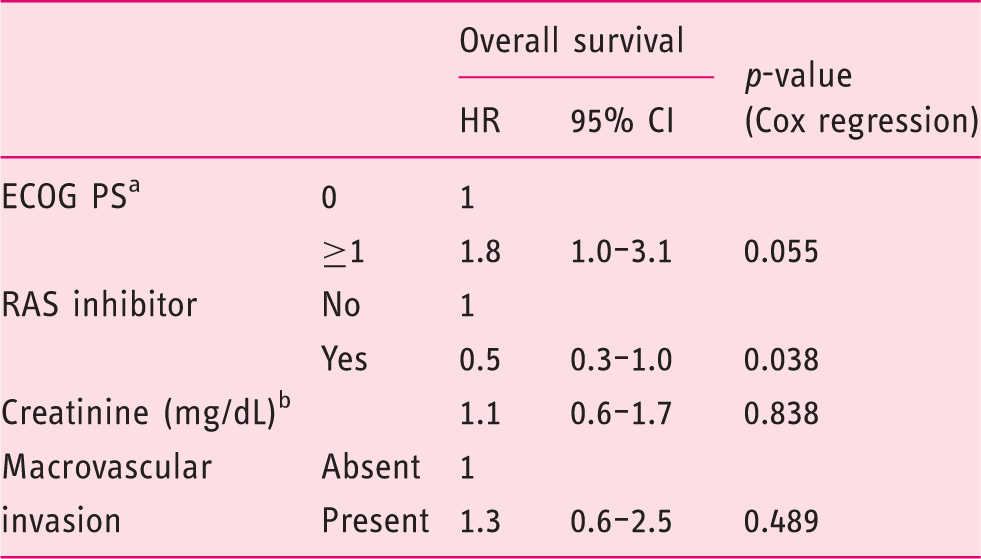
ECOG PS, Eastern Cooperation Oncology Group performance status; RAS, renin–angiotensin system.
Missing, n = 3.
Continuous variable; missing, n = 4.
Association between survival and RASi treatment stratified by sorafenib therapy
In the Vienna cohort, subgroup analysis showed that patients treated with sorafenib plus RASi (n = 14) had a median OS of 19.5 months (95% CI 5.8–33.1 months), subjects receiving sorafenib but no RASi (n = 19) had a median OS of 10.9 months (95% CI 6.0–15.8 months), patients who received RASi but not sorafenib (n = 29) had a median OS of 9.7 months (95% CI 6.6–12.7 months), and those who received neither sorafenib nor RASi (n = 94) had the worst survival of 6.0 months (95% CI 4.7–7.4 months; p = 0.043) (Figure 2(c)). Since all patients in the Mainz cohort received sorafenib treatment we could not perform similar subgroup analysis.
Discussion
We found that RASi treatment was an independent prognostic factor in a cohort of 156 HCC patients with Child-Pugh class A not amenable to surgical or local ablative therapy. We confirmed the association between RASi and OS in an independent cohort of 76 HCC patients treated with sorafenib.
A recently published retrospective study involving 153 HCC patients reported that treatment with ARB was associated with longer OS and time to recurrence after radiofrequency ablation. 18 However, these results have not been validated so far.
The RAS is involved in several tumor-promoting biological processes, such as cell proliferation, metastasis, resisting apoptosis, tumor inflammation, and immunosuppression.24,25 HCC is a complex disease as it usually develops in patients with liver cirrhosis and is then associated with portal hypertension (PHT) in the majority of cases.2,3,26 Thus, several other mechanisms may also contribute to the potentially beneficial effects of RASi on HCC survival.
First, RAS inhibition reduces tumor fibrosis/desmoplasia by blocking extracellular matrix (ECM) deposition via inhibition of both myofibroblast activation and expression of the profibrotic cytokine transforming growth factor beta (TGF-β).27–31 The desmoplastic stroma can act as a barrier to immune cells, 32 and cancer-associated fibroblasts can influence the immune system directly by inhibiting T and natural killer cells and by promoting an inflammatory pro-tumorigenic milieu. 33 Hence, the desmoplastic milieu can promote resistance to immunotherapy.34,35 Additionally, tumor desmoplasia contributes to blood vessel compression by increasing tissue stiffness and solid stress. The reduced tumor perfusion and the resulting hypoxia further support the immunosuppressive microenvironment, promote the selection of more invasive tumor cells, and lower the efficacy of certain treatment modalities.30,36,37 In several solid malignancies including cholangiocarcinoma, tumor desmoplasia occurs as a secondary event in response to tumor growth, wherein malignant cells drive stromal fibroblasts to produce and deposit ECM.27,30,38
In contrast, liver fibrosis and cirrhosis represent a procarcinogenic microenvironment that promotes development and progression of HCC, a tumor type not considered to be highly desmoplastic. 38
However, it was recently shown that blocking the activation of hepatic stellate cells and their transformation to myofibroblasts can attenuate tumor desmoplasia in HCC. 39 Sorafenib increased tumor desmoplasia by intensifying tumor hypoxia which led to increased myofibroblast infiltration and differentiation via upregulation of the stromal-derived factor 1 alpha (SDF-1α)/C-X-C chemokine receptor type 4 (CXCR-4) pathway and increased Gr-1(+) myeloid cell infiltration. Inhibition of the SDF-1α/CXCR-4 pathway or Gr-1(+) myeloid cell infiltration reduced hypoxia-mediated HCC desmoplasia and increase the efficacy of sorafenib treatment. 39
Second, the antifibrotic effects of RASi do not only reduce tumor desmoplasia but also inhibit the progression of liver fibrosis, as shown by several preclinical and clinical studies.40–46 Given the facts that most HCC patients suffer from concomitant liver fibrosis/cirrhosis,2,3 and that the non-tumoral surrounding liver tissue and the degree of liver dysfunction represent the main prognostic factors in HCC patients,4,47–50 the attenuation of liver fibrosis progression might also contribute to the improved survival associated with RASi treatment.
Third, the inhibition of angiotensin II type 1 receptor (AT1R)-mediated angiogenesis (via reducing VEGF production) represents another important mechanism.51–55 Notably, the combination of sorafenib and losartan attenuated the development of preneoplastic lesions in a non-diabetic rat model of steatohepatitis along with a reduction of hepatic neovascularization and VEGF. 21 Other preclinical studies investigated ACEi/ARB in the preventive setting,56–58 or in established tumors,59–61 in different rodent models of HCC and found that these drugs, alone or in combination with other treatments, were able to inhibit HCC development and growth, respectively. These studies proposed the downregulation of VEGF expression and the consecutive reduction of angiogenesis as potential mechanism of action.56–61 The combinations of the ACEi perindopril with vitamin K, 20 and with branched-chain amino acids, 19 were investigated prospectively in patients who underwent curative treatment for HCC, based on preclinical efficacy data on HCC prevention and tumor growth delay.62–64 Both clinical studies reported that the combination treatment but not perindopril monotherapy was able to significantly reduce tumor recurrence. This effect was associated with a downregulation of circulating VEGF and soluble VEGF receptor 2 levels in blood.19,20
Finally, patients with HCC often suffer from portal hypertension (PHT),3,26 and complications of PHT (i.e. variceal bleeding) represent a common cause of death in patients with HCC. The RAS is implicated in the pathogenesis of PHT since it increases intrahepatic resistance by inducing proliferation and contraction of hepatic stellate cells, profibrogenic TGF-β signaling, and ECM deposition.23,28,29,31 Moreover, as discussed above, angiotensin is also involved in angiogenesis, 51 which is not only a hallmark of cancer but also of PHT.65,66 Indeed, several studies of RASi for PHT showed promising results.67–70 In a meta-analysis 23 of 19 controlled trials, ACEi/ARB treatment reduced portal pressure in Child-Pugh A patients (−17%; 95% CI: −28 to −6) without adverse events. The authors concluded that the efficacy and safety of this group may be owed to a targeted effect on the local hepatic renin–angiotensin–aldosterone (RAAS) system while patients with more advanced/decompensated cirrhosis risk hypotension and renal insufficiency due to activation of the systemic RAAS. 23
This study has several limitations including the small number of patients in some subgroups and the retrospective nature of this study with all its possible shortcomings (e.g., potential impact of unmeasured confounders). However, the well-balanced baseline characteristics between the RAS inhibitors “yes” and “no” groups, the alignment of the survival of sorafenib-treated HCC patients with the survival reported in the SHARP phase III trial, 6 and the confirmation of the data in an independent cohort support the validity of our data.
In conclusion, RASi were associated with improved survival in HCC patients treated with sorafenib. Several mechanisms may contribute to the beneficial effects of RASi on survival including reduction of tumor desmoplasia and liver fibrosis, improvement of anti-tumor immunity, decrease of portal pressure, and inhibition of angiogenesis. Thus, RASi might be most effective in patients with HCC and early-stage liver cirrhosis when combined with treatment modalities that induce hypoxia and consequently desmoplasia, immunosuppression, and VEGF expression (e.g., sorafenib, TACE). ACEi/ARB might also be a reasonable combination partner for immunotherapy. These promising results should encourage prospective evaluation.
Footnotes
Declaration of conflicting interests
M.P. received travel support and speaking fees from Bayer; A.W. received lecture and travel fees from Bayer Vital; M.A.W. received consulting and lecture fees from Bayer HealthCare; F.H. received travel support from Bayer; S.B. received travel support from Bayer; J.U.M. has no conflicts of interest; D.G.D. received research grants from Merrimack, Bayer and HealthCare Pharma; R.K.J. received consultant fees from Ophthotech, SPARC, SynDevRx, XTuit, owns equity in Enlight, Ophthotech, SynDevRx, XTuit, and serves on the Board of Directors of XTuit and the Boards of Trustees of Tekla Healthcare Investors, Tekla Life Sciences Investors, Tekla Healthcare Opportunities Fund, Tekla World Healthcare Fund; P.R.G. is consultant and speakers bureau member of Bayer Healthcare; P.R.G. received consulting and lecture fees from Bayer HealthCare, Lilly, BMS, MSD, Sillajen, Arqule, Sirtex, Blueprint; M.T. serves as a consultant for Albireo, Falk, Genfit, Gilead, Intercept, MSD, Novartis, Phenex and is a member of the speakers' bureau of Falk, Gilead, MSD, Roche. He received travel grants from Falk, Roche, Gilead and unrestricted research grants from Albireo, Falk, Intercept, MSD. He is also co-inventor of a patent on the medical use of norUDCA. M.P.R. is an investigator for AbbVie, Arqle-Daiichi, Bayer, BMS, Boehringer-Ingelheim, Imclone, Lilly, Novartis, Roche, a speaker and advisor for Abbott, Bayer, BMS, Boehringer-Ingelheim, Lilly, Novartis, Roche, and received grant support from AbbVie, Arqle-Daiichi, Bayer, Roche; W.S. received travel support and speaking fees from Bayer.
Funding
MP is supported by an Erwin-Schroedinger Fellowship by the Austrian Science Fund (FWF; project number J 3747-B28). JUM is supported by grants from the German Cancer Aid (DKH 110989) and the Volkswagen Foundation (Lichtenberg program). DGD and RKJ are supported by NIH grant P01-CA080124.
