Abstract
Background
Aspirin use may reduce the incidence of pancreatic cancer (PC), but no data are available regarding its chemopreventive effects on intraductal papillary mucinous neoplasm (IPMN). We aimed to determine whether low-dose aspirin (LDA) reduces PC development and morphological changes on imaging in IPMN patients.
Methods
A cohort of 448 IPMN patients periodically followed up with imaging tests was analyzed. We used one-to-two propensity score matching to adjust for differences between an LDA group (n = 63) and a non-LDA group (n = 385). Outcomes included increasing cyst diameter, increasing main pancreatic duct (MPD) diameter, mural nodule (MN) appearance and PC development.
Results
After matching, 63 LDA and 126 non-LDA patients were selected. During follow-up (median, 5.5 years), no significant differences were found in increasing cyst diameter, MN appearance, or PC development. However, there were significantly fewer cases of increasing MPD diameter in the LDA group (4.8% vs. 12.7%; p = 0.02). After adjustment for age and sex, LDA still decreased the risk of increasing MPD diameter (hazard ratio, 0.17; p = 0.02).
Conclusion
Our results do not support a chemopreventive effect of LDA on PC development. However, LDA reduces further MPD dilation in IPMN patients.
Introduction
Pancreatic cancer (PC) is the fourth leading cause of cancer-related death and one of the 10 most common malignancies worldwide. 1 Intraductal papillary mucinous neoplasm (IPMN) is a well-defined premalignant condition of PC.2,3 However, IPMNs are often not resected because of the highly invasive nature of the surgery required and the high risk of surgical complications. 4 Therefore, prevention of PC in IPMN patients is an important clinical issue to address.
Low-dose aspirin (LDA) is generally prescribed for the secondary prevention of major cerebrovascular and/or cardiovascular events and related death. 5 Aspirin inhibits the cyclooxygenase-2 (COX-2) pathway and also has chemopreventive effects on several cancers.6,7 Most studies of aspirin-related chemoprevention have focused on colorectal cancer, and only a few studies have shown a preventive effect of aspirin use on PC development.8,9 However, no data are available on the association between aspirin use and PC development from IPMN.
Whether physicians prescribe aspirin is potentially affected by several background factors of patients. Therefore, in this study, we used propensity score-matching to adjust for differences in baseline characteristics between IPMN patients receiving LDA and those not receiving it. We aimed to determine whether LDA reduces the occurrence of PC development and morphological changes seen on imaging during long-term follow-up of IPMN patients.
Materials and methods
Study design, setting, and participants
We conducted a retrospective cohort study using data from a prospectively recorded electronic medical database (MegaOak online imaging system, NEC, Tokyo, Japan) between February 1996 and August 2015 at the National Center for Global Health and Medicine. This retrospective study was institutional review board approved, and patient consent for the study was waived. The electronic database is a searchable collection of records in which radiologists prospectively input the findings of multidetector computed tomography (MDCT) or magnetic resonance cholangiopancreatography (MRCP).
10
First, expert radiologists made the diagnosis of IPMT or IPMN (Figure 1). The search identified 830 consecutive patients with suspected IPMN. Then, our study group reviewed all images independently. However, our study group was not blinded to the participants’ clinical information.
Flowchart of subject recruitment. IPMN: intraductal papillary mucinous neoplasm; IPMT: intraductal papillary mucinous tumor; SCN: solid cell nest; SPN: solid pseudopapillary neoplasm; LDA: low-dose aspirin; MPD: main pancreatic duct.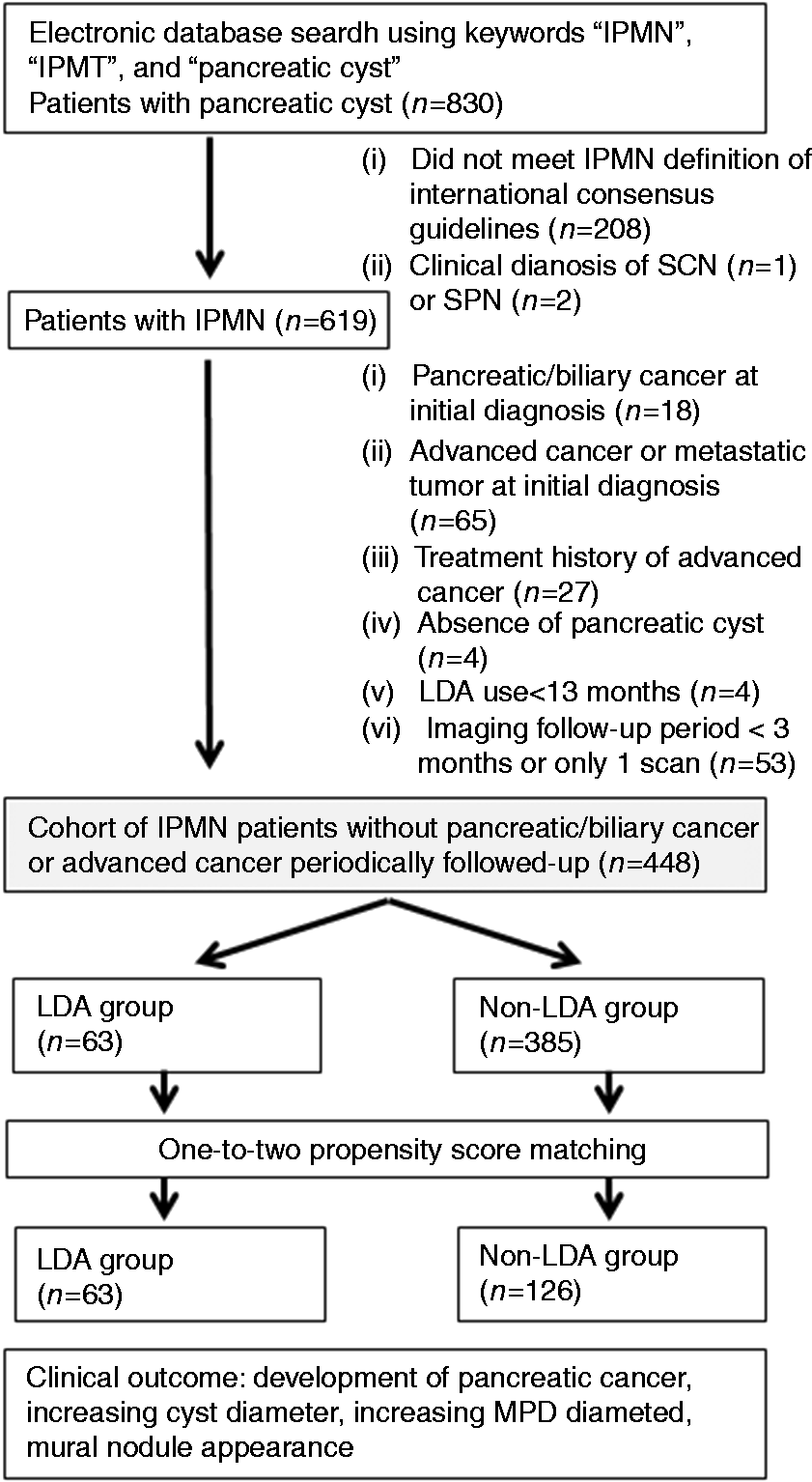
We first excluded patients who did not meet the IPMN criteria as defined by the international consensus guidelines 2012 11 of segmental or diffuse dilation of the main pancreatic duct (MPD) > 5 mm without other causes of obstruction and a pancreatic cyst communicating with the MPD as seen on MRCP or MDCT. Endoscopic retrograde cholangiopancreatography and endoscopic ultrasonography were performed at the discretion of the attending physician, in addition to MDCT or MRCP, when required for the differential diagnosis between IPMN and another pancreatic cyst. Other exclusion criteria were: (i) clinical diagnosis of serous cystic neoplasm or solid pseudopapillary neoplasm (n = 3); (ii) pancreatic cancer or biliary cancer at initial diagnosis (n = 18); iii) advanced cancer or metastatic cancer at initial diagnosis (n = 65); (iv) treatment history for advanced cancer (n = 27); (v) LDA use < 12 months (n = 4); and (vi) imaging follow-up period < 3months or one-time imaging (n = 53). This left a cohort of 448 IPMN patients who had periodic imaging follow-up for analysis (Figure 1). This study was approved by the Ethics Committee of the National Center for Global Health and Medicine.
Data collection
At each hospital visit, data on height and weight, lifestyle factors, past history, drug use, comorbidities, and laboratory tests were prospectively entered into the electronic medical records system by the physician or nurse. We evaluated 10 comorbidities: hypertension (HTN), dyslipidemia (DL), diabetes mellitus (DM), ischemic heart disease (IHD), stroke, peripheral artery disease (PA), chronic kidney disease (CKD), chronic liver disease (CLD), connective tissue disease, and chronic obstructive pulmonary disease (COPD). HTN, DL, and DM were defined by the patient taking specific medications. IHD was defined as acute myocardial infraction or angina, stroke as cerebral infraction or transient ischemic attack, CKD as receiving hemodialysis, and CLD as Child-Pugh score B or C. We also evaluated whether there was a history of acute pancreatitis, chronic pancreatitis, or cancer. The definition of daily LDA use was patients who took 100-mg enteric-coated aspirin or 81-mg buffered aspirin daily over 12 months. There were no patients taking nonsteroidal anti-inflammatory drugs (NSAIDs) daily.
Outcomes and follow-up
Patients with IPMN were periodically followed up with MDCT and/or MRCP. 10 During the follow-up period, most of the enrolled patients visited our hospital regularly (every three to six months) and were managed by experienced gastroenterologists. Because the prescription period for any drug under the health care system is limited to three months in Japan, patients needed to visit the hospital at least every three months for prescription refill. Patients were also followed-up with monitoring of serological tumor markers and laboratory data, and imaging. The images were interpreted by expert radiologists. The clinical outcomes were PC development and the morphological changes of increasing cyst diameter, increasing MPD diameter, and appearance of MN on imaging. Morphological changes on imaging were defined specifically as follows: cyst diameter increase of ≥ 10 mm compared with initial cyst diameter;12,13 MPD diameter increase by ≥ 3 mm compared with initial MPD diameter; 14 and appearance of an MN in the cyst. We defined “positive imaging changes” as having any of these three imaging changes (increasing cyst diameter, increasing MPD, and MN appearance).
Statistical analysis
Comparison of categorical data between the groups was performed using the Chi-square test. When cell counts were less than five in a 2 by 2 table, Fisher’s exact test was also performed. Continuous data were compared with Wilcoxon’s rank-sum test. Propensity score matching was used to reduce the effects of selection bias and potential confounders. 15 To estimate the propensity score, we employed a logistic regression model for LDA use that included those variables that we found to be significantly different between patients who did and did not use LDA; namely, age ≥ 70 years, sex, HTN, DM, DL, IHD, stroke, PA, CKD, CLD, COPD, and initial diagnosis year in or after 2008. Also, we included the previously reported risk factors of initial cyst size ≥ 30 mm, initial MN in the cyst, and initial MPD dilation > 5 mm that might be associated with PC development. We performed one-to-two matching analysis between LDA use and non-LDA use using the nearest neighbor method with a caliper width of 0.2 of the standard deviation of the logit of propensity score. After propensity matching, the differences between several outcomes, including PC development, increasing cyst diameter, increasing MPD diameter, and MN appearance, were compared between the two groups. Follow-up lasted from the date of initial imaging diagnosis to the date of diagnosis of any of the outcomes of PC development, increasing cyst diameter, increasing MPD diameter, and MN appearance. We censored patients at the last imaging date. The Kaplan-Meier method was used to estimate the cumulative incidence of any outcomes at 1, 5, 10, 15, and 20 years. Cox’s proportional hazards modeling was used to estimate unadjusted and age- and sex-adjusted hazard ratio (HR) and 95% confidence intervals (CI). P values < 0.05 were considered significant. All statistical analysis was performed using STATA version 13 software (StataCorp. College Station, TX, USA).
Results
Patient characteristics
Demographics and clinical characteristics of IPMN patients (n = 448).
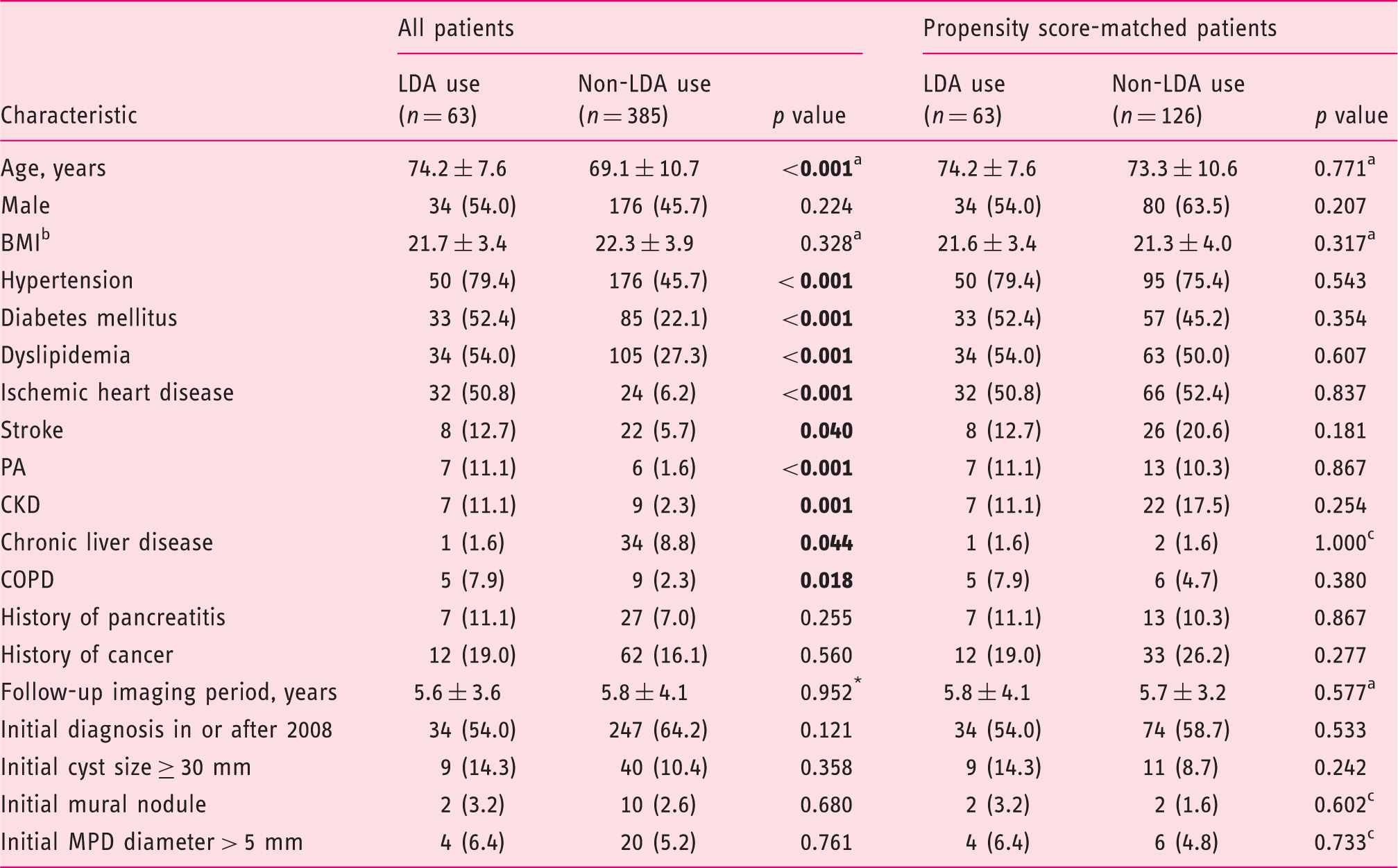
Values in parentheses are percentages. Values presented with a plus/minus sign are means ± SD. aMann–Whitney test. bBMI was analyzed before matching for 58 patients taking aspirin and 293 patients not taking aspirin and after matching for 58 and 107 patients, respectively. cFisher’s exact test.
BMI: body mass index; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; IPMN: intraductal papillary mucinous neoplasm; LDA: low-dose aspirin; MPD: main pancreatic duct; PA: peripheral artery disease.
Effect of daily LDA on PC development and morphological changes on imaging
Effect of daily low-dose aspirin on PC development and morphological changes on imaging.
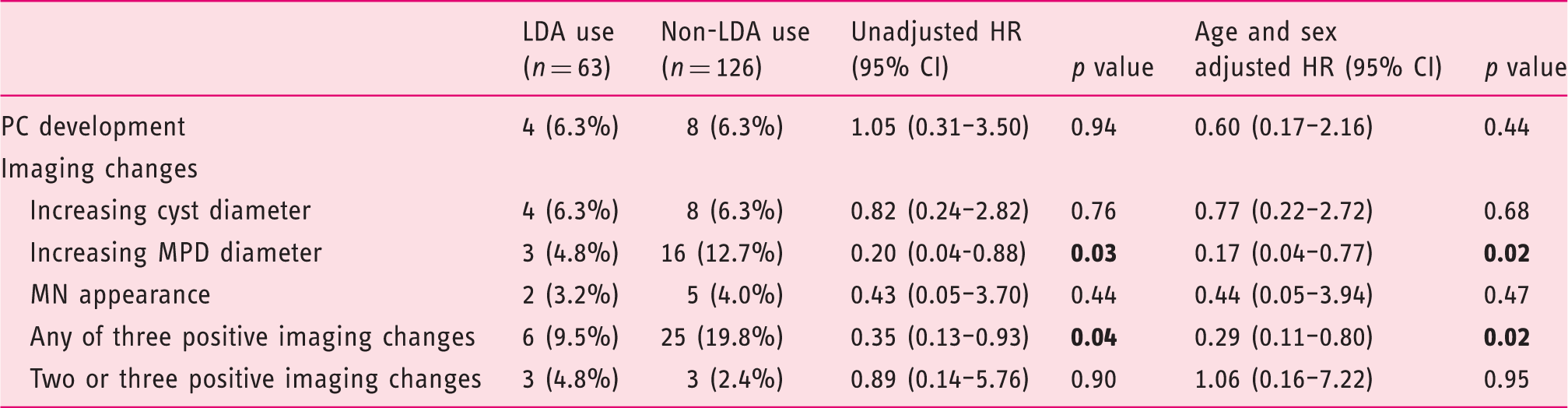
CI: confidence interval; HR: hazard ratio; LDA: low-dose aspirin; MN: mural nodule; MPD: main pancreatic duct; PC: pancreatic cancer.
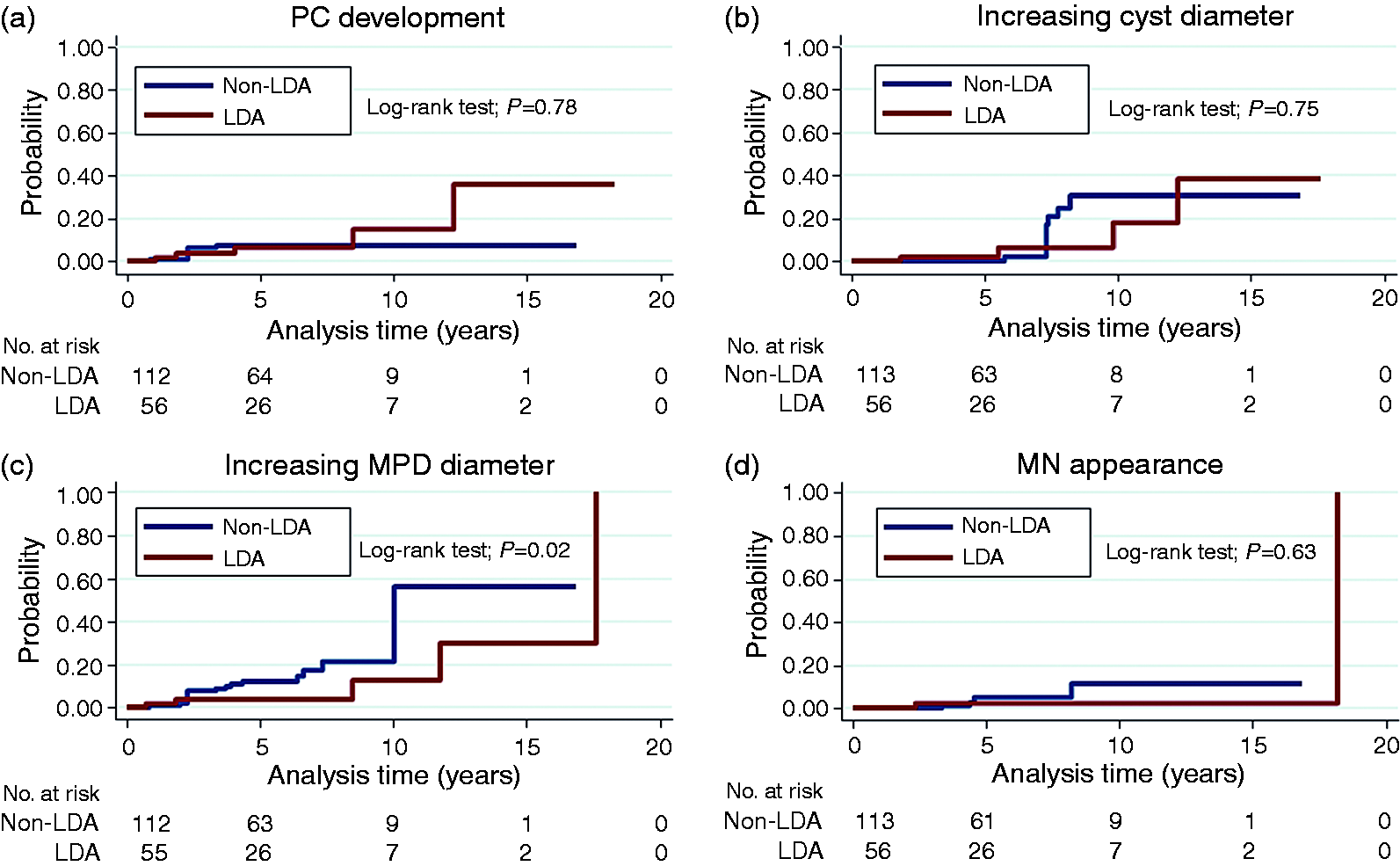
Cumulative PC and morphological changes on imaging during follow-up of IPMN patients. PC: pancreatic cancer; LDA: low-dose aspirin; MPD: main pancreatic duct; MN: mural nodule.
Discussion
This is the first study to use propensity-matched analysis to determine the effect of LDA use on PC development and morphological changes seen on imaging in IPMN patients. We found no association between LDA use and increasing cyst diameter, MN appearance, or PC development. However, compared with non-aspirin use, aspirin use decreased the risk of increasing MPD diameter (age and sex adjusted, HR 0.17, p = 0.02).
Previous studies have shown that increasing MPD diameter was a strong predictor of the development of malignancy in IPMN patients.16,17 Maguchi et al. studied 349 cases of branch duct (BD)-IPMN, including 22 surgical cases, and found a positive association between malignancy and the imaging findings of increasing MPD diameter and MN appearance. 17 In a meta-analysis, Kim et al. showed that the presence of MN, presence of MPD dilatation, and cyst size > 30 mm were risk factors for PC development, with a pooled disease ratio of 6.0, 3.2, and 2.3, respectively. 16 Furthermore, Yamaguchi et al. reported that a 2.2-mm increase in MPD diameter per year was useful to detect malignant IPMN. 14
Although there is no standard definition of increasing MPD diameter in IPMN, we defined it as further dilation of ≥ 3 mm based on the report by Yamaguchi et al. Another study revealed that MPD dilation was significantly associated with high-grade dysplasia and invasive carcinoma in a multivariate analysis. 18 Pancreatic adenoma was also associated with MPD dilation, 19 and thus aspirin use might reduce progression of pancreatic adenoma. Although MN appearance is reportedly a risk factor of PC development,17,20 our data do not show a positive association between MN appearance and aspirin use. This may be because of the small number of MN cases (n = 7) in this study. Previous studies have also shown that increases in cyst size ≥ 10 mm predicts malignancy in BD-IPMN patients.12,13 Therefore, we defined a growing cyst as a ≥ 10 mm increase in cyst diameter, but we found a null association between this increase and LDA use. Whether cyst size truly affects the risk of PC in IPMN remains unknown. Nagata et al. have stated that cyst size ≥ 30 mm was not associated with PC development and PC-related mortality. 10 Accordingly, we suggest that cyst size is neither a surrogate nor a predictive marker of PC development.
Several epidemiological studies have shown that aspirin use decreases the risk of PC development. Risch et al. reported that regular use of aspirin reduced risk of PC by almost half. 21 Rothwell et al. showed that aspirin use for > 5 years reduced the risk of death due to cancer including PC. 22 These anti-neoplastic effects of aspirin can be explained by the inhibition of COX. COX-2 exerts both cyclooxygenase activity and peroxidase activity, catalyzing the conversion of arachidonic acid to prostaglandins. 23 COX-2 expression was found in PC 24 and IPMN. 25 Therefore, aspirin, a COX-2 inhibitor, was expected to have a chemopreventive effect on invasive IPMN and PC development. In a prospective non-randomized controlled trial that evaluated the suppressive effect of NSAIDs on BD-IPMN, 26 Hayashi et al. found that NSAIDs reduced cyst growth and MN height, which is a known surrogate marker of PC development, but it did not reduce increasing MPD diameter. This discrepancy with our results may be due to the short observation period (18 months) in the Hayashi et al. study. In our study, there were also no significant differences in increasing MPD diameter between LDA and non-LDA patients within the first 18 months of observation (data not shown).
Our study has some limitations. First, this was a single-center retrospective study. Second, although we adjusted for differences in baseline characteristics between the two groups, unmeasurable confounders associated with PC development may exist, such as a history of smoking, alcohol consumption, and a family history of PC. 27 Third, the propensity score matching between LDA use and non-LDA use in our study involved a very selective, relatively elderly population of IPMN patients (mean age 74 years) compared with other IPMN studies evaluating PC development with a mean age of 60–72 years,10,28 which might lead to lack of generalizability. Fourth, the number of patients in this analysis might be too small to observe significant changes between the groups. We did not perform sample size estimation because our study was strongly based on a descriptive and exploratory retrospective design, which might result in it being underpowered. Despite these limitations, this study has several important strengths. A large number of MDCT/MRCP (1466 scans) were performed during the follow-up period, providing natural history of IPMN in non-resected patients. Levy et al. showed the natural history of IPMN in surgical patients and found many patients with high-grade dysplasia and invasive carcinoma (31%, 32/103), which is a higher rate than in our study (6.3%, 12/189). 29 This is because the individuals in the Levy study included many patients with main duct type and mixed-type IPMN (51%, 53/103) compared with our study of main duct type (5.3%, 10/189). And we could analyze IPMN patients who were LDA and non-LDA users at a matching ratio of 1:2 propensity score matching.
In conclusion, our propensity-matched results do not support a major protective effect of LDA use for PC development. However, LDA reduces further MPD dilation during long-term follow-up of IPMN patients.
Footnotes
Acknowledgments
We thank Ms Eiko Izawa, Ms Kenko Yoshida, Ms Akiko Shimizu, and Ms Hisae Kawashiro for assistance with data collection, for which no financial compensation was received.
The author contributions are as follows: N Nagata designed the study; Y Takasaki mainly wrote the manuscript; K Imbe, Y Hisada and K Sekine were the main authors of the manuscript; T Tajima, M Yanase, J Akiyama and N Uemura edited the manuscript; Dr Kazuma Fujimoto mainly contributed to editing the revised manuscripts and discussion.
Declaration of conflicting interests
None declared.
Funding
This work was supported by Grants-in-Aid for Research from the National Center for Global Health and Medicine (26A-201) and the Pancreas Research Foundation of Japan. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
