Abstract
Background
The incidence of upper gastrointestinal injury by low-dose aspirin (LDA) has increased.
Objective
We aimed to clarify the risk factors and prevention strategies associated with LDA-induced gastroduodenal ulcer in Japanese patients.
Methods
A retrospective study involving 284 LDA users who underwent oesophagogastroduodenoscopy between January and December 2010 were included. We investigated the patients’ clinical characteristics and endoscopic findings.
Results
Of 284 patients, 29 (10.2%) had gastro and/or duodenal ulcers. Male gender, peptic ulcer history, abdominal symptoms, half-dose proton pump inhibitors (PPIs), complete-dose PPIs, and nonsteroidal anti-inflammatory drugs were significantly associated with LDA-induced gastro and/or duodenal ulcers: odds ratio (95% confidence interval) 3.62 (1.06–12.27), 6.60 (1.84–23.62), 3.06 (1.12–8.40), 0.16 (0.03–0.94), 0.07 (0.01–0.61), and 9.68 (1.64–57.18), respectively. PPI significantly reduced gastric ulcers and/or duodenal ulcers (p = 0.03). The modified Lanza score for gastric mucosal lesion in the LDA cessation group was significantly lower than in the LDA noncessation group (0.53 vs. 1.02; p = 0.008).
Conclusions
Half-dose PPIs as well as complete-dose PPIs were effective for preventing LDA-induced gastric and/or duodenal ulcers. The cessation of LDA before endoscopy may lead to an underestimation of LDA-induced gastric injury.
Introduction
Low-dose aspirin (LDA; 75–325 mg daily) prevents thrombus and embolus in patients suffering from ischaemic heart disease or ischaemic cerebrovascular disease. 1 The tendency for LDA to be prescribed has increased because of the number of patients with advanced age, diabetes mellitus, and hypertension, which are all risk factors for ischaemic heart disease. However, LDA has been shown to cause gastrointestinal damage 2 and is associated with a high risk of gastroduodenal ulcers and their potentially fatal complications (e.g. gastrointestinal bleeding, perforation). 3 In case–control studies, the odds ratio (OR) of bleeding is in the range of 2.4–7.7,4,5 which is similar to that seen in a regular user of nonsteroidal anti-inflammatory drugs (NSAIDs). 6 The mortality attributable to NSAIDs or LDA use is approximately 20–25 cases per million people, of which one-third are attributable to LDA. 7
The identified risk factors for gastrointestinal ulcer bleeding with aspirin use are as follows: a history of ulcer bleeding, regular use of aspirin, advanced age (>70 years), concomitant use of NSAIDs or anticoagulants, use of dual antiplatelet therapy, infection of Helicobacter pylori (H. pylori), history of alcohol abuse, diabetes, and renal failure.6,8 Proton pump inhibitors (PPIs) are prescribed for decreasing LDA-associated gastroduodenal mucosal injury as well as NSAID-induced injury.9–11 However, in Japan, there have been few studies on the association between the risk factors and prevention of upper gastrointestinal mucosal injury by aspirin alone and in combination with other medicines.
In this retrospective study, our purpose was to elucidate the risk factors and prevention strategies associated with LDA-induced gastroduodenal mucosal injury in Japanese patients treated with LDA for arteriosclerotic disease.
Methods
This retrospective study was performed at the Aichi Medical University Hospital from January 2010 through December 2010. All endoscopic examinations were recorded digitally. Consecutive outpatients receiving LDA (75 mg) were enrolled and endoscopic findings were evaluated by an experienced endoscopist (TI) who was blinded to the study. We defined an ulcer as a mucosal deficit of >5 mm in diameter. Patients who had a history of gastrectomy were excluded from the study.
We recorded the following data from medical records: clinical characteristics, including sex, age, smoking, and drinking habits; underlying disease (hypertension, hyperlipidaemia, ischaemic heart disease, diabetes mellitus, cerebrovascular disease, and collagen disease); peptic ulcer history; concomitant drugs [gastric agents, anticoagulants (warfarin and potassium), antiplatelet drugs (ticlopidine HCL, clopidogrel sulfate, and cilostazol), NSAIDs, corticosteroids, antihypertensives, and antihyperlipidaemics]; the presence of H. pylori infection; the reason for undergoing endoscopy [abdominal symptoms (epigastric pain, heart burn, dysphagia, anorexia, nausea) or bleeding signs (anaemia, haematemesis, tarry stool)]; and endoscopic findings.
The extent of gastrointestinal mucosal injury was expressed using a modified Lanza score (MLS); a score of 0 indicating no injury, a score of 5 indicating an ulcer for gastric injury, and a score of 4 indicating an ulcer for duodenal injury. 12 H. pylori infection was determined using the rapid urease test or via histology. The criterion for the absence of pre-existing gastroduodenal ulcer was defined as no peptic ulcer history in the current medical record and no evidence of peptic ulcer scar via endoscopy. Gastric mucosal atrophy was endoscopically scored in six grades (C1, C2, C3, O1, O2, and O3; C, closed; O, opened) according to Kimura and Takemoto’s classification. 13 The presence of gastric mucosal atrophy was defined as an endoscopic score from C3 to O3.
Results
Demographic and clinical characteristics
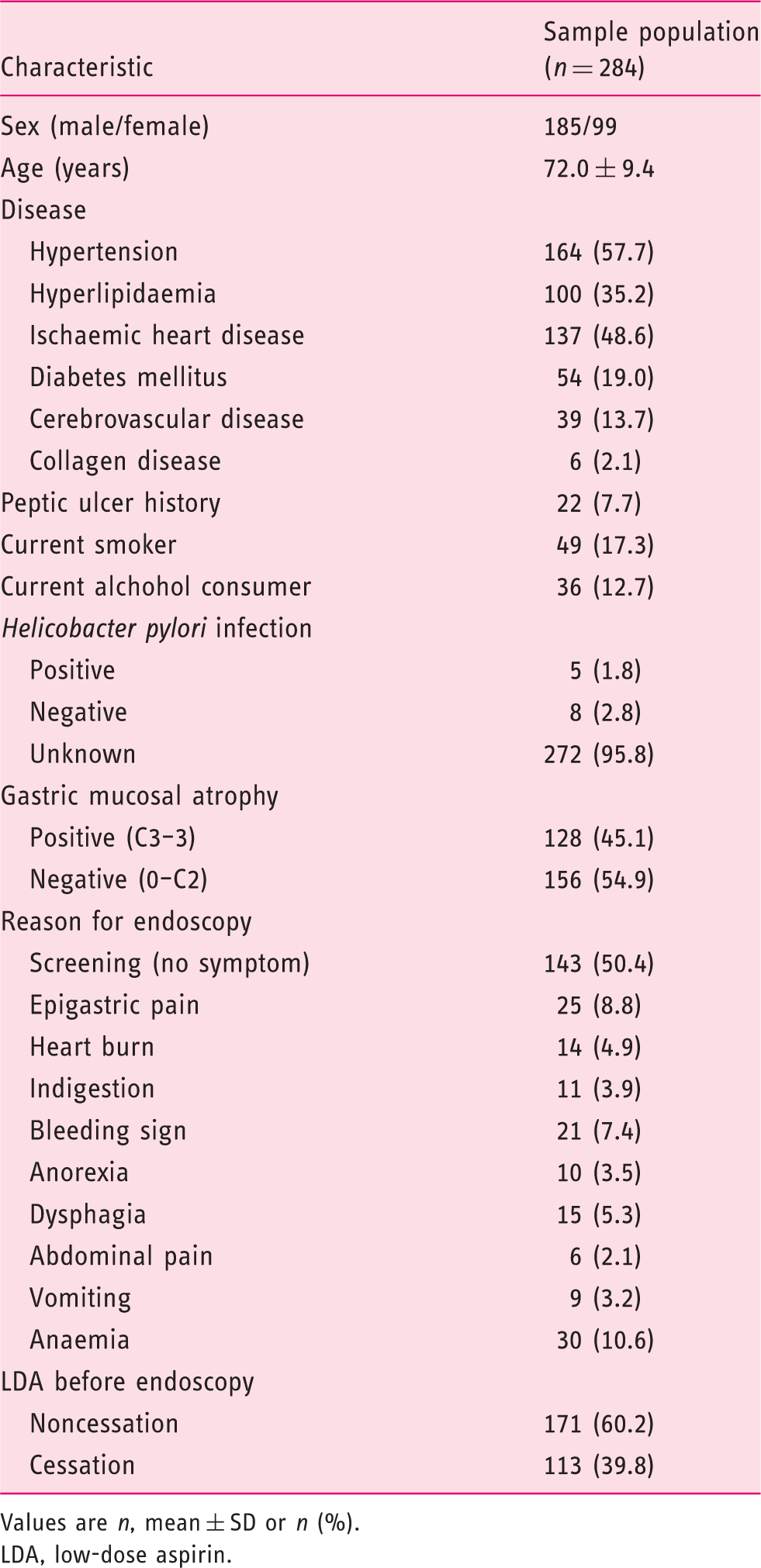
Values are n, mean ± SD or n (%).
LDA, low-dose aspirin.
Concomitant drugs
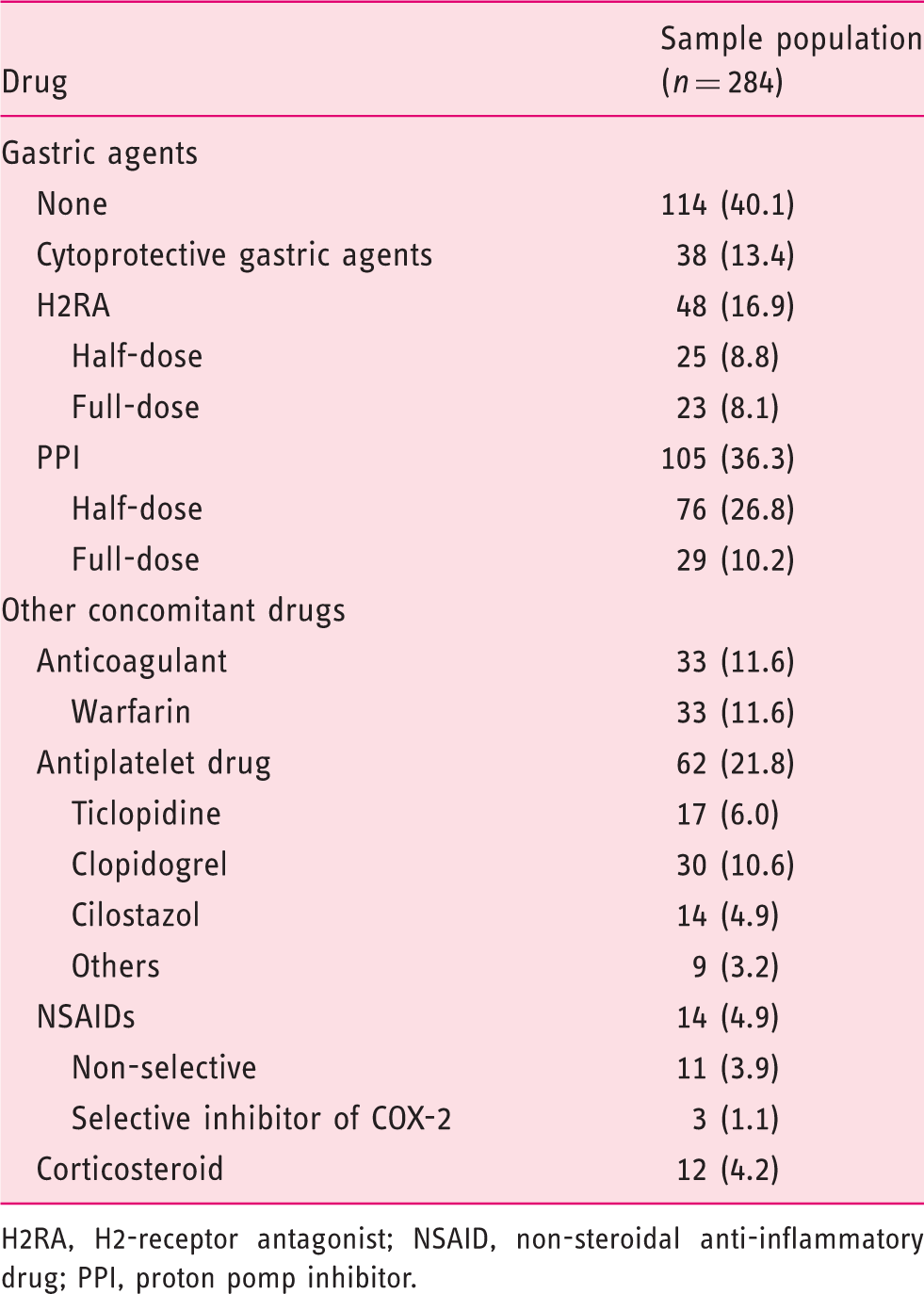
H2RA, H2-receptor antagonist; NSAID, non-steroidal anti-inflammatory drug; PPI, proton pomp inhibitor.
Multivariate analysis of the factors associated with gastroduodenal ulcers in patients receiving LDA
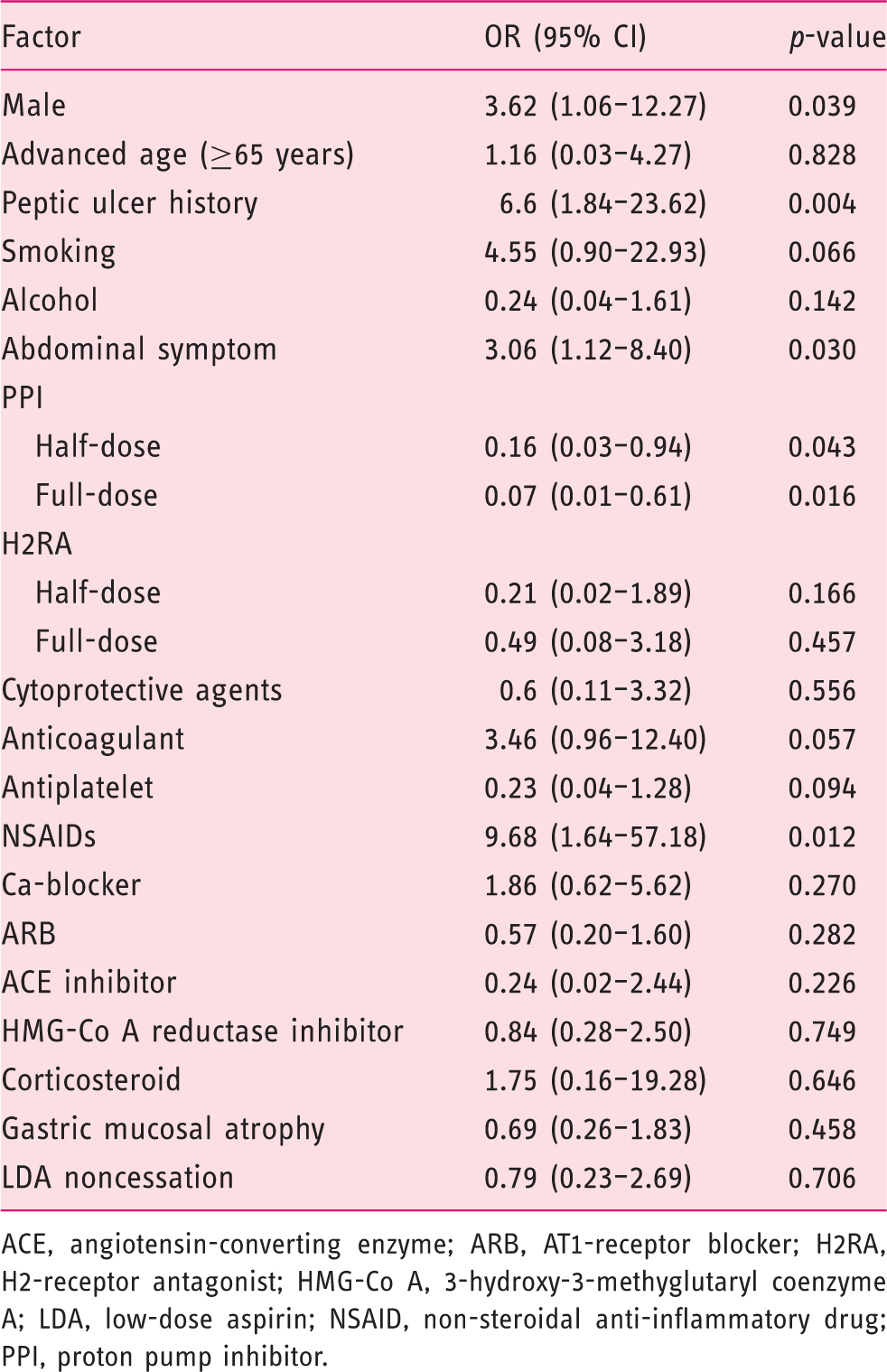
ACE, angiotensin-converting enzyme; ARB, AT1-receptor blocker; H2RA, H2-receptor antagonist; HMG-Co A, 3-hydroxy-3-methyglutaryl coenzyme A; LDA, low-dose aspirin; NSAID, non-steroidal anti-inflammatory drug; PPI, proton pump inhibitor.
MLS and LDA-induced gastric ulcer in the LDA cessation and noncessation groups
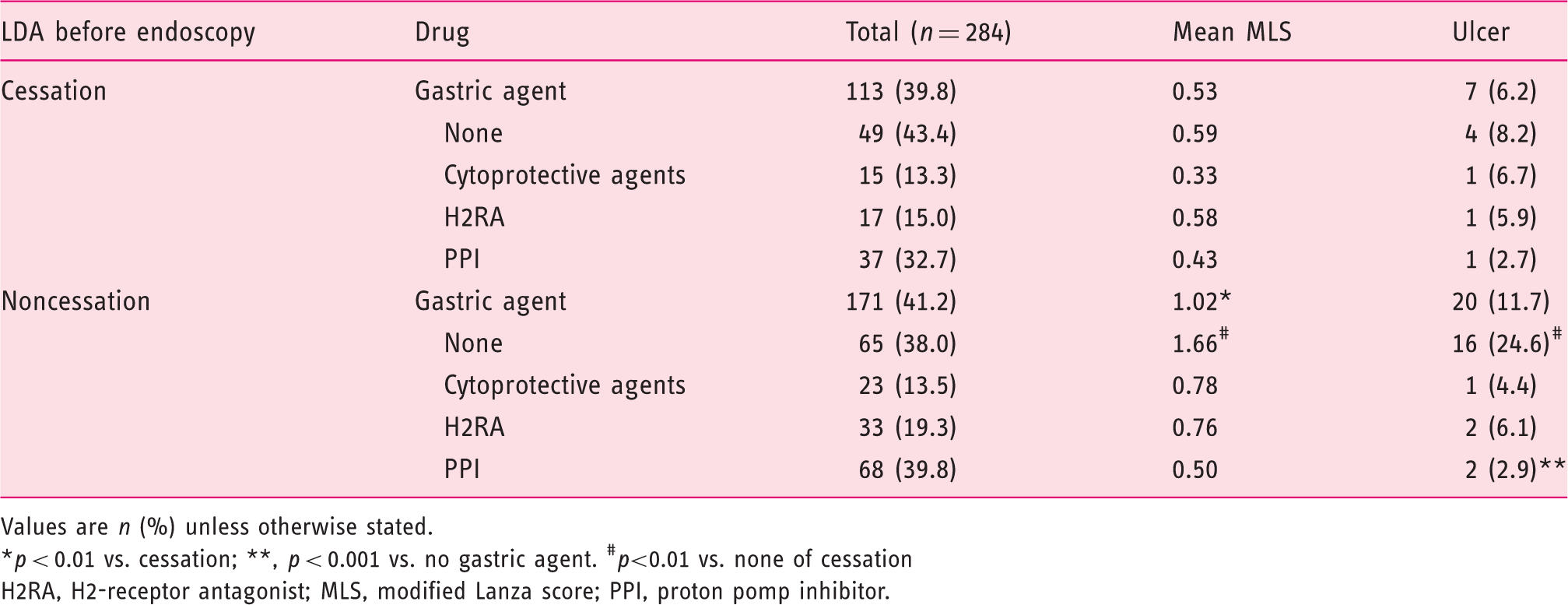
Values are n (%) unless otherwise stated.
*p < 0.01 vs. cessation; **, p < 0.001 vs. no gastric agent. #p<0.01 vs. none of cessation
H2RA, H2-receptor antagonist; MLS, modified Lanza score; PPI, proton pomp inhibitor.
Discussion
In the present retrospective study, we focused on gastroduodenal ulcers requiring treatment and investigated the association between LDA-induced gastroduodenal ulcers and clinical parameters, including combinations of medications, both antacids or other antithrombotic medicines and antihypertensive or antilipidaemic medications. The ulcer lesions were endoscopically identified in 29 (10.2%) of 284 patients who were administered LDA, and this incidence rate is similar to the results of a former prospective Japanese study. 14
Multiple logistic regression analysis revealed that male gender, a history of peptic ulcer, the presence of abdominal symptoms, and concomitant medication with NSAIDs were significant risk factors of LDA-induced gastroduodenal ulcers. These results are consistent with those of previous reports.15,16 However, in this study, the possible risk factors (i.e. advanced age, concomitant anticoagulants, antiplatelet drugs, and corticosteroids) that have been previously reported 16 were not associated with LDA-associated peptic ulcers. Gender difference has been reported to affect the clinical features and the risk factors of LDA-induced peptic ulcers, which revealed advanced age was significant risk factor only in female. 17 In the present study, the number of men with gastroduodenal ulcers was twice that of women with gastroduodenal ulcers; therefore, advanced age was not considered a risk factor for LDA-associated ulcers. Anticoagulant and corticosteroid use was not identified as a risk factor because of the small number of patients. There was a strong correlation between sex and concomitant antiplatelet use, which may be the reason for the association between male gender and LDA-induced peptic ulcer.
As reported in a previous study, 15 the OR of peptic ulcer history was high (6.6); therefore, since 2011, in Japan, the treatment of such patients with PPI has been permitted as a medical service by health insurance for the prevention of NSAID- or LDA-induced peptic ulcers. Shiotani et al. 14 reported the association between LDA-induced peptic ulcer and AT1 receptor blocker or angiotensin-converting-enzyme inhibitor. However, in the present study, there was no relation between LDA-induced peptic ulcer and antihypertensive agents, or antihyperlipidaemic agents.
A complete PPI dose and a half PPI dose are known to be effective in the treatment and prevention of aspirin-related or NSAID-related peptic ulcers.18,19 In the present study, complete PPI dose and half PPI dose prevented the development of LDA-induced peptic ulcers. In a randomized, double-blind, placebo-controlled trial, H2RA was effective in the prevention of gastroduodenal ulcers in patients taking LDA. 20 However, in the present retrospective study as well as in a previous study, no association was observed between H2RA and LDA-induced peptic ulcer. 14
According to a Japanese epidemiological study, a minimal association was observed between H. pylori infection and NSAIDs with gastric bleeding. 5 NSAIDs and H. pylori infection seem independent risk factors for peptic ulcer and bleeding. Because the current study is retrospective, in which H. pylori infection was examined in only 13 patients, the effect of H. pylori infection on peptic ulcer development could not be investigated. Previous eradication therapy was not confirmed in 280 (98.6%) patients, and 128 (45.1%) patients were suspected with gastric mucosal atrophy detected via endoscopy. This result suggests that the H. pylori infection rate seems high, because of which 15 (5.3%) of 284 patients had gastrointestinal bleeding.
In Japan, LDA treatment is often discontinued 3–7 days before endoscopy in order to decrease the risk associated with the endoscopic procedure. Tamura et al. 15 presented the association of the LDA cessation and the prevalence of LDA-induced peptic ulcers. The 7-day LDA cessation improved gastroduodenal mucosal injuries such as erosion and ulcer. In the present study, LDA-induced gastroduodenal erosion improved after LDA cessation; however, gastrointestinal ulcer did not improve after discontinuing LDA using multivariate analysis in cases including and excluding of cessation of LDA. This may be due to the short period of LDA cessation, which was 3–5 days in this study compared with a 7-day cessation in Tamura’s study. In the present study, bleeding signs were endoscopically recognized in 12 cases, and in 10 cases LDA was not discontinued before endoscopy. On the basis of the results of both Tamura’s and our study, we can conclude that a short duration of cessation LDA improves gastroduodenal mucosal injury in patients treated with LDA. LDA-induced gastroduodenal mucosal injury might be underestimated in patients with LDA cessation before endoscopy, and this is clinically important because it may prevent the prescription of gastric agents that should be prescribed in high-risk LDA users.
The interpretation of results of this study has limitations. A selection bias may be present in the patients enrolled in this study because the data were collected from LDA users who had undergone oesophagogastroduodenoscopy and not from all LDA users. A drug-selection bias may be present in this study. Moreover, because this was a retrospective study, this bias may have affected the evaluation of risk factors for LDA-induced gastroduodenal mucosal lesions. Additionally, we did not evaluate H. pylori infection for LDA-induced gastroduodenal injury because H. pylori infection was investigated in only 13 patients (4.6%) because examination of H. pylori infection is not permitted under Japan health insurance if there is no peptic ulcer. Like former studies, complete PPI dose and half PPI dose prevented the development of LDA-induced peptic ulcers, which might be suggest that drug-selection bias is relatively small.
In summary, in this retrospective study, 10.2% of LDA users had gastroduodenal ulcers. Male gender, peptic ulcer history, the presence of abdominal symptoms, and concomitant NSAID use were significant risk factors, and the use of PPI was a prevention strategies for LDA-induced gastroduodenal ulcers. The cessation of LDA before endoscopy might lead to an underestimation of LDA-induced gastroduodenal injury. Further study will be necessary to demonstrate the improvement of peptic ulcer by LDA cessation before endoscopy.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
KK received research grants from Research Organization for Gastro-Enterological Disease Treatment (NPO-ROGET) and Daiich Sankyo, and lecture fees from Daiich Sankyo and Astra Zeneca. The other authors declare that there is no conflict of interest.
