Abstract
Background
An effective and tolerable bowel preparation is important to secure quality of colonoscopies. It remains unclear if sodium picosulphate with magnesium citrate (SPMC), which is considered a tolerable bowel preparation agent, is also an effective alternative for polyethylene glycol (PEG) and sodium phosphate (NaP).
Aim
The aim of this article is to compare effectiveness of SPMC to PEG and NaP through assessment of quality of bowel cleansing measured by validated tools.
Methods
We searched electronic databases up to January 2015. Only randomised controlled trials (RCTs) were included. Two authors independently performed selection of studies, risk of bias assessment and data extraction.
Results
Thirteen RCTs were included, with overall good quality, but large heterogeneity. SPMC had slightly better quality of bowel cleansing than PEG (pooled RR 1.06; 95% CI 1.02 to 1.11). In most trials SPMC was significantly better tolerated than PEG. There were no significant differences in effectiveness or tolerability between SPMC and NaP. Side effects were similar between agents, except for dizziness (pooled RR 1.71; 95% CI 1.32 to 2.21 in favour of PEG vs. SPMC) and vomiting (pooled RR 0.35; 95% CI 0.13 to 0.95 in favour of single-dose SPMC vs. split-dose).
Conclusions
SPMC is equally effective to NaP and little superior to PEG in terms of bowel cleansing. SPMC preparations were better tolerated than PEG preparations. SPMC may be considered as standard bowel preparation for colonoscopy.
Keywords
Introduction
Colonoscopy accuracy and therapeutic safety depends heavily on the quality of bowel cleansing. 1 All current preparation strategies use a combination of dietary restrictions and laxative agents. 2
In Europe, three bowel preparation agents are commonly used, namely high-volume, osmotically balanced polyethylene glycol (PEG), low-volume, hyperosmotic sodium phosphate (NaP) and sodium picosulphate with magnesium citrate (SPMC), a low-volume osmotic laxative.2,3 Because of its taste and large volume, 5% to 15% of the patients do not complete PEG preparations. 1 Patients have less difficulty completing NaP preparations, but there are concerns regarding the safety of this agent. 4 SPMC is effective, and due to its small volume, is also easy to administer and a well-tolerated bowel preparation agent. 5
There are five previously published systematic reviews that compare SPMC with PEG and NaP as bowel preparation for colonoscopy.2–4,6 In four of them it remains unclear if SPMC is more, less or equally effective compared to PEG and NaP.2,3,6,7 Only Tan and Tjandra 4 found better effectiveness in favour of NaP. There are two previously published reviews that compare single-dose with split-dose bowel cleansing regimens.8,9 Both reviews found superiority of a split-dose regimen over a single-dose regimen, but these reviews mainly included studies about bowel cleansing regimens other than SPMC. However, all seven reviews included studies that used non-validated measurement tools for the assessment of the quality of bowel cleansing. Validated and standardised measurement tools are important to ensure an objective assessment of the quality of bowel cleansing by the endoscopist performing the colonoscopy. When studies with different and non-validated bowel cleansing measurement tools are included in a meta-analysis, it is very hard to compare results between these studies and draw reliable conclusions. As a result it remains unclear if SPMC is an effective alternative for PEG and NaP as bowel preparation for colonoscopy and if split-dose SPMC is more effective than single-dose SPMC.
Therefore, the present systematic review aims to assess the effectiveness of SPMC in comparison with PEG and NaP and single-dose SPMC in comparison with split-dose SPMC measured by validated tools. Secondarily we compare tolerability and side effects of these agents.
Materials and methods
Eligibility criteria
Only randomised controlled trials (RCTs) were included in this review. Selected studies included adults (>18 years), outpatients and hospitalised patients, who had to be prepared for colonoscopy. RCTs were included if they compared SPMC, or SPMC in combination with an additional laxative, with either PEG or NaP. RCTs comparing single-dose SPMC with split-dose SPMC were also included in this review. All included studies had to report the quality of bowel preparation assessed by a validated tool, namely the Ottawa Bowel Preparation Scale (OBPS), 10 the Boston Bowel Preparation Scale (BBPS) 11 or the Aronchick scale. 12 Unfortunately there are no validated measurement tools for tolerability or severity of side effects, so we made no restrictions to the use of the measurement tools for these outcomes. Furthermore, there were no restrictions with regard to publication date in our search, but we included RCTs published only in the English language.
Information sources
We searched the following electronic databases up to January 2015: MEDLINE (Appendix 1), EMBASE (Appendix 2) and the Cochrane Central Register of Controlled Trials – CENTRAL (Appendix 3). The references of the included RCTs were searched manually to ensure no pertinent articles were missed.
Study selection
Two authors (IvL and IM) independently evaluated titles and, when available, abstracts for eligibility in this review. When more information was required to determine whether a trial met the inclusion criteria, we obtained full-text. We resolved disagreement by discussion, and when consensus was not reached, we involved a third author (AE) for decision.
Data collection process
Two authors (IvL and IM) independently extracted data from the included trials. Again, we resolved disagreement by discussion, and when consensus was not reached, we involved a third author (AE) for decision. Data items were collected digital on the Cochrane data collection form for intervention reviews.
Data items
We decided a priori to extract the following data from each article: (1) general study information (e.g. author(s), title, study period, country, location); (2) characteristics of the study (e.g. age and sex), in- and exclusion criteria and numbers of patients in each group; (3) characteristics of the intervention (e.g. type, dosing and timing of the preparation); (4) outcome measures and measurement tools.
Our primary outcome measure is effectiveness of bowel preparation, defined as quality of bowel cleansing measured by a validated tool.
Our secondary outcome measures are: quality of bowel cleansing in each colon segment, tolerability, side effects (nausea, vomiting, abdominal pain, bloating, headache and dizziness), ability to complete the preparation and willingness to repeat the same preparation in the future. Secondary outcomes were reported only from studies that also reported our primary outcome measurement.
Risk of bias in individual studies
Two authors (IvL and IM) independently assessed the risk of bias of the included studies using the Cochrane Collaboration tool for assessing risk of bias. 13 This tool contains the following components: sequence generation, allocation concealment, blinding, incomplete outcome data, selective reporting and other potential threats to validity. We focused the assessment on the primary outcome measure and for each individual study we judged each component to be low, high or unclear risk of bias. Again, we resolved disagreement by discussion and involved a third author (AE) if necessary. Finally, we present a risk of bias summary figure to overview risk of bias assessment in all included studies.
Summary measures
Outcome measures are presented using the data and p values from the original articles. Where studies evaluated similar interventions in a similar population we formally tested heterogeneity of the data using the I2 test. Where pooling seemed appropriate in view of clinical similarities between studies, we planned to use a fixed-effect model where I2 was below 30% and a random-effect model where I2 was between 30% and 60%. 14 We did not intend to pool studies where I2 was greater than 60%. For continuous outcomes, mean differences (MD) and 95% confidence intervals (CI) were calculated. For dichotomous outcomes, relative risk (RR) and 95% CI were calculated. We presented the results as described in the original articles and did not perform any recalculations.
Results
Study selection
The search yielded 776 potentially relevant titles. Two authors (IvL and IM) independently evaluated titles and, when available, abstracts for eligibility in this review. After a consensus meeting 52 articles were selected. All of these were read full-text. We excluded 40 articles, if they were non-randomised trials, investigated another bowel preparation agent, investigated a preparation for another intervention or used a non-validated measurement tool for assessing the quality of bowel cleansing. One of the articles was excluded because it was not published in English. There was no disagreement between authors on in- and exclusion of articles. We checked the references of the remaining studies and previously published reviews and that revealed three more articles. One of them met all inclusion criteria and was also included in this review. Finally, 13 RCTs were included in this review. The process of study selection is shown in the flow diagram in Figure 1, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.
15
Flow diagram of the study selection.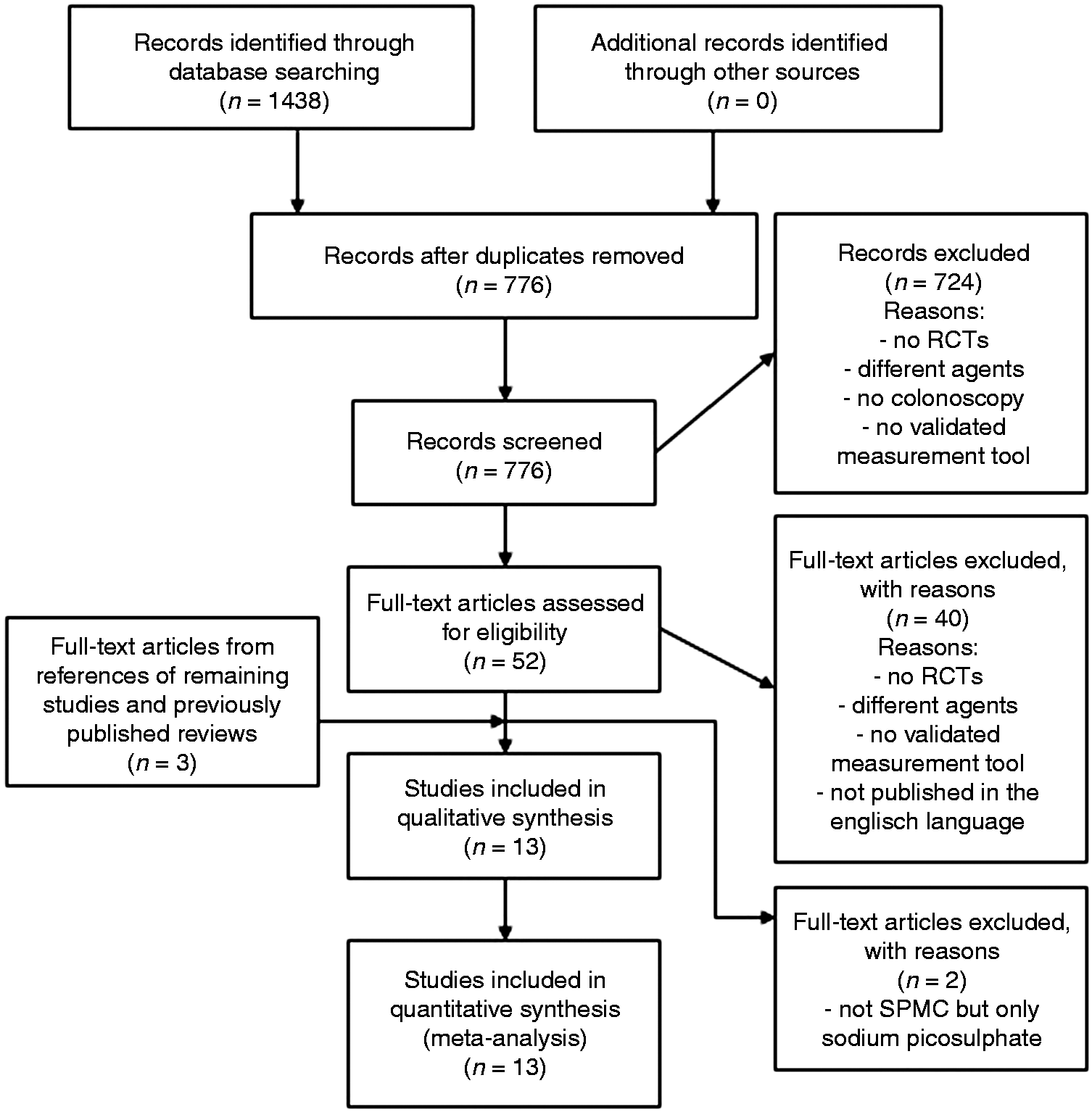
Study characteristics
Characteristics of included studies.

PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy.
Risk of bias within studies
Overall, the methodological quality of the 13 RCTs is similar and good (Figure 2). Sequence generation was unclear in two trials (15%), and concealment of allocation was not described in four trials (31%). Blinding of participants and personnel was impossible because of the nature of the intervention, but it is not likely that this affected the primary outcome in any of the trials, because this outcome was scored by the endoscopist, for whom blinding of the outcome assessment was ensured in all trials. Seven trials (54%) mentioned use of an intention-to-treat (ITT) analysis, two trials (15%) possibly contain selective reporting bias and in four trials (31%) insufficient information about possible influence of funders was given, resulting in an ‘unclear risk of bias’ score. The risk of bias summary is presented in Figure 2.
Risk of bias summary.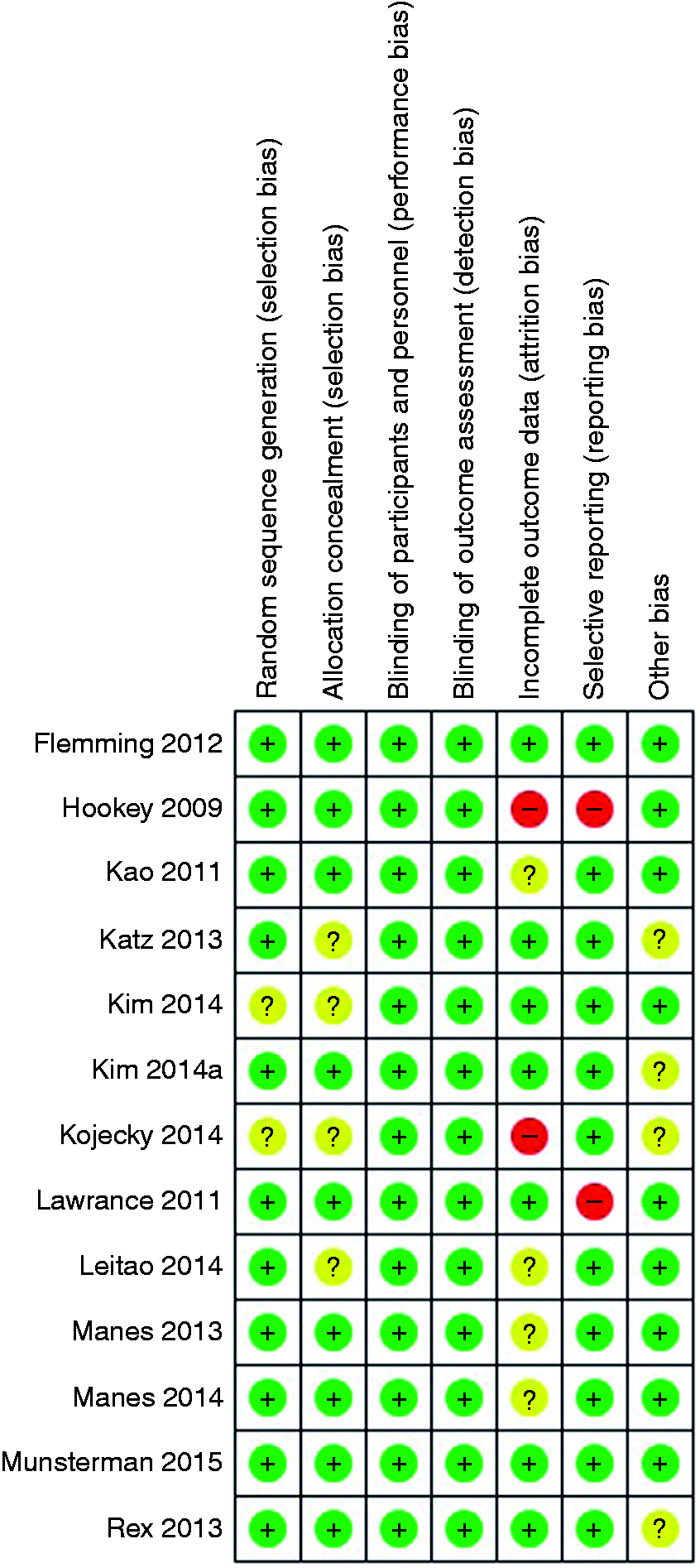
Heterogeneity
The studies vary in clinically and methodological terms. First of all, the intervention groups vary in dosing and timing of the bowel preparation agent, with or without bisacodyl and single-dose or split-dose. In addition, some outcome measures are measured with different measurement tools and most of the outcome measures are reported in different ways. Because of that, meta-analysis was feasible only with studies that used the same measurement tools and reported the results in the same way.
Synthesis of results
Quality of bowel preparation
Four trials found a significant difference in quality of bowel preparation in favour of SPMC compared to PEG19,28 or NaP.18,23 Eight trials reported no significant difference between SPMC and PEG18,20–22,24,25,27 or NaP.
17
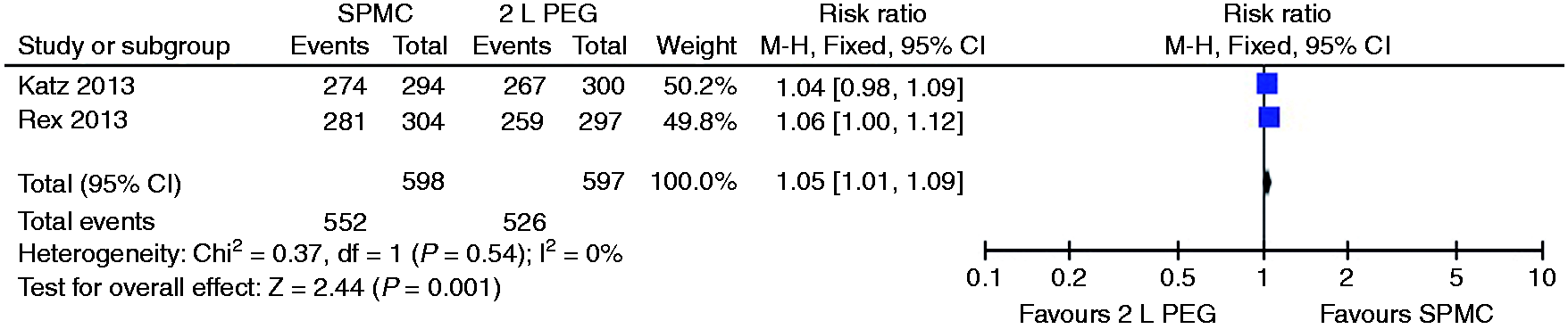
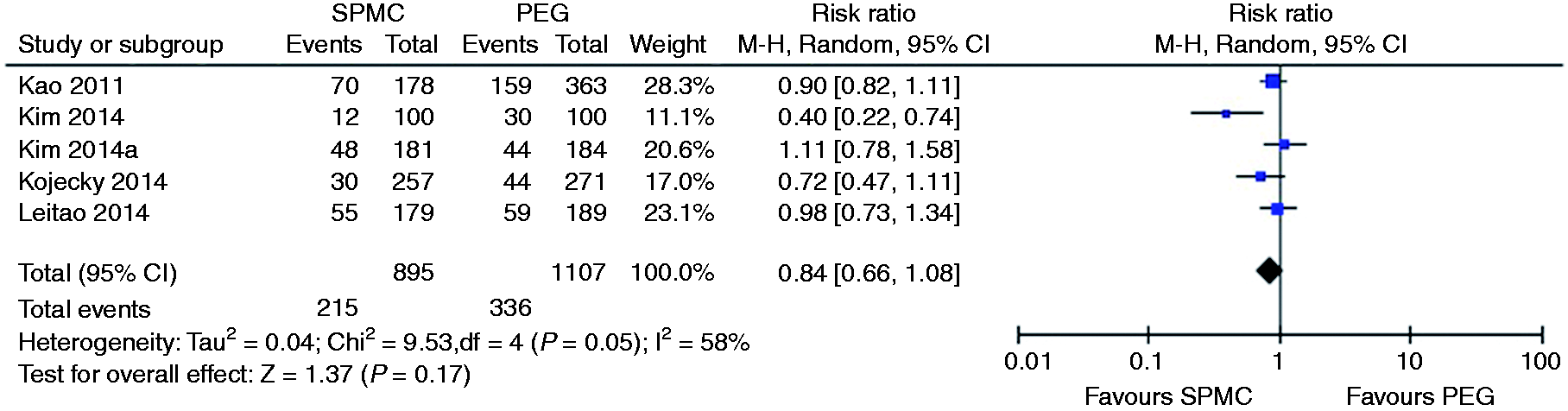
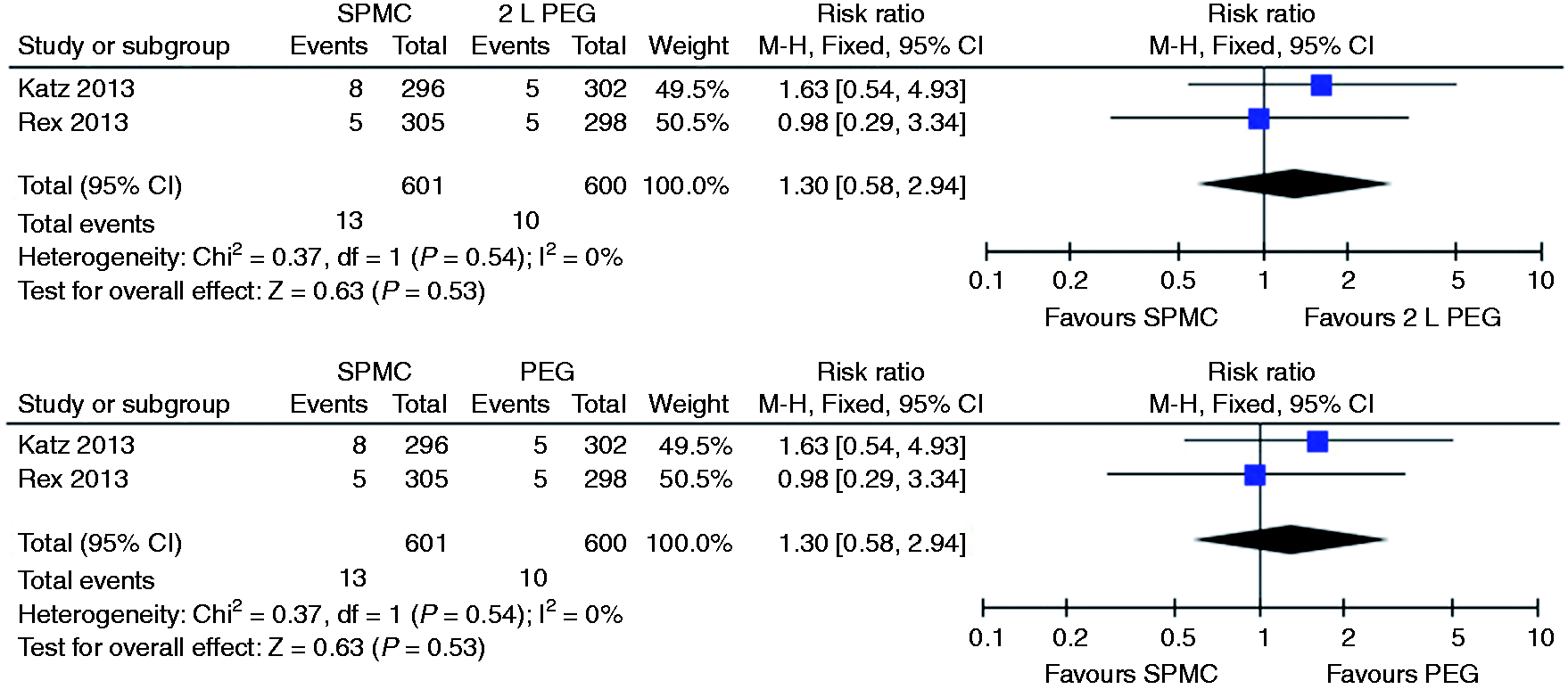
We were able to pool four studies,19,20,22,28 resulting in a small significant difference between SPMC and PEG in favour of SPMC (pooled RR 1.06; 95% CI 1.02 to 1.11). Stratifying this into SPMC compared to 4 litre PEG, no significant difference was found between the two (see Figure 3).
Relative risk for each study and pooled relative risk with 95% CI to excellent/good on the Aronchick scale for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.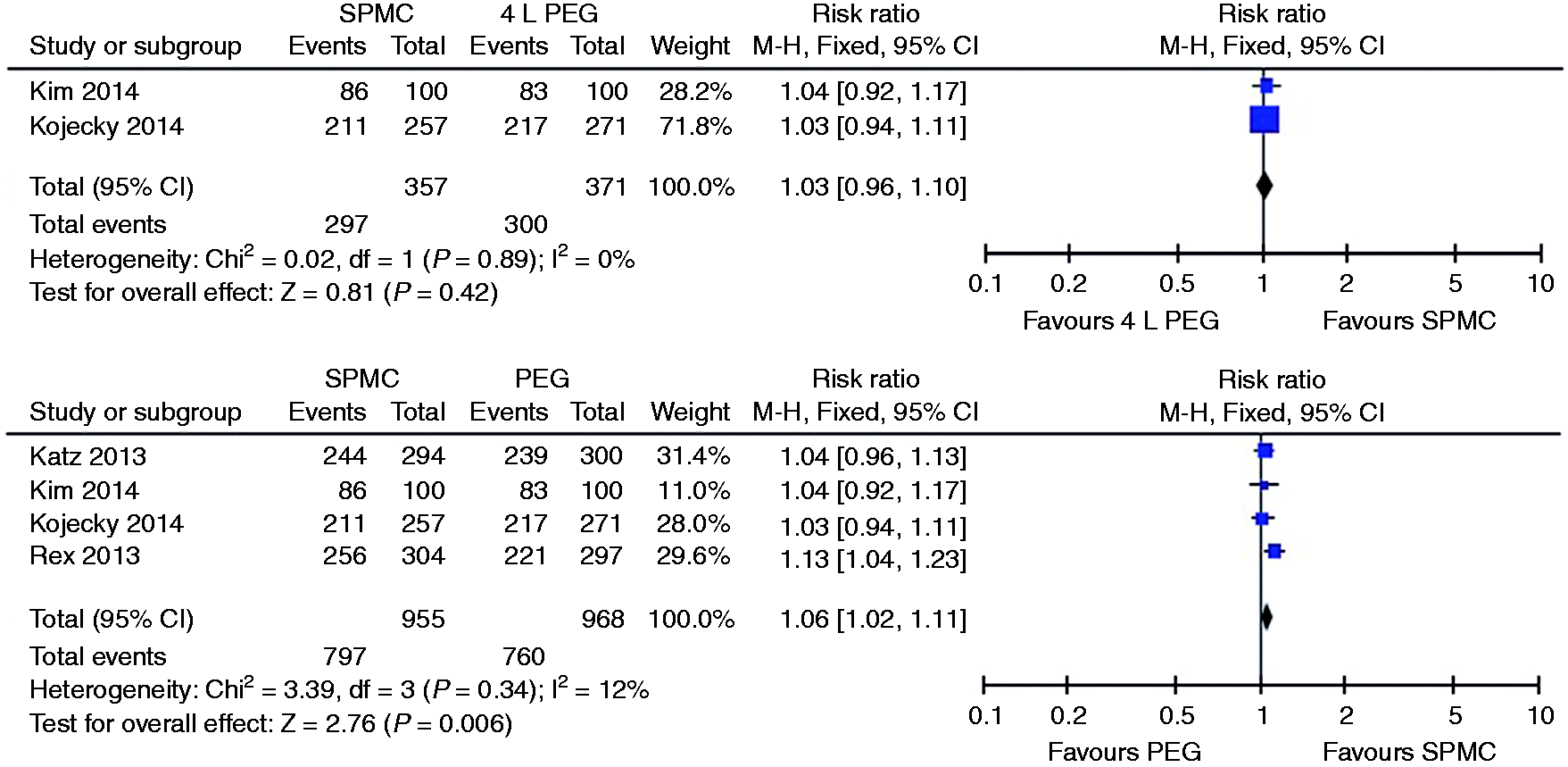
Quality of bowel preparation.

PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy; OBPS: Ottawa Bowel Preparation Scale; BBPS: Boston Bowel Preparation Scale; CI: confidence interval; IQR: interquartile range.
Quality of bowel preparation in separate colon segments
Three trials reported a significant difference in quality of bowel preparation in the right colon segment in favour of SPMC compared to PEG
28
or NaP.17,18 Eight trials found no significant difference in quality of bowel preparation in the right colon segment between SPMC and PEG18–21,23–25,27 or NaP.
23
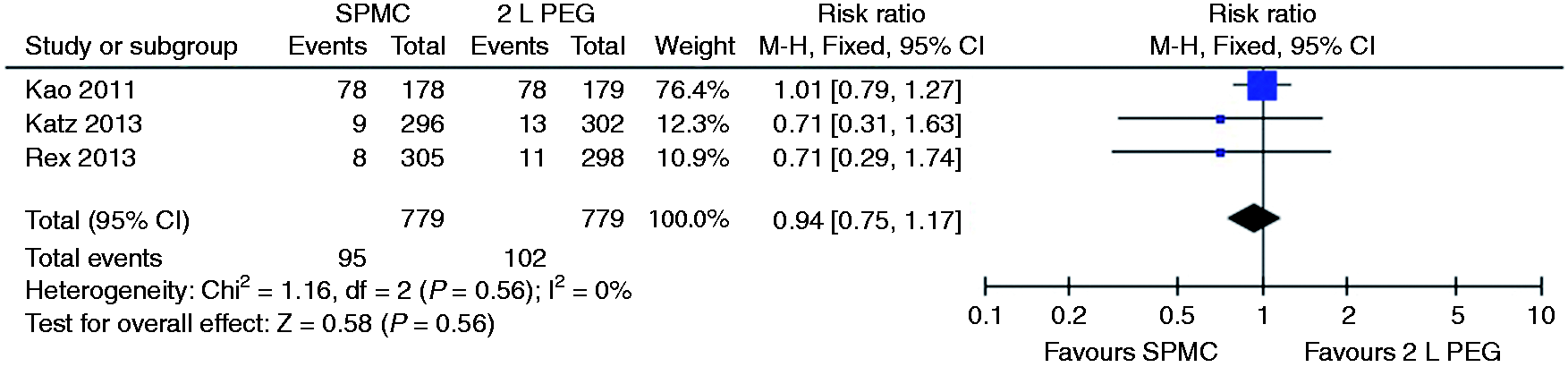
We were able to pool two studies, but no significant difference between SPMC and PEG was found (see Figure 4).
Mean difference for each study and pooled mean difference with 95% CI in quality of bowel preparation in the right colon segment on the Boston Bowel Preparation Scale for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.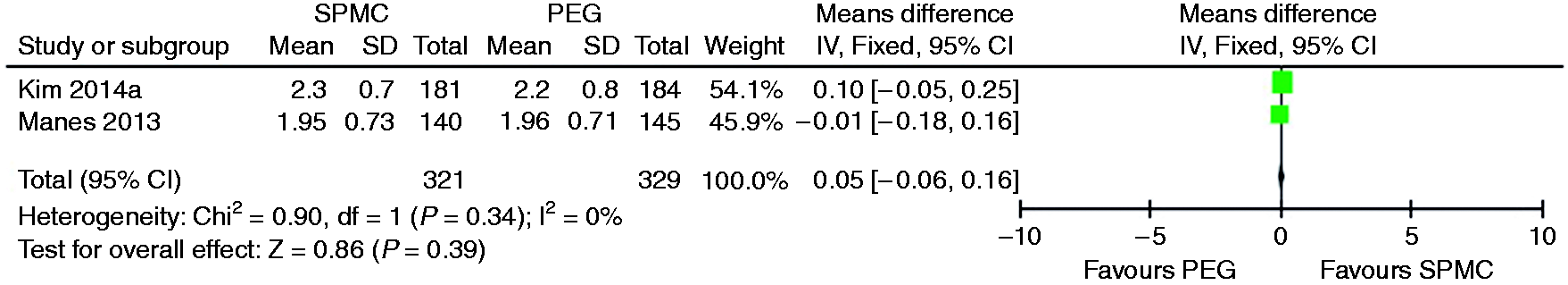
Two trials also reported a significant difference in quality of bowel preparation in the mid- and rectosigmoid colon segment in favour of SPMC compared to PEG
28
or NaP.
18
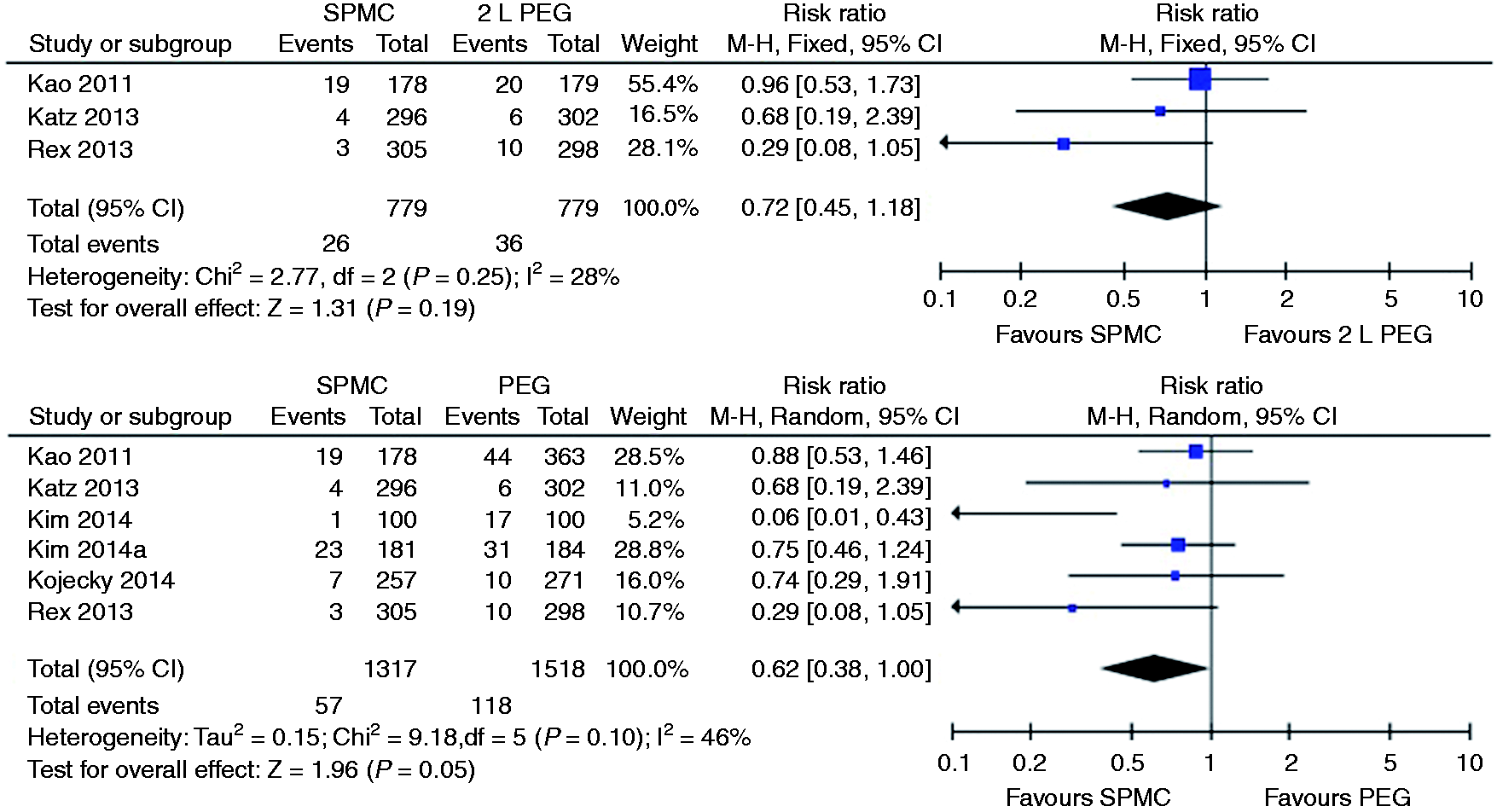
Eight trials found no significant difference in quality of bowel preparation in the mid- and rectosigmoid colon segment between SPMC and PEG18–21,23,24,27 or NaP.17,23 We were able to pool two studies, resulting in a small significant difference between SPMC and 2 litre PEG in favour of SPMC in both the mid- and rectosigmoid colon (see Figures 5 and 6).
Relative risk for each study and pooled relative risk with 95% CI to excellent/good/fair in the mid-colon segment on the Ottawa Bowel Preparation Scale for SPMC versus 2 litre PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol. Relative risk for each study and pooled relative risk with 95% CI to excellent/good/fair in the rectosigmoid colon segment on the Ottawa Bowel Preparation Scale for SPMC versus 2 litre PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.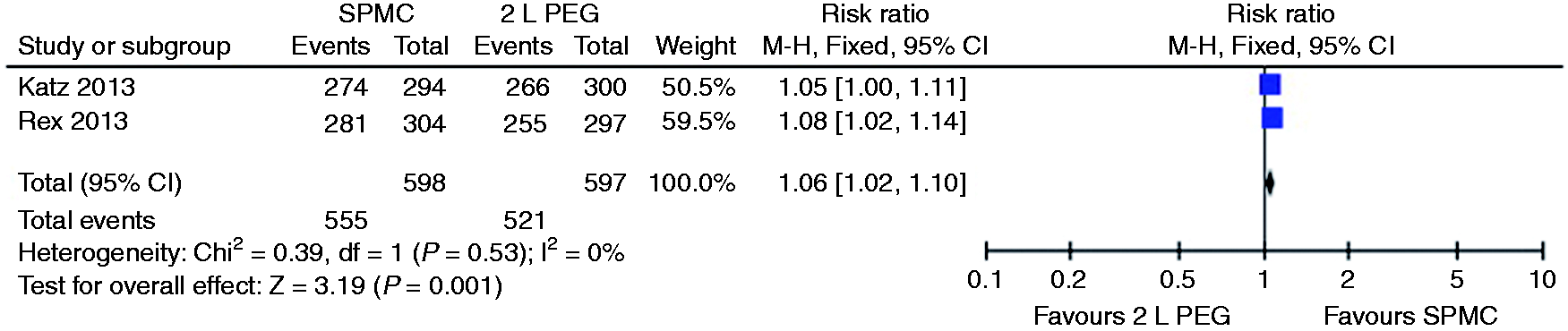
Quality of bowel preparation in each colon segment.

PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy; OBPS: Ottawa Bowel Preparation Scale; BBPS: Boston Bowel Preparation Scale.
Tolerability
Tolerability.

PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy; IQR: interquartile range.
Side effects
Side effects.

PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy; Abd.: abdominal; UNK: unknown.
Relative risk for each study and pooled relative risk with 95% CI to get nausea for SPMC versus 2 litre PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
Relative risk for each study and pooled relative risk with 95% CI to get vomiting for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
Relative risk for each study and pooled relative risk with 95% CI to get abdominal pain for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
Relative risk for each study and pooled relative risk with 95% CI to get headache for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
Relative risk for each study and pooled relative risk with 95% CI to get dizziness for SPMC versus PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
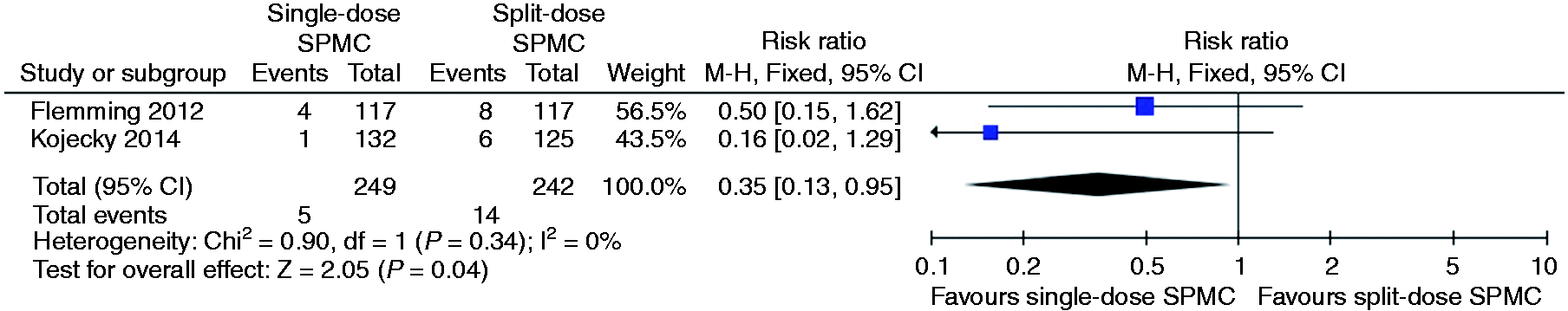
Relative risk for each study and pooled relative risk with 95% CI to get vomiting for single-dose SPMC versus split-dose SPMC. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate.
The pooled RR of the incidence of vomiting between single-dose and split-dose SPMC from two studies was in favour of single-dose SPMC (pooled RR 0.35; 95% CI 0.13 to 0.95) (see Figure 13).16,22
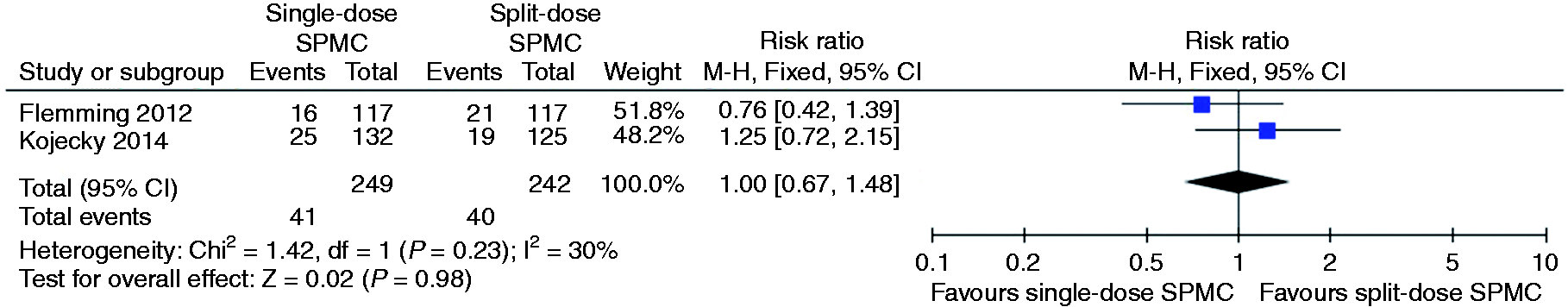
Relative risk for each study and pooled relative risk with 95% CI to get bloating for single-dose SPMC versus split-dose SPMC. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate.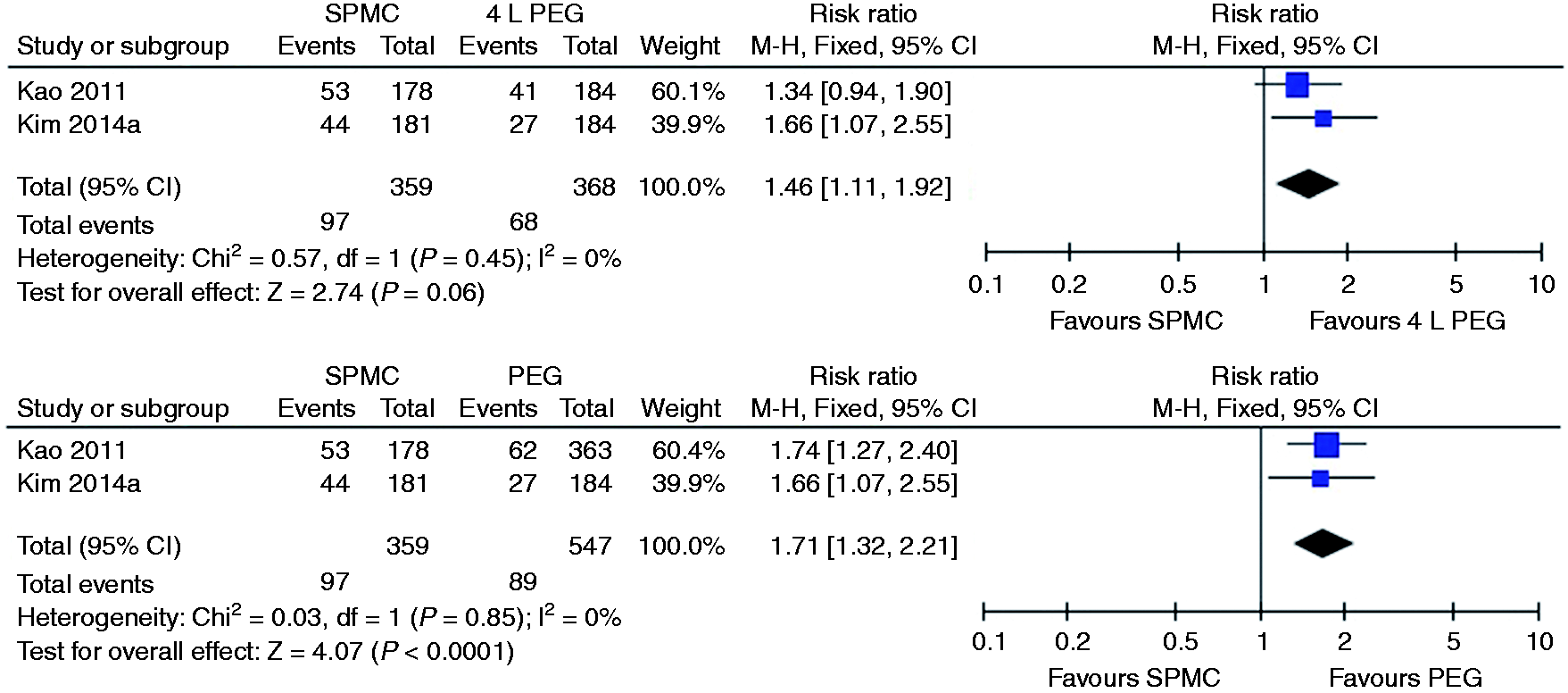
Lastly we were able to pool the two studies that measured the incidence of bloating between single-dose and split-dose SPMC, but no significant difference was found (pooled RR 1.00, 95% CI 0.67 to 1.48) (see Figure 13).16,22
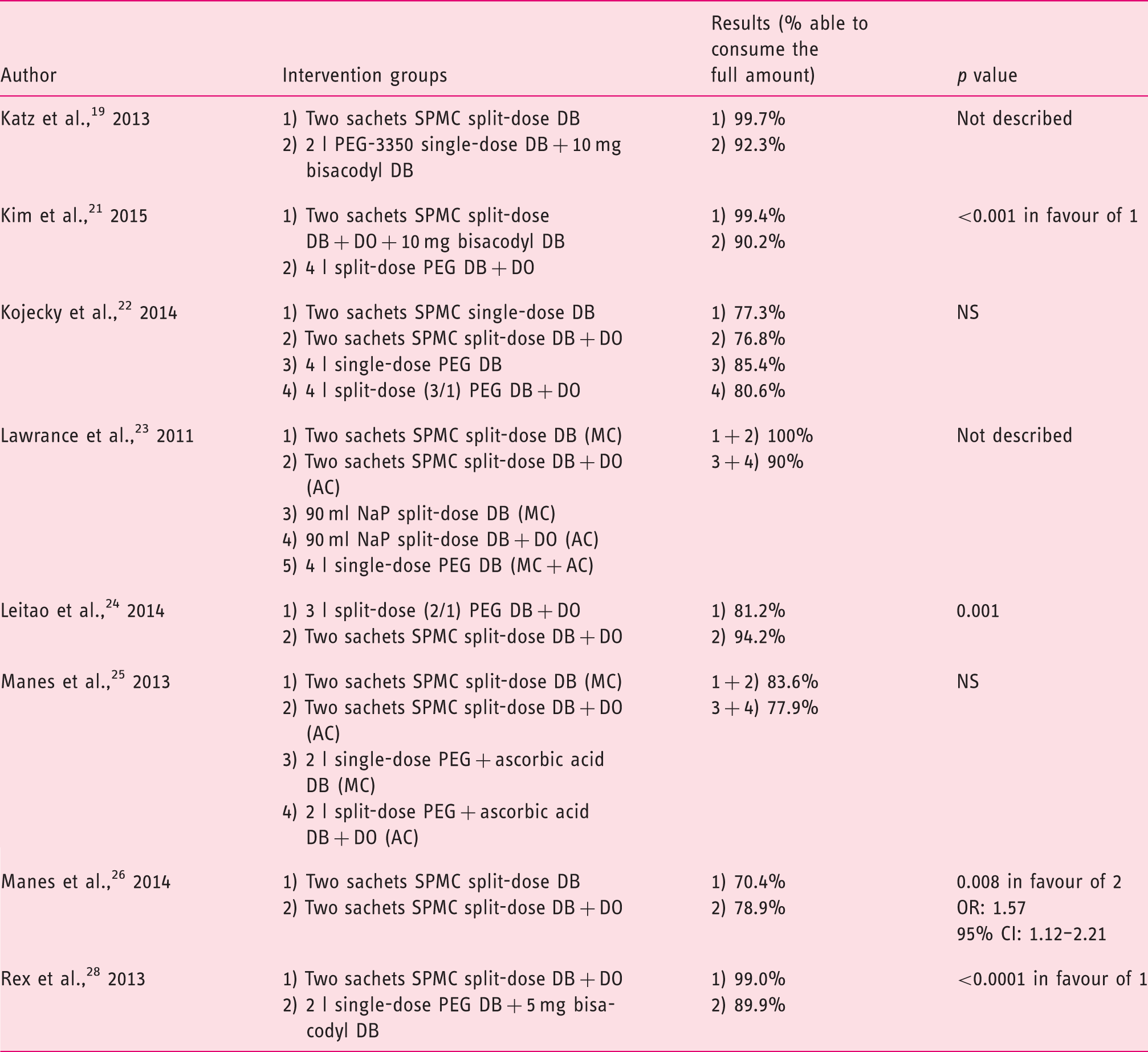
Ability to complete the preparation and willingness to repeat the same preparation in the future
Ability to complete the preparation.
PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy; OR: odds ratio; CI: confidence interval.
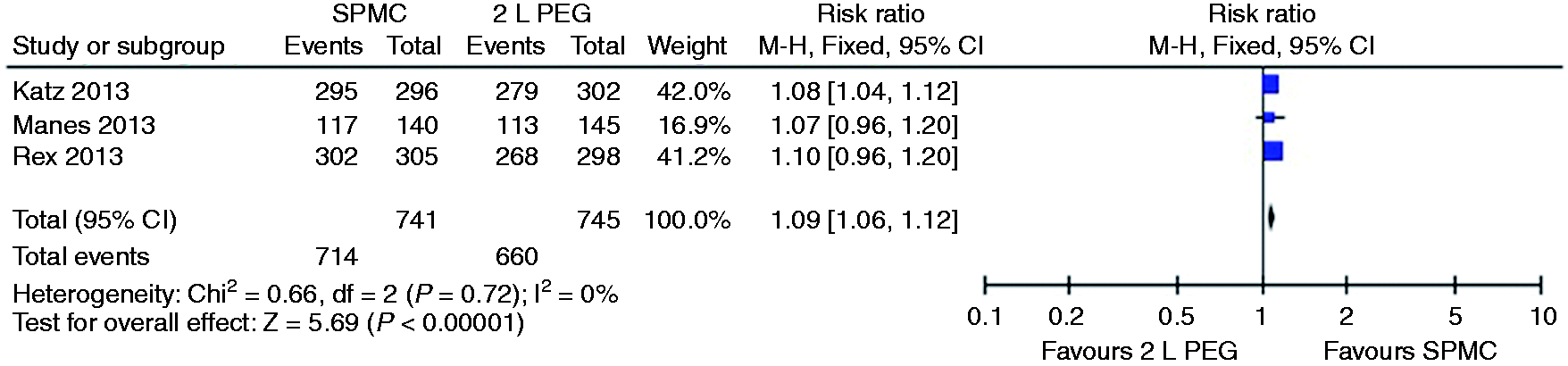
Relative risk for each study and pooled relative risk with 95% CI to complete the preparation for SPMC versus 2 litre PEG. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate; PEG: polyethylene glycol.
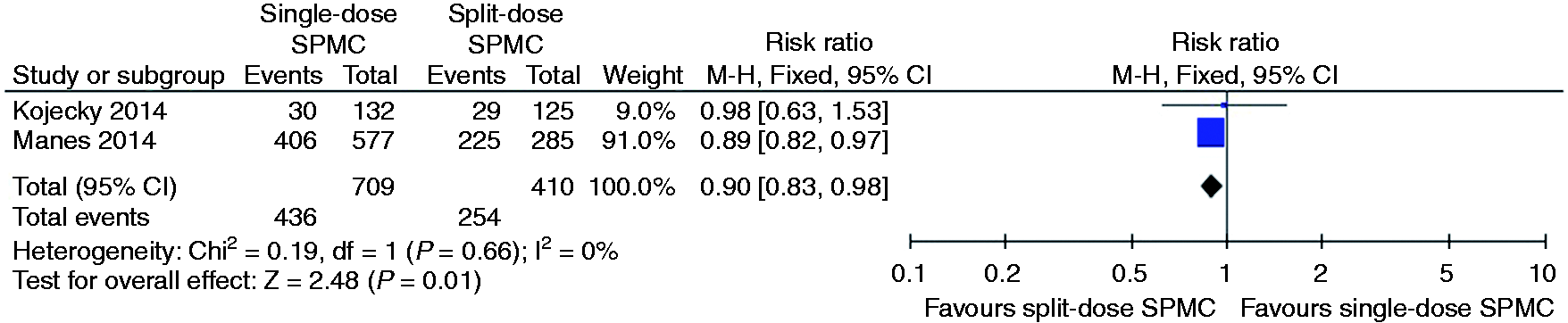
Relative risk for each study and pooled relative risk with 95% CI to complete the preparation for single-dose SPMC versus split-dose SPMC. CI: confidence interval; SPMC: sodium picosulphate with magnesium citrate.
Willingness to repeat the same preparation in the future.
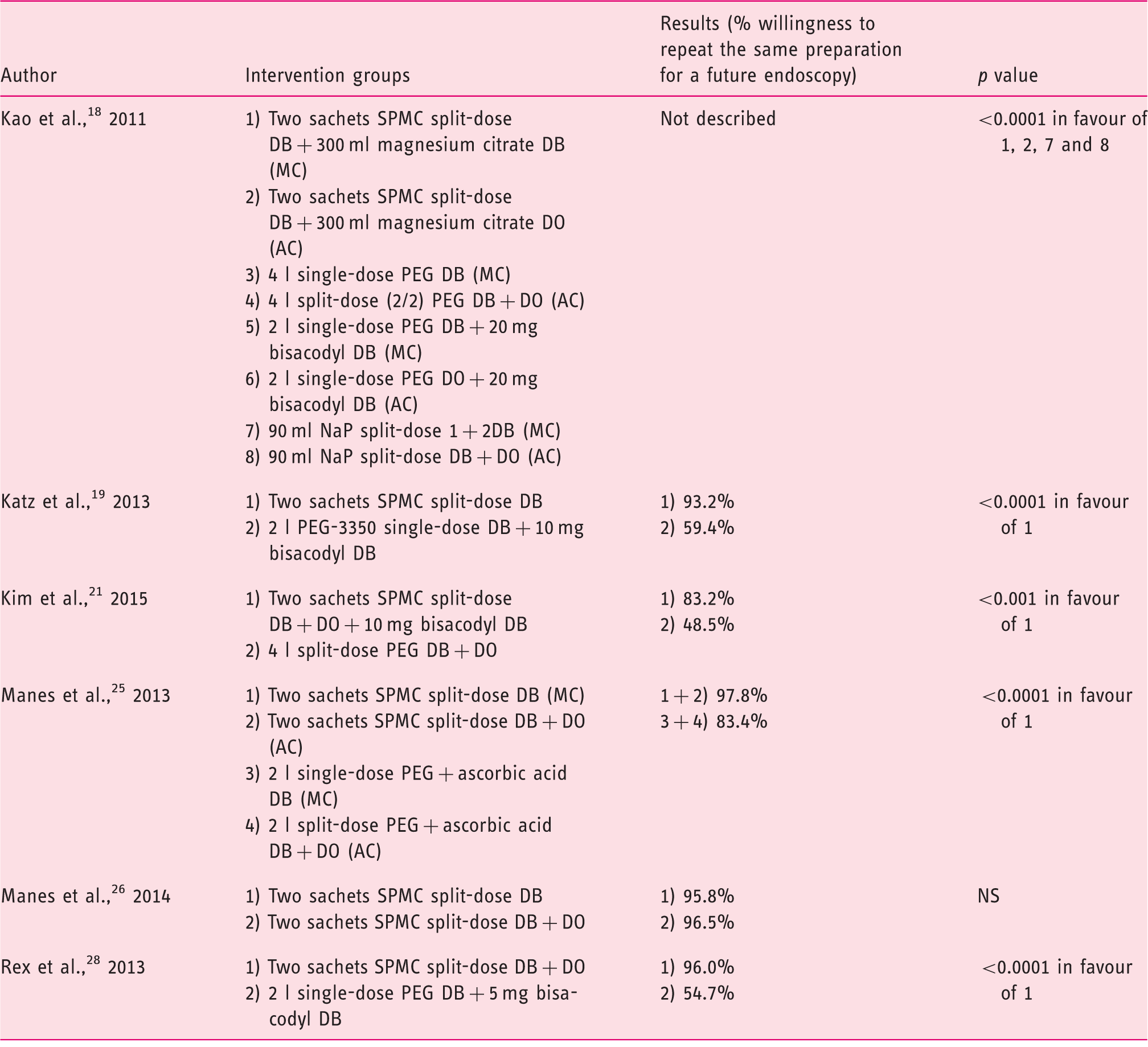
PEG: polyethylene glycol; NaP: sodium phosphate; SPMC: sodium picosulphate with magnesium citrate; DB: day before colonoscopy; DO: day of colonoscopy; MC: morning colonoscopy; AC: afternoon colonoscopy.
Discussion
This meta-analysis shows a slight significant difference in quality of bowel preparation in favour of SPMC compared to PEG. However, when stratifying the subgroup SPMC compared to 4 litre PEG the significance of this result was lost. When looking at tolerability of the bowel preparation we found that SPMC preparations are better tolerated than PEG preparations. Overall, no statistically significant differences in either quality of bowel preparation or tolerability were demonstrated between SPMC and NaP. Side effects were similar between SPMC and PEG or NaP, except for dizziness. A significant difference in the incidence of dizziness was found between SPMC and PEG in favour of PEG. Side effects were also similar between single-dose SPMC and split-dose SPMC, except for vomiting. A slight significant difference in the incidence of vomiting was found in favour of single-dose SPMC.
Our main results about quality of bowel cleansing are consistent with the conclusions in the review of Barkun et al. 2 The better tolerability was also found in the review of Hookey and Vanner, 3 Jin et al. 6 and Tan and Tjandra. 4 However, these latter found NaP more effective in bowel cleansing than sodium picosulphate or PEG. However, in the reviews by Hookey and Vanner and Tan and Tjandra, sodium picosulphate was compared instead of SPMC.
The flaw of these reviews is that they also included studies that used non-validated and subjective measurement tools for the assessment of overall quality of bowel cleansing. Our results, although generally consistent with the results in earlier reviews, have a stronger body of evidence due to the inclusion of only objective and validated measurement tools for effectiveness of bowel preparation.
Due to lack of homogeneity in measurement tools and the way of reporting outcome measures, pooling was possible only within studies that used the same measurement tools and reported the results in the same way. The meta-analysis conducted in this review provides additional information about the extent of the effect, but should always be considered carefully together with the non-pooled results.
We acknowledge several limitations in our study. First, not all databases were explored, although the main medical electronic databases were included. Second, the results we found may be influenced by the various dietary restrictions used in the RCTs. These varied from a low-fibre diet one, two or three days before colonoscopy to a clear liquid diet with or without a light breakfast or lunch, and with or without fasting for a certain period on the day before colonoscopy. A combination of both was also used. We have not considered these differences in this review because it makes it impossible to compare results. Thirdly, the findings of this review are restricted to the effectiveness and tolerability of SPMC as bowel preparation agent for colonoscopy. Safety is not included as outcome separately; however, in this review we took the side effects into account.
Several populations, such as children, elderly, patients with severe heart failure, renal insufficiency or previous colorectal surgery, were excluded in the RCTs. Therefore, we still have no knowledge about the effectiveness and tolerability of SPMC compared to PEG and NaP in these sensitive populations. Future research could therefore focus on the effectiveness and tolerability of SPMC compared to PEG and NaP in sensitive populations, and the most appropriate additional dietary restriction.
In conclusion, SPMC is equally effective as bowel preparation to NaP and little superior to PEG. Furthermore, the tolerability of SPMC is better than of PEG preparations. These findings are important for clinical practice, as the frequency of colonoscopies will increase due to recently introduced screening programs in multiple countries in Europe with more patients being exposed to bowel preparation agents. Because inability to complete the preparation contributes to poor cleansing, which may lead to incomplete visualisation of the colon, it is important that a bowel preparation agent is effective in cleansing and also tolerable for patients.2,29
SPMC has the most of these properties, and may therefore be considered as a first-choice bowel preparation agent.
Footnotes
Acknowledgements
The authors have made substantial contributions to the design of the study (IvL, IM, AE, JM, RvdH), collection of the data (IvL, IM, AE), analysis of the data (IvL, IM, AE) or drafting of the article (IvL, IM, AE, JM). All authors gave approval of the final version of this manuscript (IvL, IM, AE, JM, RvdH).
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
