Abstract
Background
Although screening colonoscopy is effective for early detection of colorectal cancer (CRC), screening rates remain low. Multiple factors are thought to be responsible for the low rates of screening colonoscopy, but bowel preparation appears to be a key deterrent. Tolerability issues with bowel preparations may lead to poor patient compliance, inadequate colon cleansing, and reduced detection of colonic polyps.
Aim
The aim of this article is to evaluate the feasibility, safety, and outcomes of minimally prepared intra-procedural hydroflush technique as an alternative to traditional bowel preparation in screening colonoscopy.
Design
We conducted a prospective feasibility study of intraprocedural hydroflush technique aided by water-jet pumps and a mechanical suction novel device following minimal bowel preparation for CRC screening.
Patients and methods
Nine procedures were performed in nine healthy individuals scheduled for routine screening colonoscopy. Preparation included mild diet restriction, laxative tablets two to three days prior to the procedure and two Fleet enemas one to two hours before the colonoscopy. The cleanliness of the colon was assessed by using the Boston bowel preparation scale. Insertion and withdrawal times were recorded.
Results
Complete colonoscopy to the cecum was performed in all patients (100%). Mean time to the cecum was 5.78 ± 2.68 minutes, and mean withdrawal time was 15.33 ± 3.94 minutes. Endoscopic visualization of the entire mucosa was achieved in all cases with no colonoscopy repeated because of inadequate preparation. Four polyps have been detected and removed in three patients. Mild mucosal erosions were seen in some areas where the suction was used extensively, similar to those that can be seen during conventional colonoscopy. Mild stiffness of the shaft of the scope was noted.
Limitations
This was an uncontrolled feasibility study of selected patients.
Conclusions
Minimally prepared colonoscopy with the intra-procedural hydroflush technique for colorectal screening is feasible. The water exchange technology compensates for the mild stiffness of the scope. This technique might increase patients’ compliance for CRC screening.
Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed cancer worldwide and the second leading cause of cancer deaths. 1 CRC largely can be prevented by the detection and removal of adenomatous polyps during colonoscopy, and survival is significantly better when CRC is diagnosed at an early stage.2,3 Despite these facts, screening rates remain low.4,5 One of the major obstacles preventing a large-scale implementation of screening programs is low patient compliance, which is limited because of embarrassment, fear of potential complications, pain and discomfort associated with the endoscopic procedure, and most of all, with bowel preparation.6,7
The ideal preparation for colonoscopy should reliably empty the colon from all fecal material, allowing optimal visualization of the entire colonic mucosa. Currently, standard polyethylene glycol (PEG) solutions and sodium phosphate (NaP) compounds are the most frequently used preparations; however, these solutions are often taken incompletely owing to the low palatability and the high volume of liquids required, which induce nausea and vomiting, with negative consequences in terms of compliance and colon cleansing.8–10
ClearPath® (Easy-Glide, Kfar Truman, Israel) is a novel device that has been developed recently and can be mounted on a conventional endoscope for washing the colon during the endoscopic procedure and removing a large amount of waste simultaneously. In a preliminary pilot feasibility study in a porcine model, we recently described the safety and efficacy of this intra-procedural cleaning device in partly prepared porcine colons. 11
The current study is a proof-of-concept study to evaluate the efficacy of a minimally prepared hydroflush screening colonoscopy program using the ClearPath device.
Patients and methods
Patients
The study included patients of 40–75 years of age who were scheduled to undergo colonoscopy for routine screening. Participants were excluded for history of inflammatory bowel disease, colonic resection, polyposis syndrome or radiation therapy to the abdomen or pelvis, or for significant comorbidity or active diverticulitis or suspicion of colonic stricture. All patients received an explanation of the procedure’s technique and confirmed their agreement to participate in the study.
ClearPath device
Technical description
The ClearPath device has been approved by the European Union (EU) and the United States Food and Drug Administration (FDA) to be used in all types of endoscopy. It has two main components, the control cabinet and the single-use disposable unit. The control cabinet includes a peristaltic pump, a control unit, and a pinch valve that enables control of the suction flow. The disposable element comprises two channels, one for supplying water for irrigation and one for suction. Both channels are connected to an irrigation and suction head that attaches firmly to the tip of the colonoscope (Figure 1). The ClearPath tube and head are attached to the entire length of the colonoscope with a polyurethane sleeve (Figure 2). When it is attached, the device adds approximately 6 mm to the diameter of the scope. Water for irrigation flows through four 0.6-mm nozzles in the distal head, and debris is evacuated through a single 18-mm2 cross-sectional aperture. A square-shaped nozzle positioned inside the suction orifice is triggered every time irrigation is activated and serves to clear the opening of aggregations of debris. Two slits on opposite sides of the suction orifice help to maintain the flow of air when the head is positioned against the colon wall. Both suction and the irrigation are controlled by foot pedals. A peristaltic pump controls the water flow. Irrigation is used at a pressure of up to two bars for cleaning, and irrigation pulses are limited to 2.5 seconds by the control unit. Suction is not time limited, but rather halted for 0.08 seconds every one second for the purpose of shaking off any debris that causes clogging along the suction line.
A view of the irrigation and suction head located at the distal tip of the tube. The four irrigation nozzles can be seen. The two slits on either side of the suction opening help to maintain airflow even when the device head is positioned against the wall of the colon. A view of the ClearPath® tube and head attached to the entire length of the colonoscope with a polyurethane sleeve.

Procedure
Subjects fulfilling the criteria for screening colonoscopy were offered to undergo the mini-preparation colonoscopy procedure. The preparation for the procedure included mild diet restriction two to three days prior to the procedure, two laxative tablets (Bisacodil) before sleeping for two to three days and one to two sodium phosphate enemas (Fleet enemas) just before the colonoscopy.
Colonoscopy was performed in accordance with the recommendations of the American Society for Gastrointestinal Endoscopy (ASGE) guidelines. 12 The bowel flushing operation was carried out by the examining physician while performing the colonoscopy. The device was inserted and advanced to the cecum while irrigation and suction are controlled by foot pedals. The rinse water was at room temperature and the mean volume was around 500 ml per procedure. Insertion and withdrawal times were recorded.
Efficacy of bowel cleansing
A clear attempt to clean the entire colonic surface area was performed. The adequacy of bowel preparation was scored by the endoscopist on withdrawal of the endoscope, using the Boston bowel preparation scale: 13 Each of the three segments of the colon (right, including cecum and ascending colon; transverse, including hepatic and splenic flexures; and left, including descending colon, sigmoid and rectum) is given a score from 0 to 3 defined as follows: 0 = unprepared colon segment with mucosa not seen due to solid stool that cannot be cleared; 1 = portion of mucosa of the colon segment seen, but other areas of the colon segment not well seen due to staining, residual stool and/or opaque liquid; 2 = minor amount of residual staining, small fragments of stool and/or opaque liquid, but mucosa of colon segment seen well; 3 = entire mucosa of colon segment seen well with no residual staining, small fragments of stool or opaque liquid. Each of the three segment scores is then summed for a total score of 0–9, where 0 is unprepared and 9 is entirely clean. Normal sedation doses of mediazoloam (3–10 mg) and phentanyl (0.05–0.10 mg) were used.
SAS software (SAS Institute, Cary, NC, USA) was used for performance of data analyses. Continuous variables were reported as mean ± SD and categorical variables as percentages. Two-sided t-test was used to compare the means of continuous variables in the two groups, and Chi-square test was used to compare the categorical variables. A p value <0.05 was considered statistically significant.
Results
Nine (eight male) healthy participants, age 44–64 years, underwent screening colonoscopy in November 2011. The colonoscopies were performed by one expert gastroenterologist (NA) using the ClearPath device.
Demographic characteristics and procedural outcomes (measurements of bowel preparation scores and procedural time) in nine patients
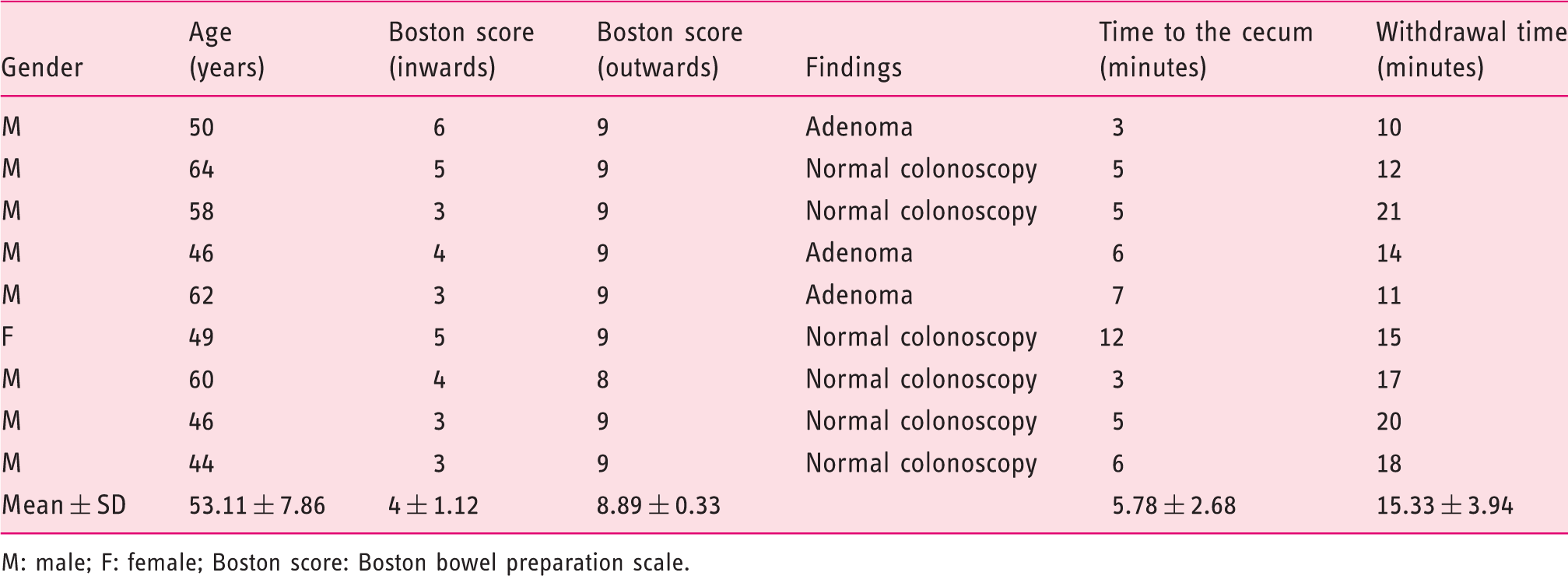
M: male; F: female; Boston score: Boston bowel preparation scale.
Cleanliness of the colon on the way in and the way out is summarized in Table 1. The Boston score on the way in was 4 ± 1.12 and 8.89 ± 0.33 on the way out. A complete visualization of the entire mucosa was achieved in all the cases.
Mild mucosal erosions were seen in some areas where the suction was used extensively, similar to those that can be seen during conventional colonoscopy. Mild stiffness of the shaft of the scope was noted. No notable clogging of the suction tubing was encountered during any of the procedures.
Discussion
In the present study, a proof of concept of mini-preparation screening colonoscopy using the ClearPath device was shown in nine healthy individuals undergoing screening colonoscopy. We have shown previously in an animal study that the ClearPath device is a simple and safe technique for intra-procedural colon cleaning owing to its highly effective irrigation and suction capabilities, enabling a clear and thorough inspection of the entire mucosa. This device has potential efficacy in several scenarios. It can be used in any case where there is a suspicion that the patient was not prepared properly, such as in the case of previous poorly prepared initial colonoscopy, or in the case of an urgent endoscopic examination when a standard full preparation of the bowel could not be performed. It also has potential in a case of gastrointestinal bleeding when a large amount of bleeding might interfere with identifying the source of bleeding. For the latter indication, a similar method has been described recently using a similar device with significantly lower flushing pressure and without a separate suction channel. 14 In the present study we present another potential use of this device, which is primarily as a mini-preparation procedure. The main advantage of this method is the reduction in the amount of solution that has to be consumed by the patients and as a result the significant reduction of the patients’ inconvenience. Since patient tolerance of the colonoscopy bowel preparation-regimen affects patient compliance and henceforth willingness to undergo repeated examinations, the currently suggested method that uses only laxative pills and enemas without the need for drinking large amounts of non-palatable liquids might undoubtedly increase patients’ compliance and promote adherence to CRC screening programs.
A potential disadvantage of the ClearPath is that is an externally attached device that slightly increases the diameter and the rigidity of the colonoscope; however, it did not actually affect the maneuverability during insertion of the colonoscope to the extent that the examination could not be performed with ease. Moreover, it has an advantage of employing the water exchange technique, that is, allowing simultaneously instillation of copious amounts of water and at the same time performing rigorous suction and by this way facilitating the sliding of the colonoscope inside the colon. Indeed, a technique of water immersion has been previously described that led to enhanced cecal intubation, shorter time to reach the cecum, and less abdominal discomfort.15,16
Gastroenterologists certainly prefer to perform colonoscopy following complete preparation of the bowel. Thus, the method described, herein, does not pretend to replace conventional bowel preparation methods. However, this method may be used as an alternative method when the patients are reluctant to undergo the preparation procedure, or when they cannot, like in urgent procedures. It can also be helpful as an additive tool for cleaning the bowel in patients prone to non-ideal preparation, e.g. previous failures, severe constipation, and hospitalized elderly patients who are not able to drink sufficient liquid.
In summary, in this study, we have established the efficacy and safety of the ClearPath device for bowel cleaning during mini-preparation screening colonoscopy. This might improve patients’ acceptance and tolerability and increase their compliance with CRC screening programs, and may be an effective solution in cases where full preparation is not possible to carry out.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
