Abstract
Background
The Crohn’s and Ulcerative Colitis questionnaire (CUCQ) has previously been validated in patients with mild to moderate Crohn’s and ulcerative colitis (UC). The aim of this study was to validate the tool in patients with acute severe UC.
Methods
We undertook a validation of the CUCQ in patients recruited to the COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial (CONSTRUCT). We carried out psychometric analysis to examine the underlying dimensions of the scale, internal consistency and construct validity. We carried out stepwise regression to examine which items accounted for the greatest variance in the scale.
Results
We obtained complete data for 270 patients. The internal consistency of the CUCQ was excellent (Cronbach’s alpha > 0.8). The CUCQ scores achieved significant correlations with two generic quality of life scales (SF-12 and EQ-5D), demonstrating good construct validity. Stepwise regression identified 16 items that accounted for greater than 95% of the variance of the CUCQ. Only three of the eight items selected for a short form in mild to moderate patients were selected for patients with acute severe UC.
Conclusions
The CUCQ demonstrated good validity in our sample of acute severe UC patients. Stepwise regression identified potential to shorten the tool, but that different items would be selected compared with less severe patients. If the tool is to be applied across the spectrum of disease it would be more appropriate to use the full 32 items in the scale. Further work to explore test-retest is required in acute patients.
Keywords
Introduction
Ulcerative colitis (UC) is a chronic incurable relapsing inflammatory disorder which presents with a multitude of symptoms impairing patient quality of life (QoL).1,2 The symptoms associated with UC are unpredictable and there can be significant variation in symptoms both over time within the same patient, and with different patients.
Measuring QoL in UC patients is important in order to assess changes in the patients’ condition over time and following treatment, and also to gain an insight into patient perception of their condition and how this compares with clinical or objective outcomes.
The aim of the COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial (CONSTRUCT) was to compare the clinical and cost effectiveness of ciclosporin and infliximab in treating steroid-resistant acute severe UC.3,4 Recent findings from other investigators illustrated no difference between cicolpsorin and infliximab in their primary outcome of treatment failures, but they did not report QoL of patients following treatment. 5 As the National Institute for Health and Care Excellence (NICE) have described that the ultimate criteria for interventions in healthcare are effectiveness and cost effectiveness in improving the survival and QoL of patients over extended periods, in undertaking the CONSTRUCT trial we chose a patient-reported outcome measure as the basis for our primary outcome measure.
Preliminary validation of the Crohn’s and Ulcerative Colitis questionnaire (CUCQ) in patients with mild to moderate disease confirmed that it met essential psychometric criteria. 6 This paper reports on the validation of the CUCQ within the context of the CONSTRUCT trial in patients with acute severe UC.3,4
Methods
We used the standard psychometric approaches for validation of the CUCQ as outlined by Streiner et al. 7 For this validation we undertook: principal components analysis to examine the underlying dimensions of the scale; calculation of Cronbach’s alpha to test the internal consistency of the scale; correlations with two generic QoL scales (SF-12 and EQ-5D) to evaluate the construct validity of the scale; and stepwise regression to explore which items contributed most to the scale. Details of the item-generation process, piloting and initial validation of the CUCQ in a sample of mild to moderate patients have previously been reported. 6
Details of CUCQ scoring
We calculated scores for the CUCQ as follows.
We scored questions with four responses as 0, 1, 2 or 3 in ascending severity. We scored questions with responses between 0 and 14 days as the actual value. We reversed the scoring of questions with wording in the reverse direction (Q7, Q22 and Q32) to code all questions in the same direction. We rescaled questions between 0 and 1 by dividing actual responses by their maximum score (3 or 14). We calculated total CUCQ scores by summing all valid responses and dividing by the number of completed questions.
The lower the CUCQ the better the respondent’s health.
We calculated the CUCQ scores only when the patient had responded to at least 75% of the questions, i.e. 24/32. If participants had completed fewer than 75% of the questions, we treated the total CUCQ score as missing. We gave equal weight to each question of the 32 questions.
Validation of the CUCQ in the CONSTRUCT randomised controlled trial (RCT)
We undertook validation of the CUCQ on the CONSTRUCT3,4 trial sample. We conducted psychometric analysis of the CUCQ in the following way.
We examined the 32 sets of response frequencies for floor or ceiling effects. We calculated the Kaiser-Meyer-Olkin measure of adequacy (KMO) and Bartlett’s test to judge whether principal component analysis was appropriate. We calculated Cronbach’s alpha (which should exceed 0.7 for good internal consistency). We calculated item-total correlations for each question (which should exceed 0.2 for good homogeneity). We undertook principal components analysis to assess the underlying structure; we considered factors important if their eigenvalues were clearly greater than 1, and individual questions as useful if their factor loadings exceeded 0.4. We assessed the construct validity of the scale by examining the Pearson correlation between the CUCQ and two generic quality-of-life questionnaires – EQ-5D and SF-12.
Ethics
We received ethical approval from the Research Ethics Committee for Wales (Ref 08/MRE09/42); National Health Service (NHS) Research & Development (R&D) approval from each participating Trust or Health Board; and Medicine and Healthcare Regulatory Agency (MHRA) approval to undertake the CONSTRUCT trial.
Results
Patient sample
Clinical and demographic characteristics of the CONSTRUCT RCT participants.
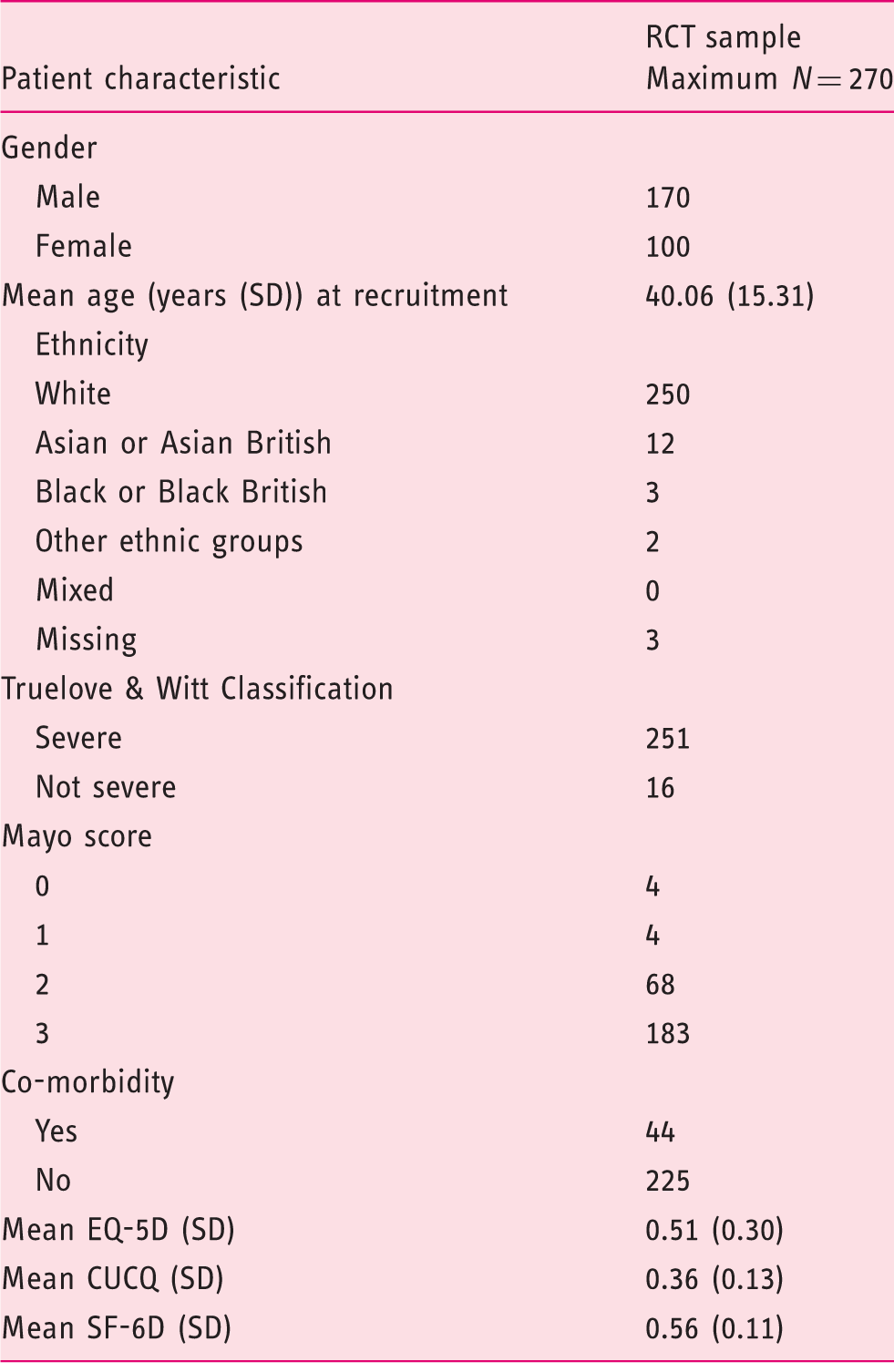
CONSTRUCT: COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial; RCT: Randomised Controlled Trial; CUCQ: Crohn’s and Ulcerative Colitis questionnaire.
Validation of the CUCQ in the CONSTRUCT RCT
We examined the data prior to undertaking principal components analysis. The KMO was 0.844; and the Bartlett’s test of Sphericity was 0.000, indicating that the data were suitable principal components analysis.
The CUCQ questions, their item total correlations and their maximum response rate (CONSTRUCT RCT sample).

CONSTRUCT: COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial; RCT: Randomised Controlled Trial; CUCQ: Crohn’s and Ulcerative Colitis questionnaire.
Principal component analysis of the CONSTRUCT CUCQ questions from the CONSTRUCT RCT sample based on a four-factor solution.

Extraction method: Principal component analysis Rotation method: Oblimin with Kaiser normalization
. Rotation converged in 15 iterations.
CONSTRUCT: COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial; RCT: Randomised Controlled Trial; CUCQ: Crohn’s and Ulcerative Colitis questionnaire.
Correlations between the CUCQ and the SF-12 MCS and PCS and EQ-5D in the CONSTRUCT RCT sample.

MCS: Mental Health Composite; PCS: Physical Health Composite; CUCQ: Crohn’s and Ulcerative Colitis questionnaire; CONSTRUCT: COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial; RCT: Randomised Controlled Trial.
Model summary of the CUCQ questions in the CONSTRUCT RCT sample.

CUCQ: Crohn’s and Ulcerative Colitis questionnaire; CONSTRUCT: COmparison of iNfliximab and ciclosporin in STeroid Resistant Ulcerative Colitis Trial; RCT: Randomised Controlled Trial.
Discussion
We previously validated the CUCQ in patients with stable or moderately active inflammatory bowel disease (IBD) 6 where it demonstrated good psychometric properties. It had not previously been applied to acute severe patients, however, so we needed to validate it on patients with severe disease. We therefore tested the validity of the CUCQ within the CONSTRUCT trial 3 on patients diagnosed with acute severe UC.
Psychometric analysis of the CUCQ in the CONSTRUCT sample of acute severe patients demonstrated excellent internal consistency (Cronbach’s alpha > 0.8). There were only a handful of questions which demonstrated item-total correlations less than 0.2, response rates of greater than 80% or ceiling effects. Principal components analysis indicated that there were four main factors with an eigenvalue of greater than 1 and which explained 42% of the variance in the data. The CUCQ scores also achieved significant correlations with both the SF-12 mental and physical component summary scores and the EQ-5D demonstrating good construct validity. Stepwise regression analysis identified that 16 questions explained greater than 95% of the variance in the CUCQ when applied to the acute severe UC patients in the CONSTRUCT RCT sample. Interestingly the questions that were identified as being the major contributors to a shorter scale were different in the acute sample to those identified in a mild to moderate sample 6 (only three of the eight questions identified for the CUCQ 8 in the mild to moderate sample were identified in the acute severe sample). This would suggest that the full 32 questions in the CUCQ would be more appropriate to use across the spectrum of disease, to ensure that the changing picture of the disease on QoL is captured.
Although initial psychometric testing of the CUCQ demonstrated good validity, more work is needed to explore the responsiveness and test-retest validity of the CUCQ in an acute severe sample. Although the CONSTRUCT RCT patients completed follow-up questionnaires, there were insufficient data to satisfactorily undertake these analyses. Further work is also needed to explore the usefulness of the individual factor scores in monitoring patient QoL.
There have been no questionnaires that have been suitable for patients who have undergone colectomy surgery. We developed an extension to the CUCQ (CUCQ+), which included 10 additional questions specifically for stoma patients, and which excluded six non-relevant questions from the CUCQ. We undertook initial piloting and testing of the CUCQ+ within CONSTRUCT but need to gather additional data to undertake a full psychometric analysis (including principal components analysis, consistency, construct validity, responsiveness and test-rest validity).
In order to be clinically applicable we aim to produce a questionnaire which could be applied across the spectrum of disease from mild to severe (including one that is applicable to patients who undergo colectomy surgery), to longitudinally assess patient disease-specific QoL over time. In order to be practical in clinical practice this questionnaire should be short, so we would aim to produce a questionnaire that captures the most important symptoms across the spectrum of disease severity. Work is already underway to recruit more patients to undertake this further validation work and development of a short combined severity form.
Footnotes
Acknowledgements
We would like to thank a number of people who helped towards the successful completion of the study: the study participants, giving so generously of their time and sharing their experiences with us, especially for completing questionnaires for up to 3.5 years: Laura Hawes, who provided useful advice from a service user perspective; the principal investigators and the research nurses, who played an invaluable role in helping to identify, recruit, randomise participants and collect data; Gaynor Demery, Jane Draper, Emma Riordan and Judy Williams, who provided administrative support to the trial; Ashley Akbari and Dr Kym Thorne, who helped develop the data collection tools and the implementation of GeneCIS; Giles Croft, Hayley Dickinson and Tracy Hughes, who supported the use of GeneCIS; Wai-Yee Cheung for her contribution to the design of the study and patient-reported outcome measures; Daphne Russell for her contribution to the design of the study; and Kathy Malinovsky and Leanne Quinn, who provided helpful guidance in the setup of CONSTRUCT.
HAH led on the patient-reported outcomes within the CONSTRUCT trial, and led on the writing of this paper. LA provided patient-reported outcomes support to the CONSTRUCT trial, and contributed to drafting this paper. AW was responsible for leading and reporting the statistical analysis of the CONSTRUCT trial data, and contributed to drafting this paper. W-YC contributed to the design of the study and patient-reported outcome measures. ACS was responsible for overall trial management of the CONSTRUCT trial, and contributed to drafting this paper. JGW conceived the idea for the CONSTRUCT trial and successfully secured funding, was the principal investigator on the CONSTRUCT trial, led on the writing of the CONSTRUCT report and contributed to drafting this paper.
The views and opinions expressed herein are those of the authors and do not necessarily reflect those of the Department of Health.
Declaration of conflicting interests
None declared.
Funding
The CONSTRUCT study (ISRCTN 22663589) was supported by the National Institute for Health Research Health Technology Assessment programme (grant number: 06/78/03).
