Abstract
Background
Treatment options for patients with opioid-induced constipation (OIC) and inadequate response to laxatives (LIR) are few.
Objective
Assess the efficacy and safety of orally administered naloxegol in patients with prospectively confirmed OIC and LIR
Methods
We analyzed pooled data from two identical randomized, double-blind, placebo-controlled, Phase 3 trials of naloxegol in patients with non-cancer pain, OIC and LIR in which naloxegol (12.5 mg,
Results
OIC response rates for the naloxegol 25-mg (
Conclusions
Naloxegol was efficacious, generally safe and well tolerated in the patients with OIC and LIR, while preserving opioid analgesia.
ClinicalTrials.gov identifiers: NCT01309841; NCT01323790
Keywords
Introduction
The prevalence of constipation in patients taking opioids for chronic non-cancer pain is 40–80%.1–3 Current treatment guidelines recommend initiation of laxative use at the inception of opioid pharmacotherapy. 4 Historically, laxatives provide suboptimal relief in 20–40% of patients with opioid-induced constipation (OIC)1,5; however, a recent survey of opioid users found that 94% of patients using laxatives routinely over a 2-week period experienced an inadequate therapeutic response. 6 Unresolved OIC may increase the overall burden of illness in patients with chronic pain undergoing opioid pharmacotherapy. 1
The greatest unmet need in patients with OIC exists in those patients with an inadequate response to laxative therapy. Conventional laxatives do not directly address the spectrum of physiologic mechanisms of OIC, and their use is limited by the potential for gastrointestinal-related side effects and electrolyte imbalances, dehydration and bowel obstruction. 5 Furthermore, the efficacy of conventional laxatives has never been conclusively demonstrated in robust, double-blind, placebo-controlled trials. Consequently, unlike prescription pharmacotherapies (naloxegol, lubiprostone or methylnaltrexone bromide),7–9 no conventional laxative possesses an approved indication for specific use in OIC. The efficacy of laxatives in patients with OIC is often suboptimal,5,6,10–12 and symptoms of constipation persist during laxative therapy in >50% of patients.5,6 Despite this, stimulants, stool softeners and osmotic laxatives are recommended as first-line laxative therapies for OIC.5,10 Although laxatives may result in improvement of specific symptoms of OIC or a brief period of relief, 13 a truly effective OIC therapy should produce rapid and consistent relief.
The clinical assessment of patients with an inadequate response to laxative therapy requires a standard definition of its characteristics. Because no standard definition exists, input and advice was solicited from an advisory board composed of gastroenterologists and palliative care specialists to provide definitions and a classification method. The ‘stool symptoms’ domain of the Patient Assessment of Constipation - Symptoms (PAC-SYM), 14 validated in patients with OIC, 15 was used as the basis for development of the ‘Stool Symptom Screener’ as a validated tool for assessment of laxative response status. 16 A patient was classified as a laxative-inadequate responder (LIR) if he/she reported using any laxatives for a minimum of 4 days within 2 weeks and had continued stool symptom ratings of moderate, severe, or very severe (in response to one or more of the four stool symptom domain questions) on the Stool Symptom Screener, used at baseline to define laxative response. 17 This definition of LIR, used in the clinical development program of naloxegol, captures stool symptoms of relevance in OIC patients.10,16
Naloxegol and methylnaltrexone are peripherally acting, µ-opioid receptor antagonists (PAMORA) that specifically target the opioid receptor mechanism responsible for OIC. 17 Methylnaltrexone (subcutaneous injection) is indicated for the treatment of OIC in patients with advanced illness and insufficient response to laxatives, or patients with chronic non-cancer pain. 8 The efficacy of methylnaltrexone (subcutaneous injection) was established in separate double-blind, randomized, placebo-controlled, Phase 3 trials.18,19 In patients with advanced medical illness who were permitted to continue their baseline laxative regimen (except ±4 hours of study drug dosing), the proportion of patients with rescue-free bowel movements was significantly higher for the methylnaltrexone group (0.15 mg/kg, alternate days) compared with placebo. 18 Similar findings for methylnaltrexone were reported in laxative-free (except for rescue medication) patients with OIC and non-cancer pain when 12 mg was administered every day or every other day, compared with placebo. 19
Naloxegol (oral tablet) is indicated for use in adult patients with OIC and chronic non-cancer pain, 9 and in adult patients with OIC and LIR in Canada 20 and the EU. 21 Naloxegol may have its greatest utility in treating OIC patients for whom conventional laxatives are inadequate. 17 In the overall patient population and in patients classified as LIR before enrollment (≥50% by trial design) in two randomized, placebo-controlled, Phase 3 trials of naloxegol, the response rates were significantly higher versus placebo in the 25-mg group for both studies and in the 12.5-mg group for one study. 17
LIR patients with OIC may experience a variety of gastrointestinal symptoms that negatively affect quality of life 6 and may be at risk for reduced management of chronic pain owing to suboptimal adherence to opioid treatment. 1 Thus, a substantial unmet medical need exists for OIC patients who do not experience adequate relief from conventional laxatives. Our objective was to demonstrate a range of positive outcomes with naloxegol in patients with prospectively confirmed OIC and documented inadequate responses to conventional laxatives.
Methods
Study design and patients
Data were pooled from the LIR populations of two identical multicenter, randomized, double-blind, placebo-controlled, Phase 3 trials conducted in the US and Europe (KODIAC-04, NCT01309841 and KODIAC-05, NCT01323790). 17 Outpatients with non-cancer pain, aged 18–84 years, receiving oral opioid medication of 30–1000 mg per day of a morphine-equivalent dose were enrolled. 17 Enrollment and randomization were designed to ensure that ≥50% of patients randomized were LIR at baseline, as previously defined. 17 As pre-specified in the study protocol, patients were further categorized as being 2X LIR if they met the above LIR definition when taking ≥2 laxative classes. Patients who were taking laxatives and did not meet the symptom criteria of moderate to very severe on the Stool Symptom Screener were classified as laxative-adequate responders (LARs). Patients who were not taking laxatives or who reported laxative use for <4 days within 2 weeks of screening were classified as laxative-unknown responders (LURs). Study patients received an electronic diary at screening to record daily the date and time of OIC symptoms, pain level, use of rescue laxatives and opioid medications for breakthrough pain.
Efficacy assessments
In both studies, the response during the 12-week treatment period, defined as having ≥3 spontaneous bowel movements (SBMs) per week and an increase from baseline of ≥1 SBM per week, for ≥9 of 12 weeks and for ≥3 of the final 4 weeks was assessed in the intent-to-treat (ITT; primary endpoint) and LIR (key secondary endpoints) populations. 17 We determined the 12-week response rates in the pooled LIR and 2X LIR populations from both Phase 3 studies. Other secondary endpoints assessed in the ITT population are also presented herein for the pooled LIR population, including response rates incorporating SBMs and symptoms; median time to first post-dose SBM; proportion of patients with first post-dose SBM within 6, 12 and 24 hours; mean number of days per week with ≥1 SBM; mean number of SBMs per week; changes from baseline in daily OIC symptoms of straining, stool consistency and the percentage of days per week with a complete SBM (CSBM); and changes from baseline in the PAC-SYM scores and Patient Assessment of Constipation - Quality of Life (PAC-QOL) scores, with improvements indicated by negative changes from baseline.14,22 Another supportive endpoint examining the proportion of patients with increases from baseline of ≥3 SBMs per week is also presented. Relevant data for individual studies are included, to show consistency of results between pooled data and data from individual studies.
Safety assessments
The type, number and frequency of adverse events (AEs) were assessed as previously described, 17 and were collated for the LIR population. Events assessed included AEs, serious AEs and AEs leading to discontinuation. AEs by system organ class and preferred term were also collated for the LIR population. Lastly, changes from baseline in the morphine-equivalent dose, Numeric Rating Scale (NRS) pain score, 23 and modified Himmelsbach opioid withdrawal score (mHOWS)24,25 were assessed. 17
Statistical analysis
In contrast to the primary analyses from the individual studies in which efficacy analyses included patients who experienced all levels of response to conventional laxative therapy,
17
here we report the results of efficacy analyses performed among LIR patients in the ITT population.
17
Safety analyses were conducted for patients in the ITT population who received ≥1 dose of the study drug. The pooled 12-week response rates and the response rates incorporating symptoms were analyzed by the Cochran-Mantel-Haenszel test, stratified by study. For the time to first post-dose SBM, treatment comparisons were conducted using the log-rank test and the median times were estimated using the Kaplan-Meier approach. Changes from baseline in mean days per week with ≥1 SBM, the mean SBMs per week, the daily OIC symptoms data (straining, stool consistency and percentage of days per week with CSBM), PAC-SYM scores and PAC-QOL scores were analyzed by mixed-model repeated measures (MMRM) with fixed effects for the baseline, treatment and treatment-time interaction. Study center was included as a random effect in all MMRM models except the PAC-QOL satisfaction domain, for which it was included as a fixed effect owing to model convergence issues.
Results
Patients
Demographic and clinical characteristics in the LIR population at baseline

Baseline was assessed during the OIC confirmation period.
Baseline was assessed at enrollment.
Calculated as the mean of the daily opioid doses (maintenance plus breakthrough) during the OIC confirmation period.
Includes headache/migraine, neuralgia, pain syndrome and other conditions.
LIR: laxative-inadequate responder; MEU: morphine-equivalent units; NRS: Numeric Rating Scale; OIC: opioid-induced constipation; PEG: polyethylene glycol; SBM: spontaneous bowel movement.
Efficacy
The OIC response rates in the pooled LIR population were higher in patients receiving naloxegol 25 mg (95% CI 1.253–2.001; Pooled response rates over Weeks 1–12 in the subpopulations of patients with LIR and 2X LIR.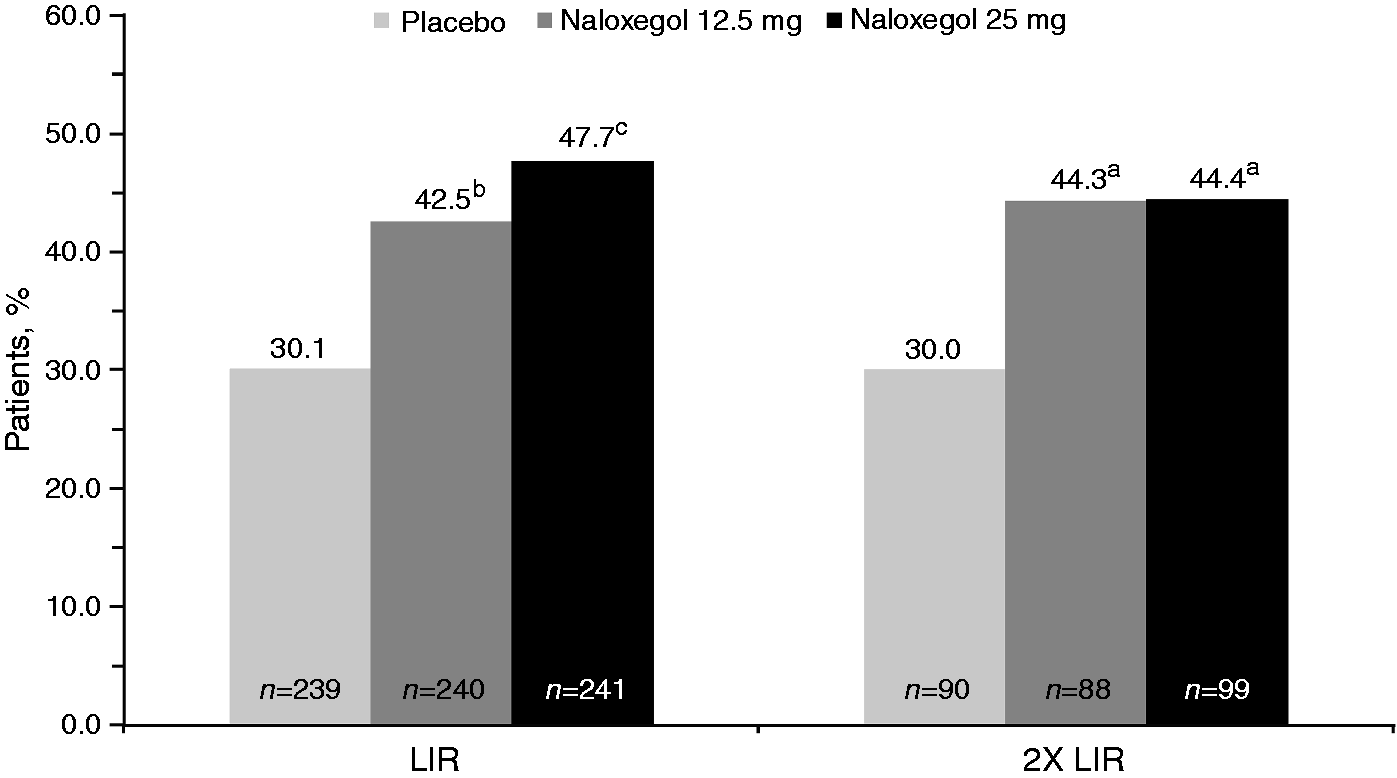
Response rates incorporating SBMs and symptoms in the pooled LIR population were also higher in patients receiving naloxegol 25 mg (95% CI 10.5–27.2; Pooled response rates incorporating SBMs and symptoms over Weeks 1–12 in LIR patients. Response rate was defined by achievement of the primary efficacy endpoint (≥3 SBMs/wk with ≥1 SBM/wk increase over baseline for ≥9 of the 12 weeks and ≥3 of the last 4 weeks of treatment); and additional symptom improvement (improvement and no worsening from baseline) in ≥1 of the following, based on mean change from baseline: straining score by ≥0.5 points, Bristol Stool Scale score by ≥1 point, mean number of days with complete SBM by ≥1 day.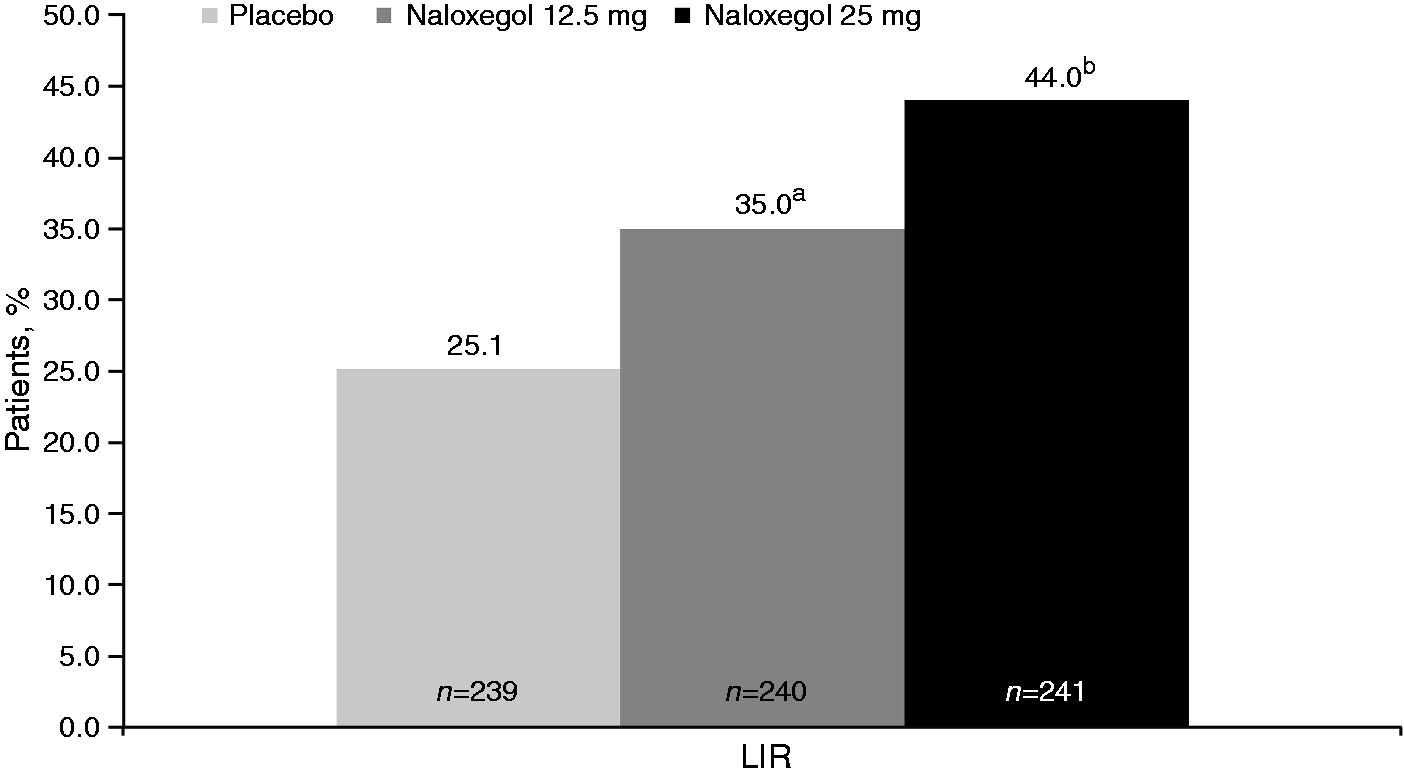
Both naloxegol doses resulted in shorter times to first post-dose SBM compared with placebo (
The proportion of LIR patients with a SBM within 6, 12 and 24 hours after the first dose is presented in Figure 3. In the pooled LIR population, the proportions of patients having an SBM within 24 hours after the first dose were 67.2%, 61.3% and 36.4% for naloxegol 25 mg, naloxegol 12.5 mg and placebo, respectively. Similar rates at 24 hours were observed in the individual studies (Supplementary Table S1).
Proportion of patients classified as LIR with first post-dose SBM within 6, 12 and 24 hours (ITT analysis set).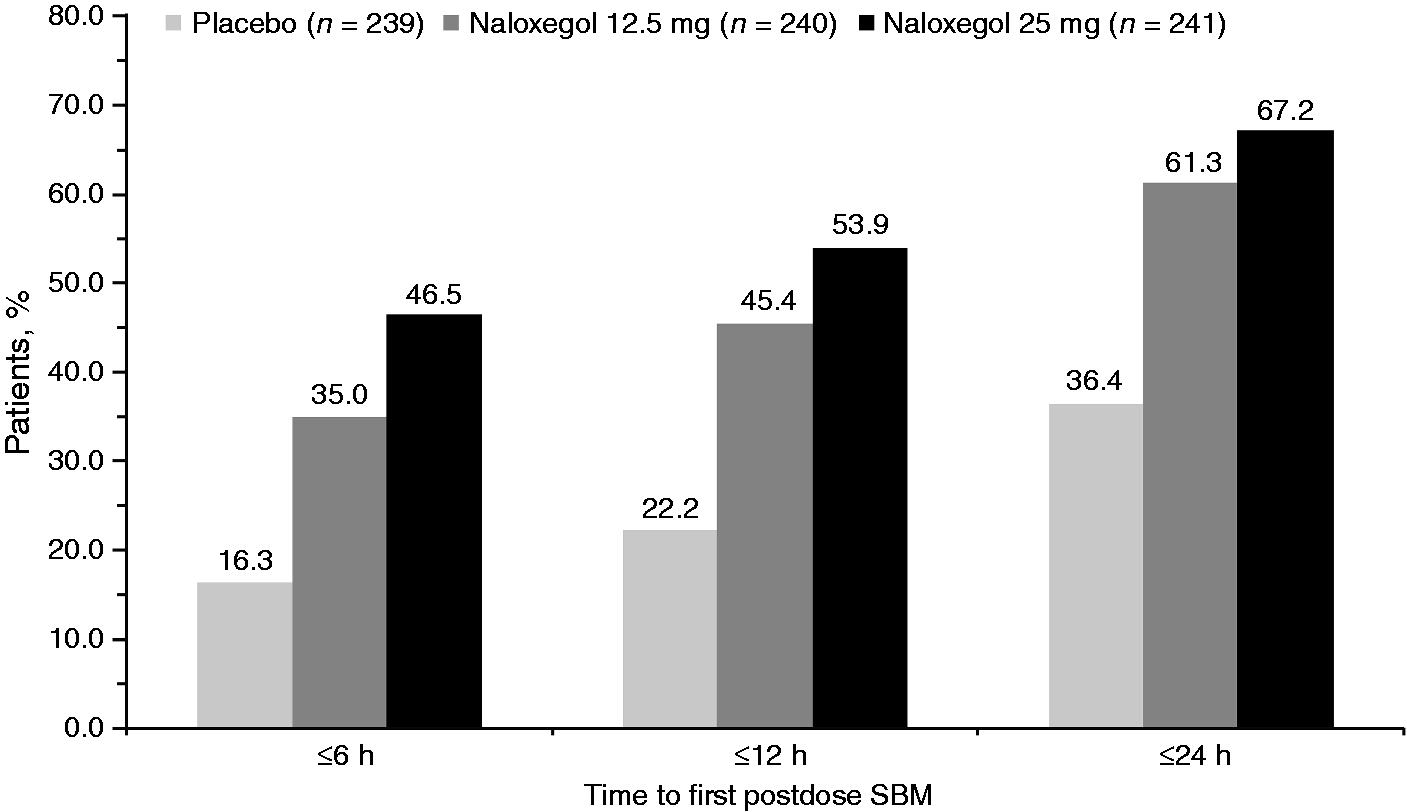
In the pooled LIR population, greater improvements in change from baseline in the mean number of days per week with ≥1 SBM during Weeks 1–12 of the study were observed for naloxegol 25 mg (
In the pooled LIR population, greater improvements in mean number of SBMs per week were observed during the Weeks 1–12 for the naloxegol 25-mg dose (
Proportion of patients with increases of ≥3 SBMs per week from baseline over Weeks 1–12, in the pooled LIR population, by baseline SBMs per week (ITT analysis set)
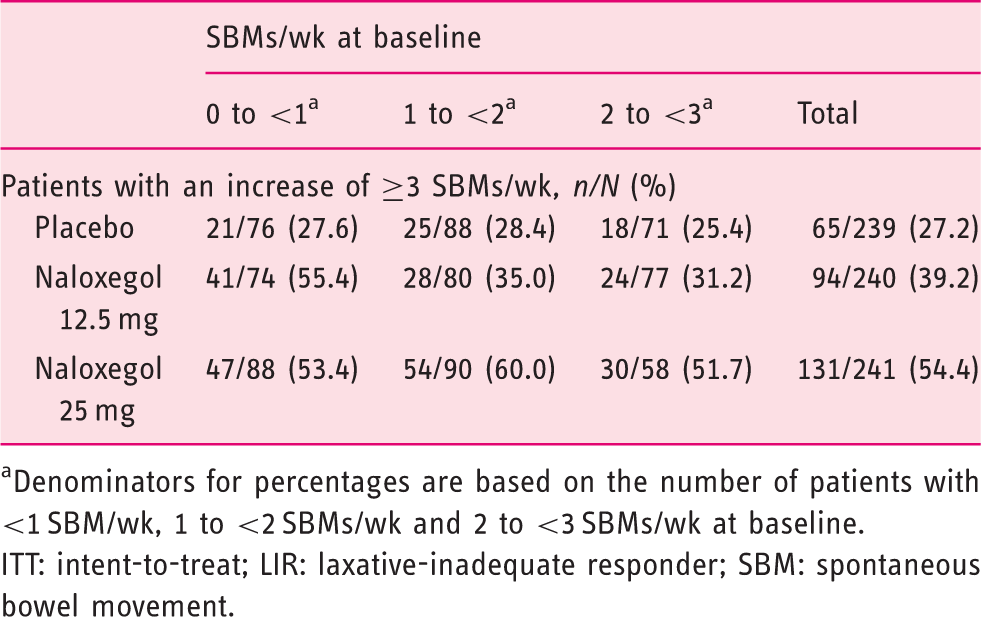
Denominators for percentages are based on the number of patients with <1 SBM/wk, 1 to <2 SBMs/wk and 2 to <3 SBMs/wk at baseline.
ITT: intent-to-treat; LIR: laxative-inadequate responder; SBM: spontaneous bowel movement.
Analysis of pooled data for daily OIC symptoms revealed improvements in straining, stool consistency, and completeness of bowel movements compared with placebo during Weeks 1–12. Greater improvements were observed for naloxegol 25 mg (
Changes from baseline in the severity of constipation symptoms, as measured by the PAC-SYM total score and subscores for rectal and stool symptoms, were greater for the naloxegol 25-mg and 12.5-mg groups, compared with placebo at Week 12 (Figure 4 and Supplementary Table S3). Changes from baseline in the severity of abdominal symptom scores were similar between naloxegol treatment groups and placebo.
Mean difference versus placebo at Week 12 in the PAC-SYM domain scores (ITT analysis set) for patients classified as LIR receiving (a) naloxegol 25 mg and (b) naloxegol 12.5 mg. Negative values indicate greater improvement with naloxegol treatment.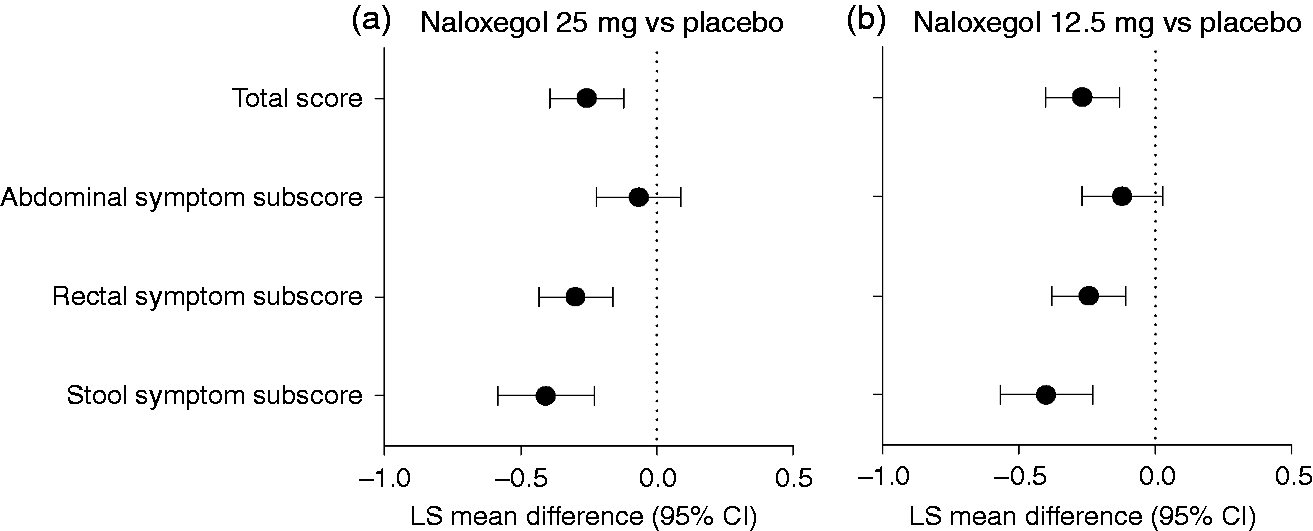
For the PAC-QOL, changes at Week 12 from baseline in the satisfaction domain were greater for the naloxegol 25-mg and 12.5-mg groups compared with placebo (LS mean difference versus placebo (95% CI): −0.50 (−0.74 to −0.26) and −0.42 (−0.66 to −0.18), respectively). LS mean changes were similar for the naloxegol 25-mg and 12.5-mg groups. Changes from baseline at Week 12 for all other PAC-QOL domains (physical discomfort, psychosocial discomfort, and worries and concerns) for the naloxegol 25-mg and 12.5-mg groups, including the total score, were comparable with placebo in the individual study populations and were not formally analyzed in the pooled population.
Safety and tolerability
Adverse event summary for the pooled LIR population (safety set)
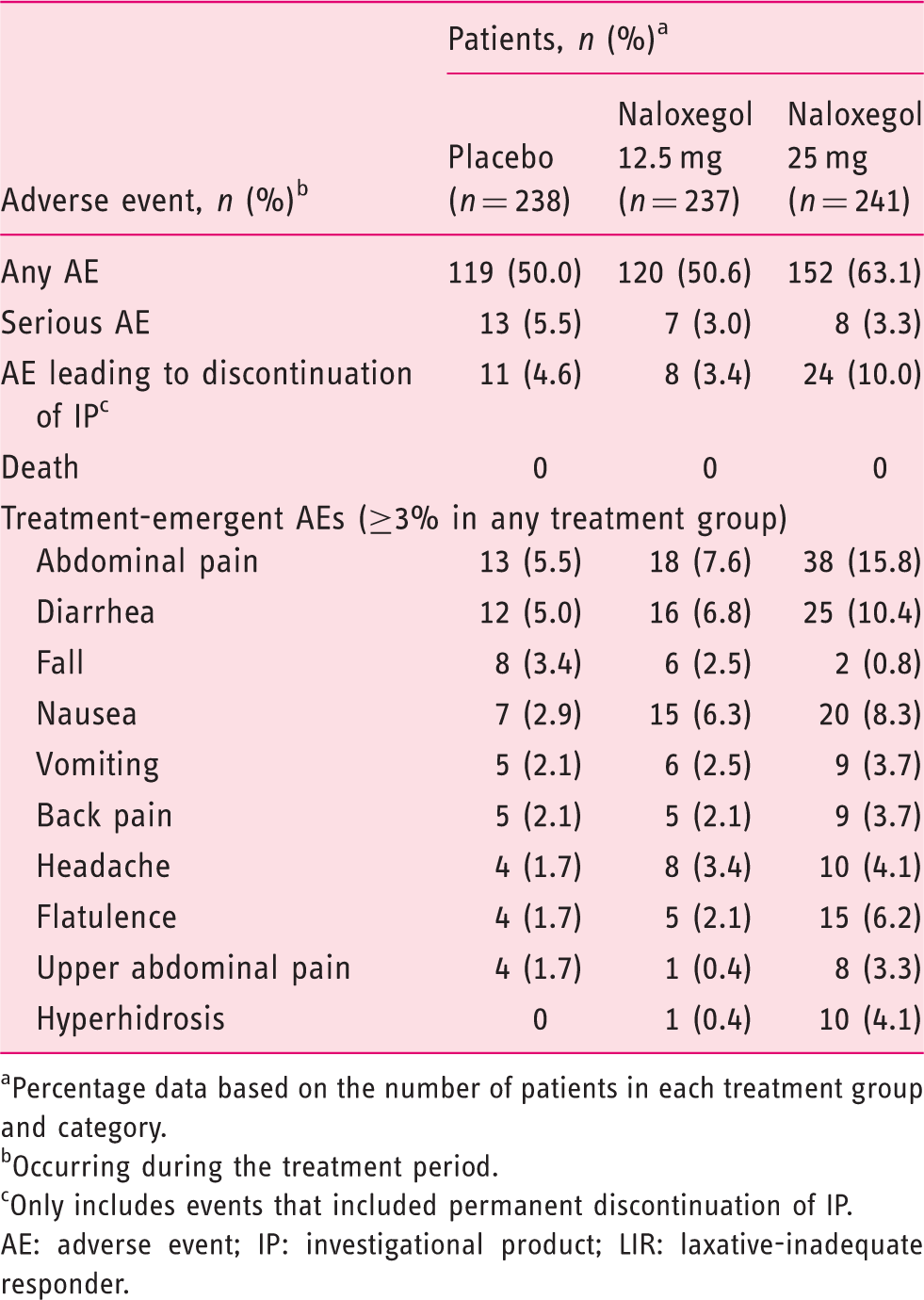
Percentage data based on the number of patients in each treatment group and category.
Occurring during the treatment period.
Only includes events that included permanent discontinuation of IP.
AE: adverse event; IP: investigational product; LIR: laxative-inadequate responder.
The most common AEs reported by LIRs receiving naloxegol were abdominal pain, diarrhea and nausea (Table 3). Flatulence, upper abdominal pain and hyperhidrosis were reported more frequently in the naloxegol 25-mg group, versus the naloxegol 12.5-mg or placebo groups (Table 3). The proportions of patients with increases from baseline in opioid dose, NRS pain score, and mHOWS were similar among treatment groups (Supplementary Table S4).
Discussion
The efficacy of naloxegol in LIR patients was previously established in two Phase 3 studies.
17
A significant treatment response in these patients was observed in KODIAC-04 for the 12.5-mg (
LIR patients experienced improvements in various measures of bowel function and OIC symptomatology, resulting from the collective pharmacokinetic and pharmacodynamic effects of naloxegol on bowel physiology. The time to first SBM was shorter for both doses in the pooled LIR and individual study populations. 17 Dose-related improvements in several measures of SBMs supported the therapeutic effects of naloxegol in LIR patients. Symptoms of straining, stool consistency and completeness of bowel movements also improved in LIR patients with OIC receiving naloxegol. The positive effect of naloxegol on the PAC-SYM total score and rectal and stool subscores and the PAC-QOL treatment satisfaction scores supported a therapeutic effect that is meaningful to patients. Furthermore, the lack of notable changes from baseline in patient-reported pain scores, opioid dose or mHOWS suggested there was negligible permeability across the blood-brain barrier.
A major strength of these investigations is the use of a standardized definition of LIR, encompassing commonly reported OIC symptoms 16 of importance to patients. The LIR assessment used a patient-friendly screening tool evaluating OIC symptomatology and laxative use, 16 building upon previous efforts to evaluate the burden of constipation in patients.14,15 The routine use of standardized definitions for OIC and its components in clinical practice should facilitate diagnosis and treatment.
A limitation of these investigations is the conservative nature of the definition used to identify LIR patients because it excludes those who were not using any laxatives within 2 weeks before screening, 17 and the lack of use may have been a result of prior ineffectiveness in these patients. Also, the small number of 2X LIR patients precludes the drawing of definitive conclusions from these data in patients who were particularly refractive to laxatives.
Conclusions
The consistency of response for naloxegol doses of 12.5 mg and 25 mg in LIR patients, together with the improvements observed in other measures of bowel function and the absence of notable changes versus placebo in pain scores, opioid dose, or opioid withdrawals in this population, suggested that the PAMORA agent naloxegol is an effective treatment option for patients with OIC who experience inadequate relief from traditional laxative therapies.
Footnotes
Acknowledgments
Editorial support was provided by Diane DeHaven-Hudkins and Erica S Wehner from Complete Healthcare Communications, Inc. (Chadds Ford, PA, USA).
Funding statement
This work was funded by AstraZeneca Pharmaceuticals LP (Wilmington, DE). This included funding for editorial support.
Conflict of interest
Author J Tack’s institution has received lecture fees from Abbott, Almirall, Aptelis, AstraZeneca, Janssen, Menarini, Novartis, Shire and Zeria; he is a consultant for AlfaWasserman, AstraZeneca, Danone, Ironwood, Janssen, Menarini, Novartis, Rhythm, Shire, Sucampo, Takeda, Theravance, Tsumura, Yuhan and Zeria. J Tack is also a board member of the Rome Foundation. He has received grant support from Novartis, Shire, Tsumura and Zeria. Authors U Diva, R Tummala and M Sostek are employees and shareholders of AstraZeneca Pharmaceuticals LP. J Lappalainen was an employee of AstraZeneca Pharmaceuticals LP at the time this work was performed and is a shareholder of AstraZeneca.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
