Abstract
Background
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder characterized by recurrent abdominal pain and/or discomfort. Probiotics have been reported to benefit IBS symptoms but the level of benefit remains quite unclear.
Objective
This study was designed to assess the benefit of
Methods
A randomized, double blind, placebo-controlled trial has been performed in 379 subjects with diagnosed IBS. Subjects were randomly supplemented with the probiotics (1000 mg) or placebo for 12 weeks. Questionnaires (gastrointestinal symptoms, stools, wellbeing, and quality of life) were completed. Primary endpoint was percentage of responders defined as having a 50% decrease in the weekly average “intestinal pain/discomfort score” for at least 4 out of the last 8 weeks of the study.
Results
There was no overall benefit of
Conclusions
In this study,
Introduction
Irritable bowel syndrome (IBS) is a ubiquitous functional gastrointestinal disorder characterized by abdominal pain or discomfort, associated with altered bowel habits, 1 reported by 10% of the general population with a female predominance. 2 It has negative impact on health-related quality of life and induces high health costs. Despite increased understanding there have been few new therapies introduced, and a combination of rare adverse effects and very low tolerance of side effects has led to the withdrawal of some effective therapies, notably alosetron and tegaserod.
Recent interest has therefore focused on drugs (e.g. lubiprostone, linaclotide) or agents like pro- and prebiotics whose actions are confined to the lumen with a perceived low likelihood of unwanted systemic effects.
There are numerous studies suggesting possible benefit of probiotics on IBS symptoms but the results are variable.3–8 This heterogeneity arises because different strains have different modes of action and also because IBS patients were often unselected and hence heterogeneous as regards bowel habit and underlying disease mechanism. Regrettably, few studies have included a mechanistic component so it is unclear exactly which of their numerous putative effects demonstrated in the laboratory are responsible for benefit in clinical practice.
Based on these preliminary data, we aimed to confirm these findings in another RCT using a larger dose of
Materials and methods
We performed a multi-center, randomized, double blind, placebo-controlled trial according to Good Clinical Practice Guidelines, Declaration of Helsinki, and approved by the Ethics Committee Ouest IV of Nantes (France). Signed written informed consent for the study was obtained from all subjects before protocol-specific procedures were carried out and subjects were informed of their right to withdraw from the study at any time.
Patients
Men and women (18–75 years of age) were recruited by French general practitioners (GPs) and in a clinical investigation center (Nantes, France). Patients were required to be diagnosed with IBS by their GP and to meet the Rome III criteria, 1 with pain or discomfort present >1 day per week; and a score for abdominal pain or discomfort >1 and <6, determined at the beginning of the study on a seven-point Likert scale for pain/discomfort scored from 0 (no pain or discomfort) to 7 (severe pain or discomfort). Lactose intolerance was excluded in subjects characterized by IBS with diarrhea (IBS-D) and mixed IBS (IBS-M) by means of a milk challenge (no symptoms after consuming 500 mL low fat milk after an overnight fast). All patients had normal full blood count and routine biochemical screen, transglutaminases IgA, fecal calprotectin, and C-reactive protein (CRP) levels. Long-standing treatments for diarrhea, laxatives, and antispasmodic drugs were not an exclusion criterion provided the doses were stable and not modified during the study. Menopausal females receiving a hormone treatment or contraception in non-menopausal female subjects must have started their treatment at least 3 months beforehand and remained on a stable dose for the entire duration of the study.
Subjects were excluded if they had chronic gastrointestinal disorders, history of gluten intolerance (celiac disease), or elevated tissue transglutaminase IgA titers, treatments likely to influence IBS in particular by modifying intestinal sensitivity or motility (antidepressants, opioids, and narcotic analgesics), antibiotic therapy in progress or prescribed in the 8 weeks before inclusion in the study, long-term treatment with analgesics or non-steroidal anti-inflammatory drugs. Subjects not willing to stop taking probiotics, prebiotics, or synbiotics in the form of dietary supplements or convenience goods were not eligible. Pregnancy, chronic alcoholism, vegetarian or vegan subjects, eating disorders such as anorexia or bulimia, and documented food allergies were also exclusion criteria.
Study design
The 12-week study included 364 subjects divided in two parallel groups. Subjects visited the clinical center at four visits (Figure 1): V0 (pre-inclusion visit), V1 (inclusion visit, allocation of the study products), V2 (4 weeks after V1, follow-up visit) and V3 (12 weeks after V1, end-of-study visit).
Study design.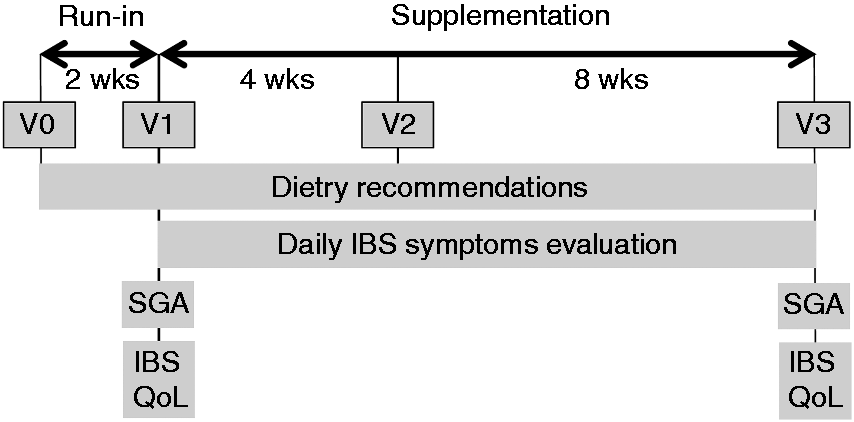
Pre-inclusion visits were planned to check inclusion and exclusion criteria and perform screening tests. Bristol stool scale (BSS) questionnaire and IBS symptoms, including abdominal pain/discomfort, were evaluated daily for a two-week period. Additionally, dietary recommendations were explained to the subject, in order to avoid eating any foods containing prebiotics and/or probiotics and any fermented milk products. At inclusion visit, investigators enrolled and randomized eligible IBS patients to consume either
Study products and compliance evaluation
Study products were presented in capsule form, packaged in plastic bottles. Active and placebo products were similar in color, form, flavor, and size. During the whole study neither the investigators nor the subjects were aware of the product tested. Subjects were instructed to consume orally two capsules of 500 mg per day in the morning, during breakfast, together with a glass of water.
The I-3856 strain is a proprietary, well characterized strain of Lesaffre, registered in the French National Collection of Cultures of Microorganisms (CNCM). The
Patients had to return all their capsule bottles, containing unconsumed capsules at V3. Compliance was calculated during the treatment period at visit V3, by comparison of the theoretical number of capsules consumed during the individual supplementation period and the number of capsules returned.
Study endpoints
The primary endpoint defined in the protocol was the percentage of responders, i.e. the percentage of patients who had an improvement of 50% of the weekly average “intestinal pain/discomfort score” compared with baseline average score (V0-V0bis) for at least 4 out of the last 8 weeks of the study (V2 to V3). This was based on previous experience suggesting
Safety endpoints
Safety of the study was assessed considering the occurrence of adverse events, safety blood parameters, and the follow-up of vital signs (blood pressure and heart rate). Adverse events were recorded throughout the study and investigators had to estimate their severity and to judge whether they were related to treatment.
Statistical considerations
Sample size was calculated to be 172 subjects per arm in order to detect a difference of 15% between active and placebo groups for the primary criteria, assuming a 47% placebo response rate, 13 using a bilateral binomial test with 80% power and alpha risk of 5%. Allowing for a 5% of drop-out, we planned to enroll a total of 364 subjects.
Data were analyzed using SAS® software version 9.3 (SAS Institute Inc., Cary, NC, USA). Results are expressed as Mean ± SD or Estimated Mean ± SE. Significance was set at p < 0.05.
Primary endpoint (percentage of responder) was analyzed using a logistic regression model. IBS symptoms and data from BSS were analyzed applying a mixed model Analysis of Covariance (ANCOVA) for repeated measurements, SGA was analyzed applying a generalized estimating equation model for correlated binary variable, and quality of life scores were analyzed using analysis of variance (ANOVA).
Primary analysis was performed on the full set of patients, in both intent-to-treat (ITT) and per protocol (PP) populations. Additional analyses were performed in subsets of IBS population (IBS-C, IBS-D, and IBS-M), based on Rome III definition. 1 Individual IBS symptoms and composite score were analyzed weekly and as area under the curve (AUC) (calculated on the 12 weeks of the study).
Results
Subjects
Baseline characteristics of subjects
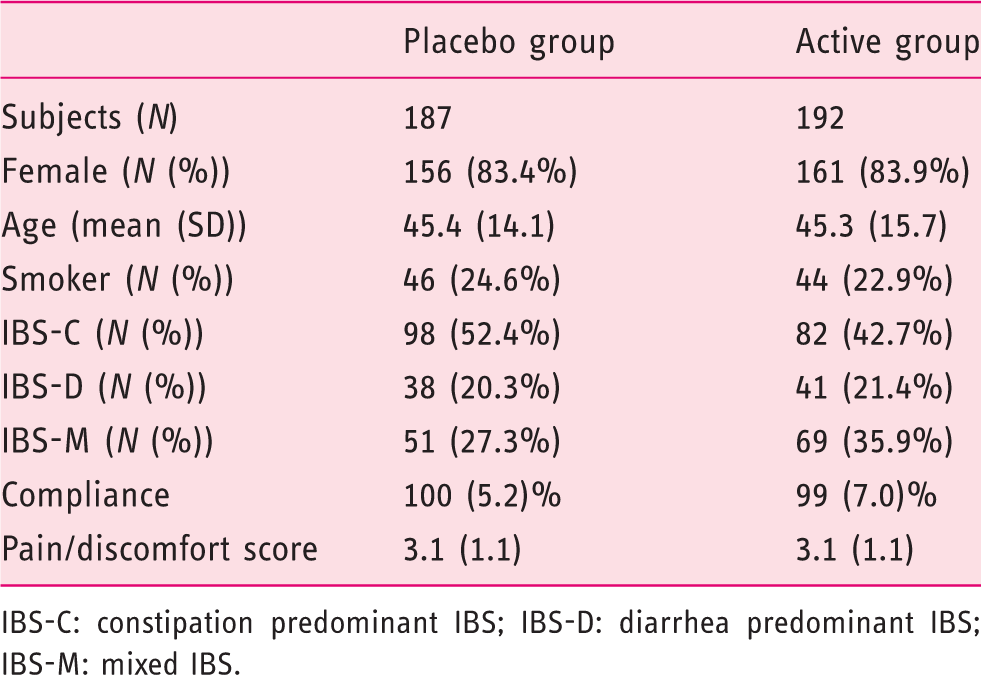
IBS-C: constipation predominant IBS; IBS-D: diarrhea predominant IBS; IBS-M: mixed IBS.
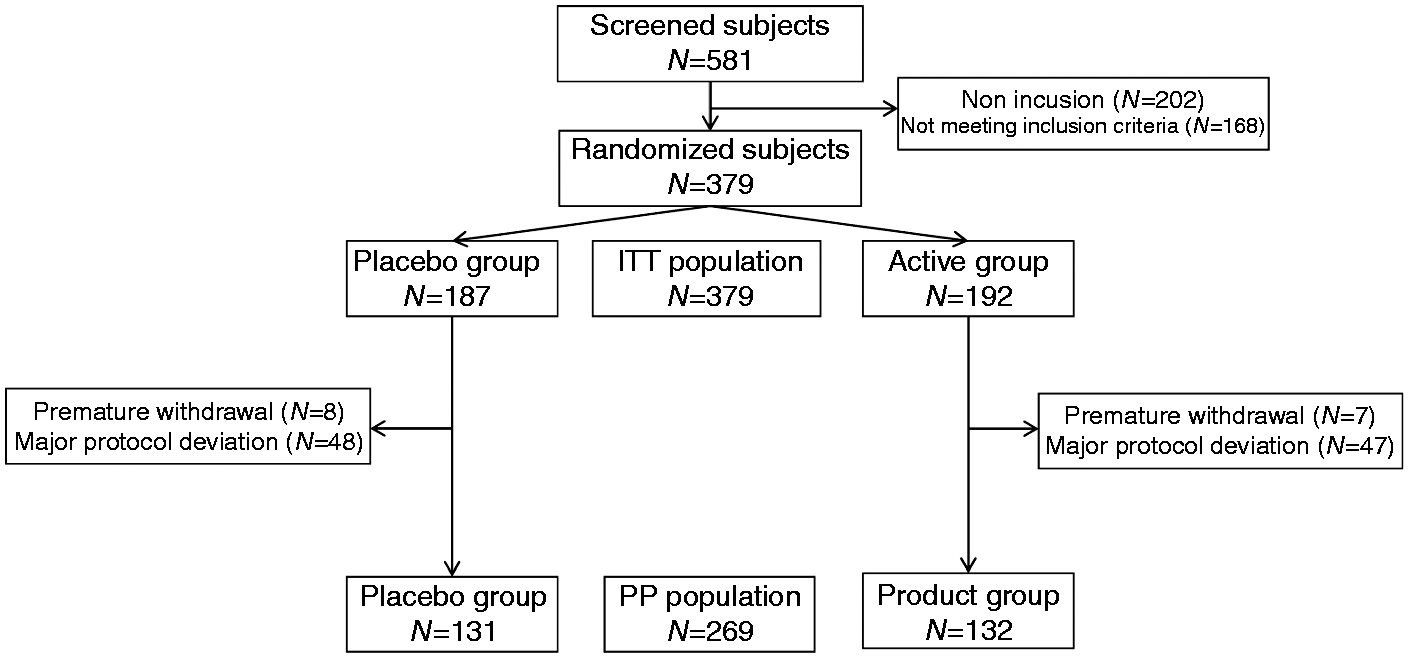
Flow chart of subjects throughout the study.
Fifteen (15) subjects did not complete the study (protocol deviation (
Results on total population
The responder status of subjects was analyzed during the last 8 weeks of the study in subjects without missing data. Forty-seven (47) subjects out of 175 in the placebo group (26.9%) and 57 subjects out of 177 in the active group (32.2%) were considered as responders, which means that they have an improvement of 50% of the weekly average “intestinal pain/discomfort score” compared with baseline average score for at least 4 out of the last 8 weeks of the study. However, the active product was not a statistically significant predictor of the responder status (
A relief in IBS symptoms was reported by subjects as soon as the first week of supplementation (27.2% of subjects of the placebo group, 33.6% in the active group), with statistically significant improvement at the end of supplementation (57.7% of subjects of the placebo group, 60.9% in the active group at the last week of supplementation,
Subgroup post hoc analysis
Baseline characteristics of subjects in the subgroups of IBS
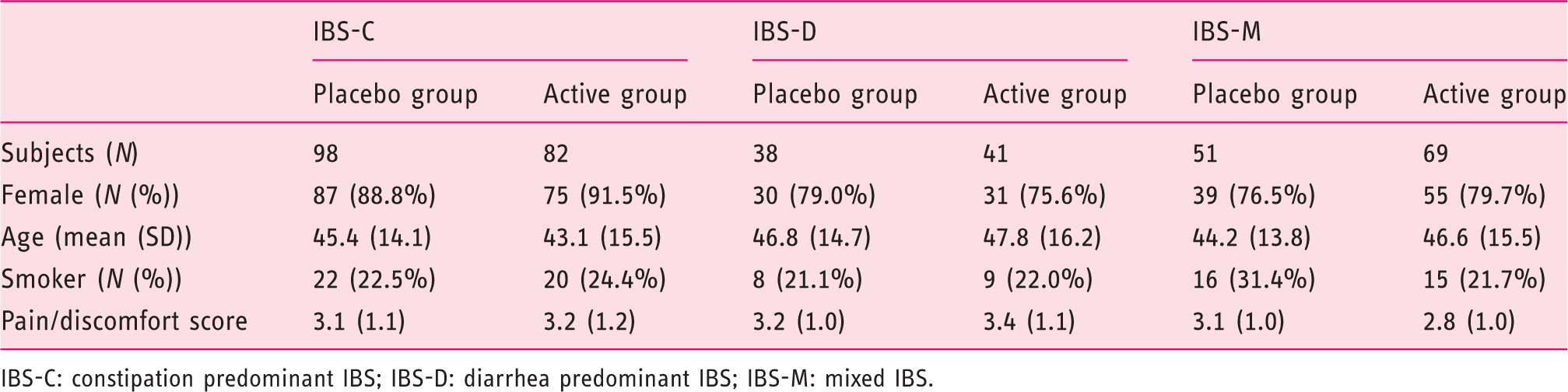
IBS-C: constipation predominant IBS; IBS-D: diarrhea predominant IBS; IBS-M: mixed IBS.
IBS symptoms scores (mean (SD)) during the study in placebo and active IBS subgroups, before (W0) and after (W12) supplementation
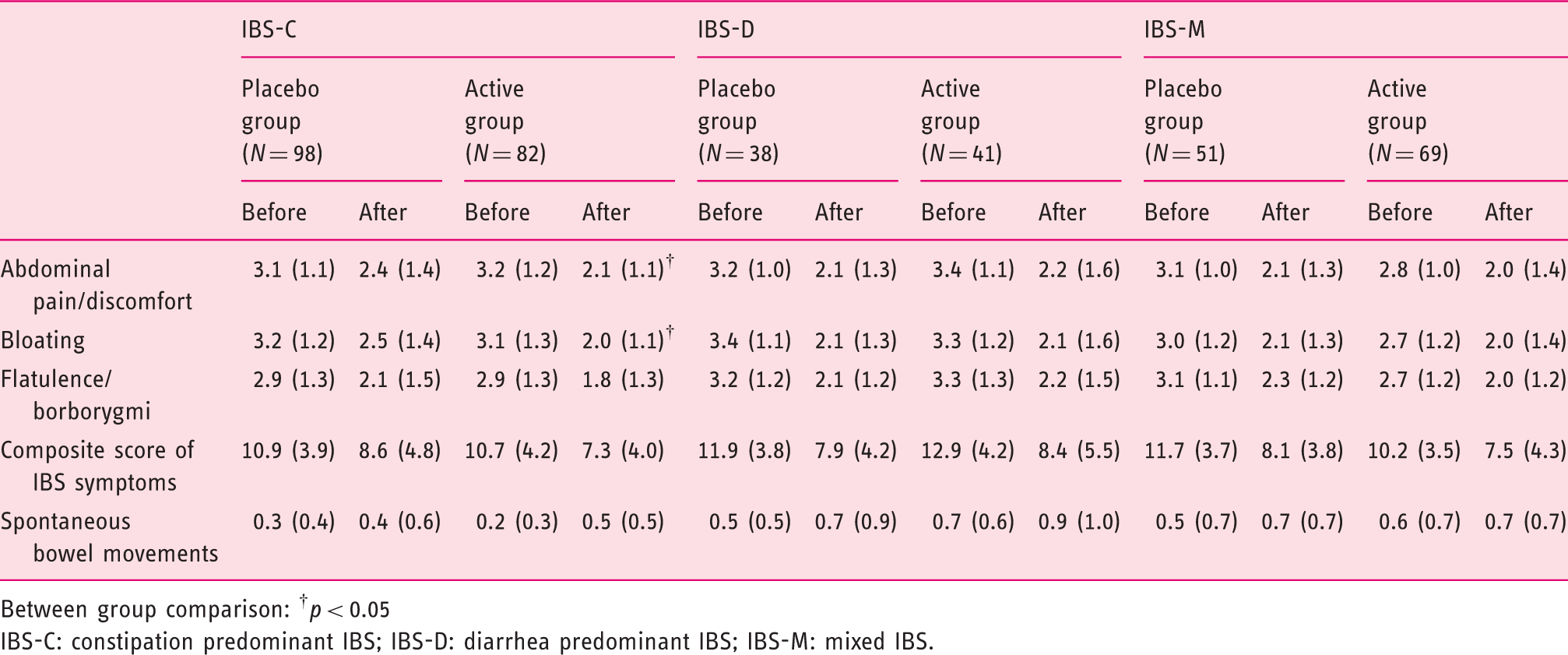
Between group comparison: †
IBS-C: constipation predominant IBS; IBS-D: diarrhea predominant IBS; IBS-M: mixed IBS.
At the end of the supplementation period, significant improvement was observed in abdominal pain/discomfort, bloating, flatulence/borborygmi, and composite score in both active and placebo groups of IBS-C subjects (Table 3) but greater on active than placebo for abdominal pain/discomfort ( Evolution of abdominal pain/discomfort (a), bloating (b) and composite score of IBS symptoms (c) in IBS-C subjects, during the 12-week consumption of active product (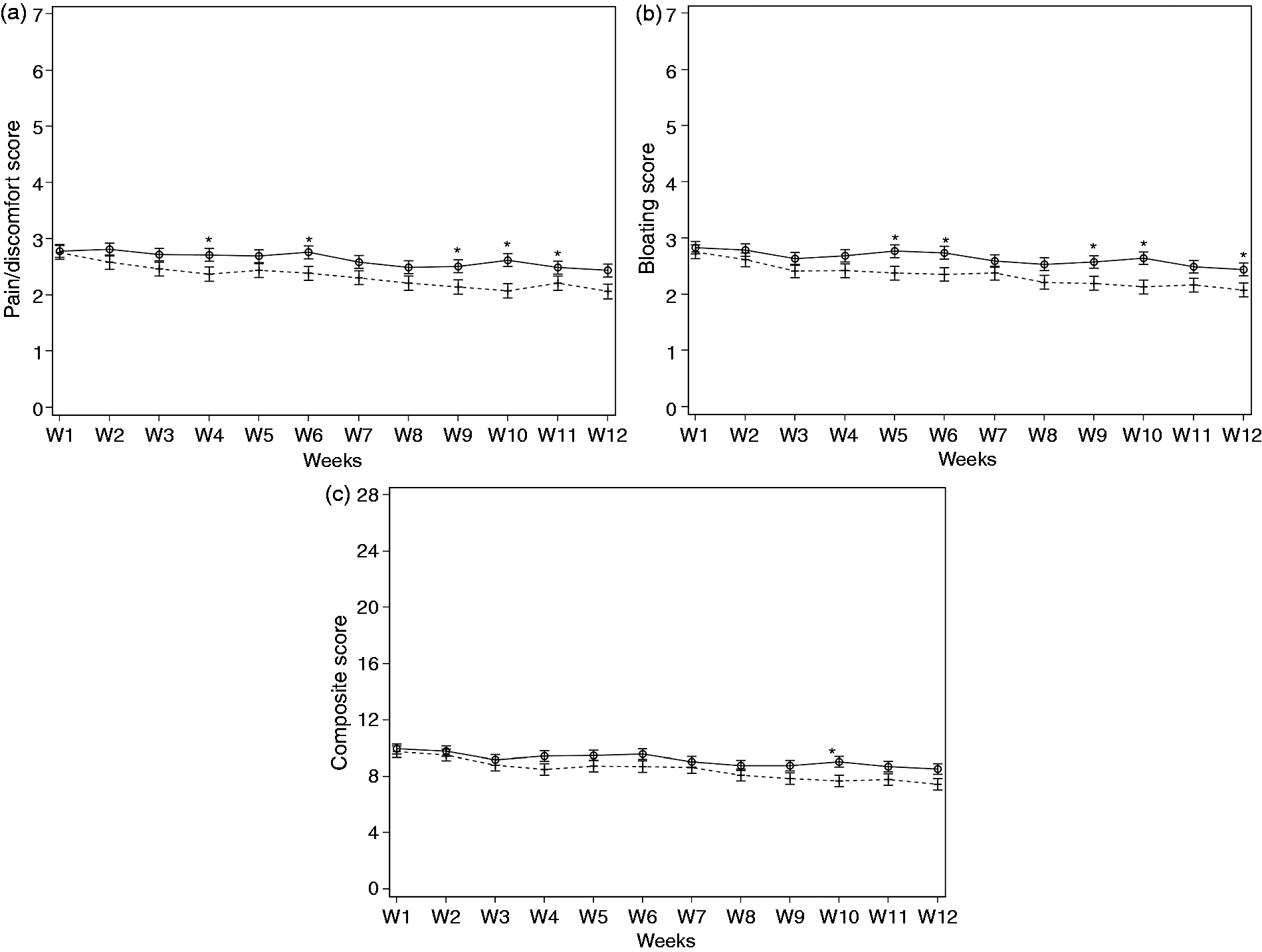
In IBS-C subgroup, the beneficial effect of AUC of abdominal pain/discomfort (a), bloating (b) and composite score of IBS symptoms (c) in IBS-C subjects over the 12 weeks of active product (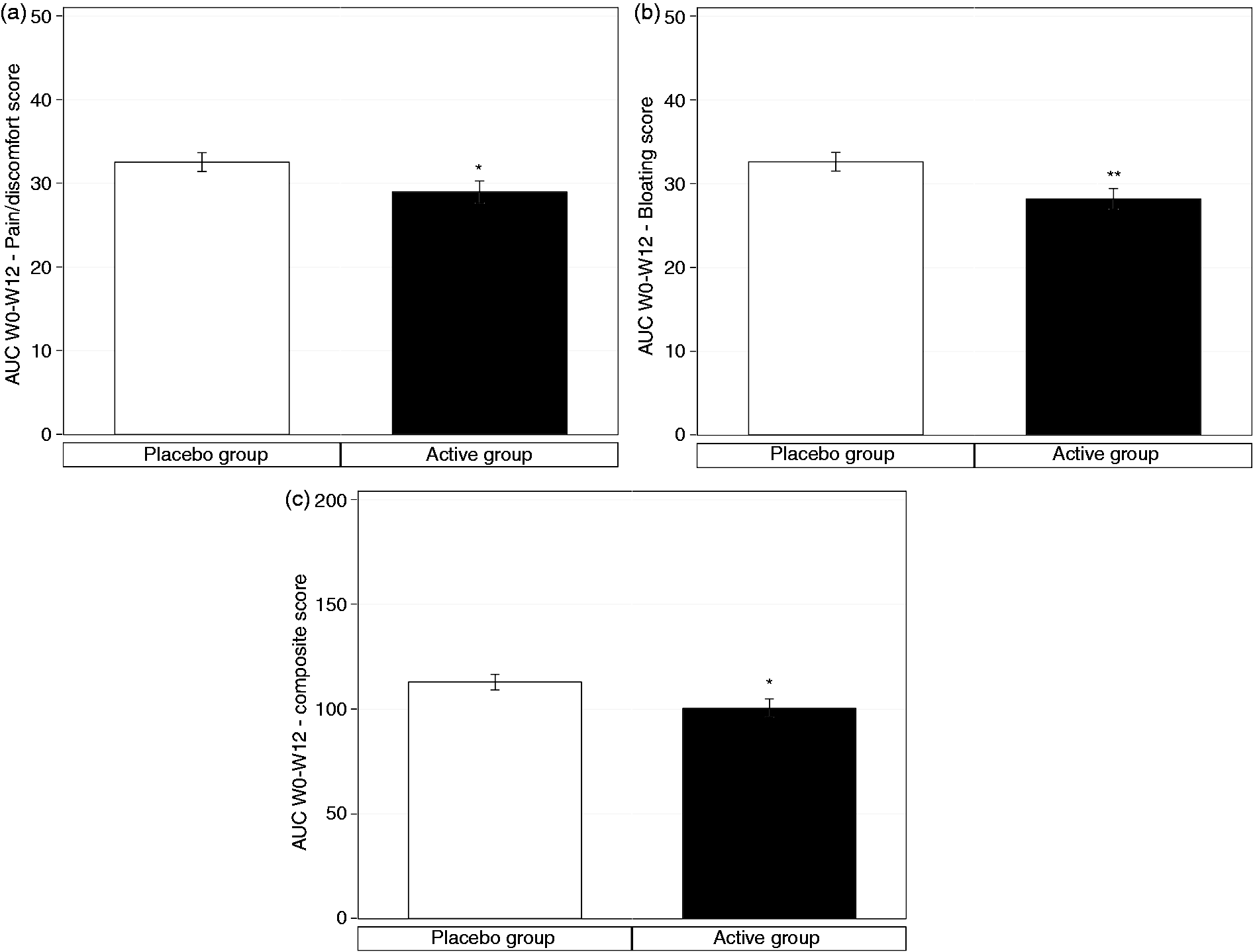
The composite score of IBS symptoms similarly demonstrated significantly lower total AUC in the active group, in comparison to the placebo group (−11.0%, diff [CI95%] = −12.46 [−23.76; −1.16],
Safety
No serious adverse event linked to the research or to the study product was recorded during the study in the placebo and product groups. Sixteen adverse events were judged as possibly linked to the research or to the study product by the investigators, and among these 16 adverse events, 14 were gastrointestinal (constipation (4 in active group, 2 in placebo group), abdominal pain (2 in active group), gastroesophageal reflux (1 in active group, 1 in placebo group), etc.): 10 were reported in the active group and 4 in the placebo group.
Discussion
The results of this randomized, placebo-controlled clinical study demonstrate that
Why
Our results provide additional data to those obtained before on the beneficial effect of
Footnotes
Acknowledgments
All authors have approved the final version of the article and the list of authors. The authors would like to thank subjects for their interest, time and compliance with the supplementation. Writing support was provided by Gunnard K Jacobson of Lesaffre Yeast Corporation.
Funding
This work was realized in the frame of the collaborative research program LEVACI supported by the French FUI fund (Fonds Unique Interministériel), the FEDER (European Fund for Regional Development), the Region Nord-Pas-de-Calais and LMCU (Lille Métrople Communauté Urbaine) in France. LEVACI was approved by the French competitiveness clusters “Nutrition-Health-Longevity” and “Vegepolys”. The writing of this paper was funded by Lesaffre.
Conflict of interest
Prof. Robin Spiller has served as an expert for Lesaffre and Biofortis and has received consulting fees and research funding from Lesaffre.
Fanny Pelerin, Amelie Cayzeele Decherf, and Dr Peter Jüsten are employees of Lesaffre.
Corinne Maudet, Béatrice Housez, and Murielle Cazaubiel are employees of Biofortis. Biofortis has received funding from Lesaffre.
Notes
The study was registered on ClinicalTrials.gov registry under identifier NCT01613456. NB: At the time of the study registration, an error was made stating that the primary endpoint was evaluated over 12 weeks when in fact the protocol specified an evaluation over the last 8 weeks of treatment. Unfortunately, this point was only rectified on clinicaltrials.gov after study completion. However, the study published in this Journal was carried out exactly according to the approved protocol and the primary endpoint was never changed, neither before nor after the trial was started.
