Abstract
Background
Effective management of irritable bowel syndrome (IBS), a common functional gastrointestinal disorder, can be challenging for physicians because of the lack of simple diagnostic tests and the wide variety of treatment approaches available.
Objective
The objective of this article is to outline a simple algorithm for day-to-day clinical practice to help physicians navigate key stages to reaching a positive IBS diagnosis and guidance on how to prioritise the use of specific management strategies.
Methods
This algorithm was based on the opinion of an expert panel evaluating current evidence.
Results
The key principles forming the foundation of this evidence-supported algorithm are: confidently naming and explaining an IBS diagnosis for the patient, followed by assessment of key patient characteristics likely to influence the choice of therapy, such as predominant symptoms, and exploring the patient agenda and preferences. Consultation should always include education and reassurance with an explanatory model of IBS tailored to the patient. Individualised lifestyle changes, dietary modifications, pharmacological therapies, psychological strategies or a combination of interventions may be used to optimise treatment for each patient.
Conclusion
The simple visual tools developed here navigate the key stages to reaching a positive diagnosis of IBS, and provide a stepwise approach to patient-centred management targeted towards the most bothersome symptoms. Establishing a strong patient-physician relationship is central to all stages of the patient journey from diagnosis to effective management.
Introduction
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder (FGID), with prevalence rates ranging between 5% and 20%, depending on the geographical region and the criteria used for assessment. 1 The defining features of IBS are the presence of recurrent abdominal pain in association with altered bowel habits (diarrhoea, constipation or both). The spectrum, duration and severity of symptoms can range from inconvenient to incapacitating, 2 and can prevent individuals from participating in everyday activities. 3
Despite the prevalence of IBS, its diagnosis and management remain as challenges for global healthcare systems.
Diagnosis
FGIDs most likely exist on a continuum rather than in isolation as separate and discrete disorders, with significant symptom overlap among these conditions. 4 Furthermore, the symptoms of IBS can mimic those associated with organic diseases, posing a challenge for diagnosis. 5 The recently updated Rome IV criteria were designed to facilitate making a positive diagnosis of IBS, based on the presence of characteristic symptoms and the absence of objective findings from a limited number of standard diagnostic tests and investigations. 4 While the majority of patients with IBS are diagnosed and treated in primary care, only a minority of general practitioners use the Rome criteria to make a diagnosis. 6 Even experts have difficulty in consistently making a positive diagnosis. 7 Differences in national guidelines and practice approaches as well as the availability and costs of diagnostic tests may also influence the diagnostic approach used. 7
Management
While a variety of different therapy options are available to target the symptoms of IBS, many have not been evaluated in high-quality, randomised controlled trials. Several recent review articles,8–10 along with multiple systematic reviews and meta-analyses,11–14 have provided detailed evaluation of the clinical evidence for different treatment approaches for the management of IBS. However, guidance in terms of how to prioritise the use of different agents is lacking.
Our objective was to develop a simplified algorithm to be used in day-to-day clinical practice to support practitioners to reach a positive diagnosis of IBS, prioritise the use of specific therapies to target predominant symptoms, and provide guidance on how to incorporate key patient characteristics into a tailored management approach.
Methods
A panel of 13 international experts in the field of FGIDs met twice to discuss and agree on the development of simplified algorithms for the diagnosis and management of IBS. The algorithms developed during this process were based on the expert opinion of the panel, taking into consideration professional guidelines, the quality of the clinical evidence for specific management strategies and the group’s experience from clinical practice.
During the first meeting, as a group, the panel participated in a series of workshop activities to: (1) define the key stages in reaching a positive diagnosis of IBS; (2) determine what patient factors physicians need to consider before starting treatment; (3) provide guidance on the sequence in which to use different therapy options, based on key patient characteristics and the quality of evidence supporting the use of specific agents in each context; (4) discuss how to assess treatment success and what this may mean to each individual patient; and (5) discuss the long-term management of IBS. The workshop format allowed all participants to provide their individual opinions equally, allowing sufficient time for group discussion and agreement. Based on these initial outputs, draft algorithms were developed. The panel then met a second time to discuss further refinement and finalisation of the algorithms. The final algorithms presented here were agreed upon by all members of the panel.
Reaching a positive diagnosis of IBS
Patient presenting with symptoms suggestive of IBS
Typically, the first point at which patients with suspected IBS will consult a physician is when their symptoms are bothersome enough to affect their daily life and warrant them seeking medical attention (Figure 1). Factors that may drive this consultation are symptom severity, number of symptoms, concomitant psychological disorders and concerns that symptoms might indicate an underlying severe disease.9,15
Simplified algorithm for irritable bowel syndrome (IBS) diagnosis.
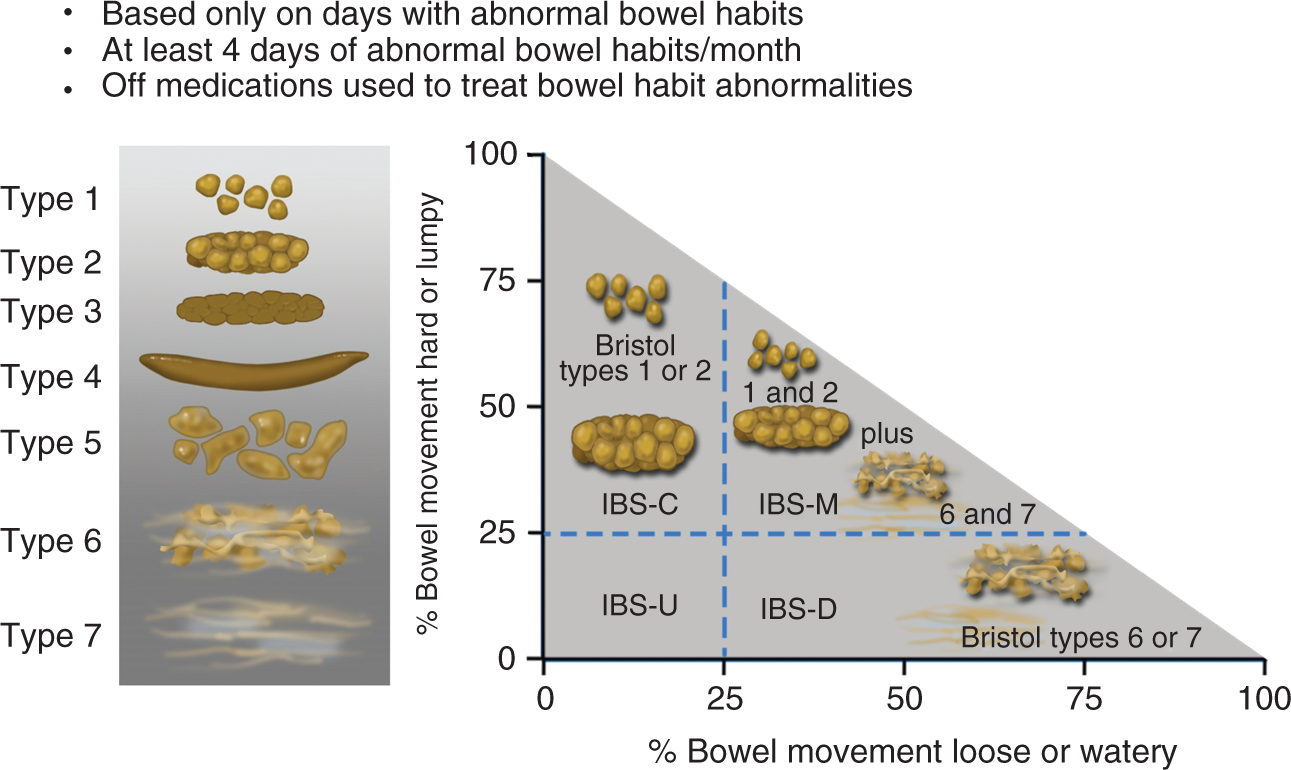
Determining the predominant disorder in bowel habit will be part of the initial clinical evaluation of the patient. The Bristol Stool Form Scale (BSFS; Figure 2)
17
should be used to record stool consistency on days when patients have abnormal bowel habits (BSFS type 1–2 or 6–7); for accurate assessment this should be performed when patients are not taking medications that alter their bowel habits (e.g. laxatives or anti-diarrhoeal agents). These assessments can be used to identify the IBS subtype (Figure 2):
4
IBS with predominant constipation (IBS-C): Patient reports that abnormal bowel movements are usually constipation (BSFS type 1 or 2). IBS with predominant diarrhoea (IBS-D): Patient reports that abnormal bowel movements are usually diarrhoea (BSFS type 6 or 7). IBS with mixed bowel habits (IBS-M): Patient reports that abnormal bowel movements are usually both constipation and diarrhoea (more than one-quarter constipation and more than one-quarter diarrhoea).
Irritable bowel syndrome subtypes.
4
Assessment and investigation (history/physical exam)
Performing a battery of tests in all patients suspected of having IBS is not warranted as most patients <50 years old have a very low probability of harbouring organic disease. 18 Limited diagnostic testing can play an important role in distinguishing IBS from other gastrointestinal (GI) conditions associated with similar symptoms (e.g. coeliac disease, inflammatory bowel disease (IBD), lactose intolerance, and microscopic colitis). 4 For the majority of patients with a clinical history compatible with IBS, the tests or investigations required will vary according to patient demographics, clinical situation and reported symptoms. 9 Tests that may be performed at this stage include complete blood count (because anaemia or an elevated white blood cell count should warrant further investigation), and C-reactive protein and faecal calprotectin in those with diarrhoea to exclude IBD. 4 Routine thyroid tests are not indicated in all patients, but can be checked if clinical suspicion of thyroid disorder is high. 4 Diagnostic testing for coeliac disease may be warranted in patients from areas with a high prevalence of the disease. A colonoscopy is indicated for all patients ≥50 years, with biopsies indicated for patients with diarrhoea or mixed bowel habits. 4
Initial patient assessments.

Factors supporting IBS diagnosis
What further questions need to be addressed to confirm a positive diagnosis of IBS? The pattern of abdominal pain or discomfort should be considered in terms of the duration, type, location, time of occurrence and its relation to defecation, e.g. whether pain is relieved with bowel movements.4,20 Other abdominal symptoms that are consistent with a diagnosis of IBS (but not present in all patients) include bloating, distention and flatulence. The presence of other FGIDs may also support a diagnosis of IBS. Non-GI symptoms that are supportive of an IBS diagnosis also include migraine headaches, interstitial cystitis and dyspareunia (Figure 3). Constant lethargy is also commonly experienced by patients
21
with IBS and it can be reassuring for patients to be informed that this is a well-recognised symptom.
Patient features supportive of an IBS diagnosis or raising concern for organic pathology.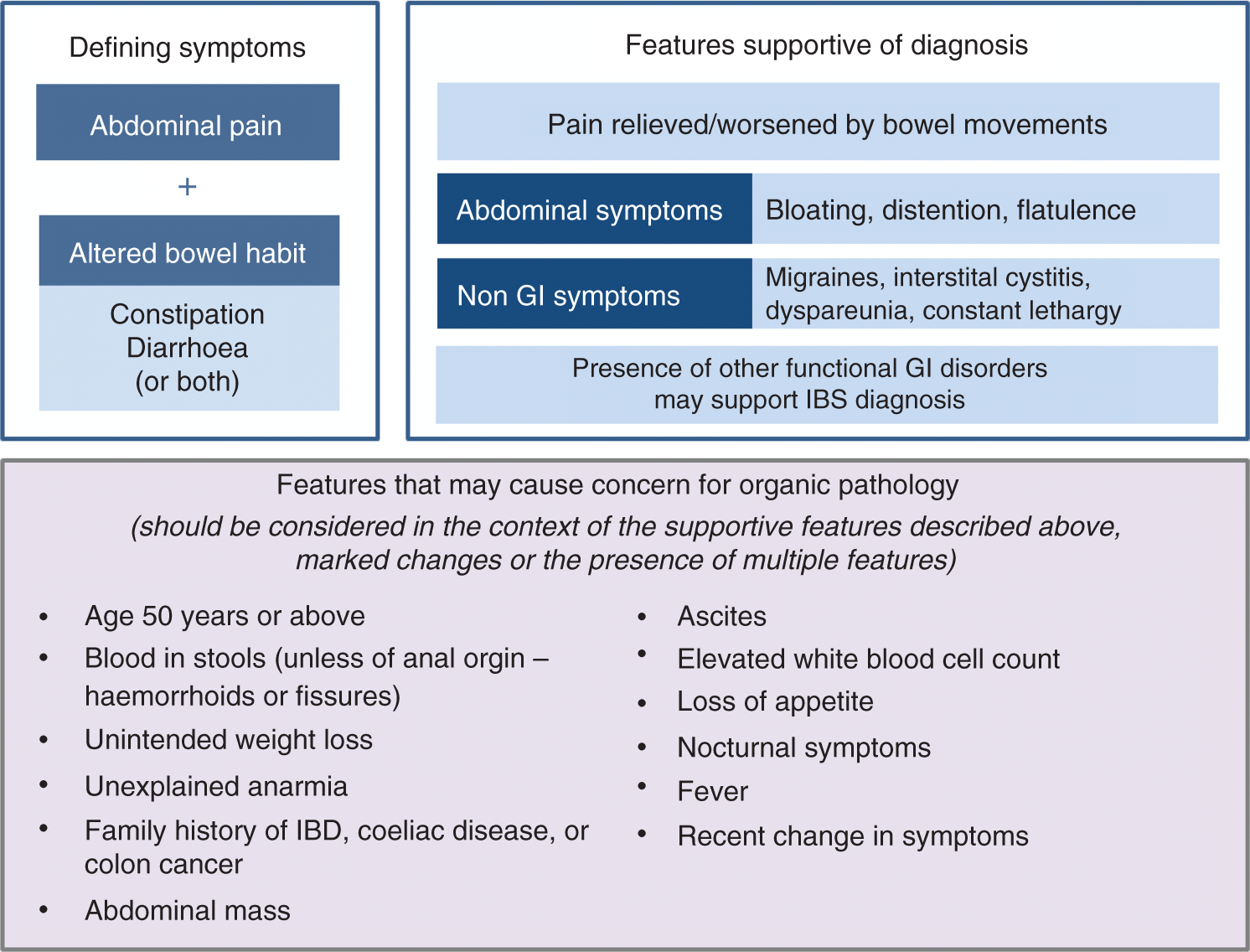
The nature and onset of symptoms is also important; for example, onset after gastroenteritis would suggest post-infectious IBS. The onset of IBS-like symptoms after an acute episode of diverticulitis has also been observed. 22 Stressful events in a patient’s history such as domestic abuse 23 or serving in the military 24 may also be linked to the risk of developing IBS.
Ruling out organic pathology
Further investigations that may be required in some patients.
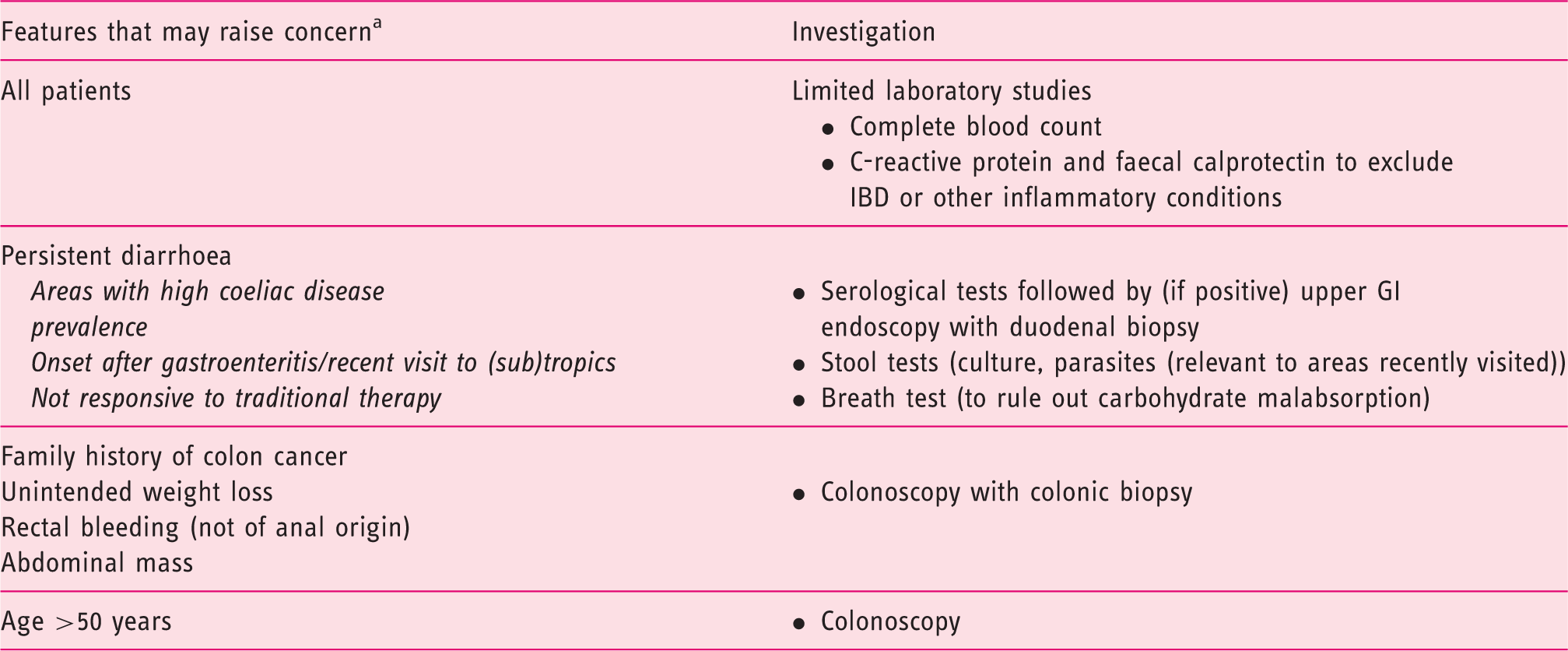
Should be considered in the context of the supportive features of IBS, marked changes or the presence of multiple features.
IBS: irritable bowel syndrome; IBD: inflammatory bowel disease; GI: gastrointestinal.
Patient vignette – example of a case of suspected irritable bowel syndrome requiring further investigation.

Management algorithm
1. Consider key patient characteristics
When deciding on an appropriate treatment strategy, it is important to understand the clinical profile of the patient, particularly in terms of the most predominant symptom. The pattern and severity of the GI symptoms experienced, along with the impact of these symptoms on daily activities and quality of life, will be the key determinants of the choice of management strategy, in addition to patient preferences (e.g. preference for non-pharmacological therapies) and treatment history (of both over-the-counter and prescription medications). Psychological comorbidities that could be contributing to the presence or worsening of IBS symptoms should also be considered (Figure 4).
Management algorithm for irritable bowel syndrome.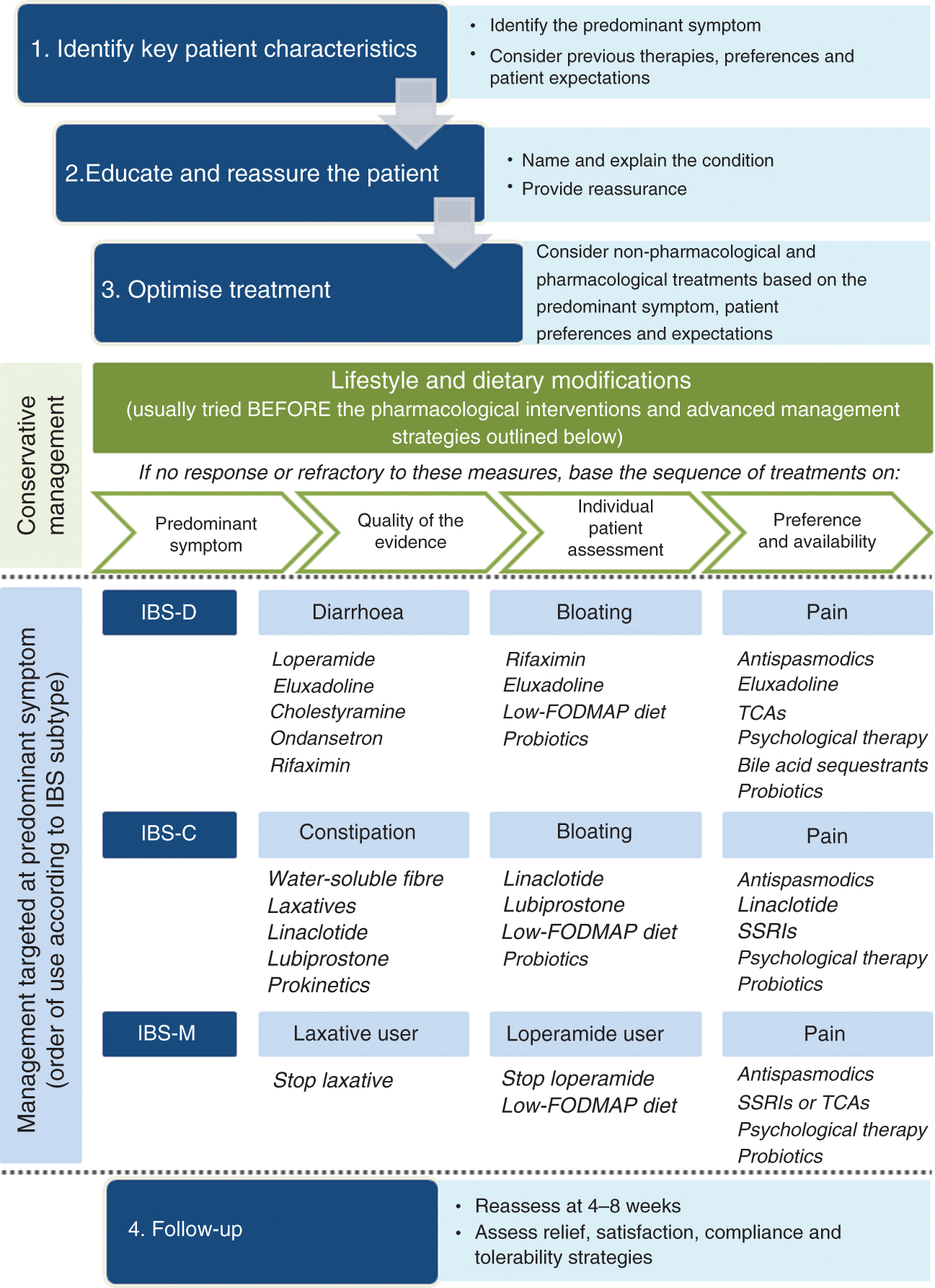
It is additionally important to explore the patient’s goals to elicit their personal perspective of their condition, and to understand what their expectations are in terms of treatment success. Knowing what patients with IBS want or expect from their medical care is critical in helping them to manage their symptoms, as failure to do so can lead to patient dissatisfaction with care, lack of compliance with prescribed treatments, and the inappropriate use of medical resources. 26
2. Educating, supporting and sharing information with the patient
Exploring the patient’s goals also helps the physician understand the level of knowledge the patient has about their condition, and in turn allows information to be shared in an appropriate patient-specific way. Many physicians may feel uncomfortable giving a diagnosis of IBS until other possible explanations (i.e. organic disease) for a patient’s symptoms have been completely ruled out.11,27 However, continued unnecessary investigations can have a negative effect on patient management by undermining the ultimate diagnosis of IBS and the patient’s confidence in their treating physician. 27 Effective management of IBS is therefore reliant on physicians being able to provide information and instruction for the patient by naming the condition through a confident diagnosis, with a clear explanation of what they believe is causing a patient’s symptoms and how they intend to target these factors with specific management strategies. 28 Providing patients with a written diagnosis of IBS rather than just verbal confirmation can also encourage them to understand and appreciate their new diagnosis.
Examples of ‘lay’ language for communicating with patients 27 .

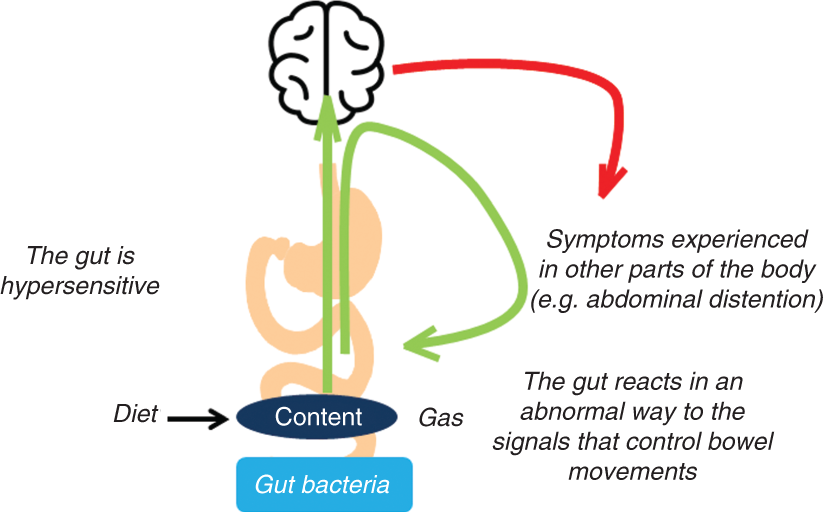
Explaining irritable bowel syndrome pathophysiology to the patient.
The more time that is spent at this important early stage in establishing a strong patient-physician relationship – getting to the root of the patient’s concerns and explaining their condition – the greater the chance of finding an effective management strategy. Key factors that can help build this relationship include active listening, not interrupting, using empathy, setting realistic patient expectations, and using nonverbal techniques such as eye contact and open body posture. 11 Patient education and reassurance can also help teach patients simple self-management strategies related to diet and stress management, and assuage symptom-related fears and anxiety. 29 Continuity of care is also important, with the development of a good patient-doctor relationship in follow-up appointments.
3. Create the optimal management strategy for each individual patient
Ideally, therapies that have been evaluated in high-quality randomised controlled trials would be the therapy of choice for patients with IBS. The reality is that many readily available treatments for IBS are either not specifically approved for the symptomatic treatment of IBS, or the evidence supporting their use is poor. However, physicians are familiar with their use and they are often inexpensive. Newly approved therapies have been developed specifically for the treatment of multiple IBS symptoms and have been assessed in high-quality clinical trials, but there is no direct evidence suggesting that newer agents are superior to traditional therapies, and it is very unlikely that comparative trials will ever be conducted to assess this. First-line usage of newly developed agents is therefore highly unlikely in the short term.
Lifestyle/dietary interventions
Regardless of subtypes or predominant symptoms, for many patients, the first-line approach of lifestyle and dietary modifications may provide relief from IBS without the need for further interventions. 28 These include promoting increased physical activity 30 and encouraging healthy eating habits such as modifying the intake of alcohol, caffeine, fat, spicy food, and gas-producing foods. 31 Investigating the possibility of carbohydrate malabsorption, restricting milk and dairy products, and modifying dietary fibre may also be considered at this stage. 31 Patients who do not respond or are refractory to the above measures may require symptom-modifying drugs or psychological treatments, 28 along with more advanced dietary interventions. 31 If suspected, functional outlet obstruction should be investigated and treated.
Pharmacological therapies for IBS management should be targeted towards the patient’s predominant symptom, which could be their dominant abnormal bowel habit, abdominal pain or bloating. With our increased understanding of the pathophysiology of IBS, pharmacological agents targeting the underlying disease mechanisms and thereby multiple symptoms of IBS associated with specific subtypes have also been developed.
A number of recent review articles,8–10 along with multiple systematic reviews and meta-analyses11–14 have provided a detailed overview of the quality of the clinical evidence for different management strategies for IBS. Taking this evidence into account, we summarise below the therapy options available for the management of different IBS subtypes according to their suitability for targeting specific symptoms and the order in which to prioritise their use (Figure 4).
Management of IBS-C
Initial management
Water-soluble fibre (e.g. psyllium) has been shown to provide overall symptom relief in IBS, 32 while the osmotic laxative, polyethylene glycol, has been found to improve stool frequency and consistency, but has not shown a significant effect on abdominal pain or bloating. 33 As the evidence base for soluble fibre for management of IBS-C is as strong as that for most pharmacological therapies, it is reasonable to use these agents as a first-line strategy because of their low cost, over-the-counter availability and favourable tolerability profile.9,32,33
Pharmacological treatments
By definition, all patients with IBS-C will experience abdominal pain and constipation, potentially along with other characteristic symptoms. The pharmacological agents available for the management of IBS-C vary from those targeting specific symptoms to those with a more global effect on multiple symptoms. The optimal therapy choice will ultimately depend on the severity of the symptoms that are most bothersome to the patient, and on local availability of treatments and patient preferences.
Agents that target multiple symptoms in patients with IBS-C include linaclotide and lubiprostone. Linaclotide is a guanylate cyclase C agonist that increases the production of cyclic guanosine monophosphate, which is proposed to reduce constipation by increasing fluid secretion and accelerating intestinal transit, and to target abdominal pain by reducing visceral hypersensitivity. 34 Although linaclotide has highest-quality evidence to support its use for the management of IBS-C, it is unlikely to be used before more traditional therapy approaches such as laxatives and soluble fibre. In phase 3 clinical trials, linaclotide was shown to significantly improve abdominal pain and discomfort, and to provide significant relief from the symptoms of IBS-C, including abdominal bloating, stool consistency and severity of straining, as well as increase the mean number of spontaneous and complete spontaneous bowel movements per week. Linaclotide is approved by the European Medicines Agency (EMA) for the treatment of IBS-C.35,36
Another agent targeting fluid secretion in the GI tract is the chloride channel activator, lubiprostone, which has been shown to provide significant relief from IBS-C symptoms, including bloating, bowel movement frequency, abdominal pain, straining, constipation severity and stool consistency. 37 Although lubiprostone has been evaluated in patients with IBS-C in phase 3 clinical trials with moderate evidence quality, it is not currently approved for this indication by the EMA.
For patients with IBS with pain predominance, antispasmodics (which include anticholinergic or calcium channel-blocking agents) are an established therapy option, as they relax gut smooth muscle. 38 Pain in IBS is thought to be in part the result of smooth muscle spasms. 9 While the quality of most studies assessing the efficacy of antispasmodics in IBS is suboptimal, the results of meta-analyses suggest benefits of these agents as a class over placebo for abdominal pain, 39 and they may provide symptomatic short-term relief. 12 However, not all antispasmodics have been shown to be effective, and the availability of specific agents varies significantly from country to country. Moreover, it is not clear whether they act differently in different IBS subgroups.
Antidepressants are also widely used for the treatment of IBS pain because of their observed effects on pain perception, mood and motility, 11 and were first introduced into the management of IBS based on the observation that depression and anxiety were frequent comorbidities among patients seen in secondary and tertiary care. 12 In a meta-analysis of randomised controlled trials of patients with IBS treated with antidepressants, these agents as a class were shown to improve abdominal pain along with global IBS symptoms. 14 The symptomatic benefit of antidepressants in IBS seems to be unrelated to the presence or improvement of coexistent depression. Patient preferences may come into play when considering the suitability of these agents, as some patients may be averse to the idea of taking antidepressants. 12 Of this class, selective serotonin re-uptake inhibitors (SSRIs) may be an appropriate choice for patients with IBS-C due to the prokinetic effects of these drugs. 11 SSRIs tend to be prescribed at dosages standard for treating mental health disorders, so may also be more suitable for patients with psychological comorbidities. 40 However, tricyclic antidepressants (TCAs) could also be used, as they are effective for abdominal pain and do not induce constipation when administered at low dosage.
Prokinetic agents, such as prucalopride, may also be considered as a treatment option specifically targeting constipation. Although prucalopride has demonstrated efficacy in the treatment of chronic idiopathic constipation, there are currently no data on the efficacy or safety for this agent in the treatment of IBS-C. 12
Management of IBS-D
Initial management
Water-soluble fibre (e.g. psyllium) has a high water-holding/gel-forming capacity that is preserved throughout the large bowel, and can act as a bulking agent to firm loose/liquid stools in patients with diarrhoea. 41 A diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) may also decrease symptoms in patients with IBS, particularly those with IBS-D.10,42 However, rigorous trials of dietary manipulations in patients with IBS are currently lacking, making it difficult to make strong recommendations on the optimal use of this approach.
Pharmacological treatments
As all patients with IBS-D experience pain and diarrhoea, the optimal therapy choice will be determined based on the severity of the symptoms that are most bothersome to the patient, local availability of treatments, and on patient preferences. The pharmacological agents available for the management of IBS-D vary from those specifically targeting diarrhoea, such as loperamide, to those targeting multiple symptoms.
Loperamide is a μ-opioid receptor agonist which improves diarrhoea by decreasing peristalsis, prolonging GI transit time, and through reduction of fluid secretion in the intestinal lumen. 9 Although loperamide is established as an effective anti-diarrhoeal agent, the evidence base supporting its use for the treatment of IBS-D is not as strong as that of other pharmacological therapies as it has not been shown to improve overall IBS symptoms. 12 Adverse effects of loperamide include abdominal cramps, constipation, bloating and nausea. However, because of its availability over the counter, established effect on diarrhoea and relatively low cost, loperamide is frequently used as a first-line therapy for IBS-D. Loperamide may also be used prophylactically when a patient anticipates episodes of diarrhoea. 11
Eluxadoline is a mixed μ-opioid receptor agonist, δ-opioid receptor antagonist and κ-opioid receptor agonist peripherally acting in the gut with minimal oral bioavailability. 43 Based on preclinical evidence, eluxadoline has been shown to reduce visceral hypersensitivity without completely disrupting intestinal motility, suggesting that peripheral δ-opioid receptor antagonism may reduce μ-opioid receptor-mediated constipation and, similar to its documented effects on central analgesia, enhance μ-opioid receptor-mediated peripheral analgesia. 43 In phase 3 clinical trials, eluxadoline was shown to provide sustained relief from the global symptoms of IBS-D over the six-month trial period, 44 and it has recently received regulatory approval for the treatment of IBS-D from the EMA, the United States Food and Drug Administration (FDA) and Health Canada.
There is increasing evidence to support a role for bile acids in the pathophysiology of IBS-D, with a subset of patients proposed to have bile acid malabsorption. 45 Bile salt sequestrants, such as cholestyramine or colesevelam, may therefore be effective against diarrhoeal symptoms in some patients with IBS-D. The efficacy of cholestyramine is linked to the degree of bile acid malabsorption with patients affected by severe malabsorption responding better than those with milder forms. 46 It is suggested that bile acid sequestrants should be considered after other therapies targeting diarrhoea have been unsuccessful.4,47
Serotonin (5-hydroxytryptamine (5-HT)) is one of the most important neurotransmitters in the gut, influencing gut motility and secretion. 48 The 5-HT3 receptor antagonist, ondansetron, has been shown to slow colonic transit. In a randomised controlled trial, ondansetron improved the characteristic symptoms of IBS-D: loose stools, frequency and urgency. However, a significant reduction in abdominal pain was not seen. 49 Patients with severe diarrhoea did not respond as well, suggesting that ondansetron treatment is most suited to patients with mild-to-moderate symptoms.
Recent evidence suggests a role for gut bacteria and antibiotics in the pathophysiology and treatment of IBS, respectively. 50 Small intestinal bacterial overgrowth has also been suggested to be associated with IBS in general, and with IBS-D in particular, although this connection is still a topic of considerable debate. 51 Rifaximin is an oral, minimally absorbed, broad-spectrum antimicrobial agent that targets the GI tract and is associated with a low risk of clinically relevant bacterial antibiotic resistance.52,53 In randomised controlled trials in patients with non-constipated IBS, two weeks of rifaximin treatment provided adequate relief of global IBS symptoms and individual symptoms of loose stools, abdominal pain, and bloating. 52 However, this effect gradually disappears and re-treatment is necessary in a large proportion of patients to retain symptom improvement, so the optimal use of this agent for long-term management of IBS symptoms is yet to be defined.
Antispasmodics and antidepressants are widely used to target pain-predominance in all subtypes of IBS; however, specific agents within these classes may be more suited to patients with IBS-D. As TCAs increase intestinal transit time, they may be preferable to SSRIs for patients with IBS-D. 11 However, data on the efficacy of these agents specifically for IBS-D are limited. Because of the potential for adverse effects, TCAs are generally prescribed for patients with IBS at dosages much lower than those used for depression or anxiety. 40
Management of IBS-M
As patients with IBS-M essentially experience both IBS-C and IBS-D, the management strategy needs to be adaptable. Physicians should target the treatment approach towards the most bothersome symptoms experienced at a particular time, and then be prepared to re-evaluate this approach based on the response to therapy. There are limited studies evaluating the efficacy of specific management strategies for IBS-M and the evidence base for the use of most treatment options in this context is low. Usually the approach is selected from those outlined in the IBS-C and IBS-D sections above, depending on the bowel habit symptom that bothers the patient the most.
Individual patient characteristics are likely to be the key determinant of how to proceed with management. Patients who are habitual users of anti-diarrhoeal or laxative agents may need to discontinue these therapies in order to normalise their bowel habits. Some patients may also benefit from further investigations in order to determine the most appropriate treatment modality. Dietary management strategies (e.g. the low-FODMAP diet) that target bloating and diarrhoea (if present), and do not precipitate constipation may also be considered.
It is also important to note that some patients may have ‘pseudo’-IBS-M due to the intermittent use of laxatives and anti-diarrhoeal agents making them appear to have the mixed subtype. For these patients, simple strategies such as reducing the dose of laxatives may be enough to improve management. Patients with IBS-C may also experience overflow diarrhoea due to severe constipation causing a blockage in the bowel. As a consequence, watery stools may pass around the blockage and leak from the bowel, causing watery diarrhoea or faecal incontinence.
Targeting bloating
While there are currently no management strategies that specifically target bloating in IBS, some of the newer therapy options designed to target specific IBS subtypes have demonstrated efficacy against bloating in addition to other key symptoms. These include linaclotide and lubiprostone for IBS-C, and rifaximin and eluxadoline for IBS-D (Figure 4).
In recent years, data have also emerged supporting the use of the low-FODMAP diet for the management of IBS symptoms. 54 The low-FODMAP diet has been shown to alleviate overall IBS symptoms as well as individual symptoms such as abdominal pain, bloating, constipation, diarrhoea, abdominal distention and flatulence in clinical trials.54,55 There is also evidence to suggest that the low-FODMAP diet may be more beneficial for patients with bloating predominance. 56 The diet appears safe for short-term use; however, further research is needed to determine the long-term health effects of dietary restriction and the potential detrimental impact on the gut microbiota, making it difficult to provide evidence-based recommendations on optimal use. 56 Recent studies have also reported comparable symptomatic benefit with the less stringent National Institute of Health and Care Excellence (NICE) diet.57,58 Again, data are limited to short-term efficacy studies.
There is a growing body of evidence to suggest that perturbations in the intestinal microbiota are linked to the pathophysiology of IBS, and that the composition of the gut microbiota of patients with IBS differs from that seen in ‘healthy’ individuals. 59 A number of meta-analyses and systematic reviews suggest that probiotics may provide relief from overall or individual (e.g. bloating and abdominal pain) IBS symptoms, and these agents have been widely used for decades.9,59 However, the conclusions of these studies vary owing to inadequate sample size, poor study design and use of various probiotic strains in the reviewed studies, 59 making it difficult to make definitive recommendations on their use or conclusions as to whether specific strains or preparations are effective against specific symptoms.9,11,12
Psychological therapies
Thoughts, emotions, and behaviours are proposed to be bi-directionally related to gut physiology and symptom manifestations in IBS. 60 Psychological and behavioural treatments can help patients with IBS control and reduce their pain and discomfort and are seen as ancillary to or augmenting medical treatments. 4 A variety of psychological interventions have demonstrated efficacy in improving IBS symptoms, including cognitive behavioural therapy (CBT), hypnotherapy, multicomponent psychotherapy and dynamic psychotherapy. 11 CBT is the most studied form of psychological therapy and is associated with overall improvement in IBS symptoms, with good short-term and long-term efficacy. 61 In clinical practice, the limited availability of therapists skilled in applying psychological therapy to GI problems, the high costs of delivering the treatment, and the practical difficulties for patients of scheduling weekly visits at a clinic may limit widespread use and suggest that these strategies are reserved for later lines of therapy.11,62
4. Follow-up
What are the key factors to consider during follow-up in order to optimise management?

Firstly, it is important to define patient expectations in terms of treatment success and if they are achieving satisfactory relief from overall or individual symptoms. The severity of symptoms can play into setting realistic expectations for therapy. For example, in patients with more severe symptoms, achieving improved management and daily functioning may be a more achievable goal and acceptable to the patient, than anticipating complete resolution of symptoms. 63
If the efficacy of a particular treatment is suboptimal there are a number of factors that could play a role. Before stopping a treatment, the physician should evaluate treatment adherence, changes in lifestyle and dietary habits, and concomitant therapies that might potentially interact with the treatment. Patients should be encouraged to contact their physician should they experience any adverse events so they can be advised on simple management strategies to optimise treatment tolerability. This does not have to take the form of a formal consultation: Being contactable by other means such as phone or email may be sufficient or even preferable for some patients. Dose adjustments to determine the minimum effective dose to minimise the potential for adverse effects can also be considered. As many of the symptom-modifying treatments for IBS target a specific mechanism, using a combination of management strategies may also be a valid approach for some patients to achieve a more global effect on symptoms.
Assessing the need to continue or interrupt treatment
Guidance on optimising the use of specific therapies.
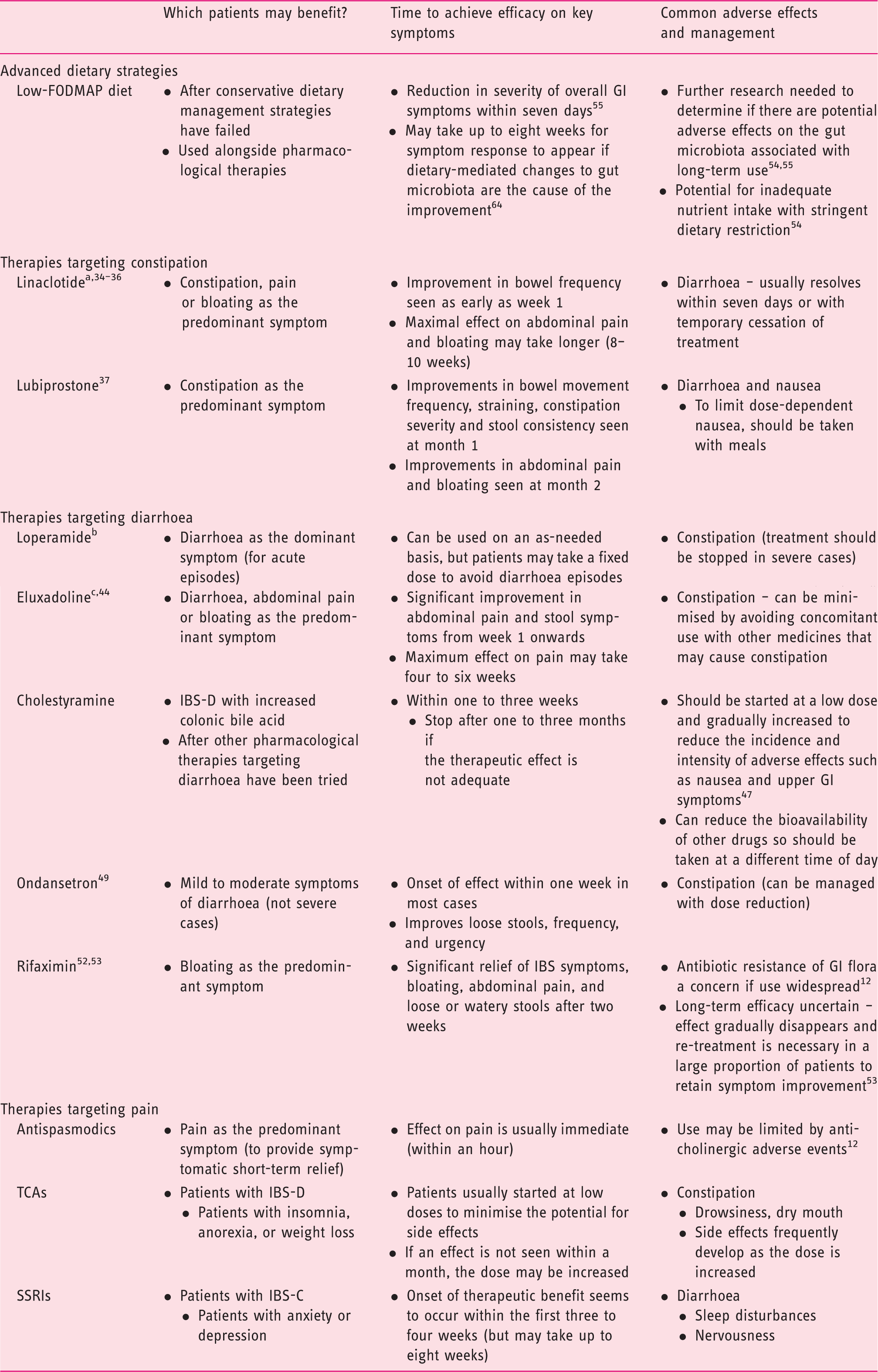
EMA-approved for the symptomatic treatment of moderate to severe IBS-C.
EMA-approved for the symptomatic treatment of acute episodes of diarrhoea associated with IBS-D.
EMA-approved for the treatment of IBS-D.
All other treatments included in the table are not currently approved by the EMA for the management of IBS.
EMA: European Medicines Agency; FODMAP: fermentable oligosaccharides, disaccharides, monosaccharides and polyols; GI: gastrointestinal; IBS-C: irritable bowel syndrome with constipation predominance; IBS-D: irritable bowel syndrome with diarrhoea predominance; SSRIs: selective serotonin re-uptake inhibitors; TCAs: tricyclic antidepressants.
Conclusions
The main challenges faced by physicians managing individuals with IBS today are the lack of simple diagnostic tests and the complex nuanced approach required for successful management. In order to simplify this process for day-to-day clinical practice, here we have developed simple visual tools to help navigate the key stages to reaching a positive diagnosis of IBS and a stepwise approach to patient-centred management targeted towards the most bothersome symptoms.
The basic tenets of IBS diagnosis and management highlighted by this simplified algorithm are very much aligned with the guiding principles proposed by recent reviews by Simrén and colleagues, 8 Enck and colleagues, 9 and Lacy and colleagues, 10 along with the Rome IV guidelines. 4 Successful management starts with the ability to make a confident positive diagnosis of IBS, along with being able to explain the underlying causes in relatable terms for the patient. These measures can help establish a strong patient-physician relationship and instil patient confidence in their treating physician. A thorough patient history can help to identify potential dietary and lifestyle triggers that can be modified as the first stage of IBS management. If symptoms are not effectively managed by these measures, pharmacological treatments can be considered, along with psychological therapies. The optimal choice of management strategy will ultimately depend on the predominant symptoms, patient preferences and a thorough understanding of the patient agenda in terms of their treatment expectations.
Footnotes
Acknowledgements
Editorial support for this manuscript was provided by Farah Dalwai from inVentiv Medical Communications, and was funded by Allergan, along with support for the organisation of the workshop meetings. The final content was developed independently by the authors without input from Allergan.
Declaration of conflicting interests
PM has received speaker’s fees from Allergan and Abbvie, and research support from Allergan and Takeda, and has served on advisory boards for Allergan, Shire and Lupin.
FM has received honoraria from Allergan.
FA has received grants from Danone, Clasado and Noventure, and served on advisory boards for Danone, Clasado and Allergan.
VA has received speaker and/or consulting fees from: Allergan, AstraZeneca, Boehringer Ingelheim, Falk, Ferring, KyowaKirin, Nordmark and Shionogi.
GB has received speaker bureau and/or advisory and/or research support from: Alfa Wassermann, Allergan, Cadigroup, Commonwealth, Danone, Falk Pharma, Ironwood, Italchimici, Lorenzatto, Malesci, Menarini, Noos, Parmalat, Shire, Synergy, Sofar, Yakult and Zespri.
MC has acted as a consultant for Allergan and Kiowa Kirin, and as a speaker for Shire and Menarini.
AE has served on advisory boards for Allergan, Almirall, Shire and Takeda.
APSH is the chair of the Rome IV Primary Care Committee. He has received fees from Allergan for advisory boards and research funding from Danone.
PL has received speaker’s fees for Abbott, Allergan, Falk, Nordmark and Shire, and served on advisory boards for Abbott and Allergan.
VS has received speaker’s fees from Alfa Wassermann, Allergan, Angelini and Valeas, and research support from Alfa Wassermann, Allergan and Shire, and has also served on advisory boards for Alfa Wassermann, Allergan, Angelini Farmaderma, Shire and Takeda.
PW has acted as a consultant for, or received research grant support from, the following pharmaceutical companies in the past five years: Almirall Pharma, Chr. Hansen, Danone Research, Ironwood Pharmaceuticals, Salix, Shire UK, Sucampo Pharmaceuticals and Allergan.
FZ has received speaker fees from Allergan, Reckitt-Benckiser, Takeda, Coloplast, Vifor pharma and Mayoli Spindler, and research support from Medtronic and Sandhill Scientific, and has served on advisory boards for Allergan and Reckitt-Benckiser.
JT has provided scientific advice to Abide Therapeutics, Alfa Wassermann, Allergan, Chr. Hansen, Danone, Genfit, Ironwood, Janssen, Kyowa Kirin, Menarini, Mylan, Novartis, Nutricia, Ono Pharma, Rhythm, Shionogi, Shire, SK Life Science, Takeda, Theravance Biopharma, Tsumura, Yuhan, Zealand and Zeria, has received research grants or support from Abide Therapeutics, Shire and Zeria, and has served on speakers’ bureaus for Abbott, Allergan, AstraZeneca, Janssen, Kyowa Kirin, Menarini, Mylan, Novartis, Shire, Takeda and Zeria.
Funding
Funding for editorial support and organisation of the workshop meetings was provided by Allergan.

