Abstract
Introduction
Although colorectal cancer (CRC) has often been regarded as a single entity, different pathways may lead to macroscopically similar cancers. These pathways may evolve into a patchy colonic field defect that we aimed to study in consecutive CRC patients.
Methods
In a single-center, observational, prospective study, consecutive CRC patients were included if surgery and a perioperative colonoscopy were planned. Personal and familial history data were collected. Tumors were studied for microsatellite instability (MSI) status, DNA repair protein expression (DRPE) and presence of BRAF and/or APC mutations. Macroscopically normal mucosa samples were tested for APC mutations. Presence and location of synchronous and metachronous adenomas and patient follow-up were analyzed. The association of two categorical variables was tested through the Fisher’s exact test (SPSS 19).
Results
Twenty-four patients (12 male, mean age 69 years) were studied. High-grade MSI (MSI-H) was found in eight tumors—these were significantly more common in the right colon (p = 0.047) and more likely to have an altered DRPE (p = 0.007). BRAF mutation was found in two of six tested MSI-H tumors. APC gene mutations were found in nine of 16 non-MSI-H tumors and absent in normal mucosa samples. There was a nonsignificant co-localization of CRC and synchronous adenomas and a significant co-localization (p = 0.05) of synchronous and metachronous adenomas.
Discussion
Sporadic CRCs evolve through distinct pathways, evidenced only by pathological and molecular analysis, but clinically relevant both for patients and their families. In non-MSI-H tumors, the expected APC gene mutations were not detected by the most commonly used techniques in a high number of cases. More studies are needed to fully characterize these tumors and to search for common early events in normal mucosa patches, which might explain the indirect evidence found here for a field defect in the colon.
Introduction
It has been proposed for several decades that most cancers start from a single cell and tumor progression occurs through genetic variability in the original clone. 1 The main molecular route to colorectal cancer (CRC), the chromosomal instability pathway, starts with adenomatous polyposis coli (APC) gene loss or beta-catenin mutations, followed by the accumulation of critical events in other genes. The time frame estimates for this sequence of mutations to occur point to the occurrence of the first event at an early age,2–4 probably during the exponential period of embryonic development, in which APC has an established determinant role. 5 CRC stem cells have been identified in humans,6,7 and it has been argued that stem cell mutations occur much more frequently in the early, exponential stage of development than in adult life. 8
The location of the epithelial colonic stem cells at crypt base was also recently demonstrated 9 and these cells seem to be the origin of CRC. 10 Intestinal crypts formation happens only at late fetal life, 11 when colonic segments are already defined, and all colonic epithelial cells arise from the pluripotent stem cells located at the crypts. Crypt development seems to emerge from a stem cells’ cooperative phenomenon, partly dependent on the Wnt pathway, and each crypt seems to contain more than one stem cell. 12 This stem cell niche appears to be the site of a clonal evolution phenomenon, including the random loss and selection of some lineages, leading to clonal extinction cycles in the crypt. Eventually, this niche succession phenomenon results in the periodic (two seven- to-19-year intervals are proposed, with a median of 8.2) loss of all but one stem cell lineage. A few mutations, namely in the Wnt pathway, may give the stem cell a selective advantage, allowing it to survive the clonal extinction cycles, with great potential for clonal progression. 13 Recent experimental data point to a relatively low mutation rate in CRC almost to its late stages, quite similar to the division and mutation rates at normal mucosa. This might be explained by an early beginning of cancer genealogy, close to conception, followed by decades of evolution in normal-appearing mucosa.13,14
In the clonal competition process, it appears that either APC loss or K-RAS activation may lead to a selection advantage for the mutated stem cell.15,16 There is evidence that even a single allele mutation in the APC gene is enough to confer some advantage in the crypt clonal competition process. 16 Mutated cells may remain phenotypically undetectable, even while in clonal expansion, until they acquire other mutations.16,17 Although CRC is regarded as a single entity, different first events may evolve through distinct pathways before they lead to macroscopically similar cancers.
The occurrence of a mutation during embryonic development leads to a genetic mosaicism, the degree of which changes according to the post-zygotic cell division stage in which the mutation occurs. 18 Therefore, the occurrence of a monoallelic gene mutation in a stem cell during embryonic development would lead to a somatic colonic mosaicism, in which the mutation would be identified in discrete colonic mucosa patches. These patches would correspond to the most likely segments for the development of adenomas, and subsequent evolution to cancer.
We therefore hypothesize that, when the most common pathway to CRC is considered, the first, monoallelic, mutation of the APC gene present in an individual’s CRC is also present in other colonic lesions and normal mucosa patches where some crypt stem cells derive from the same embryonic lineage that gave rise to the CRC stem cell. We predict that the probability of identifying the mutation will be greater in lesions and patches closer to the tumor.
Therefore, we aimed to characterize the pathways leading to sporadic CRC in a random sample and to search for mucosal patches with a mutated APC allele in those patients with a chromosomal instability pathway cancer.
Sporadic CRC was defined as that occurring in an individual with no known familial history of a hereditary CRC syndrome and no personal history of a condition associated with an increased CRC risk, namely inflammatory bowel disease.
The chromosomal instability pathway is the most common pathway to sporadic CRC, justifying 84% of the cases analyzed by the Cancer Genome Atlas Network. 19 These tumors are typically microsatellite stable. 19 Microsatellite instability (MSI) results from errors in deoxyribonucleic acid (DNA) replication that go unrepaired in the presence of mutations or epigenetic silencing of the mismatch repair genes. High-grade MSI characterizes CRC arising through another major pathway leading to CRC, the MSI pathway.
Methods
Study population
From February 2011, all consecutive patients with a histological diagnosis of a colorectal adenocarcinoma discussed at the Instituto Português de Oncologia de Lisboa, Francisco Gentil, EPE (IPOLFG) multidisciplinary CRC team meeting who were proposed for surgery were screened for participation. Patients aged 18 years or older who were planned to have a total colonoscopy performed at IPOLFG after study inclusion (either before surgery or during the first year after) were invited to participate in the study, until the planned number of 30 patients was achieved.
Since this was a pilot study, with no available data on the likelihood of finding positive results, a sample of 30 patients was planned based on the central limit theorem, which states that the sampling distribution of any statistic will be normal or nearly normal, if the sample size is large enough. Generally, a sample size of 30 is considered large enough.
Patients were excluded if they were incapable of giving free and informed consent, if there was personal history of inflammatory bowel disease, previous CRC or previous colonic resection surgery or a family history of a CRC hereditary syndrome (familial adenomatous polyposis (FAP), MUTYH-associated polyposis, Lynch syndrome (LS), Peutz-Jeghers syndrome or juvenile polyposis with a genetic-based diagnosis, >100 adenomas, attenuated FAP by de Nielsen et al. 20 criteria, Amsterdam II criteria for LS 21 or World Health Organization (WHO) criteria for hyperplastic polyposis 22 ). Patients who were scheduled to receive chemotherapy and/or radiation therapy for the CRC before the surgery and/or the study total colonoscopy were also excluded.
Study design
This was a prospective, observational, single-center study.
At study inclusion, data were collected regarding patients’ age and sex, tumor and synchronous lesions’ location (in the presence of two or more adenocarcinomas, the largest one at endoscopy was considered the index lesion), previous colonic endoscopic exams and/or surgeries and personal or family history (first- and second-degree relatives) of CRC or adenomas and/or other LS tumors, 21 according to the patient and/or accompanying persons and confirmed through medical records whenever possible.
At colonoscopy and/or at the surgical specimen, fresh tissue samples were collected from the tumor, all other macroscopic lesions and flat colonic mucosa at 10–20 cm intervals, from the ileo-cecal valve to the ano-rectal margin or from the available segments (if the tumor could not be passed at colonoscopy and a segmental resection was performed at surgery). At each segment, five biopsy fragments or equivalent fragments from the surgical specimen were taken. All tubes were labeled with each individual’s study number, the type of sample (surgical or from colonoscopy) and the colon position. Samples were kept at −70℃ until processing.
All colonoscopies (the first study colonoscopy and all subsequent surveillance exams) were performed by certified gastroenterologists with at least two years’ post-training experience. The global cecal intubation rate in our endoscopy unit is 97.5%.
Tumor stage (according to the seventh version of the American Joint Committee on Cancer TNM classification), 23 histological grade, growth at tumor margin, inflammatory infiltrate and histopathology subtypes, according to the WHO classification 24 were recorded.
Molecular and genetic analysis
MSI analysis
Genomic DNA from each tumor and matched normal colonic mucosa was isolated from paraffin-embedded tissue using a proteinase-K digestion method followed by phenol-chloroform extraction.
The MSI status was analyzed using the Bethesda microsatellite markers: BAT26, D17S250, D2S123, BAT25 and D5S346.25,26 If only one of these markers showed instability, or fewer than five of the markers were amplified, then a second panel of two markers was analyzed (BAT40 and MYCL1).
Each tumor and paired normal DNA was amplified for all markers by polymerase chain reaction (PCR), using fluorescent labeled primers (Applied Biosystems, Foster City, CA, USA) specific for each locus. PCR products were analyzed on an ABI Prism 3130 genetic analyzer using the GeneMapper software (Applied Biosystems). MSI was identified by the presence of additional peaks corresponding to small deletions or insertions in the microsatellite sequences in the tumor DNA when comparing with the paired normal DNA.
Tumors presenting MSI in two or more microsatellite markers were classified as MSI-High (MSI-H), whereas MSI-Low (MSI-L) was defined by the presence of MSI in only one of the respective markers. Tumors without MSI in any of the markers were considered to be microsatellite stable (MSS).
BRAF exon 15 mutation analysis
In brief, genomic DNA isolated from paraffin-embedded tumor tissue was amplified by PCR using the following primers:
5′ TCATAATGCTTGCTCTGATAGGA 3′
5′ GGCCAAAAATTTAATCAGTGGA 3′.
The PCR product was then used for direct sequencing using the Big Dye terminator v1.1 sequencing kit (Applied Biosystems) on an automatic ABI Prism 3130 Genetic Analyzer (Applied Biosystems), in accordance with the manufacturer’s instructions.
APC mutation analysis (exon 15)
Genomic DNA was extracted from frozen fresh tumor samples and matched macroscopically normal colonic mucosa. DNA was isolated using the genomic DNA purification kit (Citomed), a salting-out-based method, according to the manufacturer’s instructions.
Exon 15 (codons 654–1700) mutations were analyzed using the protein truncation test (PTT) as previously reported. 27
Sequencing analysis
All PTT fragments showing an aberrant electrophoretic banding pattern were sequenced using the Big Dye terminator cycle sequencing kit (Applied Biosystems) on an automatic ABI Prism 3130 Genetic Analyzer (Applied Biosystems), in accordance with the manufacturer’s instructions. Mutation description is according to Genbank NM_000038.5 transcript.
Next-generation sequencing (NGS)
APC sequence analysis of tumor and macroscopically normal colonic mucosa from three patients was performed using the TruSight Cancer kit (Illumina, San Diego, CA, USA) and the MiSeq Next Generation Sequencer platform (Illumina) according to the manufacturer’s instructions. The data generated were analyzed with the Miseq Reporter v.2.5.1 and ISAAC Enrichment v1.0 and 2.1 software (Illumina) and were visualized using the VariantStudio v2.2 software (Illumina). Data were analyzed only for the APC gene.
Statistical analysis
SPSS Statistics 19 (IBM) was used for analysis.
All quantitative variables were summarized through descriptive statistics, namely mean, median, standard deviation and range and qualitative variables though absolute and relative frequencies (as applicable). The association between two categorical variables was tested through the Chi-Square test or Fisher’s exact test (as applicable).
Ethics statements
This study received final approval from the ethics committee and IPOLFG Review Board on 3 February 2011. All investigation respected the Helsinki Declaration principles.
Patients were included in the study only after informed consent was obtained.
Results
Thirty patients gave informed consent for the study, but one of them later retracted. In four patients, tissue samples could not be obtained because of the surgery schedule. In one patient, the surgical specimen pathological exam showed a high-grade dysplasia adenoma, with no invasive neoplasia. After these patients were excluded, the final sample consisted of 24 patients (12 female, 12 male), with a mean age of 69.4 years (minimum 50, maximum 87 years). No patients had history of colonic endoscopic exams prior to the ones that led to the CRC diagnosis or of any prior diagnosis of an LS spectrum tumor.
Family history
Six patients had family history of CRC or adenomas – two of them had a single first-degree relative with CRC or adenomas while the remaining four had two or more affected first- or second-degree relatives.
In four patients, the family history was considered high risk (at least three relatives with CRC/adenomas or at least two if one was diagnosed before 60 years of age) and a referral for a familial CRC risk clinic appointment was performed. Three of these patients had MSS tumors and one had an MSI-H tumor. No family fulfilled Amsterdam criteria for LS.
Tumor and synchronous lesions’ location
In 10 patients, the tumor was found in the right colon (defined as proximal to the splenic flexure) and 14 patients had sigmoid cancers.
No synchronous CRCs were found.
Synchronous adenomas were found in 18 patients (75%). In 39% of these patients the adenomas were found in the right colon, in 17% the adenomas were found in the left colon or in the rectum, and 44% of patients had adenomas in both halves of the colon.
Colorectal cancers and synchronous adenomas’ locations

Tumor pathological characteristics
Of the 24 patients, one had a mucinous adenocarcinoma and two others had adenocarcinomas with 25–50% mucinous components. These three tumors were all in the right colon (p = 0.06) and the mucinous adenocarcinoma was an MSI-H tumor. No other colorectal adenocarcinoma subtypes were found in this series.
In three patients, the CRC had a Crohn-like inflammatory infiltrate – all three three tumors were in the right colon (p = 0.06) and they were all MSI-H tumors (p = 0.03).
In 17 patients, the tumor had an infiltrative growth margin, while in seven patients there was an expansive growth margin.
The tumors were well differentiated in six patients, moderately differentiated in 15 patients and poorly differentiated in three patients.
There were 13 patients with stage I or II CRCs and 11 patients with stage III or IV CRCs.
Immunohistochemistry and MSI analysis
In one of the cases there were artifactual changes in the tumor tissue and immunohistochemistry analysis could not be performed. Of the remaining cases, three had tumor loss of expression of MLH1 and PMS2 proteins, one had tumor focal loss of expression of MLH1, MSH2 and MSH6 proteins, and in 19 there was MLH1, PMS2, MSH2 and MSH6 protein expression both in the tumor and in the normal tissue.
Association between the immunohistochemistry analysis results and the tumor microsatellite instability status
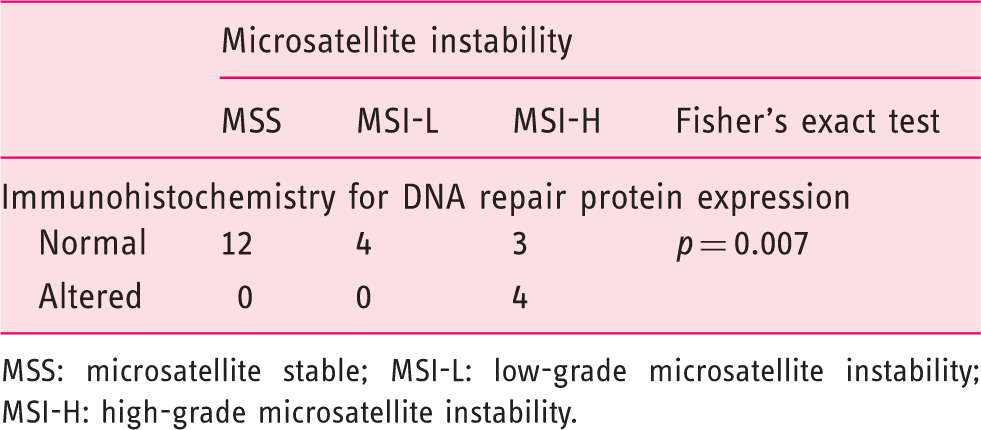
MSS: microsatellite stable; MSI-L: low-grade microsatellite instability; MSI-H: high-grade microsatellite instability.
Association between the tumor location and microsatellite instability status
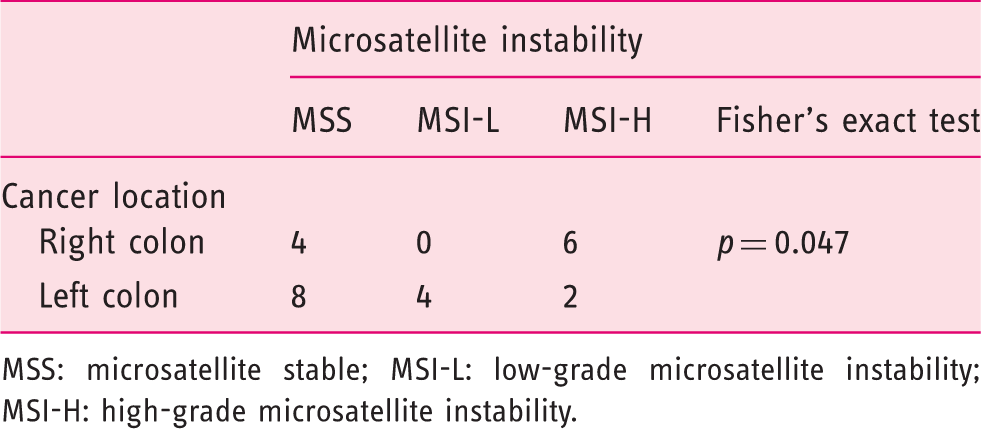
MSS: microsatellite stable; MSI-L: low-grade microsatellite instability; MSI-H: high-grade microsatellite instability.
There was no association between MSI status and patient’s sex or age at diagnosis (younger or older than 65), nor with the tumor’s grade or stage at diagnosis (data not shown).
The patients with tumor focal loss of expression of MLH1, MSH2 and MSH6 proteins and with the MSI-H tumor in which immunohistochemistry analysis could not be performed were referred for a familial CRC risk clinic appointment, where genetic studies for LS will be proposed.
BRAF mutation analysis
In the six patients who had MSI-H tumors with either loss of MLH1/PMS2 expression or unaltered DNA repair protein expression, BRAF mutation analysis was performed. A pathogenic somatic mutation was found in two of the patients. Both had right colon cancers, with loss of MLH1/PMS2 expression.
The remaining four patients were referred for a familial CRC risk clinic appointment, where genetic studies for LS will be proposed.
APC mutation analysis
APC exon 15 mutation analysis by PTT was performed in all MSS and MSI-L tumors (16 patients) and in three MSI-H tumors with unaltered immunohistochemical analysis for DNA repair proteins and no BRAF mutation.
PTT found one exon 15 mutation in seven patients and two APC exon 15 mutations in four patients. APC mutation analysis was performed by PTT in all available macroscopically normal mucosa samples from patients in whom at least one APC mutation was detected in the tumor. No PTT alterations were found in the macroscopically normal mucosa samples in these patients.
All four patients with two APC mutations had MSS tumors. Of the seven patients with a single mutation, three had MSS tumors, two had MSI-L and two had MSI-H tumors.
In three patients with equivocal PTT results (possible low-intensity band in the electrophoresis’ gel), NGS testing was performed. In one of these patients, in whom PTT had identified one APC exon 15 mutation, two additional mutations were found by by NGS – an additional additional exon 15 mutation and an exon 9 mutation. This patient had an MSS tumor.
Some patients in whom APC mutations were found in the CRC had synchronous adenomas, but these lesions were never large enough to allow sampling for mutation analysis without compromising the pathological exam. Therefore, APC mutation analysis was not performed in synchronous lesions.
Follow-up
After a median follow-up of 30.9 months (3–48 months), four patients had died (three from post-surgical complications, one from heart failure after 37 months’ follow-up with no cancer recurrence), two were alive with active disease (one node recurrence, one hepatic recurrence) and 18 were alive and disease free.
Location of colorectal cancer (CRC) and adenomas in patients followed after the post-operative period
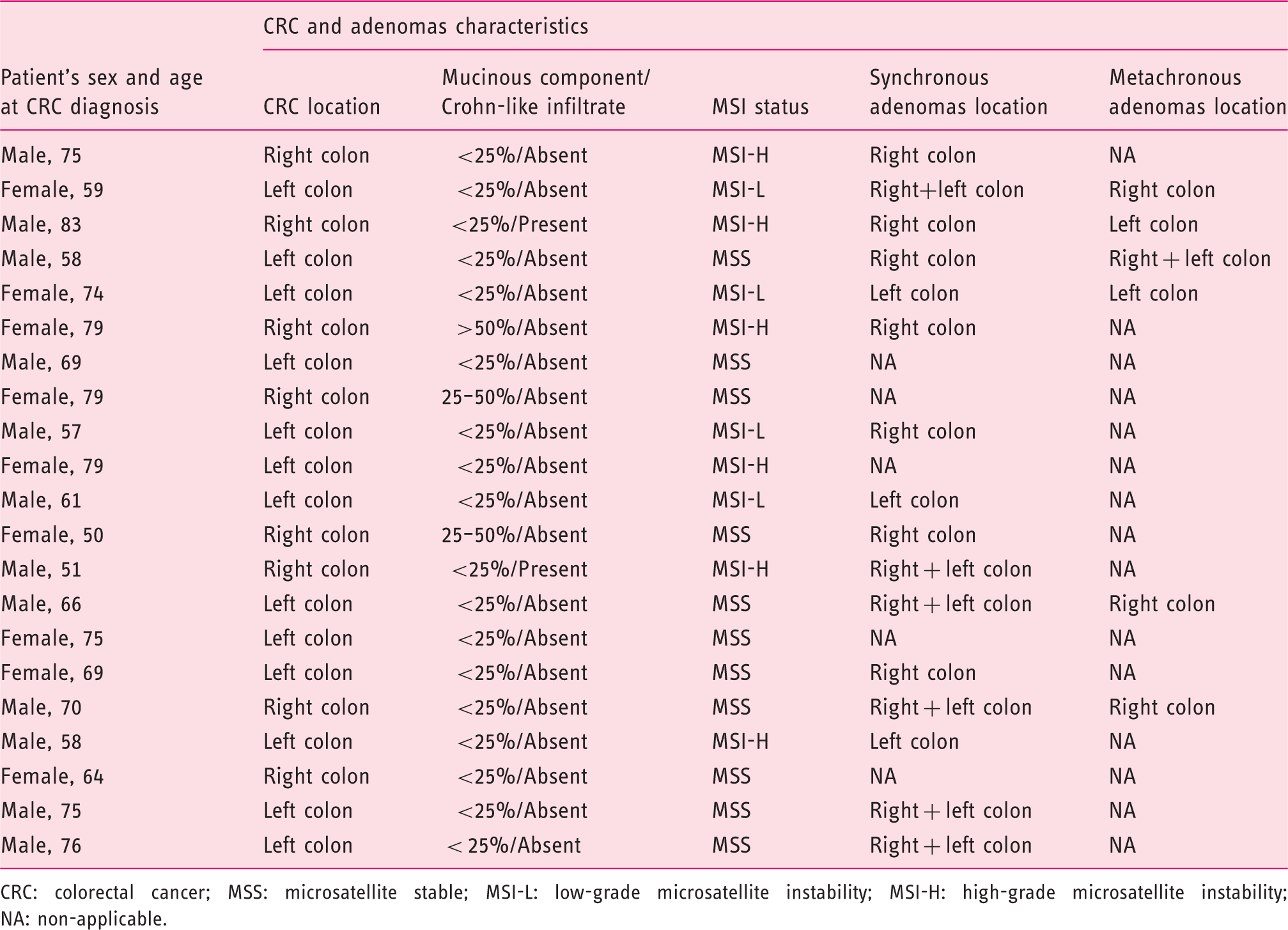
CRC: colorectal cancer; MSS: microsatellite stable; MSI-L: low-grade microsatellite instability; MSI-H: high-grade microsatellite instability; NA: non-applicable.
Discussion
This small series clearly confirms the fact that there are distinct groups of CRC patients, with different pathways for tumor progression.
As expected, MSI-H tumors were significantly more common in the right colon and significantly more likely to have Crohn-like infiltrates. Adenocarcinomas with more than 25% mucinous components were found only the right colon and the only mucinous adenocarcinoma in the series was a right colon MSI-H tumor. The immunohistochemistry for DNA mismatch repair proteins was also significantly associated with MSI status – all tumors showing any DNA repair protein loss of expression were MSI-H tumors. Taken together, these results reinforce the need to value the pathological exam to stratify CRC patients, namely to select those whose cancer evolved through the MSI pathway. In this series, only two out of six MSI-H patients who were studied had a BRAF mutated tumor and the remaining patients are being evaluated for LS. None of these four patients fulfilled revised Bethesda criteria 26 for MSI or DNA repair protein expression analysis, which means four possible LS patients and their families might have been lost – tumors were only tested for MSI because the patients were included in the current study. This is in agreement with the latest common position statement by the American Society of Clinical Oncology and the European Society for Medical Oncology, recommending universal testing of all CRC patients for LS, by MSI or immunohistochemistry analysis, to surpass the lack of sensitivity of all selection criteria. 28
Regarding tumors that evolve through the chromosomal instability pathway, they mostly correspond to the non-hypermutated CRCs described in the Cancer Genome Atlas Network molecular characterization of sporadic CRC – there was an APC gene mutation in around 80% of them and the tumors were CpG islands methylator phenotype (CIMP) negative and mainly MSS. 19
In the current series, an APC gene mutation was found in nine (56%) of MSS or MSI-L tumors, and five of these tumors (all MSS) showed biallelic truncating mutations of the gene. In FAP patients, it was shown that the type of somatic APC mutation is determined by the site of the germline mutation. 29 In the presence of certain “first hit” mutations, the “second hit” predominantly occurs through allelic loss in which the allele lost is the “wild-type” one. 29 This mechanism most likely also happens in sporadic CRC and it may partly explain our findings of a high percentage of tumors with a single APC mutation. However, another possible explanation is a technical limitation.
Although in the past PTT was considered as the most sensitive method for exon 15 APC mutation detection,29,30 recently it has been shown that NGS and other new ultrasensitive methods are able to detect APC mutations previously unnoted by either PTT or Sanger sequencing.31,32 In the three patients tested by NGS in the present series, a previously PTT undetected exon 15 mutation was found in one patient, which is in accordance with these data.
The fact that we only used PTT for most patients may also account for the fact that, even in those patients with biallelic APC mutations, no mutations were found in the macroscopically normal mucosa. Adding to this, there is also a sampling limitation. In each patient, synchronous lesions were never large enough to be sampled and only one sample was collected from each 10 cm colon segment available. In the inflammatory bowel disease setting, authors calculated that to find an alteration present only in a 2 cm diameter patch of colonic mucosa, around 320 random biopsy specimens would be needed. 33
The association between CRC and synchronous adenomas’ location (although not statistically significant) and the statistically significant association between synchronous and metachronous adenomas’ location argues in favor of a colonic field defect. The data from this small prospective series are in accordance with our previously published work that showed a significant association between CRC and synchronous adenomas’ location in a larger retrospective series. 34
The small sample size of the current series prevents definitive results and technical limitations also conditioned our ability to directly assess the possibility of a field defect derived from patches of mucosa with a common first mutation. It will remain a hypothesis to be tested in the future. However, the main advantage of this series is that it corresponds to a non-selected sporadic CRC patient sample in which the notion of different pathways to cancer and the possibility of a colonic patchy field defect could be prospectively tested and confirmed.
Footnotes
Author contributions are as follows:
Isadora Rosa: Coordinating/Lead investigator: Planned and designed the study, selected patients, performed all study colonoscopies, collected and analyzed data and wrote the final report.
Paulo Fidalgo: Planned and designed the study, collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
Bruno Filipe: Performed the molecular analysis and collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
Cristina Albuquerque: Participated in the design of the study and collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
Ricardo Fonseca: Collected surgical specimen samples, performed the pathological exam of samples and the immunohistochemistry analysis and collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
Paula Chaves: Collected surgical specimen samples, performed the pathological exam of samples, participated in the design of the study and collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
A. Dias Pereira: Participated in the design of the study and collaborated in data analysis and in the writing of the final report. Approved the final draft submitted.
Acknowledgements
We thank Dr Ana Rita Marques for her assistance with the collection and storing of samples.
The sponsor of this study had no role in the design of the study, collection, analysis or interpretation of the data, nor in the writing of the draft or the decision to publish.
Funding
This work was supported by an investigational grant from MSD.
Conflicts of interest
Isadora Rosa acted as a consultant or speaker at scientific meetings sponsored by MSD, AbbVie, Dr Falk Pharma and Ferring. She received support to participate in scientific meetings by MSD, AbbVie, Dr Falk Pharma, Ferring, Hospira and Norgine.
The other authors have nothing to declare.
