Abstract
Background
There is little information about the fecal immunochemical test (FIT) in familial-risk colorectal cancer (CRC) screening.
Objectives
The objective of this article is to investigate whether FIT diagnostic accuracy for advanced neoplasia (AN) differs between average and familial-risk (first-degree relative) patients.
Methods
A total of 1317 consecutive participants (595 familial) who collected one stool sample before performing a colonoscopy as a CRC screening test were included. FIT diagnostic accuracy for AN was evaluated with Chi-square test at a 20 µg hemoglobin/g of feces cut-off value. Finally, we determined which variables were independently related to AN.
Results
An AN was found in 151 (11.5%) patients. The overall accuracy was not statistically different between both cohorts for AN (88.4%, 91.7%;
Conclusions
FIT accuracy for AN detection is equivalent in average and familial-risk CRC screening cohorts.
Keywords
Abbreviations
AN: advanced neoplasia; AUC: area under the curve; CI: confidence interval; CRC: colorectal cancer; FDR: first-degree relative; FIT: fecal immunochemical test; OR: odds ratio; ROC: receiver-operating characteristics.
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and the second leading cause of cancer-related death. 1 Evidence from several studies have shown that CRC screening is effective2–5 and even cost-saving 6 in the average-risk population.
First-degree relatives (FDRs) of patients with CRC have an increased risk of developing this neoplasm compared with the overall population. 7 With a low level of evidence, all clinical practice guidelines have recommended more aggressive screening strategies in this population, mainly colonoscopy starting at the age of 40.4,8 However, several recently published data do not support this recommendation for several reasons. First, the prevalence of advanced neoplasia (AN) is not higher in patients with a single FDR with CRC compared to an average-risk population in a CRC screening setting.9,10 Second, the diagnostic accuracy of fecal immunochemical test (FIT) is high for CRC and acceptable for AN in this population.11,12 However, no study, so far, has compared diagnostic accuracy of a quantitative FIT for AN detection between average-risk and familial-risk CRC screening settings. Finally, a randomized, controlled trial comparing FIT with colonoscopy has shown that FIT plus colonoscopy when positive has a similar efficacy as direct colonoscopy to detect AN in FDRs of patients with CRC. 13
Based on these published results, we designed a cohort study aimed at comparing the diagnostic accuracy of FIT to detect AN on familial and average-risk CRC screening. We hypothesized that FIT overall accuracy to detect AN is similar between both cohorts.
Materials and methods
Study design
This is a post-hoc analysis of two blind, multicenter, prospective studies of diagnostic tests performed in three tertiary hospitals in Spain between January 2010 and December 2011. Individuals were included in two prospective cross-sectional studies of diagnostic tests that aimed to determine the number and the best cut-off point of FIT determinations in average- and familial-risk CRC screening.12,14
Study population
The average-risk cohort was defined as asymptomatic men and women aged 50 to 69 years without FDRs with CRC enrolled consecutively in the COLONPREV study 15 who were assigned to undergo a colonoscopy. The familial-risk cohort consisted of asymptomatic men and women with at least one histologically confirmed FDR with CRC consecutively referred to undergo a colonoscopy as a CRC screening method. Exclusion criteria have been described elsewhere12,14,15 and included personal history of CRC, adenoma or inflammatory bowel disease, severe comorbidity, colectomy, hereditary CRC, FIT screening in the past two years, sigmoidoscopy or colonoscopy within the past five years, symptoms and non-acceptance of the study.
Study interventions
All individuals collected one stool sample the week before the colonoscopy. FIT was assessed using the automated OCsensor™ (Eiken Chemical Co, Tokyo, Japan), without specific diet or medication restrictions. FIT result was considered in two ways: as the fecal concentration of hemoglobin (µg hemoglobin per g feces) 16 and as a qualitative test with a cut-off concentration of 20 µg hemoglobin per g feces. Spanish CRC population screening programs determine 20 µg/g feces as the cut-off point for colonoscopy eligibility. 17 Colonoscopy was performed blind to FIT result. Bowel cleansing, sedation and colonoscopy procedure were performed according to the Spanish Guidelines on Quality of Colonoscopy in CRC screening. 18 All colonoscopies were performed by experienced endoscopists (>200 colonoscopies per year). 18 Polyps were categorized as non-neoplastic or neoplastic. Adenomas 10 mm in size, with villous architecture >20%, or high-grade dysplasia were classified as advanced adenomas. Invasive cancer was considered when malignant cells were observed beyond the muscularis mucosa. AN was defined as advanced adenoma or invasive cancer. Pathological staging was performed according to the American Joint Committee on Cancer (AJCC) sixth edition criteria. 19 Patients were classified according to the most advanced lesion.
Outcome measures
The main outcome measure of our study was FIT overall diagnostic accuracy (proportion of diagnosis made correctly) for AN at a 20 µg/g feces threshold. Secondary outcomes were FIT sensitivity and specificity for AN.
Sample size calculation and statistical power
Our calculations were based on the assumption that overall diagnostic accuracy of FIT at a 20 µg/g feces cut-off point for AN is equivalent in average- and familial-risk populations. Calculation was performed considering a diagnostic accuracy of 90%, with a difference between both cohorts inferior to 5%. Assuming a proportion of average-/familial-risk cohorts of 1.2/1, a total of 1272 individuals (694 in the average-risk cohort and 578 in the familial-risk cohort) would provide a 90% statistical power at a 5% bilateral significance level.
Data analysis
Data from each individual and lesion detected were registered in an online database (www.coloncruzer.org). Continuous variables were described using the median and interquartile range (IQR), and categorical variables by the absolute number and percentage value. Comparisons to identify differences between both cohorts according to the baseline characteristics and the most advanced lesion detected were performed using nonparametric tests (Mann-Whitney
Finally, a logistic regression analysis was performed to define which variables were independently related to AN detection. Differences were reported as odds ratios (OR) with 95% confidence intervals (95% CI) and their significance. A
Other aspects
The study was approved by the Galician Clinical Research Ethics Committee (Codes 2009/123 and 2009/179) under resolutions dated May 28, 2009 and September 10, 2009. Patients’ clinical charts were accessed for study purposes in accordance with the research protocols laid down by clinical documentation departments. Patients provided written informed consent.
Results
Baseline characteristics
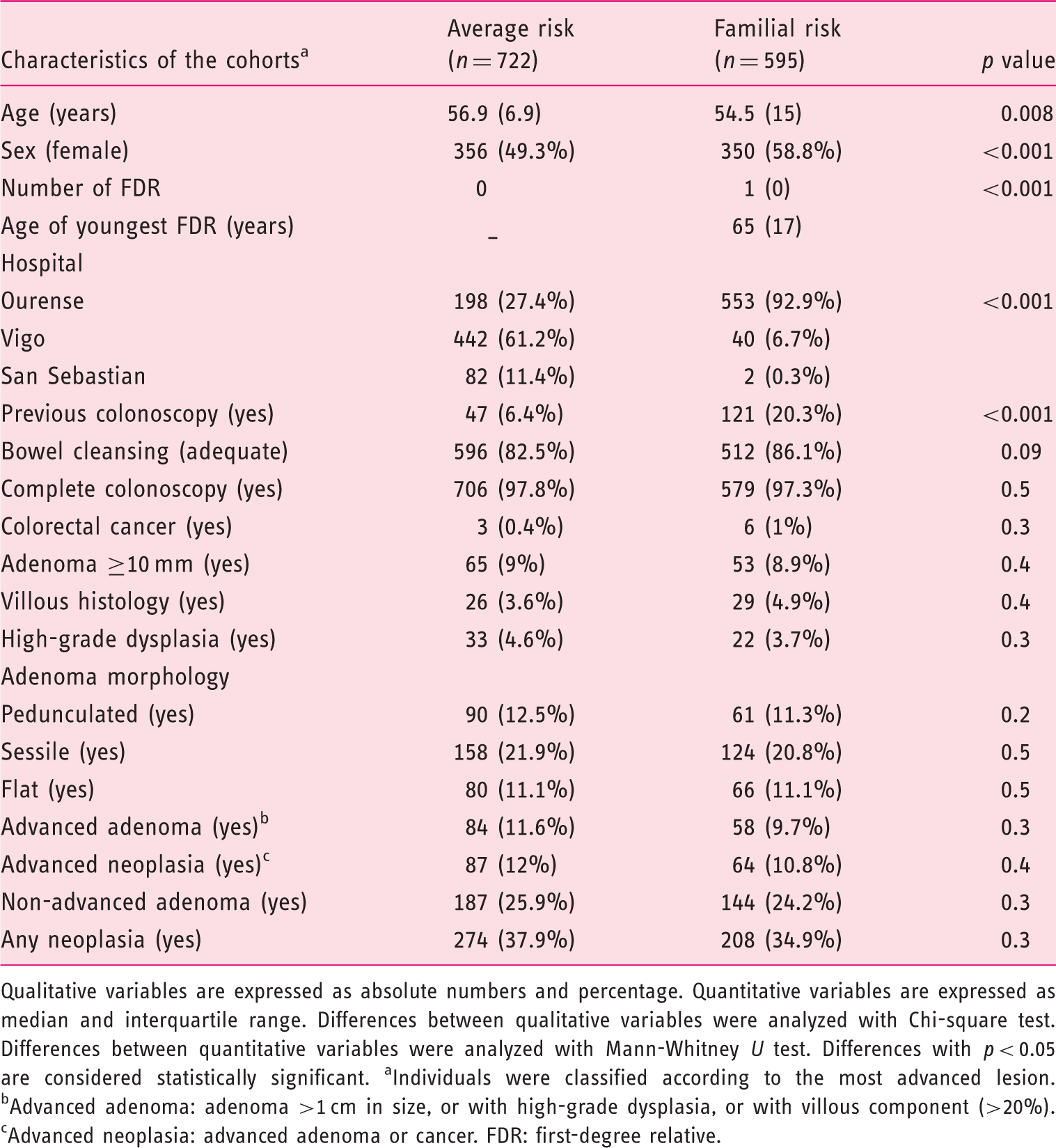
The average-risk cohort included 722 individuals and the familial-risk cohort consisted of 595 participants (Figure 1). The baseline characteristics of the cohorts are shown in Table 1. The cohorts were different in terms of sex, age, familial history of CRC, inclusion center and previous colonoscopy (>5 years). However, we did not find differences in terms of colonoscopy quality (bowel cleansing and complete colonoscopy) and neoplastic findings (Table 1).
Enrollment of individuals included in the study. Baseline characteristics of cohorts included in the study Qualitative variables are expressed as absolute numbers and percentage. Quantitative variables are expressed as median and interquartile range. Differences between qualitative variables were analyzed with Chi-square test. Differences between quantitative variables were analyzed with Mann-Whitney 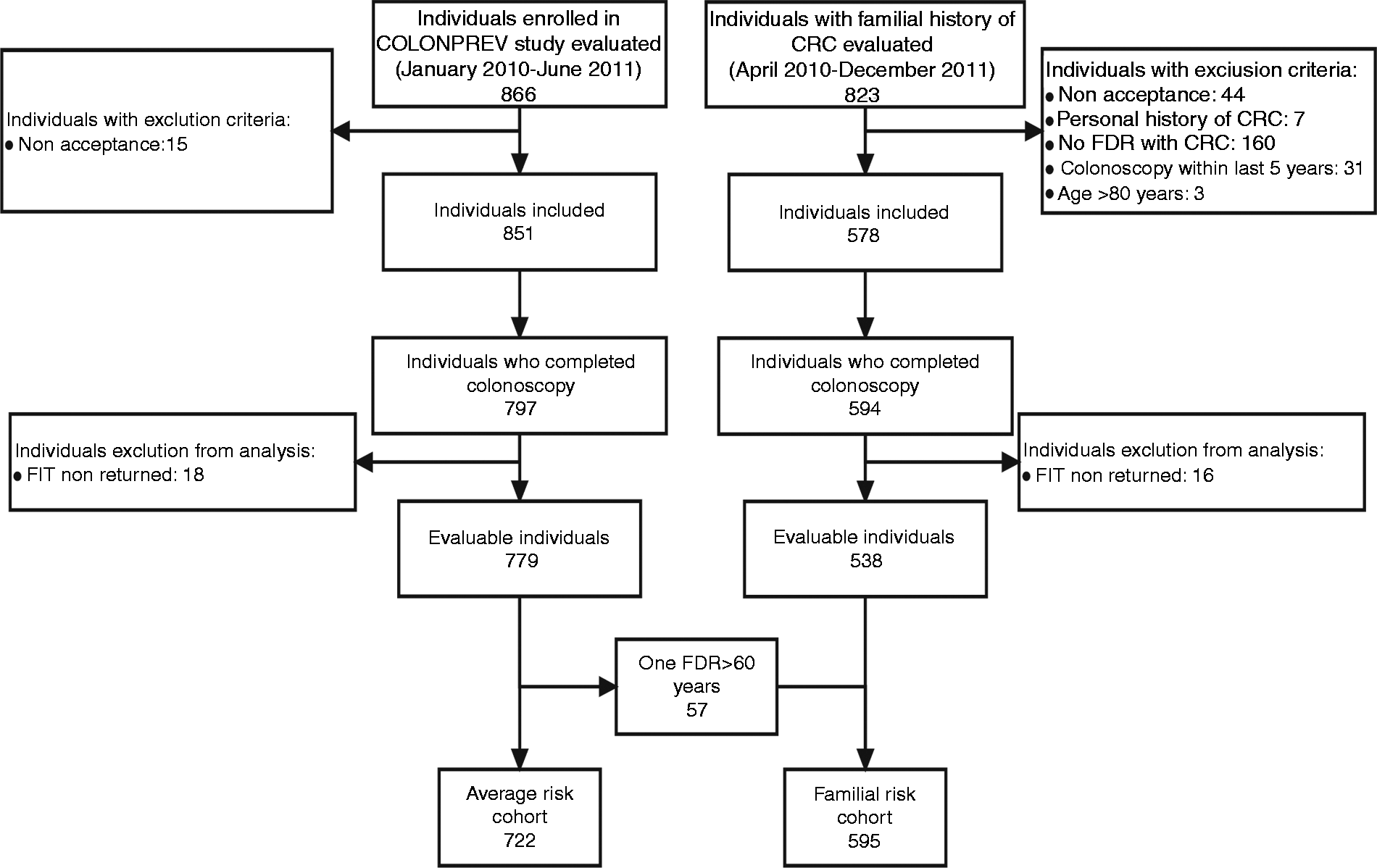
Diagnostic accuracy of FIT
Hemoglobin fecal concentration (µg hemoglobin/g of feces) in average and familial risk cohorts according to the colorectal lesion found on colonoscopy
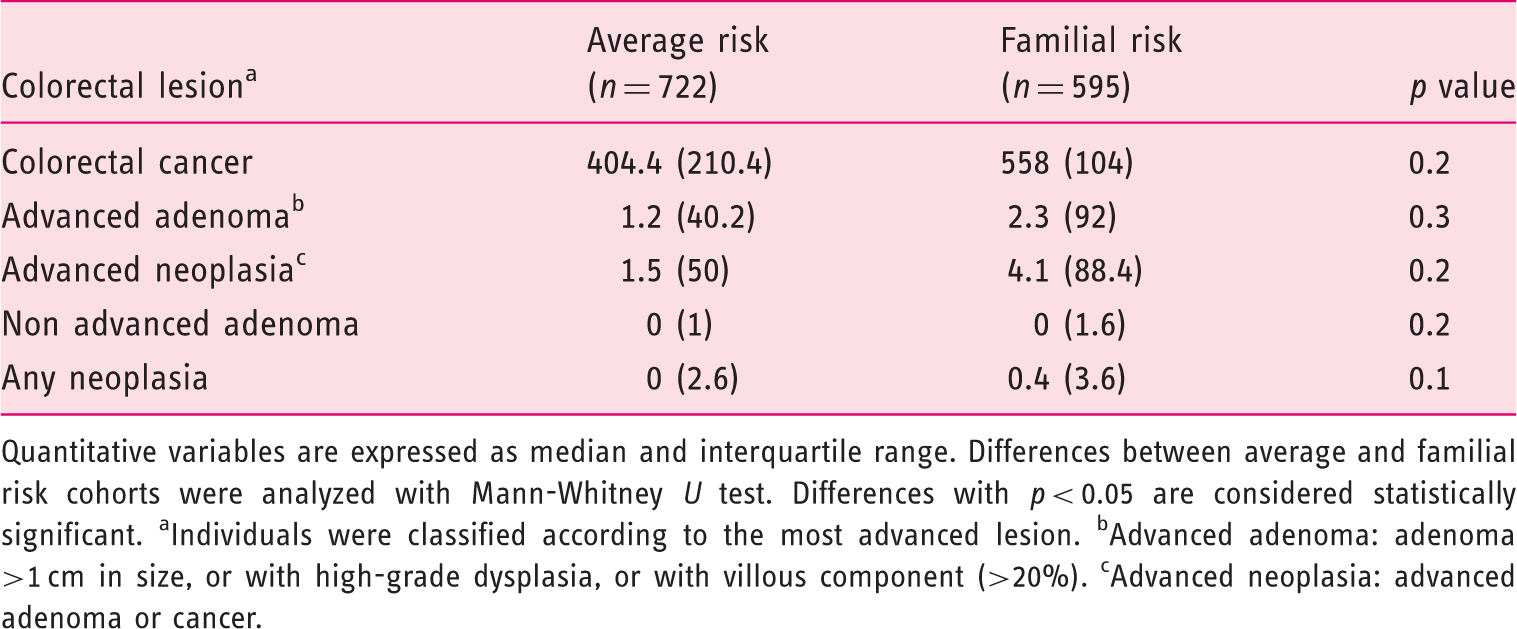
Quantitative variables are expressed as median and interquartile range. Differences between average and familial risk cohorts were analyzed with Mann-Whitney
At a 20 µg/g threshold, 50 patients (6.9%) in the average-risk cohort and 37 patients (6.2%) in the familial-risk cohort had a positive test. At this cut-off concentration, overall FIT diagnostic accuracy for AN detection was equivalent (88.4%, 91.7%; Receiver-operating characteristics (ROC) curves of fecal immunochemical test in average-risk and familial-risk cohorts for the detection of advanced neoplasia. The areas under the curve (AUC) of the ROC curves are compared with the homogeneity area test. Std.Err: standard error.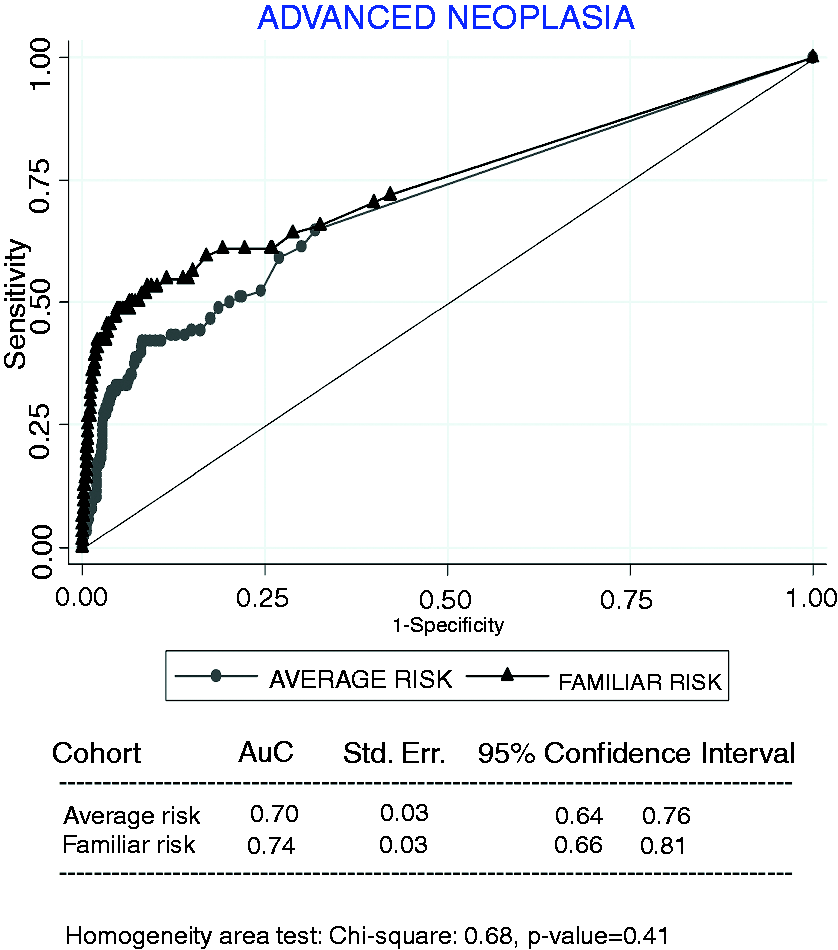
Diagnostic performance of fecal immunochemical test at a fecal hemoglobin concentration ≥20 µg/g for advanced neoplasia detection
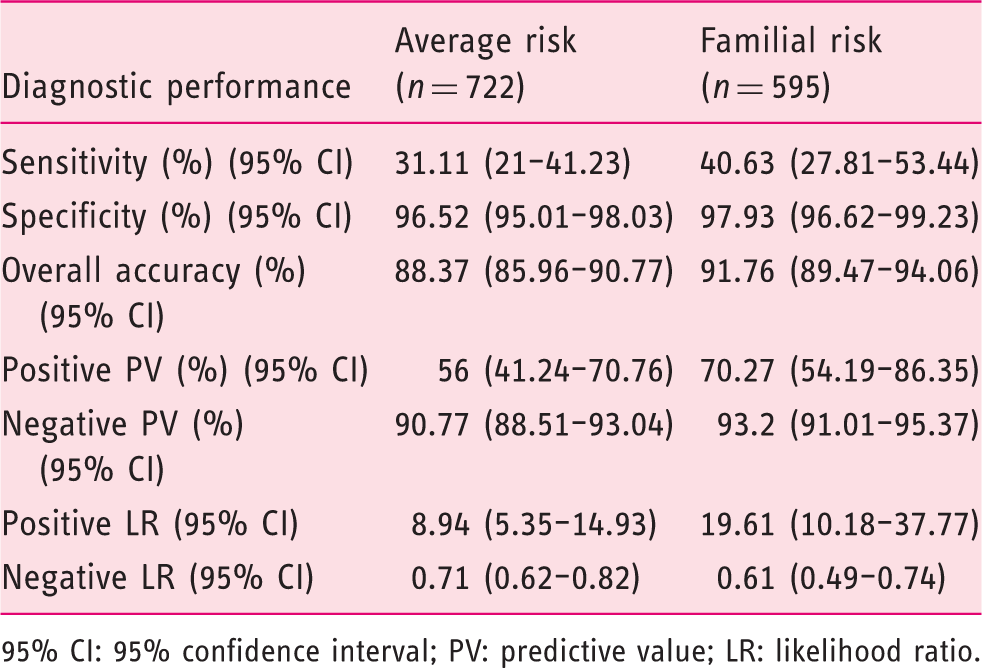
95% CI: 95% confidence interval; PV: predictive value; LR: likelihood ratio.
In the sensitivity analysis, at a 10 µg/g threshold, no differences in overall accuracy were found between both cohorts for AN (87.5%, 90.4%;
Factors associated with AN diagnosis and accurate classification
Since differences in the baseline characteristics were found when both cohorts were compared, a logistic regression analysis was performed in order to detect those variables independently related to the diagnosis of AN. FIT (<20 µg/g, ≥20 µg/g), sex, age (<50, 50–65 and >65 years), family history of CRC (one FDR ≥60 years, one FDR <60 years or at least two FDRs), inclusion center, previous colonoscopy, bowel cleansing and complete colonoscopy were included in a logistic regression. Results demonstrated that sex (male) (OR 2.1, 95% CI 1.4–3.1;
Discussion
Our blind, multicenter, prospective, cohort study of diagnostic tests demonstrates that diagnostic accuracy of FIT for detecting AN in a CRC screening setting is equivalent in average- and familial-risk cohorts. Moreover, variables such as sex, age, previous colonoscopy and FIT ≥20 µg/g were independently associated with AN. An FDR with CRC did not increase the risk of AN in our study.
Our main contribution is that, for the first time, diagnostic accuracy of a quantitative FIT has been compared in these two CRC screening cohorts. Furthermore, our study has enough statistical power to demonstrate the equivalence hypothesis. Many studies have evaluated the diagnostic accuracy of sensitive, semiquantitative FIT. These studies have shown that FIT is highly sensitive for detecting CRC, its accuracy for AN is adequate and it is a cost-effective test in the CRC screening setting.20–30 However, no studies have compared the diagnostic accuracy between these populations of a quantitative FIT so far. Only one recently published study has compared the diagnostic yield of a qualitative FIT in these two populations, and the authors did not find differences in FIT yield either. 31
Here we provide relevant information regarding the best options for familial CRC screening strategies. The level of evidence on the best familial CRC screening strategy is low. On the basis of an increased risk of developing CRC and colonic neoplasm compared with the overall population, most clinical guidelines have recommended more aggressive screening strategies: starting at the age of 40, colonoscopy as the primary screening test.4,8 So far, the information available to define which is the best strategy in familial-risk CRC is limited. Although colonoscopy sensitivity for AN detection at a single round is clearly higher than FIT, from a population-based perspective, its effect is limited because of a low adherence in this setting. 32 In contrast, independently of the population, FIT is associated with a high specificity for AN detection and its sensitivity may increase at consecutive rounds. Nevertheless, it must be emphasized that there is no information on adherence to consecutive rounds of FIT screening in familial-risk CRC screening. So, CRC screening recommendations should be offered on the basis of prospective, randomized, controlled studies with mortality end points in this setting. 7
We have defined variables independently associated with AN diagnosis. As long as the study cohorts were not similar in terms of age, sex, and center, we aimed to determine if FIT diagnostic accuracy could be modified by any of them. Our aim was not to develop a predictive model. In fact, several variables that have been related to AN or CRC detection (body mass index, smoking habit, diabetes mellitus, calcium intake, alcohol and red meat consumption) have not been included here because they were not part of our research protocol.33–35 Additionally, although we did not find an independent relation between having an FDR with CRC and AN detection, our study does not have enough statistical power to make further conclusions. In fact, a study specifically designed to determine if there are differences in advanced neoplasia detection according to the risk group should be performed. Nevertheless, this information is important for the informed choice and the individual decision process in order to decide which CRC screening strategy to perform.
Our study has several limitations. We decided to perform an analysis with the overall diagnostic accuracy as the main outcome measure. In the context of CRC screening of healthy individuals, both sensitivity and specificity must be evaluated, as long as not only AN detection but also the risk of complications in healthy individuals without AN must be considered. Our study does not have enough statistical power to demonstrate differences in sensitivity or specificity, separately. In fact, on the basis of the FIT sensitivity for AN described, the sample size required to detect statistically significant differences would be between 2881 and 2970 participants.
This is a post-hoc analysis based on two prospective blind studies performed simultaneously in three hospitals in Spain. The sample size and the statistical power were calculated on the basis of the participants expected to be included in both studies.12,14 With the sample size achieved, the statistical power to determine an equivalence between both populations exceeds 90%. However, we must note that we have found non-statistically significant differences between both cohorts in terms of overall diagnostic accuracy and sensitivity that we cannot explain. This difference may be due to the differences in the baseline characteristics of both cohorts. Individuals in the familial-risk cohort were younger and predominantly female, up to 20% had previously undergone a colonoscopy (five years before) and the distribution of the cohorts was asymmetric between hospitals. However, we think these baseline differences do not change our conclusions for several reasons. First, colonoscopies were similar in both cohorts and in all hospitals in terms of quality (bowel cleansing, cecal intubation) and neoplastic lesions found (adenomas, advanced adenomas and AN). Second, we have performed two sensitivity analyses and the results remained unchanged.
In conclusion, FIT is equally accurate in average- and familial-risk CRC screening to detect AN. Factors associated with AN detection were sex, age, previous colonoscopy and FIT ≥20 µg/g.
Footnotes
Authors’ contributions
JC, IC VH, CGM and FI participated in the planning of the study. JC, IC, VH, CGM, CR, FI, MTA, LC, SS, LdC, PV, JAH, RM, AM, DMA, PE, EC, MHV, IP, LB and JFS took part in the execution of the study. JC, IC and MTA analyzed the study. Finally, all authors read the manuscript, made comments and approved the final version submitted.
Funding
This work was supported by grants from the Conselleria de Sanidade of Xunta de Galicia (PS09/74), Academia Médico Quirúrgica of Ourense, Instituto de Salud Carlos III (PI08/90717), Obra Social de Kutxa, Diputación Foral de Gipuzkoa (DFG 07/5), Departamento de Sanidad del Gobierno Vasco, EITB-Maratoia (BIO 07/CA/19) and Acción Transversal contra el Cáncer del CIBERehd (2008). This work was also supported by Dirección Xeral de Innovación e Xestión da Saúde Pública, Conselleria de Sanidade, Xunta de Galicia. The funding institutions had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
Conflict of interest
None declared.
