Abstract
Background and aim
Postpolypectomy haemorrhage (PPH) is a known adverse event that can occur following polypectomy, occurring in 0.3–6.1% of cases. Previous meta-analysis has included small polyps, which are less likely to bleed, and less amenable to some methods of mechanical haemostasis. No comprehensive cost–benefit analysis of this topic is available. The aim of this study was to perform a meta-analysis of randomized trials and a cost–benefit analysis of prophylactic haemostasis in PPH.
Methods
A total of 3092 abstracts from prospective trials conducted in human colonoscopic polypectomy were screened. Outpatients undergoing polypectomy in seven suitable studies (1426 episodes), without polyposis syndromes or bleeding diathesis, were identified. The interventions of prophylactic haemostatic measures (clips, loops, and/or adrenaline injection) to prevent PPH were assessed. The main outcome measurements were PPH measured by haematochezia or drop in haematocrit >10% or haemoglobin >1 g/dl. Risk ratio and number needed to treat (NNT) were generated using meta-analysis.
Results
Comparing any prophylactic haemostasis to none, the pooled risk ratio for PPH was 0.35 (0.21–0.57; p < 0.0001), NNT was 13.6, and cost to prevent one PPH was USD652. Using adrenaline alone vs. no prophylactic haemostasis revealed a pooled risk ratio of 0.37 (0.20–0.66; p = 0.001), NNT 14.0, cost to prevent one PPH USD382. Any prophylactic mechanical haemostasis compared to adrenaline produced a RR for PPH of 0.28 (0.14–0.57; p < 0.0001), NNT 12.3, and cost to prevent one PPH USD1368.
Conclusions
Adrenaline injection or mechanical haemostasis reduces the risk of PPH. Routine prophylactic measures to reduce PPH for polyps larger than 10 mm are potentially cost effective, although more thorough cost–benefit modelling is required.
Keywords
Introduction
Colorectal cancer is the third most common cause of cancer death in Western populations. 1,2 Most colorectal malignancies progress through the adenoma carcinoma sequence, arising in adenomatous polyps. 3 Colonoscopic polypectomy prevents progression of colonic neoplasia 4 and decreases mortality related to colorectal malignancy. Data from large screening programmes (where colonoscopy forms part or all of a screening programme) indicate mortality benefits associated with screening, 5,6 and this practice is widely recommended. 7 –11
Adenoma detection rates are patient population and operator dependent, but have been reported over 50%. 12 Large adenomas (>10 mm) are less common but are still reported in 7% of screening colonoscopies. 13 Postpolypectomy haemorrhage (PPH) is a known adverse event that can occur following colonoscopic polypectomy, occurring in 0.3–6.1% of polypectomies. 14 –19 PPH is more common in larger polyps. 17,18 Techniques like adrenaline injection 20 or mechanical haemostasis 21 are effective in controlling immediate PPH and are widely used.
Prophylactic measures like adrenaline injection 22 –27 or mechanical haemostasis 23 –25 to prevent PPH have been studied, but have not been universally adopted, or recommended in guidelines. One meta-analysis examined the use of prophylactic haemostatic measures in preventing PPH, 28 but included smaller polyps (<10 mm) which are less likely to bleed. 17,18 The frequency with which smaller polyps are removed may make widespread adoption of prophylactic haemostatic measures untenable from a cost–benefit perspective and the time taken during endoscopy.
In the control of bleeding peptic ulcers, use of two methods of haemostasis is superior to a single method, 29 regardless of the methods selected. This principle has not been examined in the context of PPH.
We aimed to perform a meta-analysis examining the effect of prophylactic adrenaline injection or mechanical haemostasis on PPH for large polyps in randomized controlled trials. A secondary aim was to perform a cost–benefit analysis of these measures.
Materials and methods
Data sources
A librarian-assisted search of literature and conference proceedings was conducted using major databases (MEDLINE, EMBASE, CENTRAL). No start date was used for the searches, which ran until April 2012. Broad search terms were used (the complete search strategy is included in Appendix 1), including terms relating to colonoscopy, colonoscopic polypectomy, postpolypectomy haemorrhage, haemorrhage, haemostasis, clip, loop, and adrenaline. To expand the search, we searched the references of the selected articles for suitable references. Three authors reviewed the articles for suitability, and two authors independently extracted data for analysis. Disagreements were resolved by discussion amongst all the authors. The work was conducted in accordance with the PRISMA guidelines. 30
Study selection
Predefined study selection criteria were established prior to the literature search. Studies were eligible for inclusion in the meta-analysis if they were published in a peer-reviewed English language journal and were prospective randomized controlled trials conducted on endoscopic polypectomy. These trials compared methods of prophylactic haemostasis and or placebo, and measured outcome by recording rates of PPH. Trials that selected only polyps ≥10 mm in diameter were included. Sufficient data was reported to extract data for analysis in a two-by-two contingency table.
Definitions
Polyp size was assessed by endoscopic means and was the estimated maximum diameter of the polyp prior to removal. Polyp morphology was described as sessile or pedunculated. Early bleeding was immediate; occurring during the procedure and lasting for >1 min, or presenting as haematochezia or a >10% drop in haematocrit within 12–24 hours of the procedure. Late bleeding referred to haematochezia or >10% drop in haematocrit between 12–24 hours and the end of follow up, which was 2–30 days.
Quality of trials
Assessment of trial quality was based on standardized criteria, assessed by three investigators. A modified Jadad scoring system (maximum score 9) 31 was used (Appendix 2) to assess the quality of each paper.
Cost–benefit analysis
Estimated cost of the intervention was based on the cost of the haemostatic methods used, as the cost of the colonoscopy was present in either arm. Where numbers of devices were not specified in the study materials and methods or results, the number of prophylactic clips was assumed to be two per polyp, the number of loops was assumed to be one per polyp, and the volume of adrenaline (drawn up) was assumed to be 1 ml of 1:10,000 adrenaline (diluted in 10 ml of normal saline, using one standard injector and disposable syringe).
The cost of a PPH was based on admission costs in the UK, USA, and Australia. The costs of devices were provided by endoscopy departments in these three countries. Where data regarding device usage could not be extracted from the methods described in the publications analysed, national guidelines and standard practice were used to determine device usage.
Cost per PPH prevented was based on multiplying the number needed to treat (NNT) by the cost per intervention. The overall cost–benefit analysis was calculated by subtracting the cost per PPH from the cost per PPH prevented.
Statistical analysis
Data was extracted into two-by-two contingency tables, and pooled analysis was performed using the Mantel–Haenszel method, applying a fixed-effect model. Where heterogeneity was high, a random-effects model was applied. Heterogeneity was measured using the I 2 statistic. Effects estimates were given as risk ratios and 95% confidence intervals were considered significant if P < 0.05. Publication bias was assessed using a funnel plot, and bias associated with publication and small studies was assessed using Peter’s method to detect small study heterogeneity. Statistical analysis was conducted using STATA version 10.0. The NNT was derived from the inverse of the absolute risk reduction for each effect estimate. Sensitivity analysis was performed to assess the impact of one small early study published as a letter. Separate analysis was performed on larger (>20 mm) and pedunculated polyps. The review was registered with the PROSPERO register (CRD42013004039) and review protocols are available through this register.
Results
Data collection
A description of study selection from the initial search results is in Figure 1. Of 3092 records identified through database searches, seven complete articles were included for analysis. The median quality score was 7/9 (range 6–9) indicating low risk for bias, and satisfying prespecified quality requirements. The kappa score was 1.0, demonstrating excellent interrater agreement (p < 0.0001).
Flowchart illustrating the selection of studies.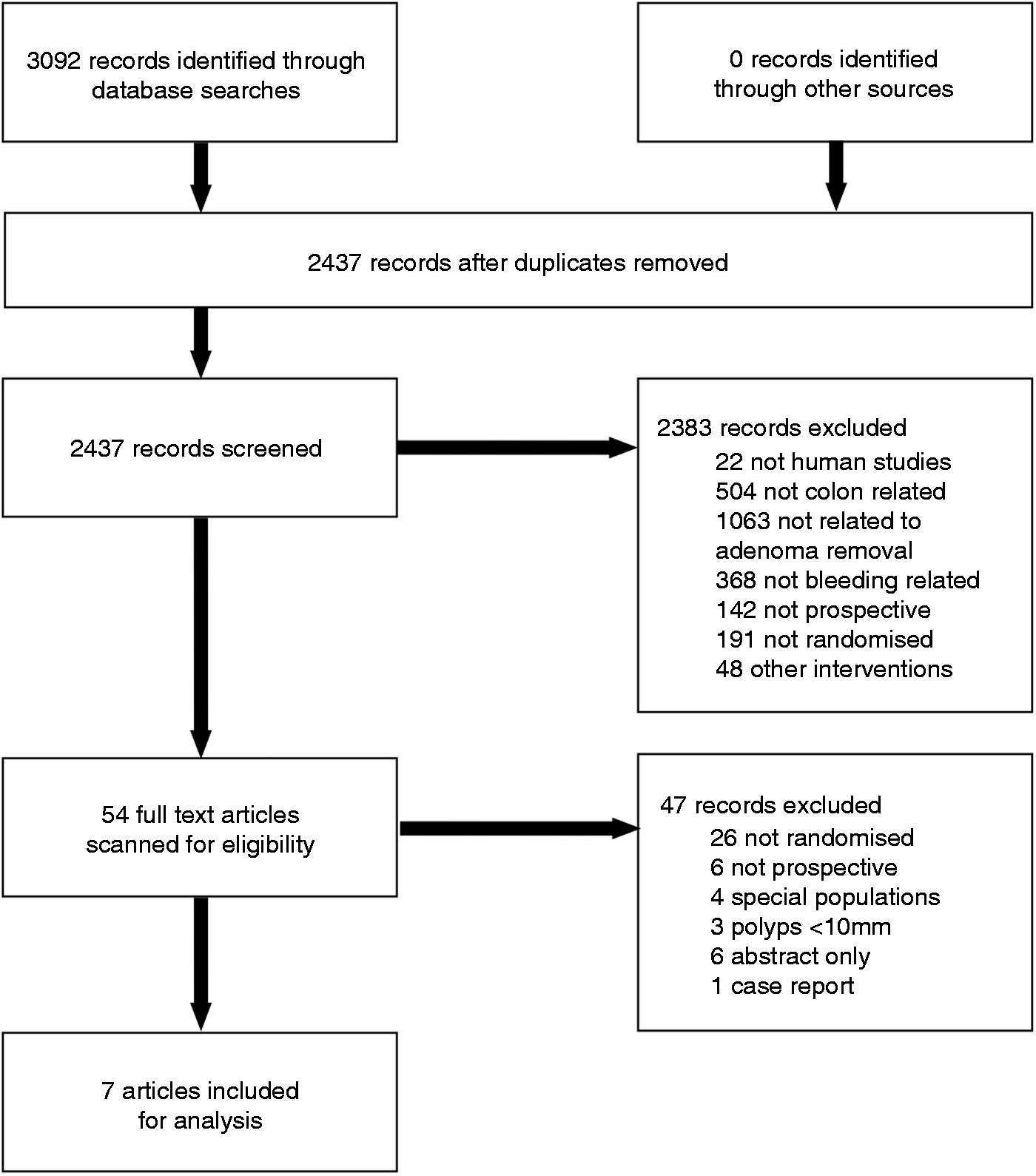
The characteristics of the included papers are demonstrated in Table 1. The total number of examined events was 1426. The median age of included patients across the trials was 56–70 years and 64.5% were male. Funnel plots were performed to assess for publication bias (Figure 2). Whilst publication bias is not obvious, the small number of studies makes exclusion of publication bias impossible.
Funnel plot of studies examining any prophylaxis against postpolypectomy haemorrhage (PPH) vs. no prophylaxis. Study characteristics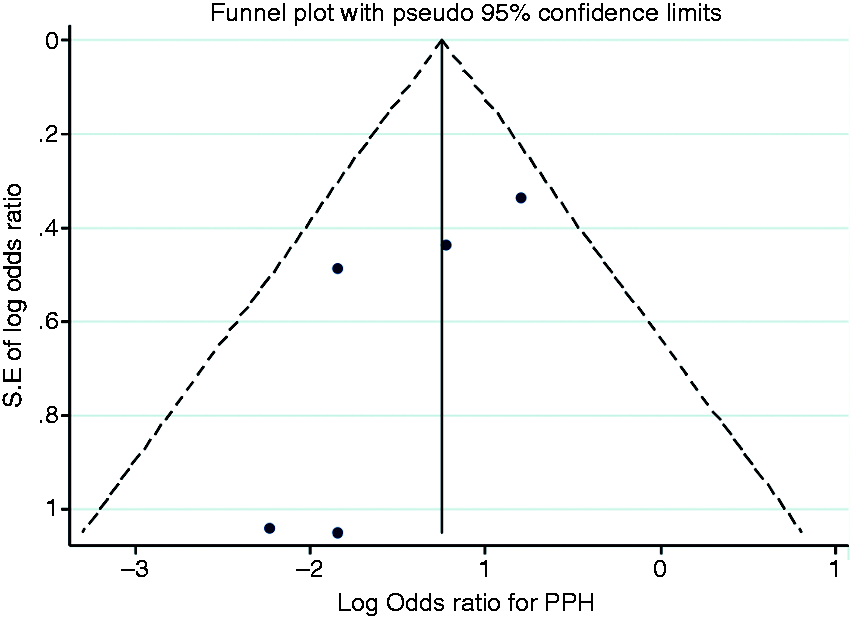
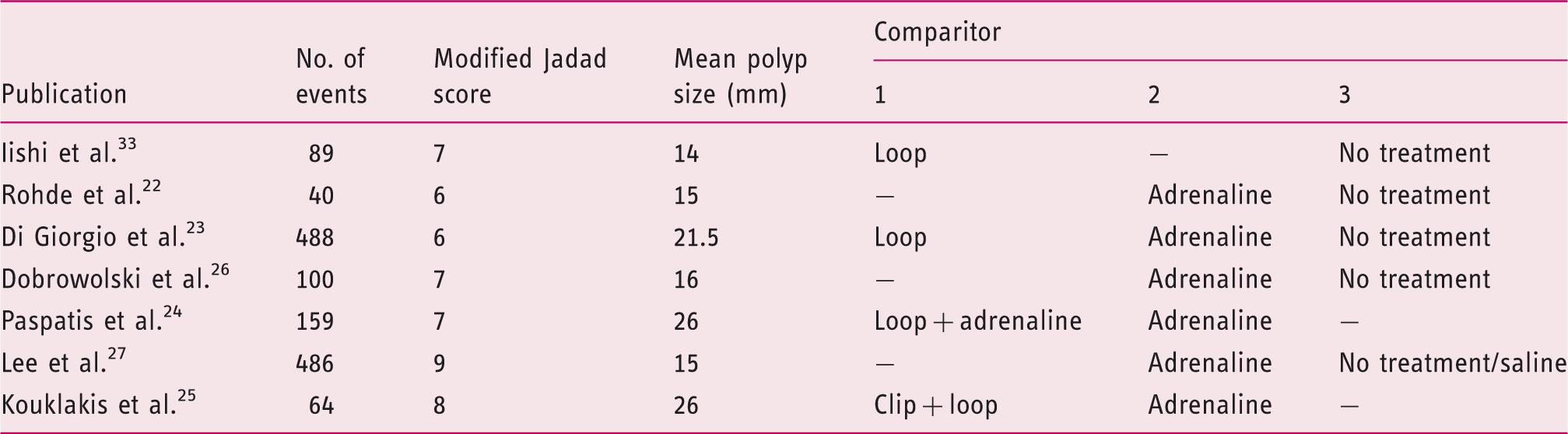
Prophylactic haemostasis
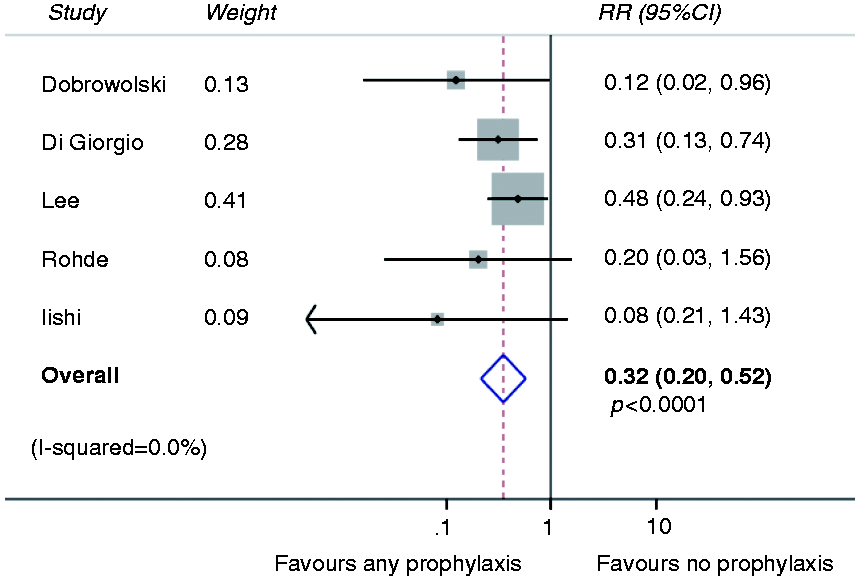
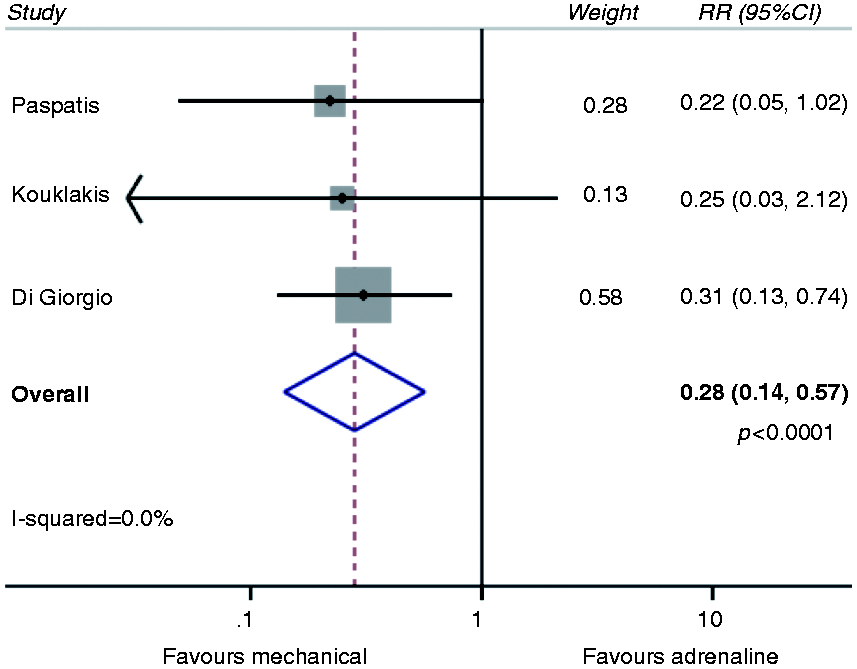
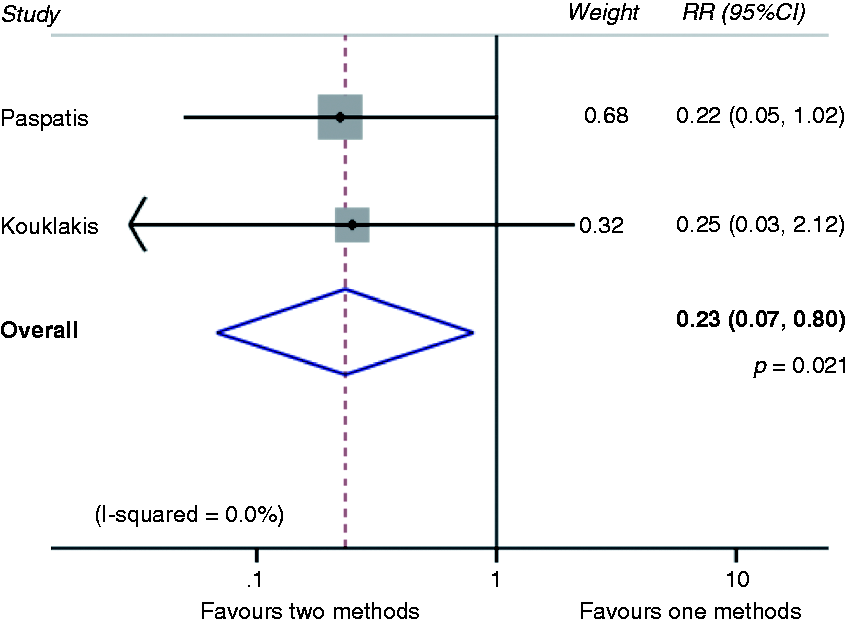
Comparing adrenaline alone with no prophylactic haemostasis revealed a pooled risk ratio of 0.37 (0.20–0.66; p = 0.001; Figure 3). When comparing any method of haemostasis to no prophylactic haemostasis, the pooled risk ratio was 0.32 (95% CI 0.20–0.52; p < 0.0001; Figure 4). The pooled risk ratio comparing any prophylactic mechanical haemostasis with adrenaline injection was 0.28 (95% CI 0.14–0.57; p < 0.0001; Figure 5). The pooled risk ratio comparing two methods of haemostasis to one method of haemostasis was 0.23 (95% CI 0.067–0.80; p = 0.02; Figure 6).
Meta-analysis comparing adrenaline vs. no prophylaxis. Meta-analysis comparing any prophylaxis vs. no prophylaxis. Meta-analysis comparing mechanical haemostasis vs. prophylactic adrenaline. Two methods of prophylactic haemostasis vs. one method.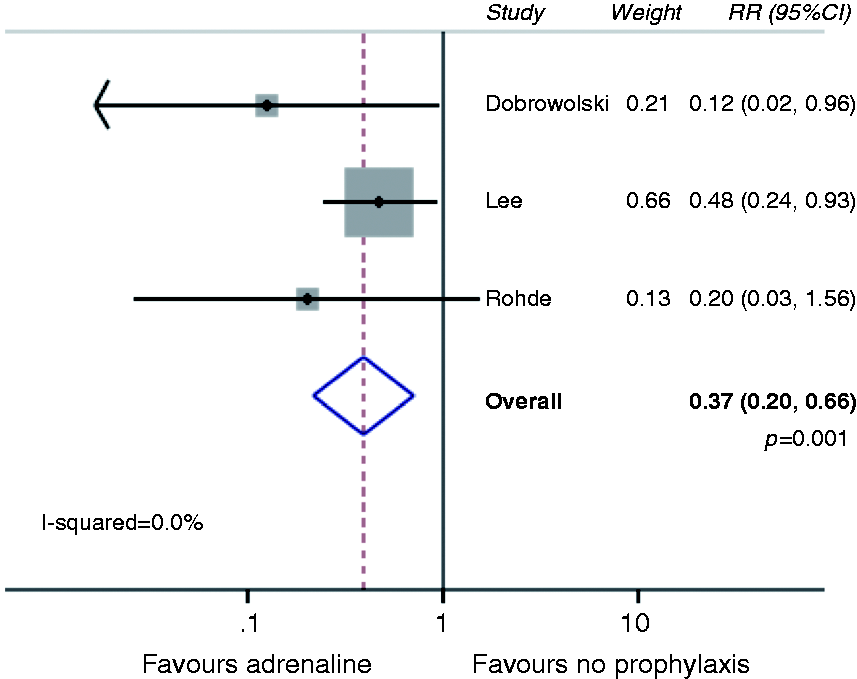
Larger polyps
As larger polyps are often associated with a higher risk of bleeding, polyps >20 mm diameter were examined as a separate group. When only larger polyps were considered, the pooled risk ratio comparing any mechanical haemostasis to adrenaline alone was 0.33 (95% CI 0.12–0.90; p = 0.03).
Cost–benefit analysis
Comparison of calculated costs
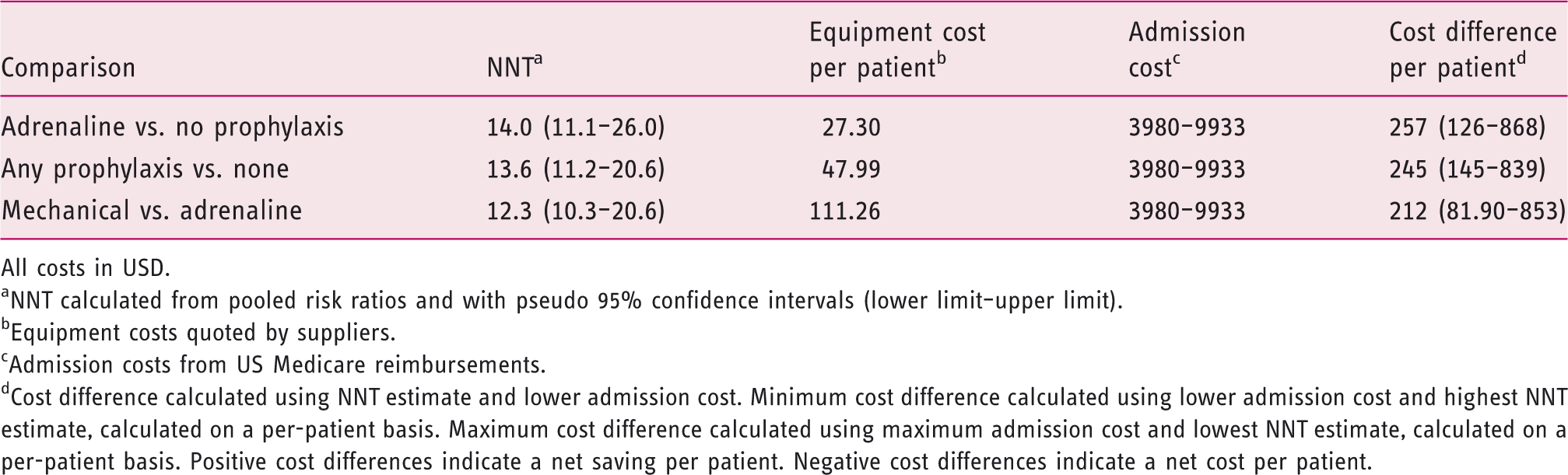
All costs in USD.
NNT calculated from pooled risk ratios and with pseudo 95% confidence intervals (lower limit–upper limit).
Equipment costs quoted by suppliers.
Admission costs from US Medicare reimbursements.
Cost difference calculated using NNT estimate and lower admission cost. Minimum cost difference calculated using lower admission cost and highest NNT estimate, calculated on a per-patient basis. Maximum cost difference calculated using maximum admission cost and lowest NNT estimate, calculated on a per-patient basis. Positive cost differences indicate a net saving per patient. Negative cost differences indicate a net cost per patient.
Cost differences based on cost estimates in the UK and Australia

Costs are in GBP or AUD. UK admission costs based on NHS tariffs 2012. Australian admission costs based on X63A and X63B DRG coding using the 2012–13 National Weighted Activity Unit.
Cost difference calculated using NNT estimate and lower admission cost. Minimum cost difference calculated using lower admission cost and highest NNT estimate, calculated on a per patient basis. Maximum cost difference calculated using maximum admission cost and lowest NNT estimate, calculated on a per patient basis. Positive cost differences indicate a net saving per patient. Negative cost differences indicate a net cost per patient.
Mechanical methods of haemostasis are more effective than adrenaline alone, but are associated with greater cost. This results in mechanical haemostasis being less cost effective in this analysis. This strategy remained potentially cost effective in the USA, saving USD212 per patient (range USD82–853) although formal Markov modelling may provide more accurate estimates. The more conservative estimate based on these figures places this as close to a cost neutral strategy. In the UK and Australia, this strategy was also closer to cost neutral, associated with a small net cost per patient in the UK.
Sensitivity analyses
A sensitivity analysis was performed to examine the effect of the inclusion of studies that did not exclude sessile polyps. The differences noted in rates of PPH comparing any method of haemostasis to no methods (RR 0.11, 95% CI 0.02–0.56; p = 0.008), and two methods of haemostasis to one (0.23, 95% CI 0.067–0.80; p = 0.02) remained unaffected. There was insufficient data to perform meta-analysis of adrenaline alone compared to no prophylaxis following exclusion of studies that included sessile polyps. A further sensitivity analysis was performed to examine the effect of the publication assessed as being of the lowest quality. 22 The differences noted in rates of PPH remained significant for all comparisons. Bias produced by small study sizes was not found to be statistically significant using Peters’ test for small study effects (Bias coefficient –64.8, 95% CI –188 to –57.9; p = 0.192).
Discussion
As screening for colorectal malignancy becomes more commonplace, the number of large polyps removed will continue to increase. PPH and the burden of readmission for monitoring, transfusion, investigation, and therapy represent a health risk to patients and a significant cost to hospitals and insurers. Prophylactic endoscopic measures designed to reduce PPH and associated admissions are available and have been rigorously tested.
Despite the availability of trials in this area, there has not been widespread adoption of prophylactic haemostatic measures to prevent PPH. This may be explained by the relative lack of frequency of PPH and the benign course that is run by most patients with PPH. Identification of prophylactic haemostatic measures in removal of polyps >10 mm as a potentially cost-effective intervention is relevant. This finding should stimulate further investigation and exploration of cost modelling to research the more expensive haemostatic techniques, and may trigger widespread adoption of this practice.
This meta-analysis confirms that prophylactic haemostatic therapy for the removal of polyps >10 mm is associated with a significant reduction in risk of PPH. Cost modelling demonstrates this practice to be potentially cost effective in a variety of healthcare settings. Injection of adrenaline is the most cost-effective method of preventing PPH, in part related to the low cost of the equipment involved.
There are several limitations to this meta-analysis. Whilst the funnel plots demonstrate that publication bias is unlikely, it cannot be excluded and the small number of publications in this area indicates that this is possible. Selection and sample bias are unlikely to have occurred due to the randomized controlled design of all of the included trials and the broad entry criteria of the included trials.
The studies varied in their definition of PPH and it is possible that reporting of outcomes was not consistent between all study groups. The consistent baseline PPH rates and effect sizes, however, suggest that this is not a significant source of bias. The PPH rate examined across all of the control arms was higher than generally reported in the literature. This is likely to be the result of both the size of polyps assessed in the included studies (polyps <10 mm were excluded) and the reported PPH rate in the literature is not exclusive of measures to ensure haemostasis.
Due to the increased cost of admission, the cost–benefit is greater in patients with catastrophic or severe adverse events. It is reasonable to consider mechanical prophylaxis or two methods of prophylaxis in patients at high risk for PPH due to polyp morphology or comorbidities. Furthermore, these patients may carry increased risk of PPH or requiring admission for PPH due to comorbidities and pharmacotherapy.
Cost estimates are highly variable and are based on figures that may not accurately represent local practice or reimbursements. These cost estimates do not include rare cases where patients may suffer further adverse events and require surgery, intensive care admission, or death. Further, transfusion and investigation costs have not been specifically addressed. These factors are theoretically addressed by the use of admission costs generated by the appropriate DRG coding which generates estimates based on pooled costs of larger numbers of patients to provide estimates of cost on a per-patient basis.
A previous meta-analysis focussed on separating early and late bleeding. 28 In the original papers, the primary end point was typically all PPH. As early bleeding, like delayed bleeding, requires assessment, investigation, and possibly transfusion and therapy, we combined these end points for our analysis.
This meta-analysis combines papers of varying quality. Inclusion of lower-quality papers may render the final results susceptible to bias. Sensitivity analysis excluding the lowest-quality trials did not demonstrate a significant change in results. As this is the only available evidence in this area, the results remain valid.
The greatest reduction in PPH was seen when comparing two methods of haemostasis to one method alone. This is less cost effective than using adrenaline injection alone, owing to the associated increase in equipment costs. For patients felt to be high risk for PPH or those with significant comorbidities, this may still prove a cost-effective strategy, although this requires separate dedicated investigation.
Trials in endoscopic haemostasis for high-risk or bleeding peptic ulcers identified similar superiority of two haemostatic mechanisms over one. Using two endoscopic methods of prophylactic haemostasis prolongs endoscopy time, particularly for patients with multiple polyps, and this may reduce uptake by endoscopists.
There are few large prospective trials comparing prophylactic haemostatic measures in polypectomy, 23,24,27 and only one comparing mechanical haemostasis to both no therapy and adrenaline. 23 However, the data regarding prophylactic haemostasis for removal of polyps >10 mm clearly indicates clinical superiority and cost effectiveness.
Three studies included sessile and pedunculated polyps.22,26,27 All three of the studies comparing adrenaline injection to placebo contained a mix of polyp morphology.22,26,27 Only one study completely reported bleeding rates by polyp morphology.26 Sessile polyps appeared to carry an equivalent or increased risk of bleeding. Recent work has shown that prophylactic mechanical haemostasis in flat lesions reduces rates of PPH. 32 Without complete data on polyp morphology, inferences regarding adrenaline injection alone for the prevention of PPH in polypectomy for pedunculated polyps can not be made. It is possible that adrenaline injection significantly decreases PPH for flat polypectomy without significantly affecting PPH in pedunculated polyps; however, there is no pathophysiological reason why this might be the case. The exact proportion of polyps that are pedunculated is unknown; however, the proportion of flat polyps is increasing (due to increased detection), perhaps making the findings of this study more applicable in a smaller proportion of cases.
Studies examining prevention of PPH should include adrenaline injection as the control arm. Future studies should separate flat, sessile, and pedunculated polyps, as mechanical measures may not be suitable for all polyp types. Further studies comparing methods of mechanical haemostasis and adrenaline and combining approaches should be performed. Cost–benefit analysis should be frequently revisited and adjusted to reflect local practices, as the costs of admissions and devices change over time and location.
In conclusion, this meta-analysis demonstrates that for polyps ≥10 mm in size, pharmacological and/or mechanical prophylactic measures to reduce PPH are effective, with low numbers needed to treat and narrow confidence intervals around risk reduction estimate. A basic cost-effectiveness analysis suggests that routine use of prophylactic techniques to prevent PPH in these larger polyps may be cost saving in some healthcare settings, although further modelling is required.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
