Abstract
Background
Although cyclosporin is effective in severe ulcerative colitis (UC), long-term colectomy rate varies between 60 and 88% among patients in whom cyclosporin initially induced remission. The aim of our study was to evaluate the long-term outcome and the optimal duration of cyclosporin therapy in acute, severe UC.
Methods
A total of 73 patients underwent i.v. cyclosporin therapy for a steroid refractory flare up of UC between 1998 and 2009. All patients were treated with 1 mg/kg i.v. methylprednisolone for 3–7 days before the administration of cyclosporin. Patients received i.v. cyclosporin of 4–5 mg/kg for 5 days following oral treatment.
Results
The mean follow up after the initiation of cyclosporin was 4.2 years. There were 20 patients who underwent early colectomy. Cyclosporin had to be discontinued due to side effects in 22 patients. Cyclosporin failed and late colectomy was performed in 14 of the 53 responders. Duration of cyclosporin treatment was significantly longer in those who avoided colectomy. The probability of avoiding colectomy proved to be 66% in case of 1-year treatment period with cyclosporin. The longer treatment period resulted in longer colectomy-free disease course.
Conclusions
Cyclosporin is effective in acute, severe UC during long-term follow up. Our data suggest that the longer cyclosporin is used, the more it is possible to avoid colectomy in the future.
Introduction
The main goal of treatment of ulcerative colitis (UC) is to induce and maintain steroid-free remission, improve the quality of life, and reduce the risks of colectomy. Corticosteroids have been the primary therapies in moderate to severe UC for years. Those who fail to respond to treatment with corticosteroids, or who present with severely active UC, should be considered as candidates for rescue treatment. 1 Our previous work revealed that anaemia, need for blood transfusion, and frequency of previous hospitalizations significantly determines the response to early parenteral corticosteroid therapy. Colectomy rate was 2.5-times higher in patients refractory to i.v. steroid therapy, which may determine both the early and late outcome and the colectomy rate of acute, severe UC. 2
Cyclosporin is one of the most effective therapeutic choice in patients with severe UC; however, long-term colectomy rate still varies between 60–88% among patients in whom cyclosporin initially induced remission. 3 The early disappointing long-term results and the serious side effects limit the widespread use of cyclosporin in the era of infliximab; however, it serves as a rapidly acting ‘bridge’ to maintenance therapy with the slowly acting agents azathioprine or mercaptopurine. 4 Although the optimal duration of cyclosporin treatment is unknown, it is usually discontinued within 3 months. 5
Since data on long-term efficacy of cyclosporin are still controversial and the optimal duration of therapy is unknown the aim of our study was to retrospectively evaluate the long-term efficacy and safety of cyclosporin therapy in patients with acute, steroid-refractory severe UC.
Patients and methods
Patient population
Demographics of the patients
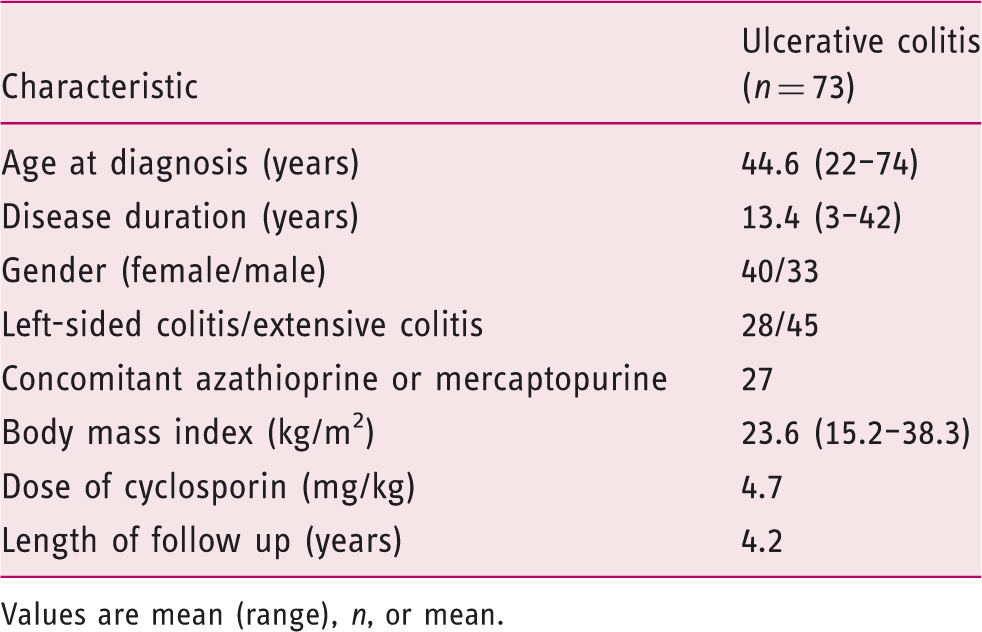
Values are mean (range), n, or mean.
Administration of cyclosporin
All patients were treated with 1 mg/kg i.v. methylprednisolone for 3–7 days before the administration of cyclosporin. Patients received i.v. cyclosporin of 4–5 mg/kg for 5 days following oral treatment at the same starting dose in the case of a good initial response. Cyclosporin levels were closely monitored, and the dose was adjusted to maintain fasting levels between 150 and 300 ng/ml and 2 hours post-dosing level of 800–1200 ng/ml. When starting cyclosporin therapy, we planned to use it at least for 6 months.
Twenty-seven patients received concomitant immunosuppression (26 thiopurines, one mycophenolate mofetil) before cyclosporin therapy. None of the patients was treated with infliximab before cyclosporin therapy.
Statistical analysis
Data analysis was carried out using Student t-test and Wilcoxon test. Proportions were analysed using Pearson’s chi-squared test, Fisher’s exact test. Kaplan–Meier survival curves were plotted for analysis with the log-rank and Breslow tests. Cox regression was applied to investigate the relationship between the cyclosporin treatment period and the risk of colectomy. p < 0.05 was considered statistically significant. For statistical analysis, SPSS version 15.0 (SPSS, Chicago, IL, USA) was used.
Results
Short and long-term efficacy of cyclosporin therapy
In this study, 53/73 (73%) patients initially responded to i.v. cyclosporin. Twenty (27%) patients underwent early colectomy within 3 months. During the >4-year follow-up period, 14 of the 53 responder patients (26%) underwent late colectomy. Cyclosporin had to be discontinued because of intolerable or severe side effects in 22/53 (42%) patients. The mortality rate was 0%. Overall, colectomy free survival was 53%. Five patients were switched to infliximab after cyclosporin failure. For maintenance therapy, azathioprine was given for 24 patients, 5-aminosalicylic acid for 18 patients, and infliximab for seven patients who avoided colectomy.
Figure 1 shows the outcomes of the rescue cyclosporin therapy in acute, severe UC patients. The demographic and clinical data of patients who underwent and who avoided surgery are detailed in Table 2.
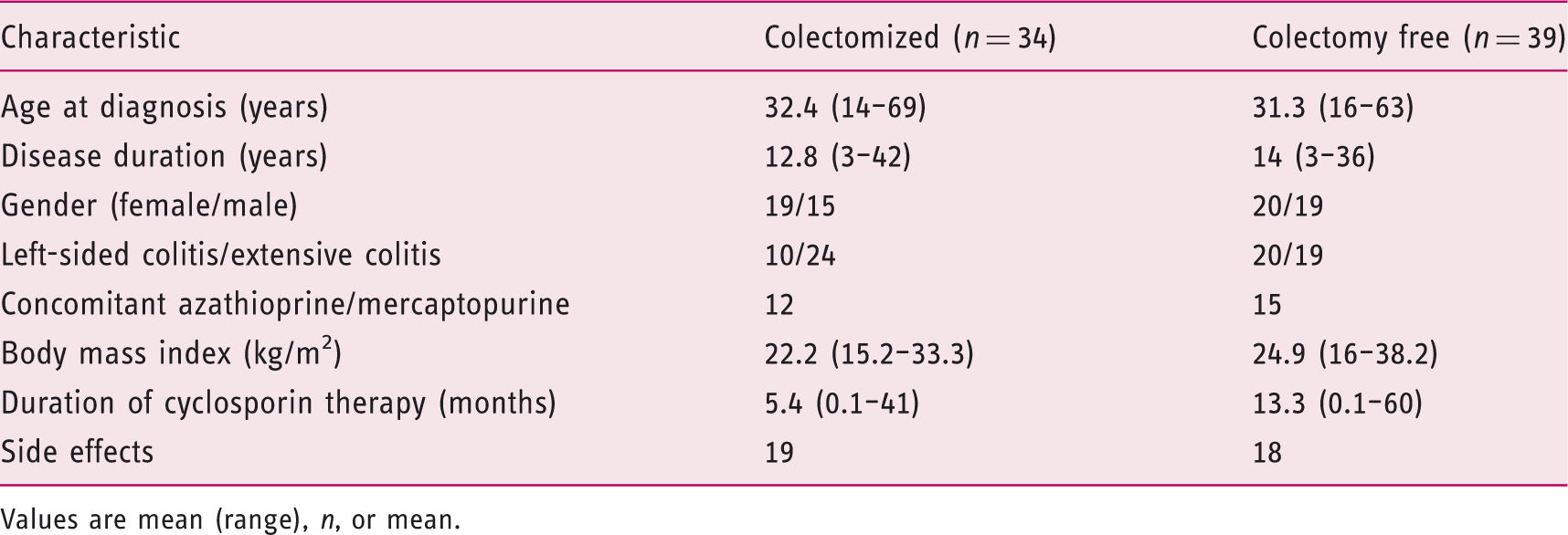
Outcomes of the rescue cyclosporin therapy in acute, severe UC patients. Demographic and clinical data of patients Values are mean (range), n, or mean.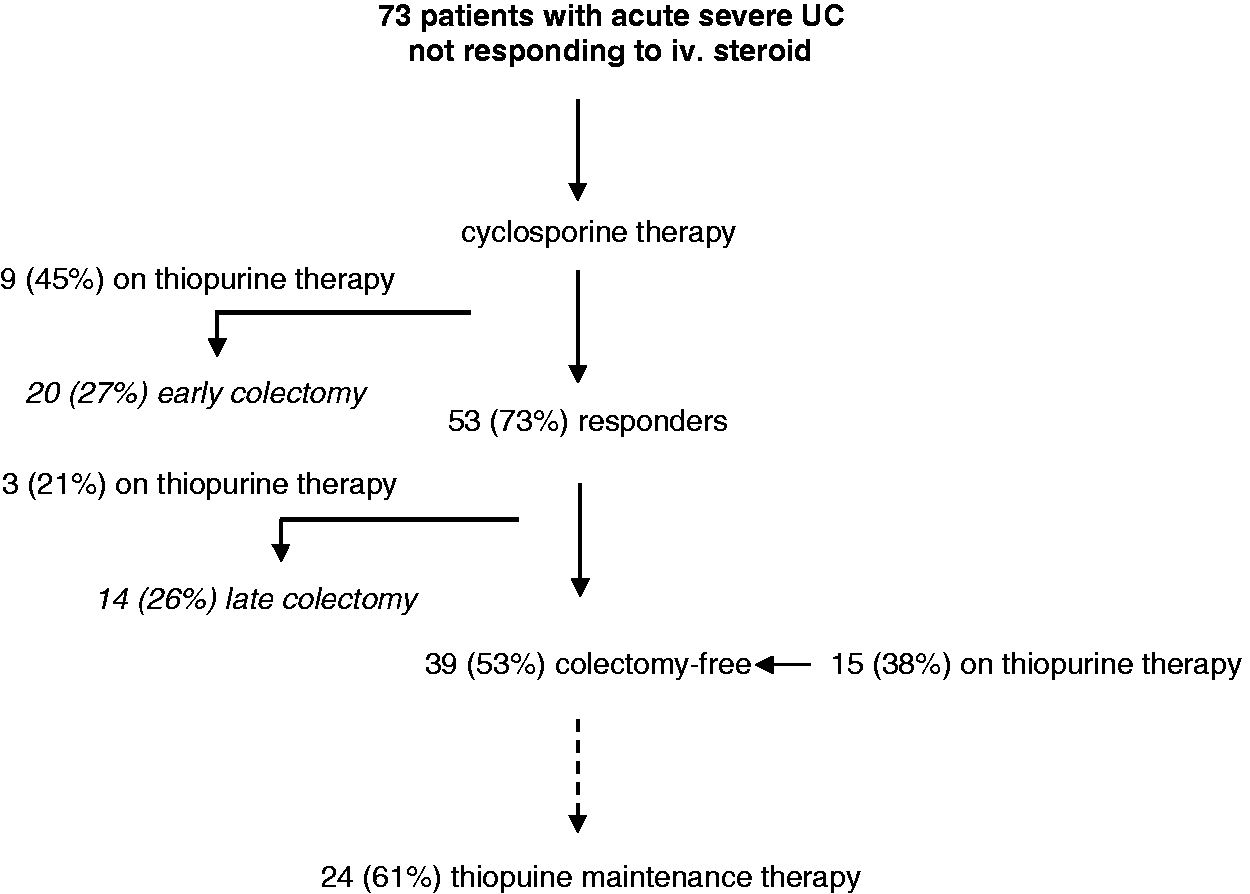
Duration of cyclosporin therapy
Duration of cyclosporin therapy was significantly shorter in those responder patients who underwent late colectomy vs. those who avoided surgery (5.4 vs. 13.3 months, p = 0.009; Figure 2). If patients were treated for a year, the probability of avoiding colectomy proved to be 66%. The longer treatment period decreased the risk of colectomy in the Cox regression model (hazard rate ratio 0.98, p = 0.013). The mean time elapsed between the discontinuation of cyclosporin therapy and colectomy was 9 months (0.1–72 months. Patients who underwent late colectomy were operated on an average of 22 months after stopping cyclosporin. Cyclosporin was discontinued because of side effects in 53% of these patients and because of loss of response in 43%. Patients who avoided colectomy were in remission, defined as Lichtiger score ≤2, for an average of 12.7 months before stopping cyclosporin. Of these patients, azathioprine was given for 24 and infliximab for seven patients for maintaining remission after the cyclosporin therapy.
Kaplan–Meier plot showing the need for colectomy in relation to the duration of cyclosporin therapy.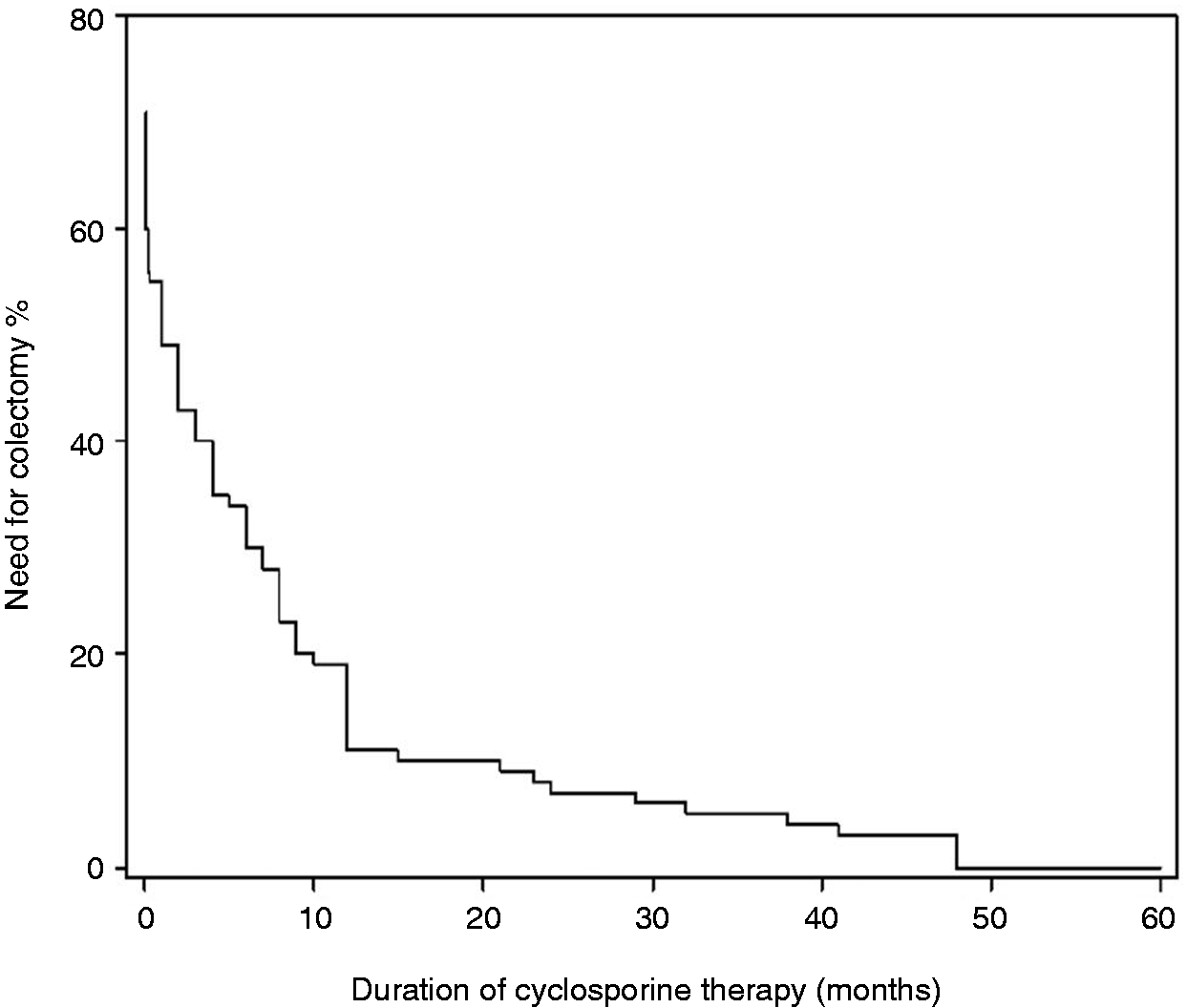
Side effects
Major side effects occurring during cyclosporin therapy
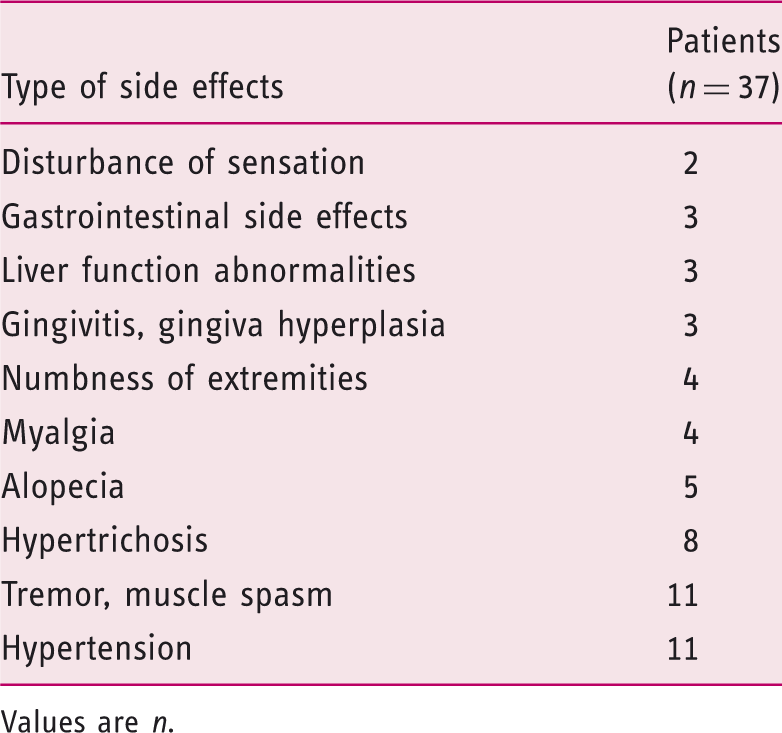
Values are n.
Discussion
This retrospective study confirmed that cyclosporin is effective in the treatment of acute, severe UC during the long-term follow up. Overall, colectomy free survival was 53% during the >4-year follow-up period. Colectomy-free survival significantly increased in case of longer duration of cyclosporin therapy. If patients were treated for a year, the probability to avoid colectomy proved to be 66%.
The published data about the short and long-term results of cyclosporin therapy are various and controversial – maybe due to the small patient numbers and the difference in the duration of therapy. Recently, some studies tried to obtain a coherent view and examined the long-term outcome of cyclosporin therapy. In the study of Mocciaro et al. 7 the colectomy rate was 29% at 3 months and 48% at 12 months in 35 cyclosporin-treated UC patients. The 2–3-year cumulative colectomy rates were 54% and 57%. In this study, cyclosporin was administered for a maximum of 3 months. Data from the UK revealed 42% of UC patients undergoing colectomy during a median follow up of 3.8 years. 8 In a Japanese study, the overall percentages of patients who had not required surgery were 72% at 1 year, 62% at 2 years, 58% at 3 years, and 48% at 5 years. 9 Unfortunately, these papers do not clarify the duration of cyclosporin therapy. Cheifetz et al. 10 treated 71 patients with cyclosporin and followed them up for a mean of 3 years. Cumulative colectomy rates were 39% at 1 year, 42% at 2 years, and 46% at 5 years. Cyclosporin was discontinued in most patients by 6 months.
In general, cyclosporin is used for inducing remission as a rescue therapy and the optimal duration of the therapy is unknown. 11 In our study, the colectomy rate after 4 years was 47%. The mean duration of cyclosporin therapy was more than 13 months in colectomy-free patients. Our results showed that two-thirds of patients can avoid colectomy if treated with cyclosporin for at least 12 months. In a recent paper of Sjöberg et al., 12 cyclosporin was given for a mean of 4.5 months: at 12 months, 77% of the patients remained colectomy-free; however, this study was not followed up for more than 1 year.
Concerns of toxicity remain the main reasons for underusing cyclosporin in the clinical practice. However, all of the studies mentioned above found the majority of the adverse events to be mild. Sternthal et al. 13 revealed major adverse events in 15% of their patients. Nephrotoxicity occurred in 5% of the patients. Serious infection occurred in 6%. seizures in 4%, anaphylaxis in 1%, and death in 2%. The most common minor adverse events were paresthesias, hypomagnesemia, hypertension, and hypertrichosis. 13 In our study, more than 70% of patients developed mild or moderate side effects and 60% of them resulted in the discontinuation of the therapy.
The main limitation of this study is the retrospective nature. However, the data compared in the study are quite reliable and they come from a single centre with an interest in the management of severe UC.
In summary, our data show similar long-term outcome of cyclosporin therapy than the recent studies. Cyclosporin seems to be effective in acute, severe UC during long-term follow up. Although the optimal duration for the treatment is unknown, our data suggest that the longer cyclosporin is used, the more it is possible to avoid colectomy in the future. Multicentre trials would be needed to confirm the optimal duration for cyclosporin.
Footnotes
Funding
This work was supported by OTKA (research proposal PD 105948; PI: Klaudia Farkas) and TÁMOP (4.2.2.A-11/1/KONV-2012-0035, 4.2.2-A-11/1/KONV-2012 0052, and 4.2.2.A-11/1/KONV-2012-0073).
Conflict of interest
The authors declare that there is no conflict of interest.
