Abstract
Background
An on-site, rapid, fingertip, whole-blood point-of-care test (POCT) is attractive for active case-finding of coeliac disease (CD) in primary care because of its simplicity.
Aim
The aim of this article is to assess the usefulness and cost-effectiveness of adult case-finding using a POCT based on deamidated gliadin peptide antibodies (IgA/IgG-DGP) in primary care for CD diagnosis.
Methods
A case-finding study for CD was conducted by using an easy-to-use, on-site, whole-blood for IgA/IgG-DGP-based fingertip POCT compared with tTG2 in 350 individuals. Sample size was calculated based on 0.28% prevalence in the reference population. Duodenal biopsies for histology, intraepithelial lymphocytes and in situ deposition of tTG2 were obtained if tTG2 and/or POCT were positive. Accuracy and cost-effectiveness of strategies using serology or POCT were calculated.
Results
Prevalence of CD was 1.14% (95% CI, 0.3–3.4), almost double what was previously observed. Four patients were diagnosed with CD. tTG2 was positive in three (0.85%) and POCT in 29 (8.2%). Sensitivity of POCT for CD was 100%, specificity 93%, PPV 14%, and NPV 100%. POCT followed by duodenal biopsy was the most cost-effective approach in our setting (standard diagnosis: €13,033/case; POCT + duodenal biopsy: €7360/case).
Conclusions
A negative POCT allows ruling out CD in primary care, making it suitable for case-finding. POCT strategy was the most cost effective.
Key summary
Established knowledge on this subject
Active case-finding is useful to detect new coeliac disease (CD) patients in primary care. Standard serology has been used for CD case-finding. A rapid point-of-care test (POCT) based on deamidated gliadin peptide antibodies (immunoglobulin (Ig)A/IgG-DGP) was shown to be cost effective at pre-endoscopy in patients assessed with anaemia. A point-of-care test (POCT) based on immunoglobulin (Ig)A/IgG deamidated gliadin peptide (DGP) has never been used as a diagnostic screening tool in a case-finding strategy in primary care, compared to standard serology. A POCT based on IgA/IgG-DGP detection was demonstrated to be suitable for coeliac disease (CD) screening in primary care identifying all seropositive CD patients while a negative test ruled out CD. The economic impact of this strategy has never been evaluated in this setting and showed to be cost effective compared with the standard approach using serology.
What are the significant and/or new findings of this study?
Introduction
The prevalence of coeliac disease (CD) using serological mass screening is around 0.2% to 1%1–4 but the number of CD patients diagnosed in clinical practice is much lower and CD remains under-diagnosed. Active case-finding is useful in terms of an increase in the number of newly detected CD patients in primary care.5–9 This approach relies on an active role being played by primary care physicians (PCPs) in selecting the individuals to be tested for CD. Standard serology (endomysial (AEA) or anti-transglutaminase 2 antibodies (tTG2)) has generally been used for this purpose. 10 In symptomatic patients the prevalence of CD is higher than in the general population, ranging from 2% to 10%.5–9 A number of studies have demonstrated that CD-related complications are reversed by a gluten-free diet (GFD). Thus, case-finding has to be encouraged.11,12
An easy-to-use, on-site, whole-blood, deamidated gliadin peptide (DGP)-based, fingertip, point-of-care test (POCT) provides rapid results, 13 making it particularly attractive for use in primary care. The Simtomax® assay detects CD with a combination of immunoglobulin A (IgA) and immunoglobulin G (IgG) antibodies against DGP, allowing the detection of CD in IgA-deficient patients. It has been recently demonstrated that Simtomax® has a similar accuracy to standard serology for biopsy-proven CD and is more reliable than another commercially available POCT based on tTG2. 14
The costs of different diagnostic approaches applied to large populations may be highly variable, having an impact on the public health system. A recent study demonstrated that the performance of a POCT (Simtomax®-IgA/IgG-DGP) pre-endoscopy in patients assessed with anaemia was cost effective. 15
The aims were to assess: (a) the effectiveness of a strategy of adult case-finding using a POCT in primary care for CD diagnosis compared with serology, and (b) whether this strategy was cost effective.
Patients and methods
Clinical enrolment criteria.

An initial diagnostic work-up was conducted to rule out other, potentially more severe diseases responsible for symptoms such as colon cancer or inflammatory bowel disease.
Viral, metabolic, neoplastic and autoimmune liver diseases were previously ruled out.
ALT: alanine transaminase; AST: aspartate transaminase; CD: coeliac disease; Hb: haemoglobin; IgA: immunoglobulin A.
Study procedures
POCT (Symtomax®) and tTG2 testing were conducted in parallel on patients meeting the enrolment criteria. Individuals with positive POCT and/or tTG2 testing were referred for duodenal biopsy to assess histopathology, intraepithelial lymphocyte (IEL) flow cytometry, and in situ deposition of tTG2 antibodies. A blood sample was obtained for confirmatory tTG2 and AEA and CD genetic markers (human leucocyte antigen (HLA)-DQ2.5, HLA-DQ2.2, and/or HLA-DQ8). Patients with negative POCT and standard serology but with persistent severe symptoms were also referred for duodenal biopsy (Figure 1). A detailed description of these techniques has been previously described in detail16–20 (Appendix 2).
Flowchart of the study procedures. CD: coeliac disease; IEL: intraepithelial lymphocyte; IgA: immunoglobulin A; PCP: primary care physician; POCT: point-of-care test; anti-tTG2: anti-transglutaminase 2 antibodies.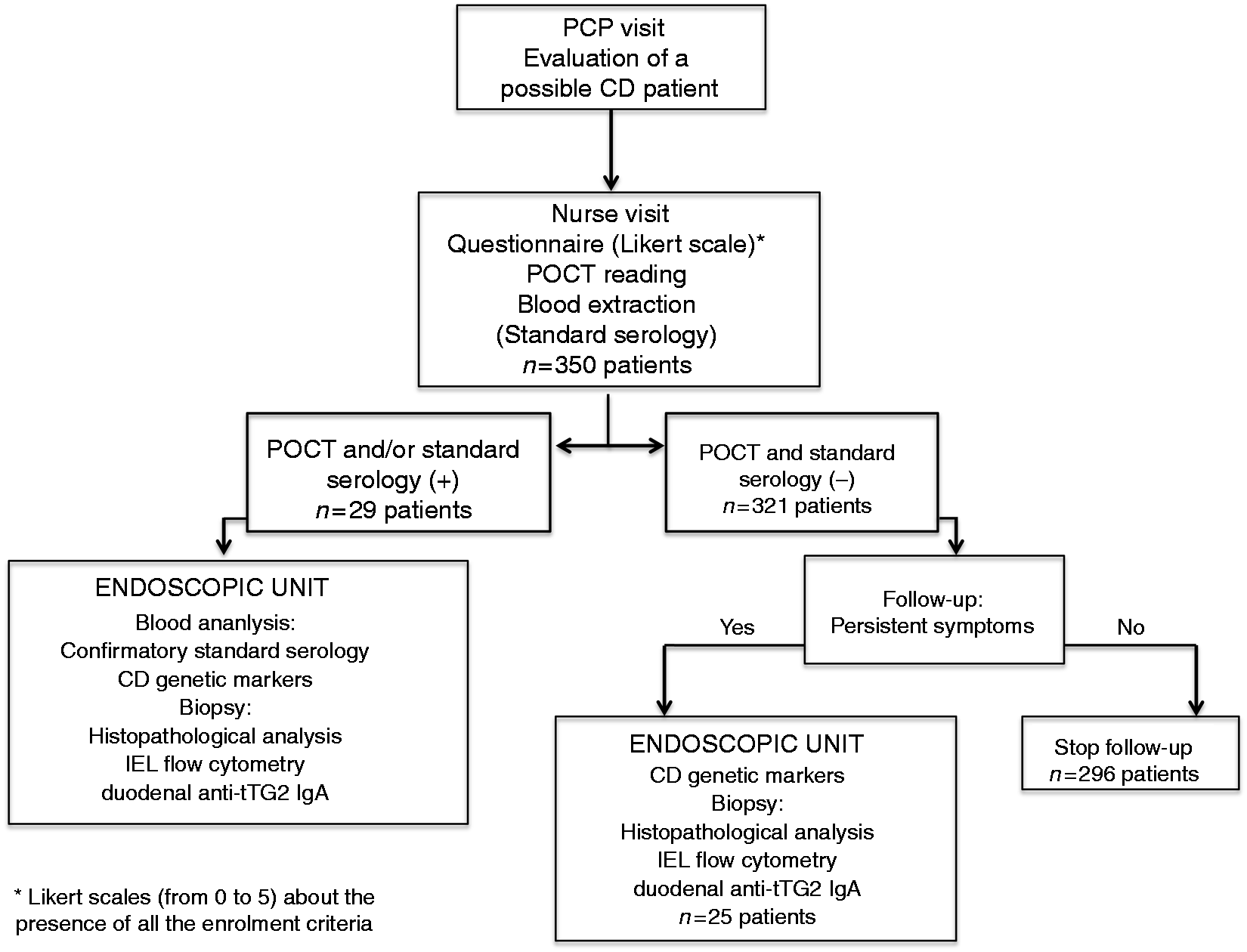
POCT
IgA anti-DGP, IgG anti-DGP, and total IgA analyses are embedded in Simtomax® (Augurix SA, Switzerland). A detailed description of the development of the device has been previously described.14,15 In the test, secondary gold-conjugated antibodies bind to the patient’s antibodies to form detectable complexes that are captured by the test in lines A and B. The CT is the control line. A CD-positive result is indicated by detection of both the CT and A lines (Figure 2(a)). IgA deficiency is indicated by absence of the B line.
(a) Point-of-care test (Simtomax®). CT: control line; line A: IgA/IgG deamidated gliadin peptide; line B: total IgA. (b) Results of the present study. CD: coeliac disease; IgA: immunoglobulin A; IgG: immunoglobulin G.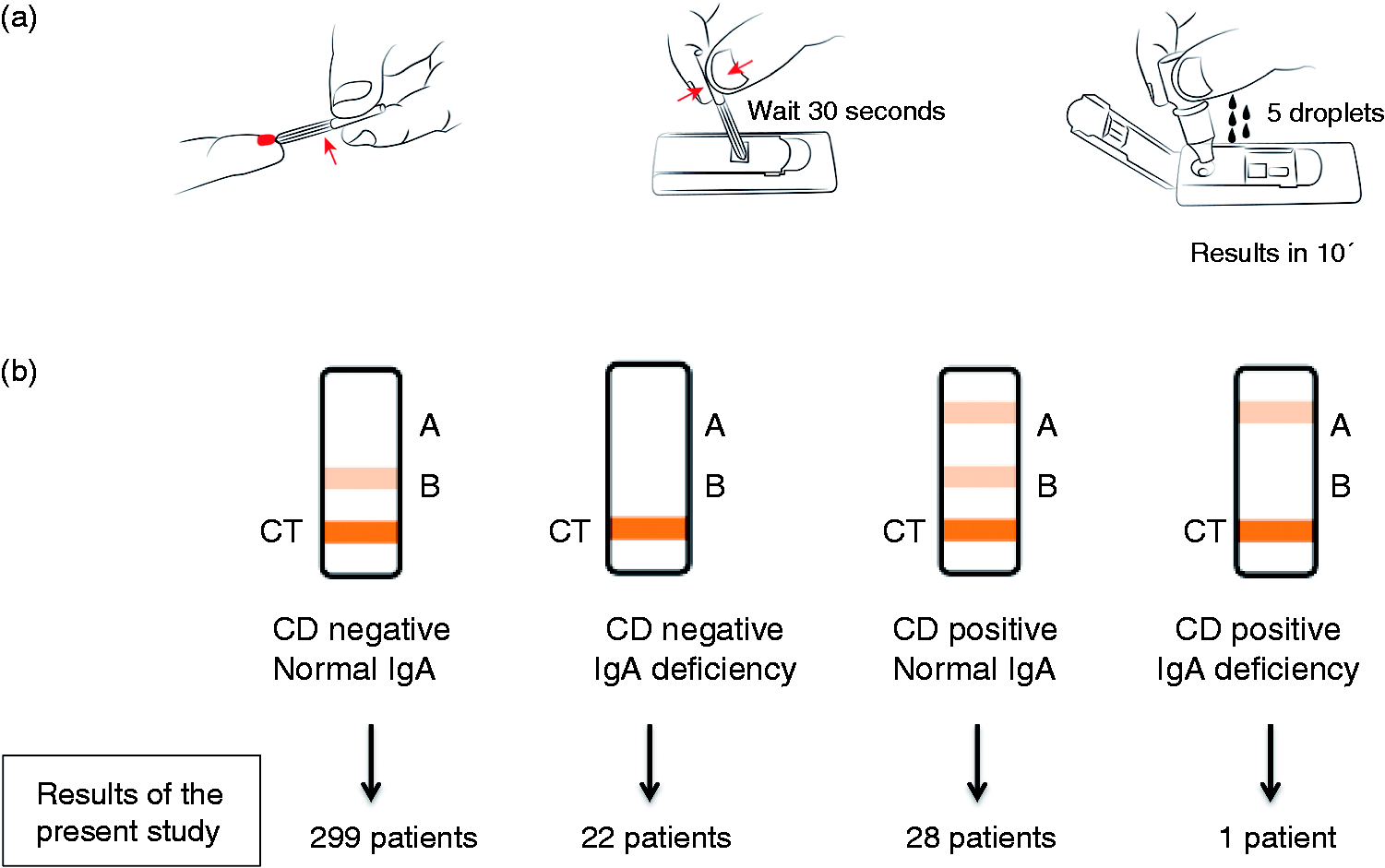
Definition of CD
CD was considered in patients with (a) positive IgA/IgG tTG2/AEA and duodenal changes of the CD spectrum (from lymphocytic enteritis to atrophy), and (b) negative IgA/IgG tTG2/AEA but duodenal atrophy together with CD cytometric pattern plus positive immunofluorescence (IF) staining of tTG2 IgA deposits.19,21
Calculation of the costs
Costs of three diagnostic approaches based on POCT or serology as screening for CD were calculated as follows: (a) standard diagnosis: serology followed by endoscopy in positive patients, (b) POCT followed by confirmatory serology in positive POCT and endoscopy in seropositive patients, and (c) POCT followed by endoscopy in positive patients (Table A.1 in Appendix 3).
Statistical analysis
The prevalence of CD in the general adult population detected by serological screening in our area is 0.28% (1:357). 22 CD prevalence in symptomatic patients and in risk groups is estimated at 3% to 10%.5–9,22,23 If the prevalence of CD was around 3%, a sample size of 350 patients would enable us to obtain a 95% confidence interval (CI) with a precision of ±1.8%. 24
In order to ascertain whether the ‘educational programme’ had an impact on the number of diagnoses, we performed a post hoc analysis to compare the CD cases in the study period with those diagnosed during the previous 18 months and cases diagnosed by the non-participant PCPs in the study period. The Z test was used to compare proportions. Prevalence of CD (95% CI) in different periods was calculated. The diagnostic accuracy of the POCT was assessed by means of sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV), and their 95% CIs, taking CD cases as the gold standard.
Ethical considerations
This study was conducted according to the ethical guidelines of the 1975 Declaration of Helsinki. Ethical approval was obtained from the Hospital Universitari Mutua Terrassa Ethical Committee on 25 February 2014, and all patients signed the informed consent.
Results
Serological, genetic and duodenal markers of the four patients fulfilling the diagnostic criteria of coeliac disease.
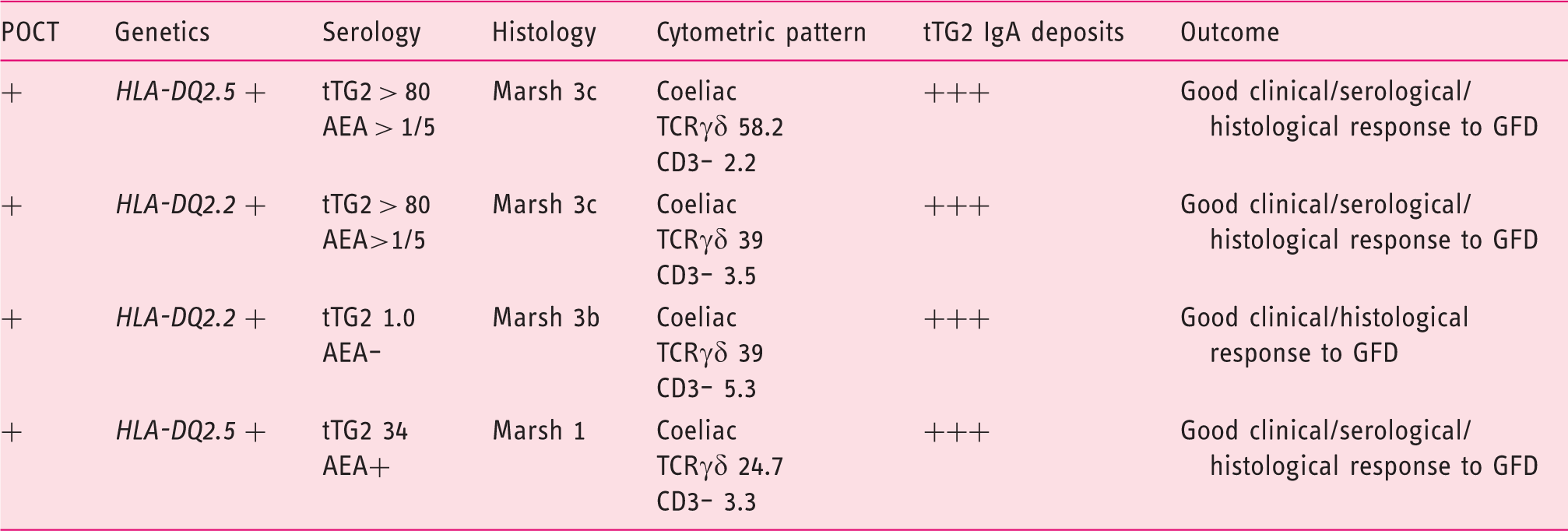
TG2: anti-transglutaminase 2 antibodies; AEA: endomysial antibodies; GFD: gluten-free diet; HLA: human leucocyte antigen; TCR: T cell receptor.
Complete coeliac cytometric pattern: TCRγδ > 8.5% and CD3– <10%.
Genetic and duodenal markers of the 10 patients with positive POCT and negative standard serology (t-TG2, AEA) with duodenal markers of potential CD.
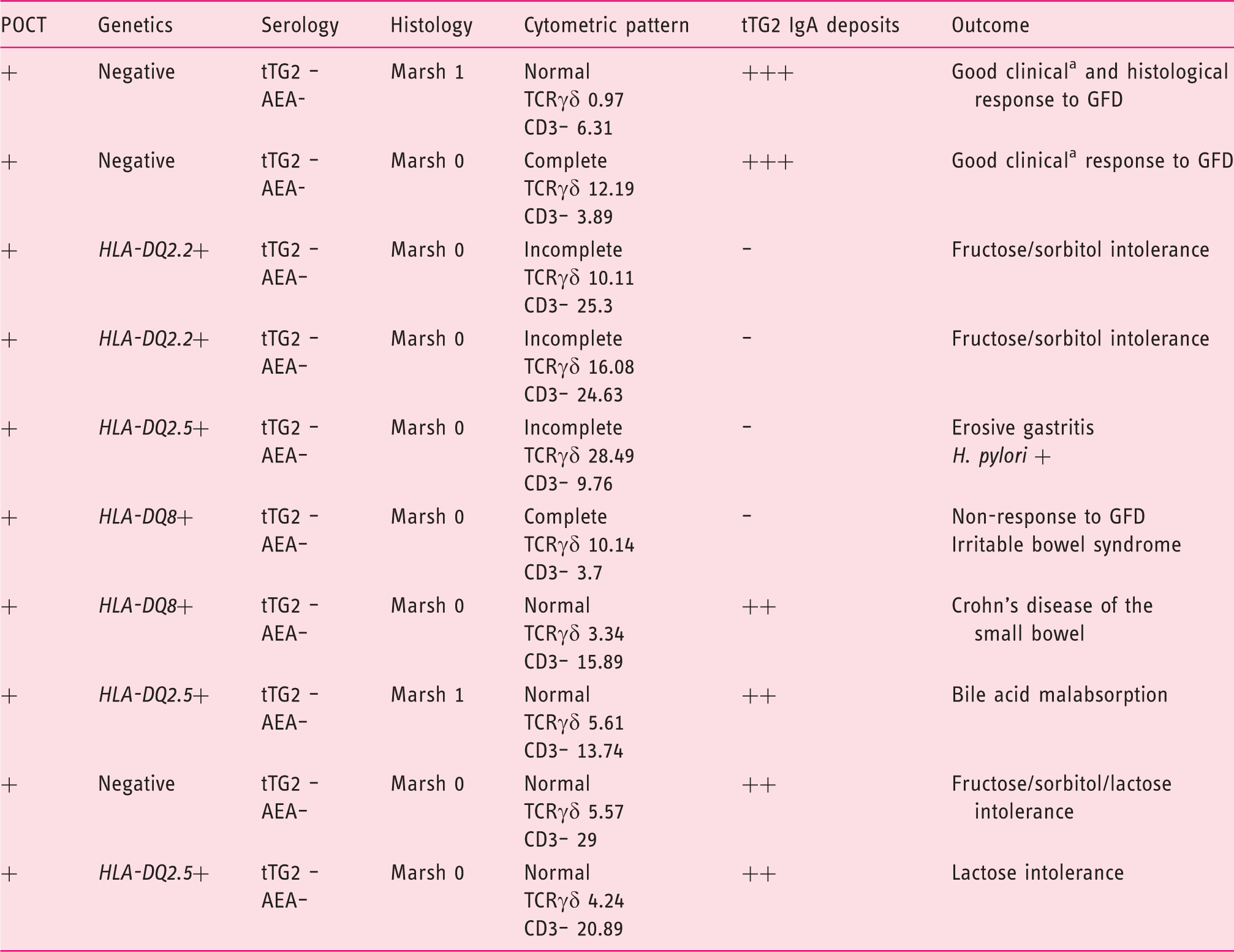
tAEA: endomysial antibodies; CD: coeliac disease; GFD: gluten-free diet; HLA: human leucocyte antigen; POCT: point-of-care test; TCR: T cell receptor; TG2: anti-transglutaminase 2 antibodies.
Complete coeliac cytometric pattern: TCRγδ > 8.5% and CD3–<10.
Incomplete coeliac cytometric pattern: selective increase of TCRγδ > 8.5%.
Chronic relapsing diarrhoea.
Genetic and duodenal markers of potential CD of the five patients with negative POCT and negative standard serology (t-TG2, AEA).
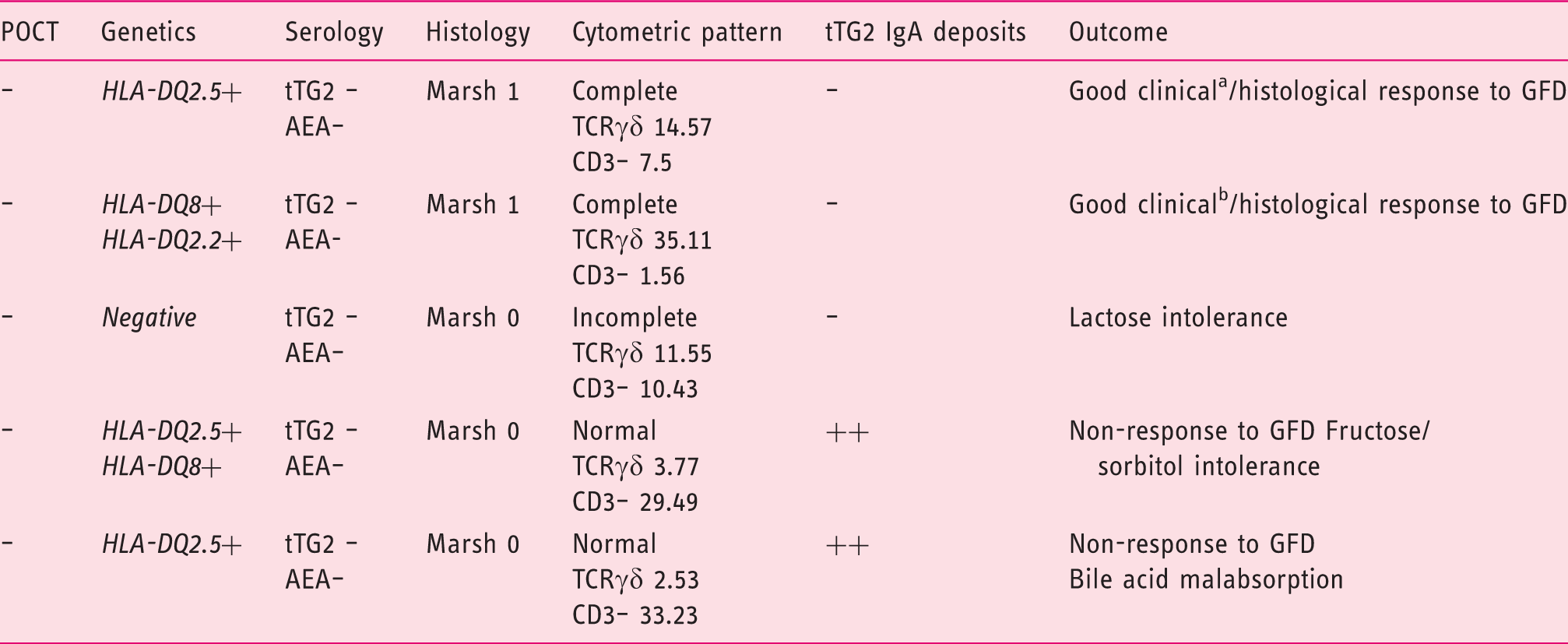
AEA: endomysial antibodies; CD: coeliac disease; GFD: gluten-free diet; HLA: human leucocyte antigen; POCT: point-of-care test; TCR: T cell receptor; tTG2: anti-transglutaminase 2 antibodies.
Complete coeliac cytometric pattern: TCRγδ > 8.5% and CD3– <10.
Incomplete coeliac cytometric pattern: selective increase of TCRγδ > 8.5%.
Relapsing diarrhoea. bRelapsing diarrhoea + iron-deficient anaemia.
No differences were found in the percentage of potential CD markers between patients with negative and positive POCTs, not fulfilling the criteria of CD (10 of 25 patients – 40% – with positive POCT and five of 25 patients – 20% – with negative POCT; p = 0.216).
Accuracy of the DGP antibody POCT compared with standard serology
The four patients fulfilling the criteria of CD had a positive POCT, but only three of them had positive standard serology (tTG2 and AEA). None of the patients with negative POCT fulfilled the diagnostic criteria of CD. This resulted in a sensitivity of POCT detecting CD of 100% (95% CI: 40–100%), a specificity of 93% (95% CI: 89–95%), NPV 100% (95% CI: 98.5–100%) and PPV 14% (95% CI: 4.5–33%).
Cost-effectiveness analysis
The total costs of the three diagnostic strategies, the detailed procedures and the number of cases diagnosed with each diagnostic strategy are detailed in Figure 3. The use of POCT followed by endoscopy and duodenal biopsy in positive patients demonstrated the best cost-effective result with a total cost of €7360.63 per diagnosed case (n = 4).
Total costs and detailed procedures of the three diagnostic strategies. anti-tTG2: anti-transglutaminase 2 antibodies; POCT: point-of-care test. Tariffs of the National Health Services of the Catalan Government are provided in Table A.1 (Appendix 3).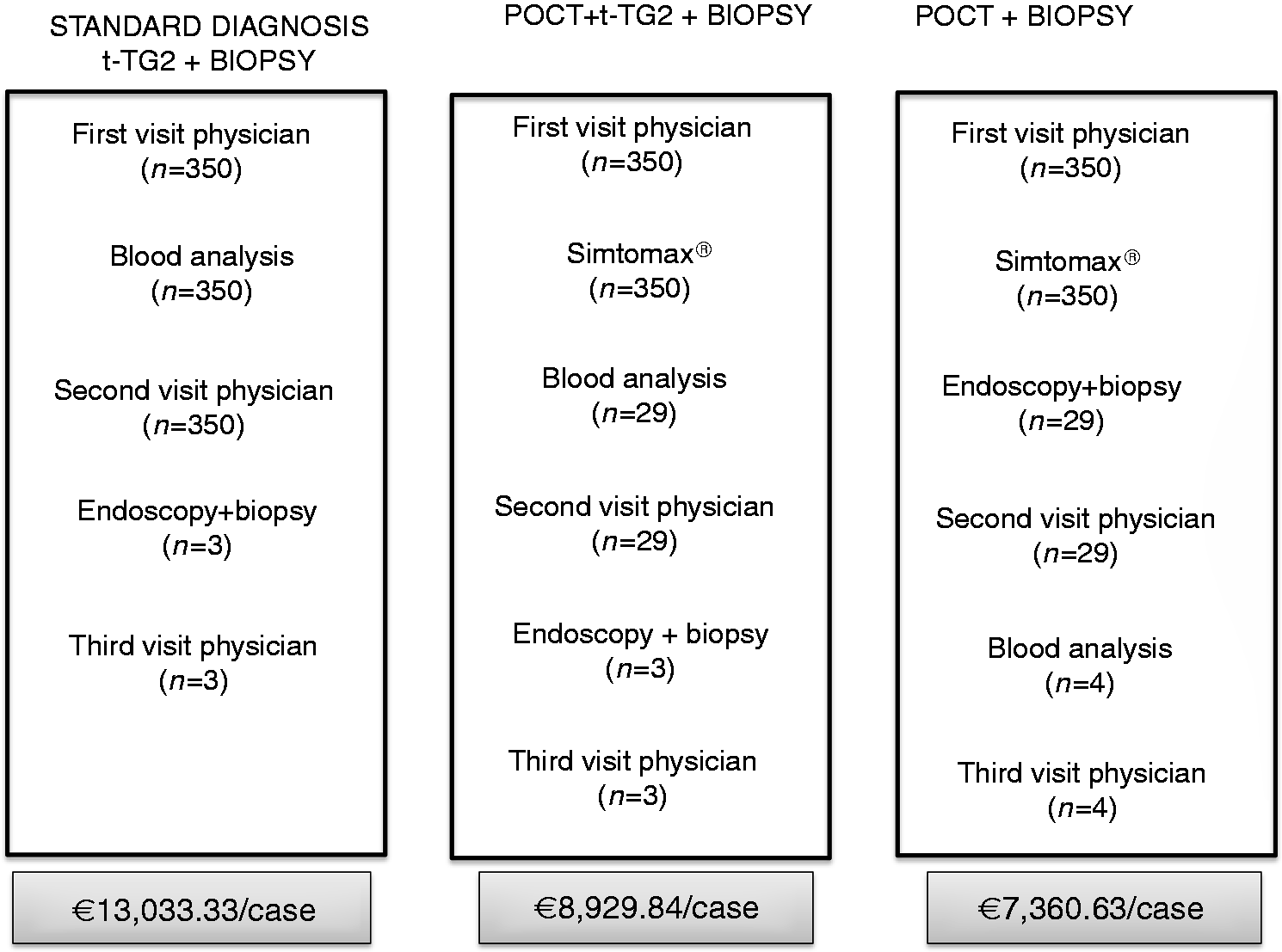
Prevalence of CD in the ‘case-finding’ and control cohorts
The prevalence of CD in the case-finding cohort was 1.14% (95% CI: 0.44–2.90), which represents 1:87. This figure is 3.8 times higher than that observed in the general adult population in the same geographical area by using a mass screening serological strategy. 21 During the previous 18 months before the initiation of the case-finding programme (1 October 2012 to 31 March 2014), 1050 serum IgA tTG2 determinations were performed and seven patients fulfilled the diagnostic criteria of CD, disclosing a prevalence of 0.67% (95% CI: 0.33–1.37), which represents 1:149. Six patients had atrophy and one patient had a Marsh 1 lesion. In the case-finding study using POCT, CD was diagnosed 1.7 times more often than in the previous period of time; however, differences were not statistically significant.
In addition, 24 PCPs that did not participate in the study requested 624 standard serology tests to rule out CD during the study period. Six out of the 624 patients fulfilled the diagnostic criteria of CD (two with atrophy, four with Marsh 1). The prevalence of CD in this cohort attended by PCPs not participating in the case-finding study was 0.96% (95% CI: 0.44–2.08), representing 1:104.
Discussion
The present study showed better diagnostic accuracy for a POCT based on IgA/IgG-DGP detection than standard serology for CD diagnosis. However, the low prevalence of CD in this case-finding cohort means that the difference in favour of POCT was due only to a single case diagnosed with POCT and not with standard serology. Thus, it can be considered that both serum IgA tTG2 and DGP-based POCTs are good diagnostic tools for CD screening in primary care. Similar diagnostic accuracy of both tests was demonstrated in the pre-endoscopy setting.14,15
The low PPV (14%) of the POCT Simtomax® was due to the detection of 7% false-positive patients for both CD diagnosis and IgA deficiency, suggesting that the cut-off for IgA/IgG-DGP detection is very low, ensuring that no cases of CD are missed. To ascertain whether this false-positive IgA/IgG-DGP detected by POCT could reflect a potential CD, the duodenal biopsy was analysed not only for histopathology but also for IEL subsets (T cell receptor (TCR) γδ+ and CD3–) and tTG2 IgA deposits. 19 Though the majority of false-positive patients had normal duodenal mucosa, 40% of them had duodenal markers of potential CD. However, similar results were obtained in a subgroup of 25 patients with negative POCTs and persistent symptoms in whom a duodenal biopsy was performed. Therefore, we realized that the majority of these patients did not have CD.
It is remarkable that some symptomatic patients may have a seronegative ‘coeliac-lite’ disease and may benefit from a GFD. 25 This was the case for two patients in the POCT-negative and two more in the POCT-positive group showing a good response to a GFD. In seronegative patients with a doubtful diagnosis of CD, the performance of a duodenal biopsy is mandatory. In these cases, the presence of duodenal CD markers together with positive genetic predisposition and histological lesion of the CD spectrum (either lymphocytic enteritis or atrophy) could be of help in deciding whether to start a GFD.19,25 Regardless, POCT based on IgA/IgG-DGP has no more accuracy for ‘coeliac-lite’ diagnosis than IgA tTG2.
The strategy of using the Simtomax® POCT for CD screening was more cost effective than a strategy based on tTG2 according to the tariffs of the National Health Services of the Catalan government. The immediate referral of the patients with positive POCT to the Endoscopic Unit for duodenal biopsy allowed us to reduce the time delay from the visit of the PCP to CD diagnosis. In addition, the POCT strategy allowed us to reduce the cost per patient to almost half compared with the cost using standard serology, even though 7% more patients in the POCT group than in the standard diagnosis had to undergo duodenal biopsy. However, it should be taken into account that prices may vary among countries, organisation of healthcare institutions and/or insurance companies.
The study also demonstrated that the case-finding strategy using a IgA/IgG-DGP POCT in patients at risk for CD is suitable for CD diagnosis in primary care since it allowed for the detection of four times as many cases of CD (1:87 (1.14%)) than that found in the general population of the same area (1:357 (0.28%)). 22
The post hoc analysis to assess the impact of the ‘educational programme’ on the number of diagnoses showed that PCPs participating in the POCT study diagnosed 1.7 more CD cases than those diagnosed in the previous 18-month period. Differences were not statistically significant but the sample size was not calculated to find differences between the two periods. In fact, taking into account the CD prevalence in our geographical area, more than 2000 patients per group would be needed to find differences.
The 0.96% prevalence of CD in the cohort attended by PCPs not participating in the case-finding study (using tTG2 for screening) was intermediate between those found in the POCT study (1.14%) and in the previous period (0.67%). Therefore, it seems that PCPs’ declining participation was also influenced by the ‘educational programme’ and by the active CD findings developed by their colleagues. A similar positive influence of an active CD case-finding environment was previously demonstrated by an Italian case-finding study in primary care. 6
The prevalence of CD in other case-finding studies using standard serology for CD screening ranged from 2% to 10%, a figure higher than in our study. However, in these geographical areas the prevalence of CD in the general population was 1:100, which is clearly higher than that reported in our country.5–9,16 Thus, the prevalence of CD in high-risk groups essentially depends on the prevalence of CD in a specific geographical area, as well as on the type of risk group included. In this sense, first-degree relatives have the highest risk of CD10,23 while patients with only one digestive symptom have the lowest CD risk. 9 In our study, the most frequent inclusion criteria were digestive symptoms, which accounted for 60% of the total cohort; the majority of them had only one digestive symptom.
The present study has some limitations. The most important is the use of tTGA as the main parameter for CD diagnosis. The availability of duodenal histopathology in all the patients would be desirable in future studies. This would provide valuable information deeper in the CD iceberg in seronegative patients. Another limitation is the number of included patients, which precluded establishing firm conclusions regarding the impact of the ‘educational programme’ on the number of new CD diagnoses between periods. At any rate, a POCT detection was demonstrated to be suitable for CD screening in primary care. POCT identified all seropositive patients while a negative test ruled out CD. It is easy to use, allows early diagnosis, and is cost effective compared with the standard approach using serology.
Footnotes
Acknowledgements
ME is the guarantor of this work.
Author contributions are as follows: ME, MR and FFB: design of the study, coordination, acquisition of data, statistical analysis and interpretation of data, manuscript writing, critical revision and approval of the final draft. MLl, JC, GM, MP, AC, AC, MI, PRR, ET, BA, CF, MM and JR: acquisition of data and approval of the final draft.
Declaration of conflicts of interest
ME has received funding from Tillotts Pharma Spain S.L.U. The other authors have nothing to declare.
Funding
This work was supported by a research grant from Tillotts Pharma Spain S.L.U., which had no input in the study design, access to the study data, or interpretation of results.
Informed consent
All patients signed informed consent to participate in this study.
Ethics approval
This study was conducted according to the ethical guidelines of the 1975 Declaration of Helsinki. Ethical approval was obtained from the Hospital Universitari Mutua Terrassa Ethical Committee on 25 February 2014.
