Abstract
Ceruminous adenocarcinoma (CAC) represents an exceedingly rare malignant glandular neoplasm originating from the ceruminous glands within the external auditory canal (EAC), constituting approximately 5%–10% of all EAC malignancies. Owing to its insidious onset and nonspecific symptoms, diagnosis is frequently delayed. This report delineates a case of right EAC CAC in a 39-year-old male, focusing on the diagnostic trajectory, multimodal therapy, and short-term outcomes. The patient presented with a one-month history of recurrent bloody purulent discharge from the right ear, intermittent otalgia, and mild conductive hearing loss. Otoscopy revealed a cauliflower-like, ulcerated, and bleeding mass on the floor of the right EAC. Computed tomography (CT) showed a mass within the right EAC encroaching upon the superficial parotid lobe and the facial nerve’s vertical segment, with no intracranial extension. Cervical ultrasound demonstrated multiple enlarged lymph nodes in right levels 2–4. A pathological biopsy confirmed CAC, with immunohistochemical staining positive for CK7, EMA, and partially for S-100. The patient underwent radical surgery, including right subtotal temporal bone resection (with EAC and mastoid removal, reconstructed using temporalis fascia and a skin graft), right total parotidectomy, and right cervical level 2–4 lymph node dissection. Intraoperatively, the tumor infiltrated the facial nerve sheath, but the nerve parenchyma was preserved; the sheath was dissected free without nerve resection or anastomosis. Postoperative adjuvant radiotherapy (60 Gy in 30 fractions) was administered. Recovery was uneventful. At 3-month follow-up, the EAC wound had healed completely, and CT/magnetic resonance imaging showed no residual or recurrent tumor.
Keywords
Introduction
Ceruminous adenocarcinoma (CAC) of the external auditory canal (EAC) is a rare malignant neoplasm arising from the epithelial and myoepithelial cells of the ceruminous gland ducts in the cartilaginous EAC. Despite an estimated overall incidence of 0.003%, it is highly malignant, with slow growth and a propensity for local invasion. Postoperative recurrence rates range from 40% to 60%, and advanced cases may metastasize to lungs and bones, posing a significant threat.1,2
As the predominant malignant glandular tumor of the EAC, it accounts for roughly 70% of all ceruminous gland tumors here, with higher prevalence along the posterior and anterior walls.3,4 Atypical early symptoms—intermittent ear pain, mild hearing loss, purulent/bloody discharge, or crusting—often lead to misdiagnosis as chronic otitis externa, cerumen impaction, or papilloma, causing delay.5,6 Approximately 60% of patients are diagnosed at intermediate or advanced stages, with potential invasion of the middle ear, facial nerve, or skull base, complicating treatment. 7
Histopathologically, Wetli’s 1972 classification categorizes ceruminous gland tumors into four types: ceruminous adenoma, pleomorphic adenoma, adenoid cystic carcinoma, and CAC. The latter is distinguished by high invasiveness, cellular atypia, increased mitotic figures, and stromal infiltration. 1 Despite advances in surgery and radiotherapy, prognosis remains poor, with a 5-year survival rate around 30%.2,4
Patient overview
A 39-year-old male presented with a one-month history of purulent and bloody secretions from the right EAC. His medical history included 5 years of chronic right external otitis, intermittently treated with ofloxacin ear drops. He denied any history of head/neck radiotherapy, chemical carcinogen exposure, or HPV infection.
Physical examination revealed a cauliflower-like mass (approx. 1.0 cm × 0.5 cm) on the floor of the right EAC. The mass was hard, bled easily on contact, and partially obstructed the canal, obscuring the tympanic membrane. The left EAC was patent with an intact tympanic membrane. The patient exhibited signs of right facial paralysis: absence of the right frontal wrinkle, incomplete eyelid closure (3 mm scleral show), shallow right nasolabial groove, and drooping of the right mouth corner with deviation to the left upon smiling, consistent with House-Brackmann grade IV. 8 Neck examination revealed two enlarged, hard, poorly defined, immobile lymph nodes (approx. 2.0 cm × 1.5 cm) in right regions I–III.
High-resolution CT of the temporal bone revealed a soft tissue mass (CT value ~45 HU) in the right EAC, encroaching upon the superficial parotid lobe and the vertical facial nerve segment, evidenced by facial nerve canal enlargement and bony wall absorption. The mastoid air cells were patent without bony destruction (Figure 1). Preoperative magnetic resonance imaging showed a well-defined, heterogeneously enhancing soft tissue mass (approx. 1.2 cm × 1.8 cm × 2.0 cm) within the right EAC, extending anteriorly to involve the superficial parotid lobe and intimately contacting the vertical facial nerve segment, which appeared contiguous. No invasion into the middle ear, temporomandibular joint, skull base, or distant metastasis was detected.
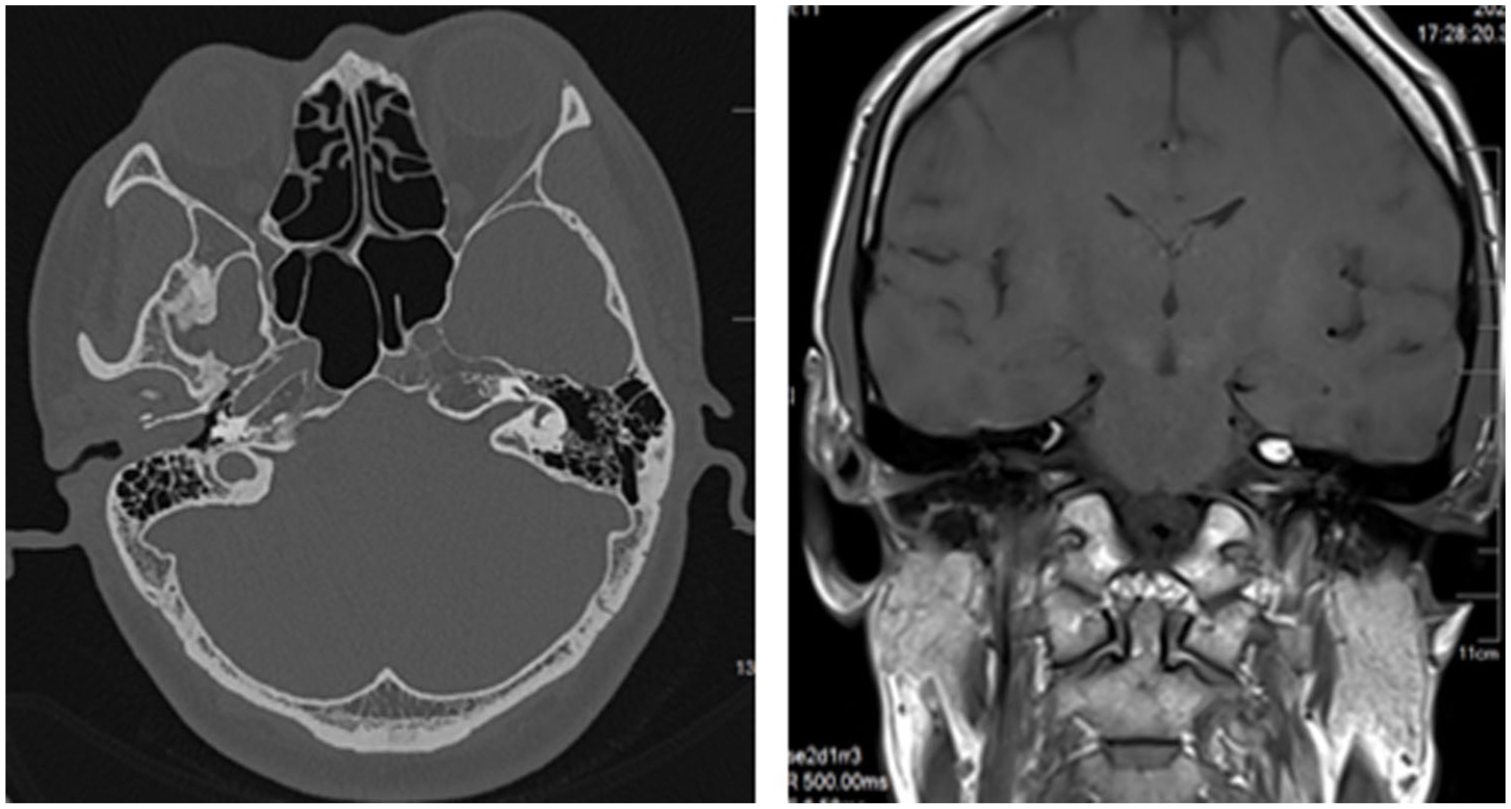
CT and MRI imaging delineates a space-occupying lesion within the right external auditory canal.
Neck ultrasound identified five enlarged lymph nodes in right cervical regions I–III, with thickened cortices and obliterated medullae; calcifications in two nodes raised suspicion for metastasis (Figure 2).

Neck color ultrasound reveals enlarged lymph nodes in the right cervical region.
Pathological biopsy of the EAC mass showed atypical hyperplasia of cerumen gland duct epithelial cells arranged in glandular/cribriform patterns, with an elevated mitotic count (~5/10 HPF). The diagnosis was “right EAC CAC with cervical lymph node metastasis (moderately differentiated)” (Figure 3).
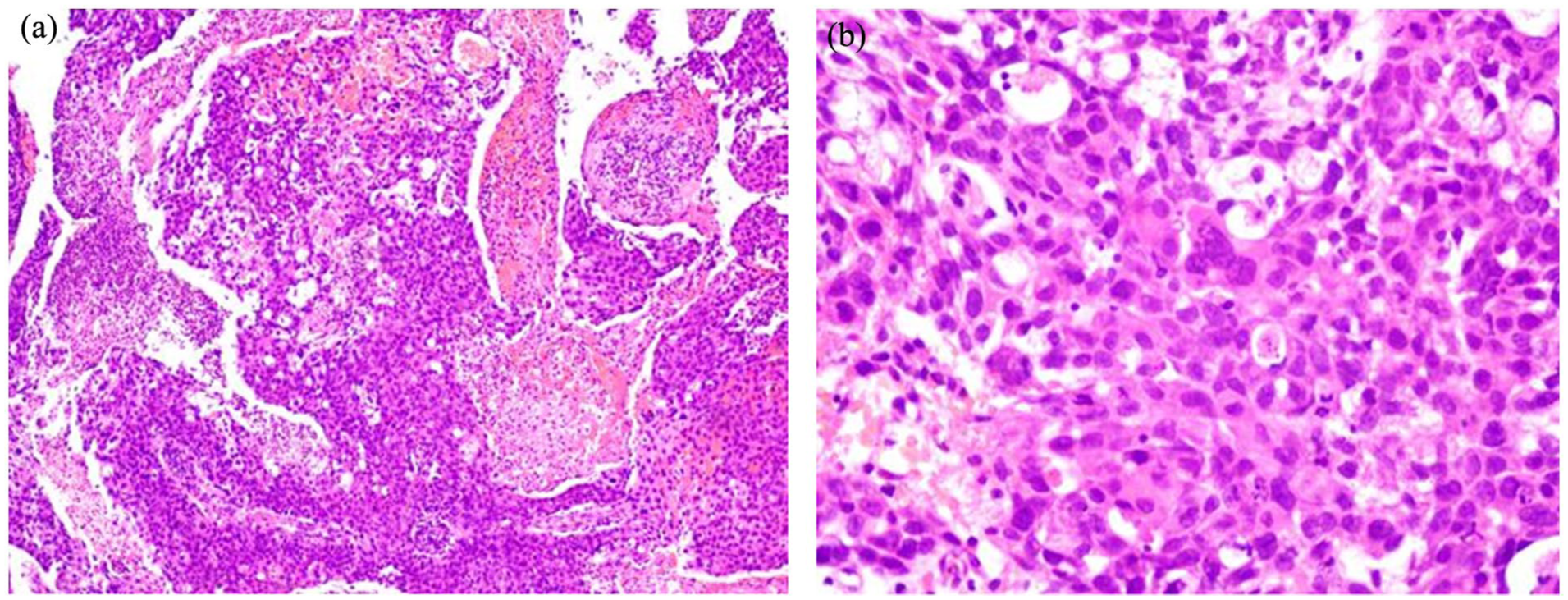
(a) HEx100 tumor cells were distributed in a patchy pattern, with patchy necrosis observed in the stroma. (b) HEx400 tumor cells show large, deeply stained nuclei with irregular shapes, megakaryocytes, and frequent mitotic figures, with medium-sized, eosinophilic cytoplasm.
Surgical treatment and procedure
An S-shaped incision (approx. 12 cm) was made, starting 1 cm anterior to the tragus, descending along the helix crus to the earlobe, angling below the mastoid, and ending 2 cm below the mandibular angle, exposing the parotid region and cervical lymph nodes.
Subtotal temporal bone resection (STBR) was performed: en bloc resection of the cartilaginous and bony EAC along with adjacent mastoid cortex. The uninvolved tympanic membrane and ossicles were preserved. The lateral temporal bone and EAC defect was reconstructed with a temporalis muscle fascia flap covered by a split-thickness skin graft, and a meatoplasty was performed to maintain patency.
The skin, subcutaneous tissue, and platysma were incised. The parotid fascia was separated to expose the superficial parotid lobe. Using the main trunk of the facial nerve (located between the mastoid’s anterior edge and EAC cartilage, ~2.5 cm deep) as a landmark, a nerve dissector was used to gently separate tissue distally, exposing the temporal, zygomatic, buccal, marginal mandibular, and cervical branches (Figure 4). During dissection, the tumor was found infiltrating the facial nerve sheath at the vertical segment. The sheath was carefully dissected and removed with the tumor, while the underlying nerve fascicles were preserved intact. No nerve resection or anastomosis was required.
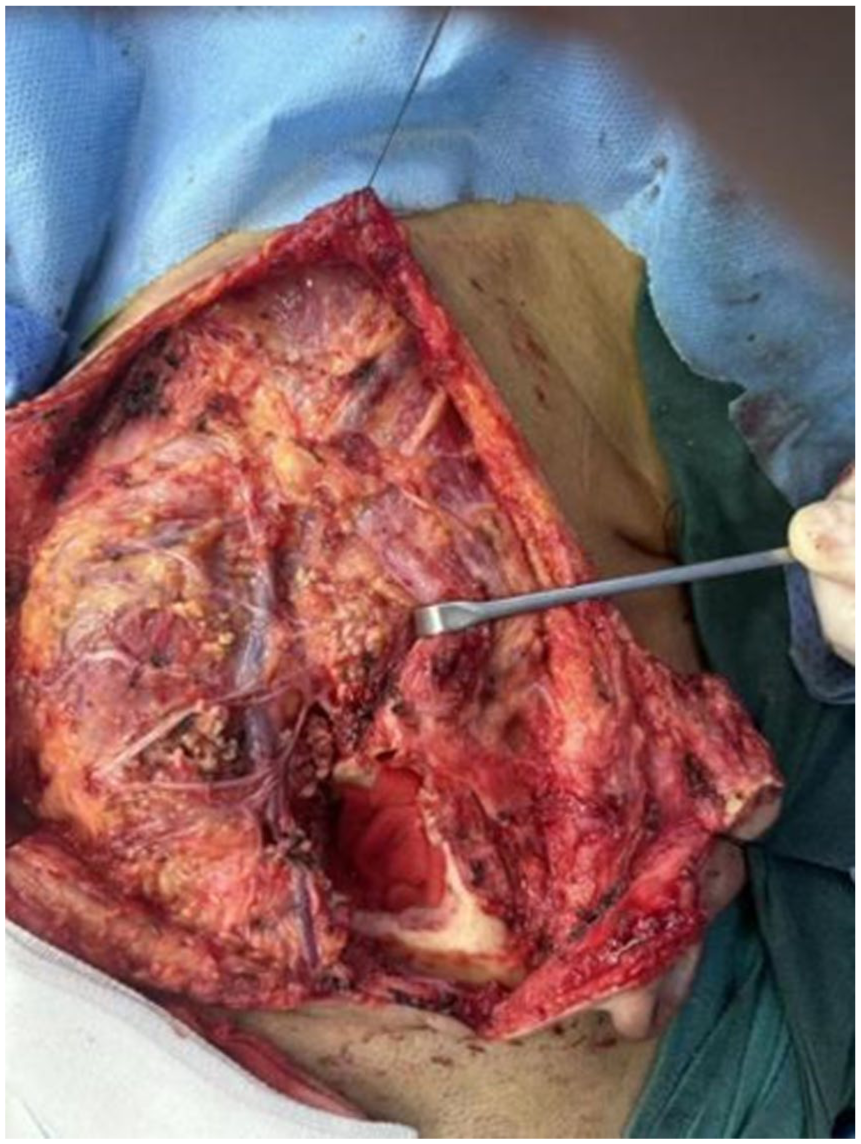
In the right surgical field, the main trunk and its branches of the right facial nerve are observed to be intact.
It is discovered that the tumor has infiltrated both the superficial and deep lobes of the parotid gland. However, the facial nerve sheath remains undamaged (the nerve parenchyma is not involved). Subsequently, a complete resection of the superficial and deep lobes of the parotid gland is carried out (as depicted in Figure 5).

This image presents the surgical cavity after parotidectomy, revealing the well-preserved vertical segment of the facial nerve.
The tumor had infiltrated both superficial and deep parotid lobes. A total parotidectomy was performed, preserving the facial nerve (Figure 5). Dissection continued along the mandible margin to expose right cervical levels II–IV. Five enlarged, hard, mildly adherent lymph nodes were dissected, preserving the internal jugular vein and accessory nerve.
Postoperative management and pathological results
Radiotherapy protocol
Two weeks post surgery, the patient received three-dimensional conformal radiotherapy: total dose 60 Gy in 30 fractions (2 Gy/fraction, 5 fractions/week). Oral mucosa was protected with Kangfuxin solution gargles.
Pathological findings
The right EAC mass was confirmed as CAC (moderately differentiated; 1.0 cm × 2.5 cm × 1.8 cm), invading the deep parotid lobe and facial nerve sheath but not the skin. Postoperative pathological stage was T4N2M0. 8
Metastasis was found in two of five dissected right cervical level II–IV lymph nodes, with one node showing a cancerous embolus (Figure 6).

Postoperative pathological examination of the right cervical lymph nodes confirms the presence of cancer metastasis: (a) HEx100 tumor cells were found in the lymph nodes in a patchy distribution; (b) HEx400 cancer thrombus is found in the blood vessel.
Follow-up outcomes
One month post surgery
The incision healed well. Facial paralysis improved to House-Brackmann grade III.
Six months post surgery
Otoscopy showed no recurrence in the right EAC surgical cavity. Neck ultrasound confirmed no enlarged lymph nodes. Chest CT showed no distant metastasis.
One year post surgery
Facial paralysis further improved to House-Brackmann grade II. The patient resumed normal work.
Discussion
Ceruminous gland adenocarcinoma of the EAC originates from the ductal epithelial or myoepithelial cells, constituting 15–20% of all EAC malignancies. Its main subtypes are adenoid cystic carcinoma and ceruminous gland ductal carcinoma. 9 Etiology may involve chronic inflammatory stimulation (as in this case), genetic anomalies (e.g. TP53 inactivation, EGFR overexpression), and radiation exposure. 10 Pathologically, moderately differentiated CAC is prone to perineural invasion (e.g. facial nerve), with high local recurrence (~30%) and neck lymph node metastasis (~25%) rates. 11
Surgical management depends on tumor extent. Tumors confined to the EAC without bone invasion may undergo lateral temporal bone resection. For limited bony erosion, STBR—as performed here—is indicated, involving en bloc removal of the EAC, tympanic bone, and partial mastoid. Extensive skull base involvement may require total temporal bone resection. Goals are complete excision with clear margins and neurological preservation where possible. Contraindications include unresectable disease or severe comorbidities. Complications include facial nerve paralysis, hearing loss, cerebrospinal fluid leak, vertigo, Frey syndrome, and wound issues. 12
When the parotid gland is invaded, total parotidectomy is standard. In this case, total parotidectomy was performed with meticulous facial nerve preservation, as the nerve sheath was infiltrated but the parenchyma was intact. When nerve parenchyma is compromised, nerve grafting (e.g. great auricular nerve) may be considered. 13 For patients with neck lymph node metastasis (cN1), dissection of levels II–IV improves survival (5-year survival ~60%). 14 Here, 2 of 5 nodes were metastatic, warranting postoperative radiotherapy.
CAC is radiosensitive; adjuvant radiotherapy reduces local recurrence rates from ~40% to ~15%. 15 Indications include positive/close margins (<5 mm), advanced stage (T3/T4), lymph node involvement (pN+), adenoid cystic carcinoma subtype, and neurovascular/parotid invasion. 9 Postoperative radiotherapy (60 Gy) in this case resulted in no recurrence at 1-year follow-up.
Key prognostic factors include margin status, T-stage, and distant metastasis. Adenoid cystic carcinoma has late local recurrence and distant metastasis (often pulmonary), with acceptable 5-year but declining long-term survival. Ductal carcinoma is more aggressive with earlier metastasis and poorer survival. Overall 5-year disease-specific survival is 60%–80%. Patients with early-stage (T1/T2) tumors and negative margins have >85% 5-year survival, while those with advanced-stage (T3/T4) or positive margins may have <50% survival. Temporal bone resection causes permanent ipsilateral hearing loss. If the facial nerve is preserved, outcomes are favorable; if sacrificed, functional recovery typically plateaus at House-Brackmann grade III–IV even with grafting. Intraoperative nerve monitoring is essential. 16
Conclusion
This case illustrates the management of advanced CAC involving the parotid gland and cervical lymph nodes. Its nonspecific symptoms and location often lead to delayed diagnosis and early invasion of critical structures. A multidisciplinary strategy—comprising precise imaging, radical surgery aiming for clear margins with neural preservation, and adjuvant radiotherapy—achieved good local control and functional recovery (House-Brackmann grade IV to II). Given high recurrence and metastasis risks, particularly for adenoid cystic carcinoma, lifelong surveillance is mandatory. A treatment paradigm centered on radical surgery followed by tailored radiotherapy appears effective for locally advanced disease, balancing oncologic control with quality of life.
Footnotes
Ethical considerations
The study was reviewed and approved by the Ethics Committee of Ganzhou People’s Hospital.
Consent for publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data presented in this study are available on request from the corresponding author.*
