Abstract
Dermatomyositis is a rare autoimmune condition known to be frequently associated with an undiagnosed malignancy. Although it is strongly linked to adenocarcinomas and other solid tumors, dermatomyositis has been scarcely described with sarcomas. We present a 63-year-old patient with simultaneous presentations of Kaposi’s sarcoma and dermatomyositis. This case highlights Kaposi’s sarcoma as a potential malignancy associated with dermatomyositis and the complexities of managing multimorbid patients.
Introduction
Dermatomyositis (DM) is an immune-mediated myopathy characterized by skeletal muscle weakness and skin changes. 1 It is thought to be mediated by the activation of the complement cascade, subsequently leading to a humoral response against the capillary and arteriolar endothelium. 2 While the exact cause of DM remains unknown, it has been hypothesized that various genetic, immunologic, and environmental factors are involved. 3 Additionally, DM has been associated with numerous malignancies, most commonly breast, ovarian, and lung cancers. 4 We therefore report a rare case of paraneoplastic DM in a patient with disseminated Kaposi’s sarcoma (KS).
Case presentation
A 63-year-old male patient presented to the dermatology clinic complaining of a 2-week itchy skin eruption. His past medical history revealed significant comorbidities, including longstanding human immunodeficiency virus (HIV) infection treated with dolutegravir/rilpivirine and end-stage renal disease secondary to Goodpasture syndrome requiring thrice weekly hemodialysis. Before starting dialysis, he was hospitalized and unsuccessfully treated with high-dose pulse intravenous methylprednisolone for Goodpasture syndrome. After discharge, his steroids were slowly tapered in the following months. Steroid tapering was fully completed 2 weeks after his first dermatology consultation. At his initial visit, his viral load was undetectable, but his CD4 count was abnormally low at 165 cell/mm3, likely associated with steroid-induced immunosuppression. Physical examination revealed erythematous and squamous lichenoid plaques predominantly localized on the scalp, back of the neck, elbows, dorsal aspect of proximal, and distal interphalangeal joints as well as upper back with relative sparing of the face and abdomen. Additionally, one singular 1.2 cm lobulated violaceous plaque was visualized on the upper back (Figure 1). Considering the patient’s immunosuppressed and HIV positive status, the infiltrative violet plaque immediately raised concerns of KS. As for the squamous plaques, numerous hypotheses such as drug eruptions, acquired ichthyosis, HIV related rashes, and DM were included in the initial differential diagnosis. Punch biopsies of both types of lesions were performed and sent for histopathological analysis.

Initial Kaposi’s sarcoma on the left upper back.
Histopathology of one violaceous nodule revealed dermal proliferation of spindle cells with extravasated red blood cells and perivascular plasma cells. Immunohistochemistry staining also detected the presence of human herpesvirus-8, thereby confirming KS. Biopsy of one of the erythematous plaques demonstrated vacuolar interface dermatitis with the presence of perivascular lymphocytic infiltrates, necrotic keratinocytes, and mucin deposits in the superficial dermis, compatible with DM.
At his follow-up appointment, the patient’s condition significantly deteriorated. We noticed the emergence of at least 15 new vascular violaceous lesions compatible with KS on the scalp, arm, trunk, and oral mucosa. Examination also revealed palpable inguinal lymph nodes of which a biopsy confirmed the presence of KS. Furthermore, a bronchoscopy detected endobronchial KS lesions. His erythematous lesions also became more pronounced and itchier, particularly on the scalp, the upper back (recognized as shawl sign), the lateral thighs (Holster sign), the elbows (Gottron sign), as well as the dorsal aspects of metacarpophalangeal and interphalangeal joints (Gottron papules; Figure 2). He also reported new-onset fatigue and proximal muscle weakness of the lower limbs. Examination of nail folds was unremarkable. He was subsequently admitted for further investigations. A complete blood count showed lymphopenia while C-reactive protein and creatine kinase levels were normal. Antinuclear antibody titer was 1:80, while extractable nuclear antigens and a myositis panel came back negative. DM paraneoplastic to KS was ultimately diagnosed.
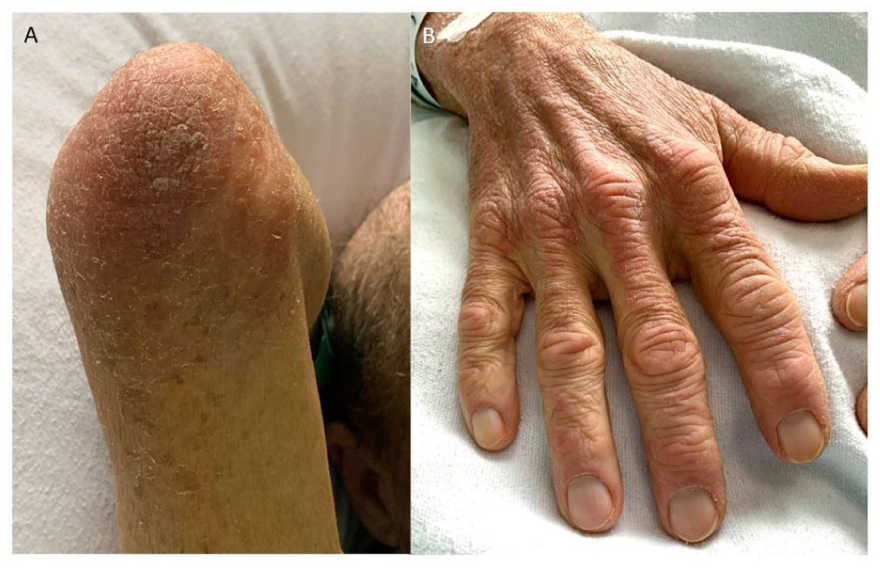
(A) Erythema affected the redness over the backs of the patient’s elbows corresponding to Gottron sign. (B) Red papules on the dorsal aspects of the patient’s metacarpophalangeal and interphalangeal joints corresponding to Gottron papules.
Due to his comorbidities, the patient refused further investigations and opted to discontinue dialysis and to seek palliative care. To relieve scaling and itching, we proposed continuing daily topical steroid applications on affected regions along with moisturizing cream. One week later, he unfortunately succumbed to his comorbidities.
Discussion
Only a few cases of patients presenting with both DM and KS have so far been described in the literature. In most of these cases, KS was deemed as a consequence of the immunosuppressive regimen used to treat DM. In fact, an onset of KS in an immunocompromised patient is among its most typical presentations. There are four major clinical subtypes of KS: the classical, endemic, iatrogenic, and HIV-AIDS-related types, with the latter two being directly linked to an immunocompromised state.5,6
Our case, however, differs from the previously described cases of DM associated with KS. In our patient, both conditions presented concurrently, whereas previous reports typically described an apparent DM months to years before the KS diagnosis. 7 Notably, Simeoni et al. 8 documented the development of KS 8 months after initiating high-dose systemic steroids and cyclophosphamide for the patient’s underlying DM. Likewise, Liang et al. 9 reported on a patient who received a definitive diagnosis of KS 6 months following immunosuppressive drugs for DM. However, in retrospect, the authors state that suspect skin lesions for KS may have already been present at an earlier time, but initially went unrecognized. In the case described by Alghanim and Gasmelseed, 7 their patient presented with lesions compatible with KS that were first noticed 1 month after commencing DM treatments. Due to the short time interval between both onsets, the authors favored the diagnosis of paraneoplastic DM. In our case, the simultaneous appearance of both lesions as well as the absence of new immunosuppressive drugs in the patient’s medication list are more suggestive of DM being paraneoplastic to KS.
Paraneoplastic syndromes arise due to the production of signaling molecules such as autoantibodies, cytokines, peptides, or hormones by malignant cells. 10 These syndromes frequently exhibit heterogeneous manifestations and multi-systemic involvements. Paraneoplastic syndromes are most commonly associated with solid internal malignancies, particularly lung cancer. 11 With regards to KS, paraneoplastic syndromes are exceedingly rare, and only a small number of cases have been discussed in the literature. Other than DM and autoimmune hemolytic anemia, three other paraneoplastic syndromes have been described with KS: two cases of scleroderma as well as singular reports of myasthenia gravis and paraneoplastic autoimmune multi-organ syndrome. 12
In conclusion, our case highlights the notion that DM can develop as a paraneoplastic syndrome of KS. It further reiterates the importance of searching for malignancies in patients presenting with seemingly isolated DM, especially if the patients are of older age.
Footnotes
Acknowledgements
None.
Ethical considerations
Institutional review board approval was not required due to the retrospective and singular nature of case reports.
Consent to participate
Informed consent was obtained taken from the patients’ family for the publication of images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
