Abstract
Henoch-Schönlein purpura is the most common systemic vasculitis in children, characterized by IgA deposits in small vessels. The etiology is unknown, but Henoch-Schönlein purpura typically follows an upper respiratory infection, or less frequently other infective or chemical triggers. The classic tetrad of symptoms includes palpable purpura (mandatory criterion), arthralgias, abdominal pain, and renal involvement. However, the cutaneous rash of Henoch-Schönlein purpura is not the presenting sign in approximately one-quarter of patients. Moreover, the other typical manifestations can present isolated or nuanced; for that reason, a prompt diagnosis may be challenging. Other clinical findings such as subcutaneous edema in hands, ankles, and feet, are quite common at pediatric Henoch-Schönlein purpura onset. Edema occurring in other locations (i.e facial and genital swelling), is uncommon, but can be a helpful additional clinical sign of Henoch-Schönlein purpura. To our knowledge, only two cases of lumbar swelling as Henoch-Schönlein purpura presentation signs have been described in literature so far.
Keywords
Introduction
Henoch-Schönlein purpura (HSP) is a small-vessel IgA-immune complex vasculitis, 1 which represents the most common form of systemic vasculitis in children. HSP typically occurs between the age of 3 and 15 years, 2 with an incidence peak between 3 and 7 years. In Europe, the annual incidence is 13–20 per 100,000 children; 3 a higher rate has been reported in several Asian studies. 4 Most studies show a slight male predominance. 5 HSP occurs primarily in fall, winter, and spring; 6 approximately 50% of HSP cases are preceded by an upper respiratory tract infection 7 (either viral or Streptococcic), vaccinations or insect bites. Symptoms vary in timing and severity, 8 but fortunately, the course of HSP is usually benign and self-limiting. According to Ankara 2008 pediatric consensus criteria developed by the main international rheumatology societies (EULAR, PRES, and PRINTO 9 ), the mandatory criterion is purpura, as more than three-quarter of affected children present the classic cutaneous rash at HSP onset.10,11 Moreover, in the classic tetrad of symptoms are also included abdominal pain, arthralgia, and renal involvement. However, either purpura or other typical clinical manifestations can present isolated or nuanced, and this can delay diagnosis and treatment. For this reason, other clinical findings should be considered. In fact, subcutaneous edema, particularly in the hands, ankles, and feet, is a common finding at HSP onset. Edema occurring in other locations, such as facial and scrotal edema, has been reported.5,12,13 In the literature, a few cases of lumbar edema as HSP presentation sign have been reported, so far.14,15 Even if lumbosacral swelling is much more uncommon, it could be considered as a helpful additional sign of HSP.
Case reports
Patient 1
A 3-year-old boy was referred to our hospital for left ankle swelling and refusal to walk. A few days before, he had sickness, abdominal pain with vomit, rhinitis, and catarrhal cough. Not traumatic events were reported. His family past illnesses history was unremarkable. Physical examination revealed hyperemic pharynx, three small petechiae, respectively on the left buttock, soft palate and chest, a significant swelling of the external peri-malleolar left ankle and of the dorsal region of the left foot, with ecchymotic suffusion and pain on active and passive movements. Ultrasound (US) showed a moderate edema of periarticular soft tissues, without joint effusion or synovial thickening. Blood and urine tests were negative. Nasopharyngeal swabs for SarsCoV2 and group A beta-hemolytic Streptococcus were negative, too. After 24 h of observation, his clinical conditions improved, and he was discharged.
Few days later, he was readmitted to our emergency department for a remarkable painful, warm swelling of the back in the paravertebral lumbar region (Figures 1 and 2). Subcutaneous edema of the right ankle and of both knees was also present. Blood and urine tests were negative.

Painful, warm swelling of the back in the paravertebral lumbar region in Patient 1.

Painful, warm swelling of the back in the paravertebral lumbar region in Patient 1.
During hospitalization, he presented fleeting swelling with a deep marked migrating livedo.
He required on-demand oral acetaminophen for abdominal pain for a total of five doses. Corticosteroid therapy was not necessary. In the suspect of an angioedema in HSP, an oral antihistaminic therapy was started with partial benefit, so after 72 h was stopped.
Autoimmunity tests were negative and C1q-ihibitor and tryptase serum levels were normal. Platelets count and coagulation tests were in range, except for a moderate reduction of plasma factor XIII activity (53%, n.v: 70%–140%). Nasopharyngeal swab for an enlarged panel of respiratory virus resulted positive for Rhino/Enterovirus. Viral serology for EBV, CMV, Parvovirus B19, Borrelia, Rickettsiae, Leishmania, and Toxoplasma gondii were negative. Fecal occult blood resulted positive in two samples. Coproculture was negative. Urine tests maintained normochromic, without proteinuria. Abdominal US resulted normal; the US of skin and soft tissues showed a minimal imbibition.
The patient never manifested an evident and typical purpuric rash and progressively recovered. He was discharged with a presumptive diagnosis of HSP. The planned follow-up is still ongoing.
Patient 2
A 4-year-old boy was referred to our hospital for right ankle and lumbar swelling in a recent onset of cutaneous vasculitis, without fever or macrohematuria. In the last 10 days, he had been treated with oral corticosteroids (betamethasone 1 mg/kg/die for 4 days) for a cutaneous rash. Physical examination at admission revealed petechiae and ecchymosis especially on the lower limbs and on the buttocks, with some infiltrated elements. Swelling of the eyelids, left hand, right ankle (Figures 3 and 4) and lumbar region were present, too. Paravertebral lumbar region US was performed and showed an hyperechogenic thickening of superficial soft tissues with edematous aspect. Abdominal US resulted normal. Blood tests showed leukocytosis, negative C-reactive protein, and elevated D-dimer (4397 ng/mL). Peripheral blood smear showed activated lymphocytes. Platelets count and coagulation tests were normal. Rapid antigen test for severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) was negative, too.

Right ankle swelling in Patient 2.

Left wrist/hand with ecchymotic suffusion and edema in Patient 2.
During hospitalization, blood tests were repeated and showed a progressive normalization of white blood cells count, a reduction of D-dimer and a normal peripheral blood smear. Platelets count and coagulation tests were normal, except for a significative reduction of plasma factor XIII activity (32%). Autoimmunity tests were negative. Viral serologies for CMV, EBV, Adenovirus, Parvovirus B19, and HHV6 were negative. Fecal occult blood was positive in three samples. Urine tests maintained normochromic, without proteinuria.
Due to the worsening of clinical conditions, abdominal pain with increase of facial and lower limbs edema and appearance of a significant scrotal edema, a genital US was performed and showed a thickening of scrotal tunics. For this reason, a corticosteroid intravenous (IV) therapy with methylprednisolone (0.8 mg/kg/die) was started. After 6 days of IV therapy, due to improvement of general conditions and disappearing of the purpuric lesions, oral steroid therapy with prednisone (1 mg/kg/die) with a progressive decalage was started. The boy was discharged with a presumptive diagnosis of HSP. The planned follow-up is still ongoing, but oral steroid therapy was stopped after 10 days.
Discussion
During 2020, 10 children with a median age of 4.6 years were hospitalized in our Tertiary Care Pediatric Hospital (Regina Margherita Children Hospital, Turin) with a diagnosis of HSP. Herein, we describe two children who had an atypical presentation of the disease.
According to Ankara 2008 pediatric consensus criteria for HSP developed by the EULAR, the PRES, and the PRINTO, 9 the mandatory criterion is purpura (usually palpable and in clusters) or petechiae, with lower limb predominance and without thrombocytopenia or coagulopathy. Patients also must have one or more of the following: abdominal pain, arthritis or arthralgia, renal involvement (proteinuria, hematuria), and anatomopathological findings of leukocytoclastic vasculitis or proliferative glomerulonephritis, with IgA immune-complex deposition within the vessel walls at immunofluorescence studies on skin or kidney biopsy. More than three-quarter of children present the classic cutaneous rash at HSP onset;10,11 however, the typical clinical manifestations can present isolated or nuanced.
In our two patients, both the lower limb skin rash and the renal involvement were not present. Both displayed abdominal pain with positive fecal blood samples and one presented migrating arthralgia.
Subcutaneous edema, particularly in the hands, ankles, and feet, is a common finding at HSP onset; edema occurring in other locations, such as facial and scrotal edema, has been reported too.5,12,13 Only a few cases of lumbar edema as HSP presentation sign have been reported in literature so far.14,15 Interestingly, in our cases, the vasculitis affected the lumbar region, as an unusual position. The observed lumbar swelling, as well as other subcutaneous edema, are likely the result of leakage from blood vessels into the surrounding tissue due to the vasculitic inflammatory process.
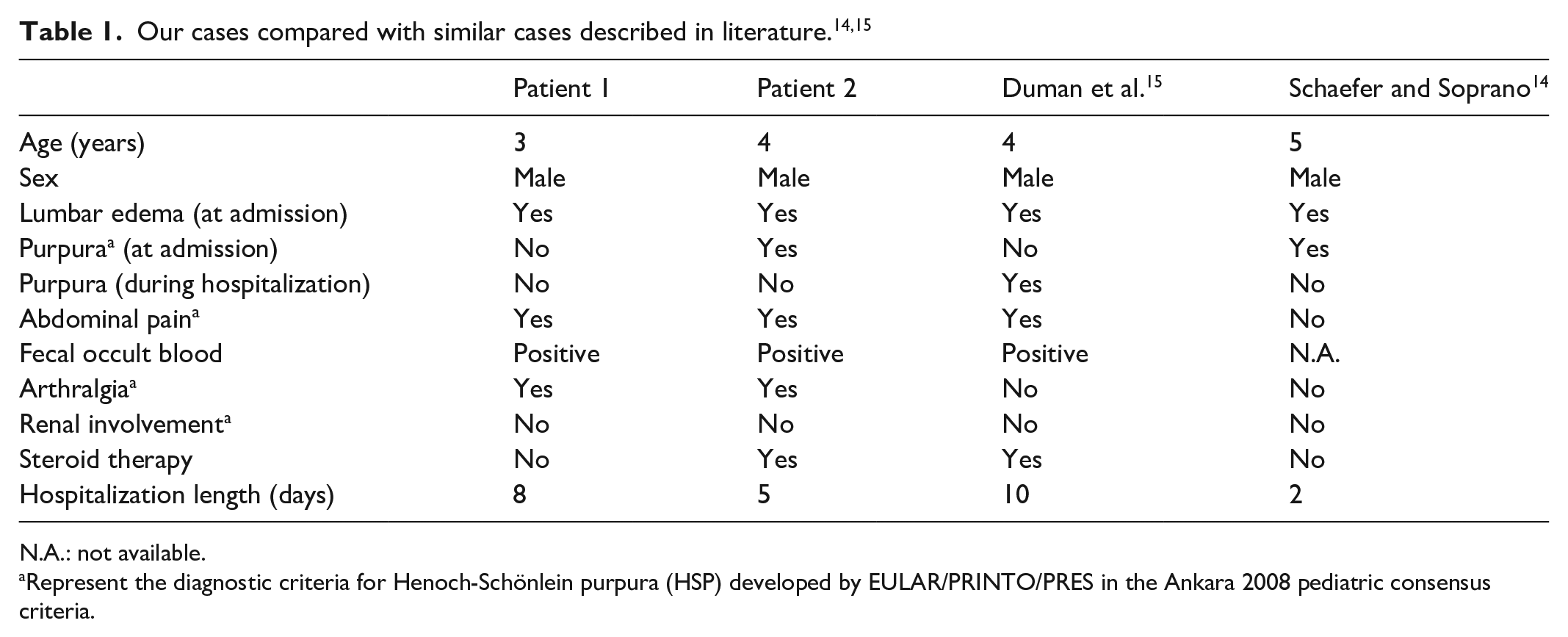
Table 1 compares our cases with other two similar cases described in literature. All the four patients are male, with a 4-year-old median age. Two out of four did not present the typical cutaneous rash at admission, and one of them never presented it. Both our patients presented migrating swelling, also in uncommon locations such as in the paravertebral lumbar region, described as a minimal imbibition of soft tissue at US images, similarly to the two cases described in literature. None of the cases presented renal involvement, while abdominal pain was described in three of them and associated with fecal occult blood finding. Steroid therapy was necessary only in two of the four patients, according to clinical conditions. They all required short hospitalization with a median length of 6 days.
N.A.: not available.
Represent the diagnostic criteria for Henoch-Schönlein purpura (HSP) developed by EULAR/PRINTO/PRES in the Ankara 2008 pediatric consensus criteria.
As the clinical picture of HSP is often incomplete, the diagnosis can be missed when based on clinical signs only. 16 The measurement of plasma factor XIII (F XIII) activity has been proven to be helpful in the diagnosis of HSP even before the rash development. It has also been postulated that F XIII activity correlates with the severity of abdominal symptoms. 17 In both our cases, a moderate to significative reduction of plasma F XIII activity was observed, with a progressive normalization in the subsequent follow-up. In Case 2, a more significant reduction of F XIII activity was correlated with more severe abdominal pain and worsening of general conditions that required steroidal IV therapy.
Although signs and symptoms may vary in timing and severity, 8 the course of HSP is usually benign and self-limiting. Both our patients were discharged respectively after 8 and 5 days of hospitalization with a planned follow-up, which is still ongoing.
Conclusion
In conclusion, we suggest lumbar edema as an additional useful clinical sign to direct the diagnostic workup in children with nuanced classic HSP presenting symptoms.
Footnotes
Acknowledgements
The authors would like to thank Professor Ramenghi, chief of Pediatric Department and Professor of Pediatrics at University of Turin and all doctors and nurses who helped them in treating these patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.