Abstract
The following case report details the case of a 40-year-old Caucasian patient who presented with dyspnea following a serologically confirmed mild-to-severe pulmonary infection with SARS-CoV-2. Chest computer tomography revealed a solitary ground-glass pulmonary nodule in the lower right lobe, measuring 2.1 cm in diameter. Video-assisted thoracoscopic surgery wedge resection revealed well-circumscribed lymphoid aggregates adjacent to the round, smaller airways, bronchioles, and blood vessels. IgKappa B exhibited a monoclonal polyclonal pattern, in contrast to the behavior exhibited by IgKappa A and IgLambda. In the following discussion, the lymphoid lesion was considered in the context of lymphoid hyperplasia, accompanied by an early infiltration of low-grade extranodal B cell lymphoma of the bronchus-associated lymphoid tissue (BALToma).
Keywords
Case presentation
A 40-year-old Caucasian woman with no smoking history was admitted with dyspnea and complaints of fatigue. After she had been afflicted by a serologically confirmed mild-severe pulmonary SARS-CoV2-infection 4 weeks ago. It was found that no other active or past infectious diseases, such as tuberculosis or influenza, could be clinically detected. A review of the patient’s medical history revealed no previous illnesses or autoimmune diseases. Furthermore, typical serum markers for lymphoma were not elevated. Physical examination, complete blood count, and liver and renal function tests were totally within physiological limits. Chest computer tomography revealed a subpleural, single ground-glass pulmonary nodule in the right lower lobe with the size of 2.1 × 2 × 1.6 cm. She underwent a video-assisted thoracoscopic surgery wedge resection of this suspicious pulmonary nodule. We analyzed the resected lung tissue by multiscale X-ray phase-contrast computed tomography (Figure 1) and scanning electron microscopy (Figure 2). Histopathological examination revealed well-circumscribed lymphoid aggregates adjacent to the round smaller airways, bronchioles, and blood vessels. Lung parenchyma with tumor-like interspersing of lymphoid tissue aggregates was seen, with germinal centers with mantle zones of variable width (ki67, CD10, CD20, Bcl2). Large networks of follicular dendritic reticulum cells (CD23-positive) were repeatedly seen (Figure 3). In the interfollicular regions and high-endothelial venules (HEV), there were aggregates of small lymphocytes, often primarily B cells (CD20) mixed with abundant intermixed T cells (CD3 and CD5 positive) and some plasma cells and histiocytes. Cell proliferation index of the lymphoid aggregate was less than 5% (ki67). LEF1 was partially positive, while cyclin D1 was negative. The morphological and immunohistochemical findings were interpreted as lymphoid hyperplasia of bronchus-associated lymphoid tissue (BALT), subject to confirmation by molecular analysis. Heteroduplex analysis was used to examine the B lymphocytes for the presence of polyclonal and clonal populations. For this purpose, three multiplex PCRs for heavy chains (IgH) and three multiplex PCRs for the light chains (Ig–κ/λ) and then analyzed by capillary gel electrophoresis. Immunoglobulin heavy chain (IgH) gene rearrangement study revealed polyclonality of the lymphoid lesion, whereas the analysis of IgKappa B was monoclonal, polyclonal pattern in IgKappa A und IgLambda.

Paraffin-embedded lung tissue was analyzed by multiscale XPCT. (a–c) XPCT-scan could reveal the peribronchial, nodular infiltration of a lymphoid aggregates with GCs (rendered GCs in c) in the BALT. (d) XPCT scan demonstrates the perivascular spreading of lymphoid cells within and surrounding BALT. (e) Segmentation of vascularity is characterized by altered severely abnormal-appearing blood vessels (yellow) which are directly linked to the upstream larger blood vessels (f).

(a–c) Scanning electron micrographs of resected lung tissue illustrated the substantial infiltration of a peribronchiolar and perivascular (white asterisk) lymphoid aggregates (blue arrows) and prominent vascular proliferations with intussusceptive pillars (d, arrowheads).

Immunohistochemical assessment of the peribronchial and perivascular lymphoid infiltrate revealed strong CD20- and Bcl2-positivity of germinal centers (CD20, CD23) and the paracortical lymphoid neoplasia with scattered T cells (CD3). Proliferation was slightly elevated in the paracortical zone.
Despite the unusual nature of this B cell lymphoproliferation with repeated monoclonal protein bands in IgK, the differential diagnostic classification was challenging due to the inconclusive immune profile. The differential diagnosis of nodular lymphofollicular hyperplasia and partial infiltration by a marginal cell lymphoma was particularly relevant in this case. Other B cell lymphomas, such as SLL or mantle cell lymphoma, could be excluded with confidence. In addition, an association of a previous inflammation with COVID-19 disease seems conceivable. Taken together, the lymphoid lesion was interpreted as a lymphoid hyperplasia with an early infiltration of by a low-grade extranodal B cell lymphoma of the bronchus-associated lymphoid tissue (BALToma). The patient underwent extensive clinical and radiological staging procedures. Subsequent follow-ups will be conducted at regular intervals to monitor the patient’s condition.
Discussion
Primary lymphomas of the lung are rare, whereas BALT lymphomas are the most common entity, accounting for approximately 75%–90% of all cases. The median age of presentation is 62 years. Females are more commonly affected, making up 55% of patients. Approximately 10% of patients suffer from autoimmune disease, 45% are smokers, while 19% have a known preexisting lung disease. 1 The overall prognosis for patients with mucosa-associated lymphoid tissue (MALT) lymphoma of the lung is good. The 10-year survival rate is over 70%. 2
Lymphoid organs are commonly classified as primary (or central), such as the thymus and bone marrow, or peripheral secondary lymphoid organs (e.g., lymph nodes, spleen, tonsils, Peyer’s patches); and are primarily responsible for inducing local immune responses. Tertiary lymphoid tissues (TLTs), on the other hand, are inducible ectopic lymphoid portions that develop de novo at sites of chronic inflammation in nonlymphoid organs.
Like lymph nodes, TLTs initiate adaptive immune responses and coordinate local tissue immunity. They form highly dynamic structural morphological entities ranging from lymphocyte aggregates to highly organized clusters with networks of follicular dendritic cells and germinal centers. 3 They certainly share structural and functional features with conventional secondary lymphoid organs: T cell zones, B cell zones, marginal zones of antigen-presenting cells, reticular stromal networks, and high-endothelial veins (HEVs; Figure 4). However, the developed lymph node is quite different from the tertiary inducible lymphoid tissues in terms of plasticity. During ontogenetic lymph node development, maturation and accumulation of lymphoid progenitors from the mesoderm occur at more or less anatomical predilection sites, where germinal center structures form via HEV around preformed sinusoidal structures. 3 In contrast, TLTs can induce a functional lymphoid aggregate within a very short time of a few hours. An important cell population that plays a regulatory role in the neogenesis of TLS is regulatory T cells (Tregs). An effective humoral and cellular immune response is based on primary (thymus, bone marrow) and secondary lymphoid organs (e.g. spleen, tonsils, lymph nodes).3,4 In 1973, John Bienenstock introduced the term BALT as part of MALT. All of his studies were performed in rabbits.4,5 In a morphologic study of lungs from many different species, the frequency of BALT varied widely, from 100% in rabbits to 30% in pigs and none in cats. 6 In a study of many adult human lungs with a pathologic diagnosis of chronic bronchitis and bronchiectasis, BALT was found in 8% when the bronchus was obstructed by a tumor, 7 whereas no BALT was found in 34 adult human lungs without inflammation. 6 In the lungs of children who died of sudden infant death syndrome, 34% had BALT, and in those who died of other causes, 44% had BALT on histologic sections.8,9 In a recently published study, 10 BALT structures were compared with pulmonary draining lymph nodes in children from birth to 13 years of age. The number of BALT decreased after 3 years of age, coinciding with an accumulation of memory T cells. B cells generated a skewed antibody response to several respiratory pathogens. Gould and Isaacson 11 studied 102 fetal lungs and 12 infant lungs whereby fetal lungs showed BALT in 77% of cases and all cases of fetal lungs with BALT-like structures were associated with choriomeningitis or postpartum pneumonia. 11 Lymphoid aggregates can be induced in structures known as inducible bronchus-associated lymphoid tissue (iBALT), which are often located in peribronchiolar or perivascular spaces. It is well documented that BALT is an obvious sign of chronic stimulation, for example, in smokers 82%, in nonsmokers 14%. 12 In a more detailed study, Hogg et al. could show a correlation of BALT formation with the stage of chronic obstructive pulmonary disease (COPD).13,14 The severe stages of COPD have been shown to be associated with the emergence of remodeled and dendritic cell-rich alveolar–lymphoid interfaces. Although chronic antigen stimulation is generally associated with the formation of iBALT and MALT lymphoma 15 by increasing the risk of lymphomatous transformation, there is controversial evidence for the role of bacterial and viral infections. 16
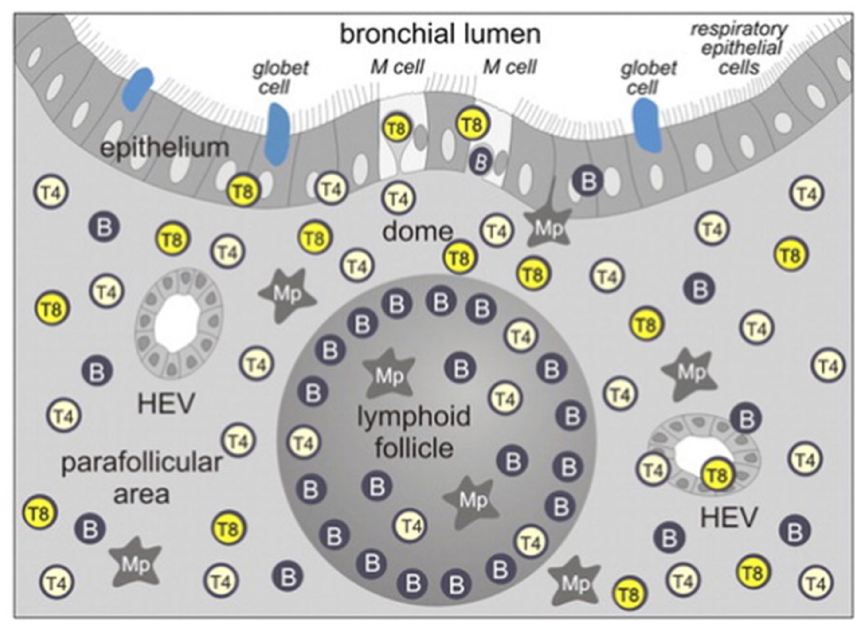
Schematic drawing of BALT in distribution of lymphocyte subsets is shown as in BALT of children. Three main compartments are: (1) Central follicular vein with accumulation of B lymphocytes. (2) The parafollicular area with mainly T lymphocytes and specialized HEV and (3) the dome area with a specialized epithelium lacking goblet cells. Various efferent and afferent autonomic nerves are indicated. There are efferent lymph vessels that drain to the regional parabronchial lymph nodes (with permission by Pabst 19 ).
In this study, we hypothesize a clinical evidence between the preceded severe SARS-CoV2-infection and the formation of the nodular lymphoid hyperplasia with the initial formation of a low-grade MALT lymphoma. The recently described SARS-CoV2-related systemic vascular inflammation and endothelial dysfunction17,18 is likely to contribute to this prolonged pulmonary inflammation by a recruitment of perivascular and peribronchial inducible BALT tissue 19 accompanied by vascular alterations and the formation of intussusceptive angiogenesis. It has been demonstrated that angiogenesis and lymph angiogenesis play a pivotal role in the formation of blood tissue. The dynamics of flow and shear forces within HEV in newly formed blood vessels have been shown to be instrumental in the recruitment of new lymphocytes and their subsequent self-organization.20,21 It is evident that several questions have yet to be answered, including the nature of the cells that initiate the BALT anlagen, the contribution of epithelial-derived mediators and local DC networks, the role of T follicular helper cells in iBALT formation and function, and the mechanisms by which Tregs control iBALT organogenesis. 22 The efficient priming of T cell responses directed against unrelated airborne antigens is a crucial process that requires dendritic cells for its sustained presence. 23
Conclusions
Further clinical investigations are needed to elucidate the role of viral and bacterial infections in the formation of BALT tissue and the involvement in the lymphomatous transformation of iBALT.
Footnotes
Ethical considerations
The study was approved by and conducted according to requirements of the ethics committees at the Hannover Medical School (vote Nr. 9022 BO K 2020). All participants gave written informed consent.
Author contributions
MA, JR, JF, TW, SEV, TS, RP, and DDJ made substantial contributions to the conception of the work. MA, JR, JF, TW, and SEV made significant contributions to the data analysis and interpretation. MA, TS, TW, and DDJ made significant contributions to the design of the work and the interpretation of data. MA drafted the original manuscript. MA, RP, SEV, and DDJ and other authors substantially contributed to the revision of the manuscript drafts. All authors have approved the submitted version of the manuscript and agreed to be accountable for any part of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been made possible in part by CZI grant DAF2020-225394 by MA and DDJ.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
