Abstract
Giant cell tumors of the bone are typically benign, locally aggressive lesions that rarely metastasize. In this report, we describe the case of a 48-year-old woman presenting with a giant cell tumor of the bone of the hip joint, which demonstrated aggressive histological features and recurrent behavior. The patient developed multiple unresectable pulmonary metastases despite several wide local excisions. Although pulmonary metastases from giant cell tumors of the bone are usually associated with a relatively favorable prognosis, guidelines for the optimal treatment of metastatic giant cell tumors of the bone remain limited. This case highlights the unpredictable nature of giant cell tumors of the bone and emphasizes the need for alternative treatment options in those with resistance to traditional therapies.
Keywords
Introduction
Giant cell tumors of the bone (GCTBs) are rare and account for ~5% of primary bone tumors.1,2 Although generally benign, they are often locally aggressive, with a tendency to infiltrate surrounding tissues and recur. 3 Metastasis is infrequent, occurring in 1%–9% of cases, most commonly to the lungs, but it can also involve the brain, kidneys, liver, and skin.1,3,4 While localized GCTB is associated with a good prognosis, the development of pulmonary metastases may portend a worsened outcome, particularly in cases of true malignant transformation.3,5,6 Nevertheless, the prognosis remains favorable compared with that of other metastatic cancers.3,5,6 We present an interesting case of a patient with GCTB of the hip joint who developed multiple pulmonary metastases.
Case presentation
A 48-year-old woman presented with a right hip fracture, and the radiographic imaging revealed an osteolytic lesion with cortical breakthrough involving the head and neck of the right femur (Figure 1). Multiple biopsies of the hip lesion established the diagnosis of GCTB. Histopathology showed an aggressive-appearing giant cell-rich lesion with secondary aneurysmal bone cyst (ABC) changes (Figure 2). The mononuclear stromal cells (neoplastic cells) exhibited increased cellularity and mitotic activity (up to seven mitoses per 10 high-power fields). An antigen Ki-67 (Ki-67) stain indicated a high proliferative index, with diffuse labeling in ~40% of cells. Importantly, markers for malignancy, including tumor protein p53 (p53) immunohistochemistry, were negative for mutation, and fluorescence in situ hybridization for mouse double minute 2 homolog (MDM2) amplification was also negative.
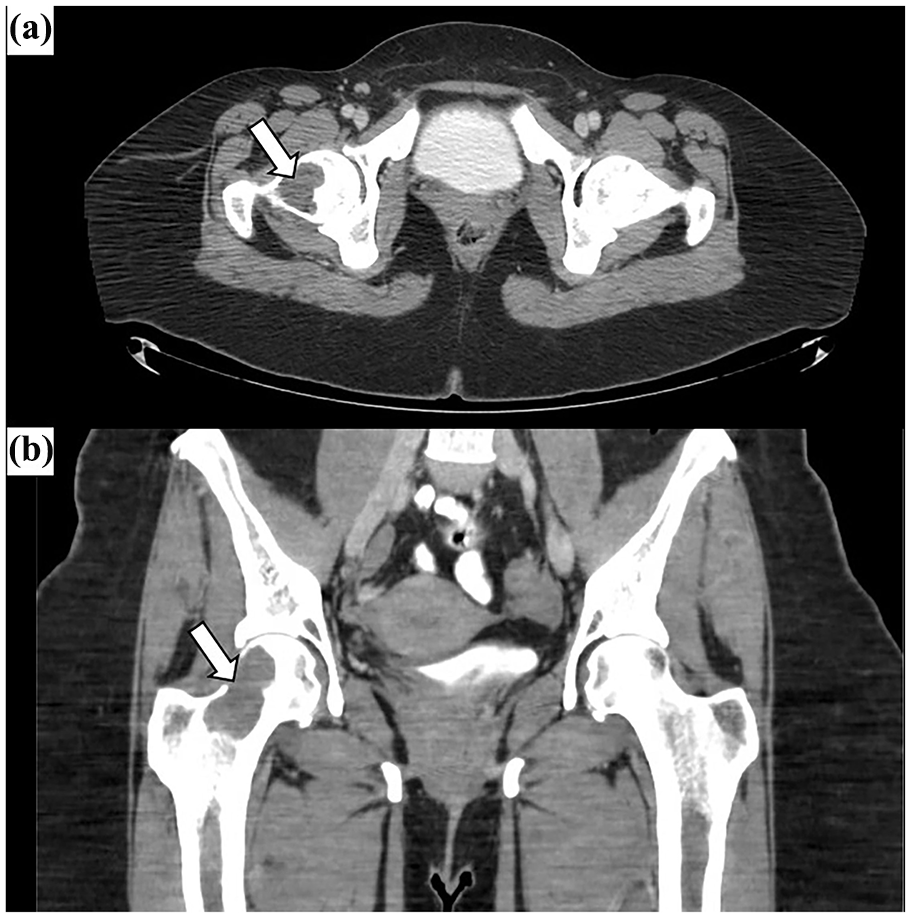
Computed tomography of the pelvis. Axial (a) and coronal (b) views demonstrate an osteolytic lesion (white arrow) in the right femoral head and neck, with evidence of cortical breakthrough.

Histopathology of the primary GCTB from the right femur. The section reveals a cellular proliferation of mononuclear stromal cells admixed with numerous osteoclast-type multinucleated giant cells within a hemorrhagic background. The mononuclear stromal cells exhibit mild nuclear atypia (b, blue arrow) and multiple mitotic figures (a, black arrows); however, no atypical mitoses are identified. (c) Panel illustrates associated secondary aneurysmal bone cyst changes. Immunohistochemical staining for p53 is negative, consistent with a wild-type expression pattern (no mutation; d), while the Ki-67 stain reveals a high proliferation index (e).
The patient underwent initial surgical management consisting of wide local excision of the primary tumor and reconstruction with a total hip endoprosthesis, which provided significant pain relief. Histopathological examination of the resected tissues reconfirmed GCTB with extensive ABC changes and moderate cytologic atypia. Atypical mitoses were absent, and the cytologic atypia did not meet the criteria for osteosarcoma or other malignancies. However, surgical margins were positive. A staging computed tomography (CT) scan of the chest performed showed no evidence of pulmonary nodules (Figure 3) or mediastinal lymphadenopathy.

Initial staging CT scan of the chest. Representative axial views (a, b), obtained at the time of the initial major surgery, demonstrating clear lung fields, and no evidence of pulmonary nodules or mediastinal lymphadenopathy.
Despite initial surgical management, the patient experienced local recurrence after 2 years, necessitating further complex surgical interventions over time. Pathological examination of tissue from these subsequent resections and biopsies consistently showed recurrent giant cell tumor, with persistently positive surgical margins. The tumor’s histology showed increased cytologic atypia in later specimens. Furthermore, later biopsies demonstrated a significant escalation in proliferative activity, with Ki-67 expression exceeding 60%, highlighting increasingly aggressive local disease. Ultimately, attempts to achieve local control led to a final disarticulation procedure. The specimen from this surgery revealed aggressive GCTB with extensive necrosis and positive deep soft tissue margins.
Subsequently, during routine surveillance ~2 years and 2 months after her initial surgery, a follow-up CT scan of the chest revealed multiple new solid pulmonary nodules (Figure 4). To characterize these, an endobronchial ultrasound-guided fine-needle aspiration (FNA) of a mediastinal lymph node (station 12R) was performed. Cytologic examination of the FNA confirmed metastatic giant cell tumor, composed of mononuclear cells, and osteoclast-type giant cells (Figure 5). The immunoprofile of the metastatic cells showed cluster of differentiation 68 positivity, special AT-rich sequence-binding protein 2 negativity and negativity for S100, desmin, and anti-cytokeratin 1 (AE1)/AE3, consistent with metastatic GCTB, and importantly, showed no cytologic features of high-grade sarcoma. These findings indicated metastatic spread of the aggressive, but histologically non-sarcomatous, GCTB.

Follow-up surveillance CT scan of the chest. Representative axial views (a, b) revealing the development of multiple new, bilateral solid pulmonary nodules (white arrows), indicative of metastatic disease.

Endoscopic ultrasound-guided fine-needle aspiration of station 12R lymph node showing mononuclear cells and a multinucleated giant cell on cytology smear (a, Diff-Quick stain) and tumor cells on cell block section (b, H&E stain). Immunoreactivities for CD68 (c) and SATB2 negativity (d) on cell block sections, respectively.
Further staging investigations, including a bone scan and magnetic resonance imaging, showed no evidence of other extrapulmonary metastases. Given the extent and number of pulmonary metastases, surgical resection was deemed technically challenging and associated with high morbidity. Consequently, the patient initiated systemic therapy with denosumab, aiming to control disease progression.
Despite denosumab therapy, the patient’s clinical condition deteriorated due to progressive metastatic disease, leading to respiratory failure, severe sepsis, and death.
Discussion
GCTB typically occurs in young adults, involving long bones and presenting with symptoms like pain and restricted movement, as seen in this case.1,6,7 Radiographically, they appear as eccentric, lytic lesions, and histologically, they are characterized by mononuclear stromal cells and osteoclast-like giant cells.4,7,8 While H3F3A (H3 clade 3A (histone H3.3)) mutations are common and aid in distinguishing GCTB from mimickers like chondroblastoma or telangiectatic osteosarcoma,7,10 the critical challenge lies in predicting their biological behavior.
The pathogenesis of GCTB involves H3F3A-mutated neoplastic stromal cells that recruit osteoclast precursors via receptor activator of nuclear factor kappa-Β ligand (RANKL) overexpression, leading to osteolysis.11,12 While this molecular understanding has paved the way for targeted therapies like denosumab, the triggers for particularly aggressive behavior or metastasis, often in the absence of frank malignant transformation (as indicated by the absence of p53 mutation and negative MDM2 amplification status in this case), remain incompletely understood.9,11
This case distinctively illustrates the aggressive potential of a GCTB that, despite lacking definitive histological features of malignancy throughout its course, led to a fatal outcome. Several pathological features observed from early on hinted at their aggressive nature: an initial Ki-67 proliferative index of ~40% which later escalated to >60%, the presence of secondary ABC changes (often associated with more aggressive GCTBs), increasing cytological atypia over successive recurrences, and, crucially, persistently, positive surgical margins after multiple resections. These factors are recognized contributors to local recurrence, which this patient repeatedly experienced.9,13 The comprehensive, immunohistochemical, and molecular workup on the primary tumor (including negative p53 immunohistochemistry for mutation, negative MDM2 amplification status for the primary tumor, and an immunoprofile of the metastatic lesion that included S100 negativity) was essential in repeatedly confirming the diagnosis as GCTB and excluding primary malignant GCTB or dedifferentiation into a high-grade sarcoma, even as the tumor’s clinical behavior became increasingly worrisome.
Metastasis in GCTB occurs in a small percentage of cases, most commonly to the lungs.1,3,4 Consistent with various reports, the pulmonary metastases in this patient, detected ~2 years and 2 months after the initial surgery despite a clear baseline chest CT, retained the classic histology of GCTB, as confirmed by FNA of a mediastinal lymph node. 10 This phenomenon of “benign appearing” metastases behaving aggressively underscores a clinical conundrum. While such metastases often have a more indolent course than other sarcomatous metastases, they can, as in this instance, lead to significant morbidity and mortality.10,16 The delayed appearance of metastases in this case highlights the critical need for long-term surveillance with regular imaging, particularly for patients with high-risk features such as large tumor size, local recurrence, or specific histological markers of aggression.3,14,15
The management of GCTB with an aggressive profile, as exemplified by the protracted surgical course and eventual metastatic progression in our patient, is particularly challenging. In our reported case, despite an initial wide local excision and subsequent attempts at controlling local recurrence through further resection, persistently positive surgical margins and increasing histological atypia led to a disarticulation. This clinical trajectory highlights the formidable difficulty in achieving durable local control with surgery alone in some GCTBs, especially when faced with such inherent biological aggressiveness. When local control is elusive or metastatic disease develops, systemic options become critical.17,18
Denosumab, a RANKL inhibitor, is indeed a key therapeutic option for unresectable or metastatic GCTB and is the only Food and Drug Administration-approved medical therapy for this condition.19,20 While many patients experience tumor stabilization or regression with denosumab, resistance to the therapy, either primary or secondary, remains a significant clinical challenge, as was evident in this case where denosumab was initiated for unresectable pulmonary metastases. The mechanisms underlying denosumab resistance or insufficient response in GCTB are not fully elucidated but are actively investigated. One contributing factor is that denosumab primarily exerts a cytostatic effect on the neoplastic stromal cells rather than a direct cytotoxic effect. This means that while the therapy can halt tumor growth, it does not necessarily kill the tumor cells, allowing for potential regrowth if the therapy is stopped or if the tumor cells are inherently resilient.21,22 Additionally, the bone formation and sclerosis induced by denosumab can complicate surgical removal of residual tumor cells. This sclerosis can trap tumor cells, making complete surgical excision difficult and potentially leading to recurrence if denosumab is discontinued. 22 Tumor heterogeneity within GCTB may also play a role, as some neoplastic stromal cell populations might be less dependent on RANKL signaling or may develop alternative survival pathways, contributing to resistance. 21 The National Comprehensive Cancer Network guidelines recommend denosumab for patients with unresectable disease and suggest continuation of the therapy until disease progression. 20 However, the limitations of denosumab in a subset of GCTBs, such as the aggressive progression seen in our patient despite therapy, underscore the need for identifying predictive biomarkers of response and developing alternative systemic therapies for patients whose tumors are refractory to RANKL inhibition.23,24 This case, therefore, serves as a sobering reminder of the potential for GCTBs to behave in an aggressive manner, leading to relentless local recurrences and fatal metastatic disease, despite histologically no malignant transformation.
Conclusion
This case underscores the unpredictable behavior of GCTBs and the critical importance of vigilant, long-term surveillance, even following extensive initial treatment of the primary tumor. Although usually classified as benign, GCTBs can manifest with aggressive histological features—such as high proliferative rates and increasing cytological atypia—and pursue a relentless clinical course characterized by multiple local recurrences requiring complex surgical interventions, as detailed in this patient’s experience. The development of pulmonary metastases, even after a significant disease-free interval, remains a critical concern. Our case illustrates the limitations of current therapeutic options including denosumab in the face of such aggressive disease progression. It also highlights the urgent need for more effective treatment strategies and robust predictive biomarkers to improve the prognosis for individuals with this rare and challenging oncologic condition.
Footnotes
Ethical Considerations
Our institution does not require ethics approval for reporting individual cases or case series.
Consent for Publication
Retrospective written informed consent was obtained from the patient’s legally authorized family representative for the publication of this case report.
Author Contributions
M.T., S.A.F., P.J., J.L., Y.D.A., and M.A.A.: all authors participated in conceptualization, formal analysis, methodology, investigation, resources, and writing and editing of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
